Abstract
Synthesis, characterization, molecular docking and evaluation of antibacterial, antiproliferative, and anti-inflammatory properties of new pyridinyl substituted triazole derivatives
Author(s): Bettadapura Gundappa Krishna, Balladka Kunhana Sarojini, Chenna Govindaraju DarshanrajEthyl 1-(pyridin-3-yl)-1 H-1,2,4-triazole-3-carboxylate derivatives were synthesized by reacting 3-aminopyridines with ethyl-2-chloroacetoacetate followed by amination and reaction with different aldehydes under microwave conditions. The novel compounds were evaluated of their in vitro antibacterial, antiproliferative and antiinflammatory activities against pathogens of medical importance. The newly synthesized compounds were subjected to molecular docking studies for the inhibition of the enzyme L-glutamine: D-fructose-6-phosphate amidotransferase [GlcN-6-P] (EC 2.6.1.16). The newly synthesized compounds were characterized by analytical, IR, 1H NMR, 13C NMR and LCMS. Among the screened compounds, Ethyl1-(2-bromo-5-fluoropyridin-3-yl)-5-(2,5-difluorophenyl)- 1H-1,2,4-triazole-3-carboxylate, Ethyl1-(2-bromo-5-fluoropyridin-3-yl)-5-(6-bromo pyridine-2-yl)-1H-1,2,4- triazole-3-carboxylate and Ethyl1-(2-bromo-5-fluoropyridin-3-yl)-5-(2,4-dichlorophenyl)-1H-1,2,4-triazole-3- carboxylate, emerged as most active compounds against all the tests.
Select your language of interest to view the total content in your interested language
Google Scholar citation report
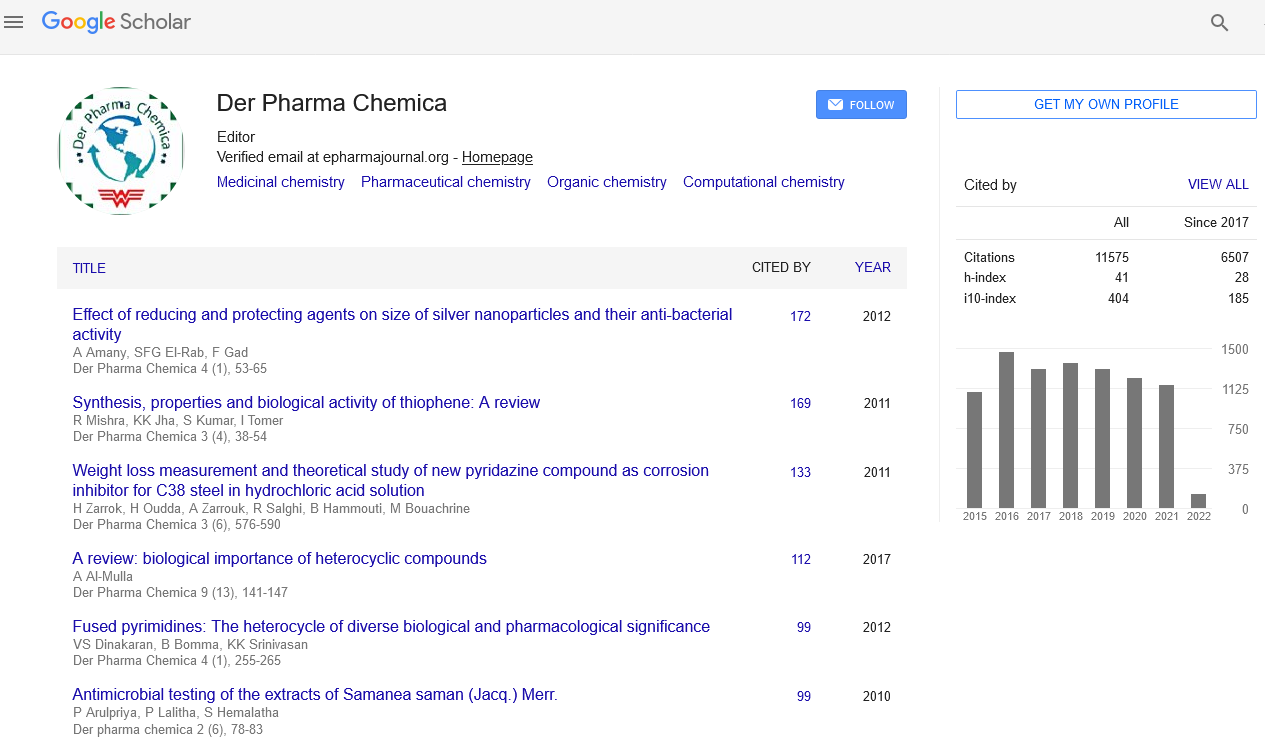
Citations : 25868
Der Pharma Chemica received 25868 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons
DOWNLOADS