Abstract
Synthesis, reactions, structure-activity relationship of 2-benzimidazole analogs as anticancer agents and study their molecular docking
Author(s): Nahed F. Abdel-GhaffarA new series of benzimidazolederivatives ,1,2,3 are reported herein. Their reactions with some nucleophiles gave 4 and 6.The reaction of 3 with sodium azide gave 5and with primary amines gave7a-c. Treatment of 7c with acrylonitrile, cinnamaldehyde, phenylisothiocyanate, gave 8,9and 10. Treatment of 10 with chloroacetic acid and/or malonic acid gave 11 and 12. The benzimidazol-2-thione derivative 2 reacted with acetylacetone to give the dicarbonyl derivative 13 which on hydrazinolysis gave the pyrazole derivative 14. Reaction of 2 with benzalacetophenone derivative, anthranilic acid, sodium nitrite and/or thiourea gave 15,16,17and 18 respectively. The formation of the 2-benzimidazolyl pyrimidine thione derivative 19 was also investigated. Some of the new synthesized derivatives were screened for their antitumor activities and theirmolecular docking and the results were encouraging.
Select your language of interest to view the total content in your interested language
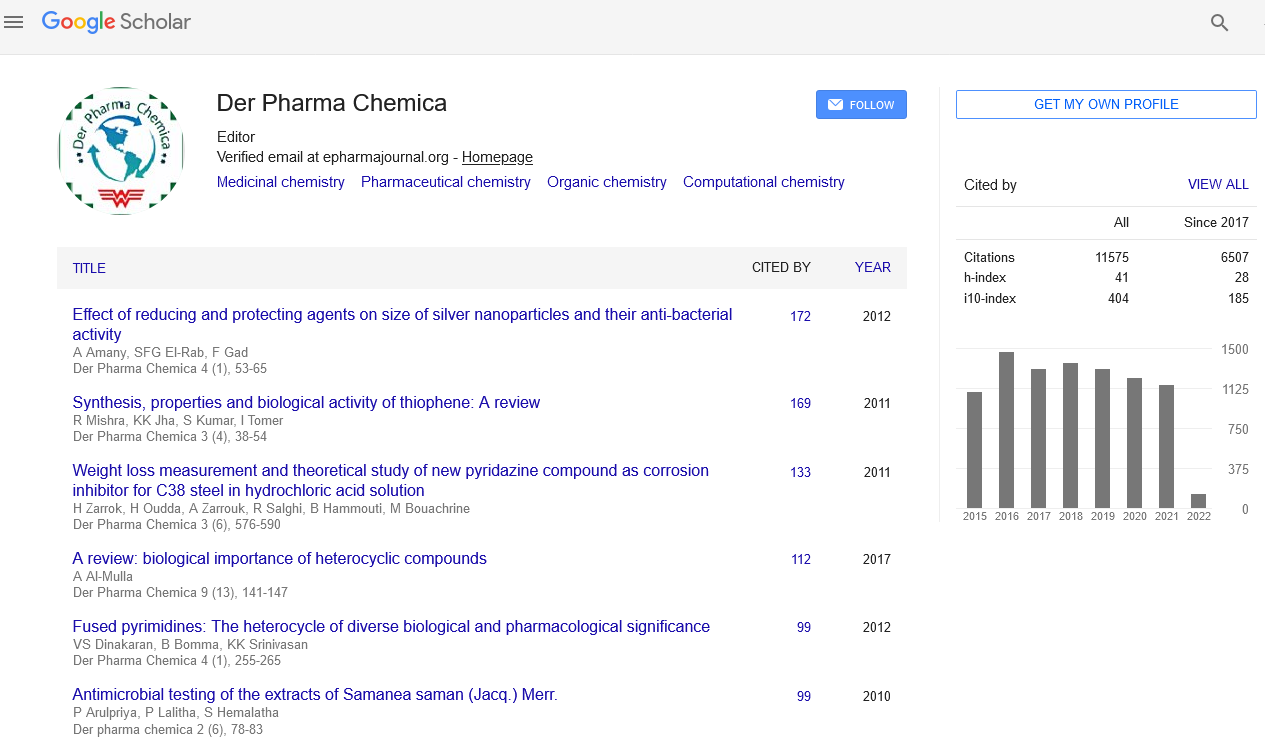
Google Scholar citation report
Citations : 25868
Der Pharma Chemica received 25868 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons
DOWNLOADS