404 Page not found!!
We weren't able to find this page. Here are some other open options you might take up!!Welcome
Discover the convenience of the Immediate Bitwave app for efficient trading anytime, anywhere.
The primary mission of the Der Pharma Chemica is to become the premier source of high quality research from whole of the world. All authors not only receive fast and comprehensive peer-review but also the article promotion to researchers working in the same field.
Der Pharma Chemica A is peer-reviewed and is published in electronic version. The language of the Der Pharma Chemica is English.
Der Pharma Chemica publishes research and review articles, rapid and short communication and book reviews. The current emphasis of the journal includes (but is not limited to) the following areas: medicinal chemistry,pharmaceutical chemistry and computational chemistry.
Der Pharma Chemica is a peer-reviewed journal that is devoted to the dissemination of new and original knowledge in all branches of Chemistry and Pharmacy. The chemistry theme may include Electrochemistry, Computational chemistry, Phytochemistry, Synthetic Drug chemistry, Nano chemistry, Instrumentation analysis; Spectroscopic methods, Chromatographic techniques and the pharmacy theme may include Pharmaceutical analysis, Biopharmaceutics, Industrial Pharmacy etc. Accepted articles may be theoretical or they may report experimental results especially in chemistry and pharmaceutical knowledge. The journal meets analytical operations including samplings, measurements, data analysis and data interpretation of all the experimental results.
The journal Der Pharma Chemica, which deals with traditional analytical procedures, must provide unique applications of the most recent, approaches in the major fields of chemistry and pharmacy sector. The main objective of this journal is to provide a wide range of knowledge in chemical and pharmaceutical sciences to all over the world. Der pharma Chemica is gladly invite the researchers to submit their respective manuscripts who are dealing with chemistry and pharmacy discipline.
OPEN ACCESS POLICY
Der Pharma Chemica publishes all its articles in full open access format which are easily accessible for scientific community.Researchers have free and unlimited access to the full-text of articles published in Scholars Research Library Journals ie Readers can access the published articles freely without login and password. It gives the authors research high visibility, availability and more citations.
You can send your submission online here :- www.scholarscentral.org/submissions/der-pharma-chemica.htmlJournal Der Pharma Chemica is Abstracted/Indexed in:
CNKI
Chemical Abstract Services (USA),
SCOPUS
EMBASE
SCIMAGO
Indian Science Abstract,
HINARI
DOAJ
Open J Gate
Genomics Journalseek
Google scholar
Select your language of interest to view the total content in your interested language
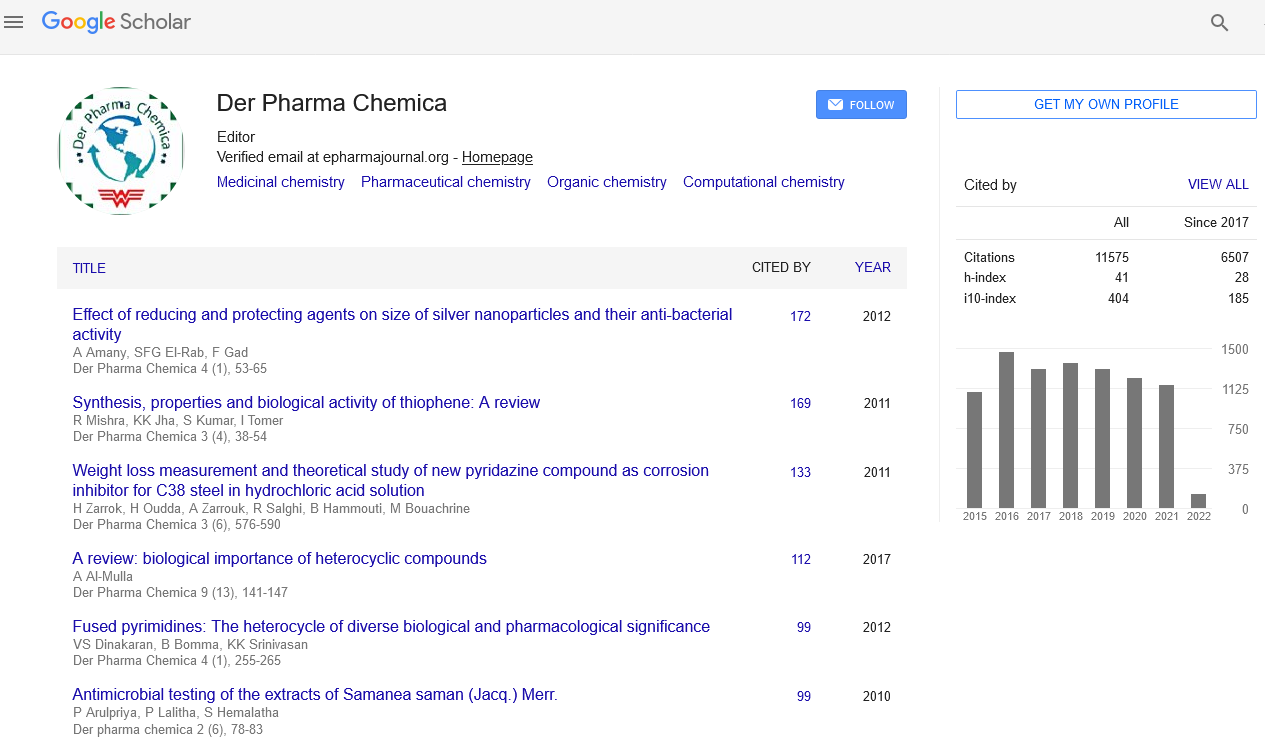
Google Scholar citation report
Citations : 25868
Der Pharma Chemica received 25868 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons
DOWNLOADS