Abstract
Synthesis, Antitubercular Activity and DNA-binding Study of some 2-Amino-3-cyano-4H-chromen-4-ylphosphonates
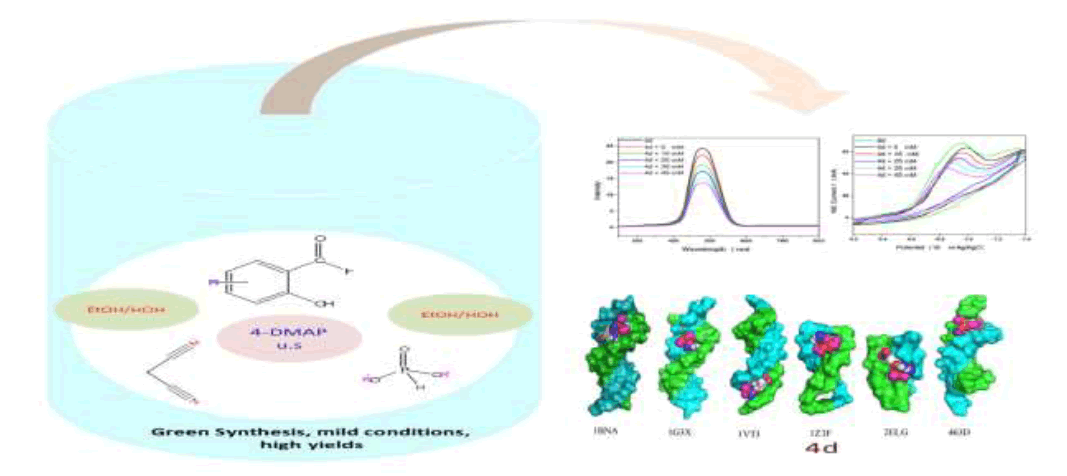
Author(s): Gulzar A Khan, Gowhar A Naikoo, Javeed A War, Umar Jan Pandit, Mehraj Ud Din Sheikh, Imran Khan, Ratnesh Das(2-Amino-3-cyano-4H-chromen-4-yl)-phosphonates 4a-e were synthesized on reacting substituted salicylaldehydes, malononitrile, and diethylphosphite via ultrasound irradiation in presence of catalytic amount of 4-Dimethylaminopyridine (DMAP). This protocol is an environmentally benign preparation and gives excellent yields of the target compounds (88-97%). The structures of new compounds were elucidated by spectroscopic techniques, including Fourier Transform Infra-Red (FTIR), Proton (1H-NMR), Carbon-13 (13C-NMR), Phosphorus- 31 (31P-NMR) Nuclear Magnetic Resonance and Liquid Chromatography/High Resolution Mass Spectrometry (LC-HRMS). All the compounds were screened for antitubercular activity against Mycobacterium tuberculosis H37 Rv using disc diffusion susceptibility method. The in vitro results indicate that all the compounds are potent. Among these 4d bearing nitro group at position C-2' is most effective and, gave a Minimal Inhibitory Concentration (MIC) value of 10 μg/ml in reference with Isoniazid at the same concentrations. DNA binding study of compound 4d with salmon milt DNA (sm-DNA) was confirmed with UV-Visible, fluorescence, cyclic voltammetry and molecular docking. It was revealed that 4d has a strong affinity towards sm-DNA and binds at DNA minor groove with a binding constant (K) 5.317 × 105 mol-1. Molecular docking calculations predicted strong affinity of 4d towards sm-DNA with a binding affinity (ΔG) -7.4 kcal mol-1. Non-covalent interactions, H-bonding and van der Waals forces were predicted as the driving forces of interaction. The compound 4d exhibited selective affinity towards adeninethiamine (A-T) base pairs.
Graphical Abstract
Select your language of interest to view the total content in your interested language
Google Scholar citation report
Citations : 15261
Der Pharma Chemica received 15261 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons



