Abstract
Synthesis, Characterization and Applications of New Hydrotalcite-Like Nano and Innovative Materials
Author(s): Radji G, Bahmani A, Ezziane K, Bettahar N, Sellami MHydrotalcite-like (layered double hydroxides, LDHs) belong to the class of inorganic lamellar compounds, and have a great capacity for insertion of anions. The basic structure of LDHs compounds are similar to a natural hydrotalcite Mg6Al2(OH)16. CO3. 4H2O, in this structure magnesium (divalent cation) was substituted with aluminum (trivalent cation) with a general formula: 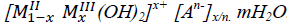 . In recent years, the LDHs compounds have received considerable attention; anion exchange properties have been extensively studied. The interst LDHs magnetic and electrochemical properties permit heterogeneous catalysis and applications in the treatment of polluted water. The LDH compounds were used as adsorbents or catalysts in reaction of degradation to non-biodegradable products. In this work, carbonated sample LDHs of type NiAl-CO3, NiCuAl-CO3 and CuAl-CO3 were synthesized and calcined at various temperatures. The products were used as catalysts in the phenol degradation with hydrogen peroxide. The carbonated samples are obtained by coprecipitation at constant pH with a molar report R (R=MII/MIII) equal to 2.0. The calcination leads to the nanometric size oxide compounds. The as-prepared LDHs-compounds “carbonated” and calcined are characterized by XRD, DTA/TG and FTIR. The calcination occurs at several temperatures and some parameters were tested as catalyst mass, oxidant H2O2 volume, temperature and time of contact.
. In recent years, the LDHs compounds have received considerable attention; anion exchange properties have been extensively studied. The interst LDHs magnetic and electrochemical properties permit heterogeneous catalysis and applications in the treatment of polluted water. The LDH compounds were used as adsorbents or catalysts in reaction of degradation to non-biodegradable products. In this work, carbonated sample LDHs of type NiAl-CO3, NiCuAl-CO3 and CuAl-CO3 were synthesized and calcined at various temperatures. The products were used as catalysts in the phenol degradation with hydrogen peroxide. The carbonated samples are obtained by coprecipitation at constant pH with a molar report R (R=MII/MIII) equal to 2.0. The calcination leads to the nanometric size oxide compounds. The as-prepared LDHs-compounds “carbonated” and calcined are characterized by XRD, DTA/TG and FTIR. The calcination occurs at several temperatures and some parameters were tested as catalyst mass, oxidant H2O2 volume, temperature and time of contact.
Select your language of interest to view the total content in your interested language
Google Scholar citation report
Citations : 15261
Der Pharma Chemica received 15261 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons



