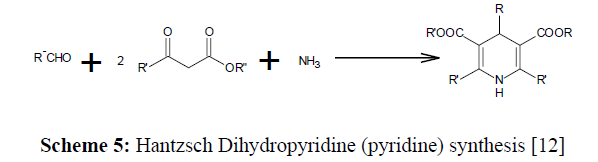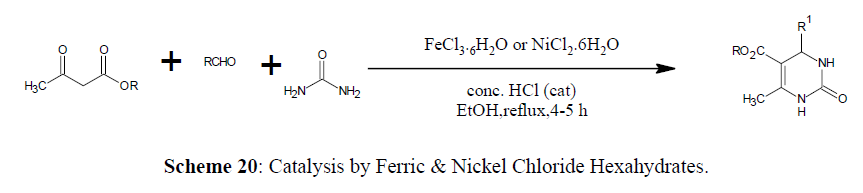Review Article - Der Pharma Chemica ( 2021) Volume 13, Issue 10
A Revision of the Biginelli Reaction: A Combined Experimental and Pharmacological Investigation
Shweta S Jadhav1, Kavita T Daware1, Kshitij S Varma1, Khushbhu Patil2, Shital J Patil1, Ganesh U Chaturbhuj3 and Tushar N Lokhande1*2Department of Chemistry, School of Science, Navrachana University, Vadodara, Gujarat, 391410, India
3Department of Pharmaceutical Sciences and Technology, Institute of Chemical Technology (ICT), Mumbai, India
Tushar N Lokhande, Post Graduate Department of Pharmaceutical Chemistry, Mahatma Gandhi Vidyamandir’s Pharmacy College, Panchavati, Nashik 422003, India, Email: tusharlokhande@hotmail.com
Abstract
Multicomponent reactions, characterized as synthetic procedures that connect three or more substrates in a highly regio and stereo-selective way to deliver structurally complex organic molecules, have seen a tremendous growth in use in the field in organic synthesis. We highlighted the recent advancements in the the synthesis of 3,4-dihydropyrimidin-2(1H)-ones which is a multicomponent Biginelli reaction. Several important Biginelli reactions and advances are also reported.
Keywords
Multicomponent reaction, Biginelli reaction, Organocatalysts, Ionic liquids, Solid phase, Material chemistry
Introduction
In the field of chemistry, A multi-component reaction (or MCR) in which a mixture of three or more compounds undergo chemical reaction to combine into a single product which is also known as a ‘Multi-component Assembly Process' (or MCAP) [1]. Three or more than three reactants are blended in a single reaction vessel to form a new product in this synthetic process. MCRs are noted for having virtually no by-products and containing almost all of the substrates in their final products. MCRs have led to high demand in various research areas of lead compound discovery and combinatorial chemistry. The necessity of rapidly finding and discovering leads caused innovative synthetic methods, leading to the optimal synthesis [2,3]. MCR reactions are performed so that the predominantly main product is formed while side products are avoided. The interpretation is influenced by the characteristics of starting materials and functional groups used, as well as the reaction parameters like solvent, temperature, catalyst, and concentration. Such considerations are crucial in the development and discovery of novel MCRs [4].
History and Development
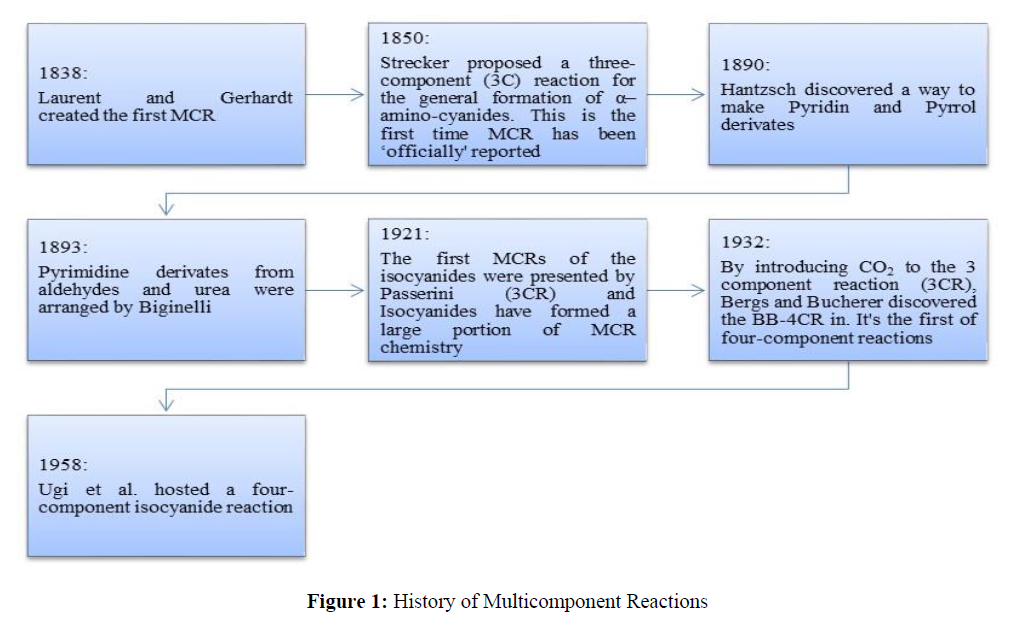
MCRs (multicomponent reactions) are chemical reactions in which the reaction yields highly selective products with a high chance of obtaining molecular diversity in a short amount of time [5] while preserving the main stream of atoms in the starting material since they allow multiple bonds to be formed in a single step [6]. MCR is a capable synthetic method for an operative and quick organic makeover. The chemical moiety to be targeted is accomplished in a single pot, unaccompanied step, allowing for molecular multiplicity and collasion by simply modifying the responding factors [7,8]. MCRs are becoming more relevant in organic chemistry and medicine as a result [9] (Figure 1).
History of Multicomponent Reactions
The following are MCR's benefits
• Use of basic, readily available materials as a starting point.
• Multiple options for rapidly constructing and developing highly complex molecules in a single phase.
• Financial benefits, such as raw material budgets, energy budgets and human capital.
• Environmentally friendly, use of solvents that are not harmful to the environment, and molecule frugality.
• Simple workflow, well-organized catalyst recovery and re-use.
• Ease of experimentation
• Constructing a product with a high degree of stereo selectivity and yield.
• It conserves both resources and time.
• Skips a couple of steps.
• Synthetic solution that is environmentally friendly.
• Stay away from potentially hazardous by-products.
• Automation's probability.
• Stages of intermediate departure and purification.
• Include opportunities for mono- and bi-functional catalysts in the develop entirely green processes [10].
Multicomponent Reactions with Carbonyl Compounds
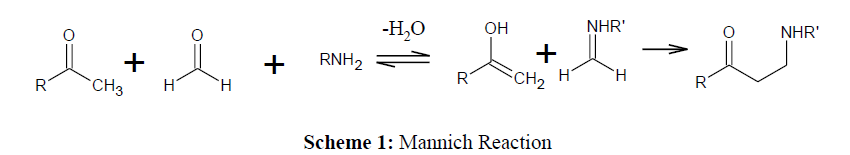
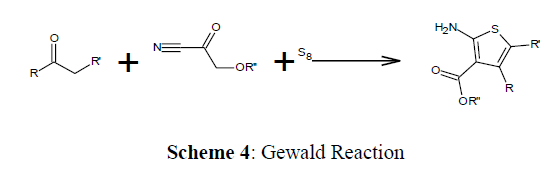
Carbonyl compounds are derivatized into the more reactive intermediates that can react with a nucleophile, is the Mannich Reaction. One of the initials multicomponent reactions to be published (Scheme 1-5)
Scheme 5: Hantzsch Dihydropyridine (pyridine) synthesis [12]
This reaction can occur if one of the carbonyl compounds interacts with the amine more quickly to form an imine while the role of a nucleophile is played by other carbonyl compound. Preforming the intermediates is an alternative in circumstances where at the same rate, both carbonyl molecules will react as the nucleophile otherwise lead to imines, resulting in a typical multistep synthesis [11].
As shown by a variety of name reactions, Carbonyl compounds were significant in the discovery of multicomponent reactions in the early phases:
Biginelli reaction
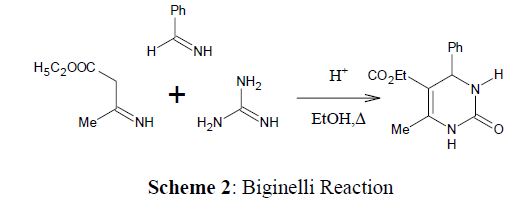
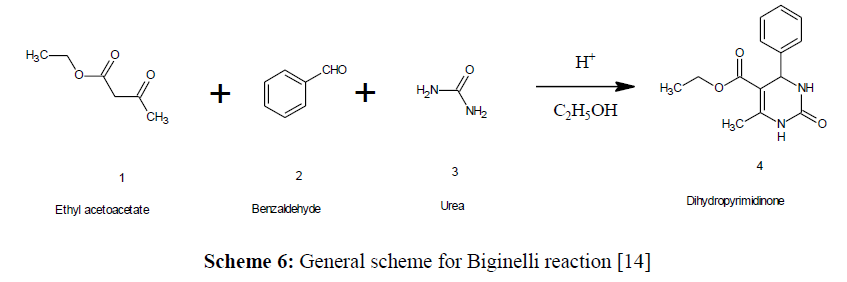
The Biginelli reaction [13], which produces Dihydropyrimidinones from aldehyde, ethyl acetoacetate, and urea, is a typical multicomponent reaction discovered by Pietro Biginelli, an Italian chemist in 1893 (Scheme 6).
Scheme 6: General scheme for Biginelli reaction [14]
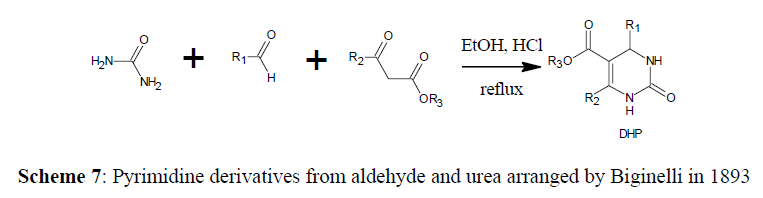
Revealed that the ethyl acetoacetate, urea, and an active aryl aldehyde (as shown in scheme) undergoes cyclocondensation with the presence of an acid as a catalyst (Bronsted acids or Lewis acids, for example trifluoroacetate hydrate [15] and boron trifluoride [16]) for the first time in 1893, a year after discovery of dihydropyridine (DHP) by Hantzsch [17] which gave 3,4-dihydropyrimidin-2(1H)- ones as major products (Dihydropyrimidines or Biginelli adducts) [18]. This method latter recognized as the Biginelli reaction or Biginelli condensation [19] (Scheme 7).
In reaction, the aldehyde portion is widely used. In general, ortho, meta, and para-substituted aromatic aldehydes give best results and donor groups reduce electron density from the meta- or para-positions are frequently used providing good yields of the desired products, but aldehydes with bulky ortho-position substituents give lower yields, where aliphatic aldehydes yield moderate results [20,21]. Furan derivatives and pyridine rings are examples of heterocyclic aldehydes that can be employed in Biginelli reaction. Various substrates used in Biginelli reaction are alkyl acetoacetates, acetoacetates with β-Keto-substitution and thioesters. Cyclic or acyclic diketones may also be used as a substrate.
In terms of structural variation, the urea portion of the Biginelli reaction has the most restrictions. Substituted ureas, produce strong yields. Thioureas along with their substitutions are also commonly used, but when compared to ureas, yields are often lower and reaction times are longer [22]. Dihydropyrimidinones received a lot of publicity by reason of the wide range of therapeutic and pharmacological activities like, antibacterial, calcium channel blockers, antihypertensive agents, antiviral, anti-inflammatory, antitumor and α-adrenergic antagonists [23].
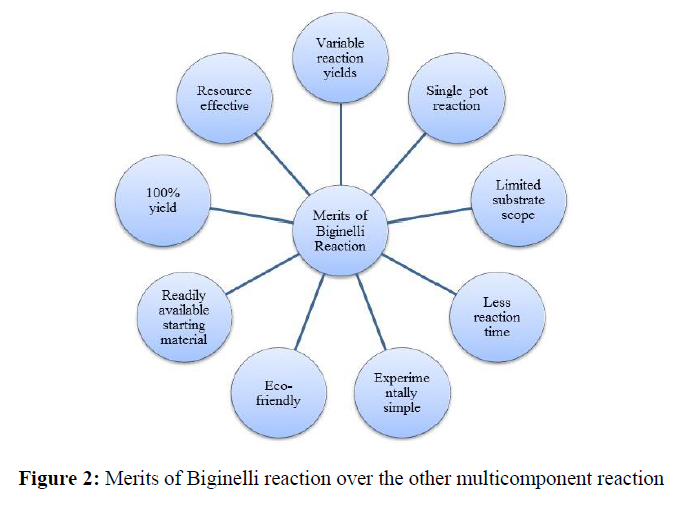
Merits of Biginelli reaction over the other multicomponent reaction (Figure 2)
Reaction Mechanisms
The Biginelli reaction consists of a set of bimolecular events that provide the desired dihydropyrimidinone [24]. Following are the different mechanisms reported.
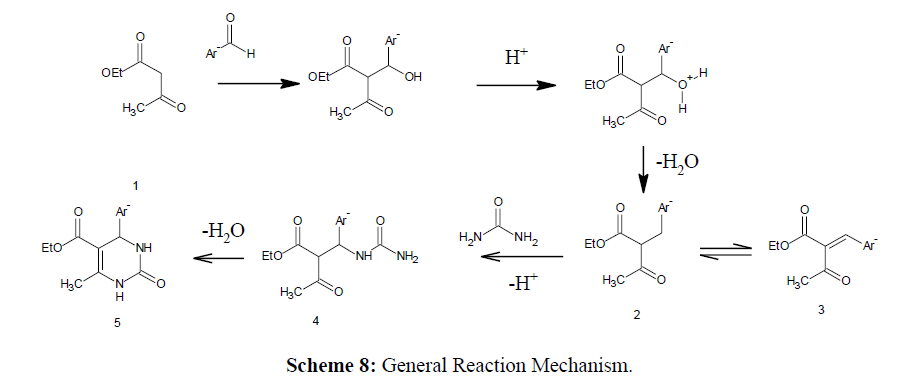
According to a mechanism put forwarded by Sweet in 1973, the step that leads to the carbenium ion (2) and is the rate-limiting step is the aldol condensation of ethylacetoacetate (1) and the aryl aldehyde. Intermediate (4) is produced by urea due to the nucleophilic addition, which easily dehydrates to provide the desired product (5) [25] (Scheme 8, 9).
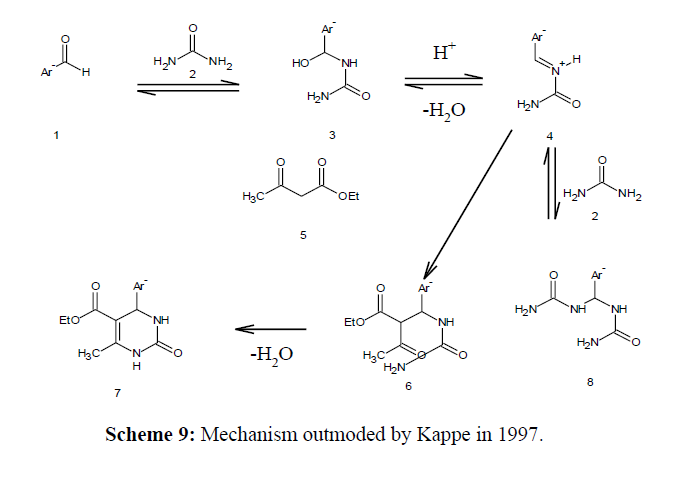
In 1997, Kappe supplemented this mechanism
The initial step in this mechanism is the nucleophilic addition of urea to the aldehyde, which is rate determining step [25,26]. The ensuing condensation process is catalysed by the addition of acid, followed by formation of imine nitrogen. Then the β- ketoester joins the imine bond, results in closing the ring due to amine's nucleophilic attack on the carbonyl group. This final step results in formation of Biginelli compound, which results in a second condensation. Three additional Protonated intermediate processes have been postulated as a mechanism [27].
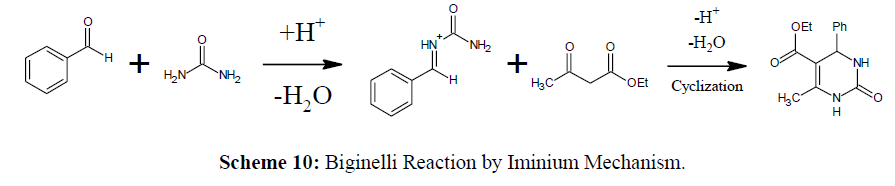
Iminium Mechanism (Ethyl Acetoacetate with Benzal-Bisurea)
In the iminium mechanism, urea and aldehyde reacts to produce iminium intermediate and later the nucleophillic addition of β- keto ester give DHPM (Dihydropyrimidinones) [28] (Scheme 10).
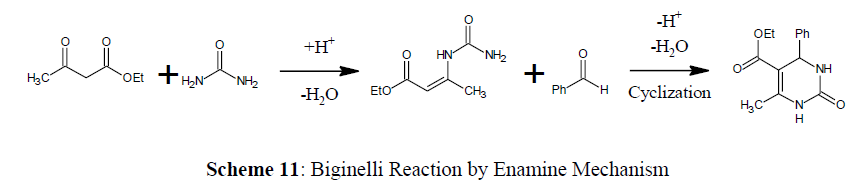
Enamine mechanism (Benzaldehyde with ethyl β-carbamidocrotonate)
In this mechanism β-keto ester and urea create a CN bond to form enamine intermediate, which further reacts with the aldehyde to form DHPM [29] (Scheme 11).
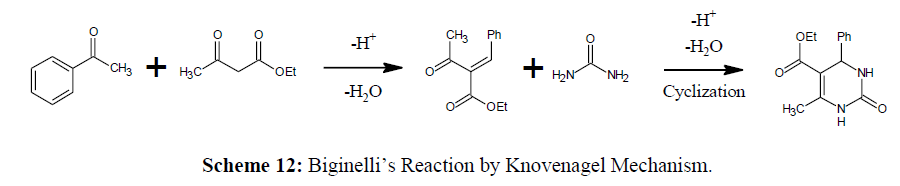
Knovenagel Mechanism (Ethyl Α-Benzalacetoacetate with Urea)
A Knoevenagel type mechanism is used in the third reaction mechanism [30]. The combination of the aldehyde with the β-keto ester produces a carbenium ion as intermediate, and it forms the DHPM when interacts with urea (Scheme 12).
Different intermediates were discovered on each of the three paths. They supplemented their research with a basic theoretical research into the formation of C-C and C-N bonds, concluding that the iminium route was preferable compare to the other two. The generation of C-C bonds was determined to be the rate-determining stage (RDS) in the process followed by nucleophilic addition of the β-keto ester.
Mechanistic studies [28]
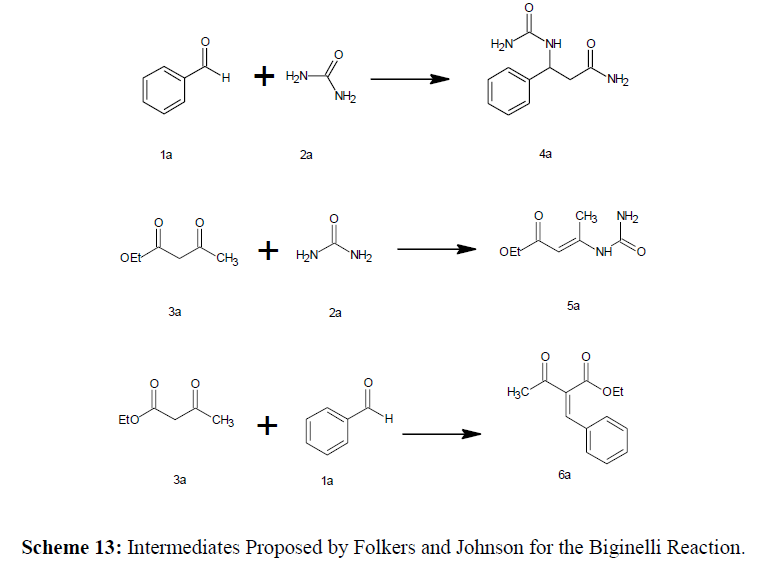
Folkers and Johnson Proposed Mechanism (1933)
In 1933, Folkers and Johnson made the first attempts; only the iminium method was used to get the DHPM with some similarities to the Mannich Condenstion. During the 1930s, Folkers and Johnson proposed that intermediates 4a, 5a, and 6a could be formed. Benzaldehyde (1a) and two equivalents of urea (2a) will produce N,N-benzylidenebis-urea (4a) by intermolecular condensation. The enamine is a intermediate for 5a is formed by condensation of ethyl acetoacetate (3a) and urea (2a), while the Knoevenagel adduct which is intermediate for 6a formed by condensation of 3a and 1a. (Scheme 13)
For the generation of dihydropyrimidine (DHPMs), different combinations of the three reaction components were investigated which are
• The teramolecular process in which Benzaldehyde, ethyl acetoacetate, and two equivalents of urea are combined.
• The reaction of benzaldehyde (1a) and methyl β-carbamidocrotonate (5a)
• The arrangement of ethyl acetoacetate (3a) and benzal-bisurea (4a),
• The reaction of ethyl α-benzalacetoacetate (6a) and urea (2a)
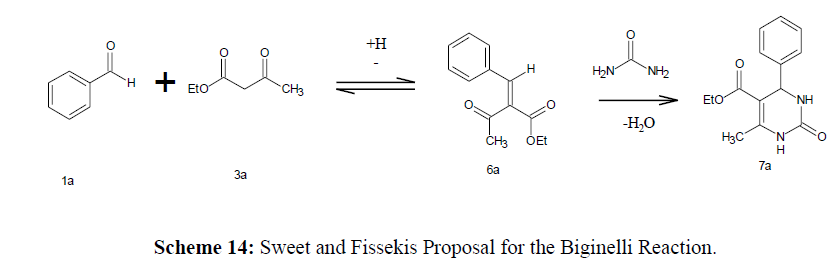
Sweet and Fissekis Proposed Mechanism (1973)
The second mechanism was proposed by Sweet and Fissekis in 1973. The Knoevenagel pathway produces a carbenium ion intermediate, which is used in the process. A method comprises a Knoevenagel condensation between benzaldehyde (1a) and ethyl acetoacetate (3a). (Scheme 14).
The carbenium ion formation is the rate-limiting acid catalyzed Knoevenagel reaction step and is the basis for the mechanism. It was suggested that carbenium ion intermediate react with urea (2a) to form an adduct compound, which yield the Biginelli DHP (7a) by intramolecular condensation. The fact that 6a, which was prepared independently, reacts with urea (2a) under acidic conditions to produce (7a) supports this process.
Organocatalysts in the Biginelli Reaction
Organic Acids as Catalysts for Biginelli Reactions
Metal-based catalysts which are being phased out in favour of organocatalysts [31]. Organocatalysts have low to no toxicity, the use of such catalysts is increasing because they are simple to work with and have a low sensitivity to ambient oxygen or water, and are usually inexpensive [32]. Organocatalysts with any of these desirable properties are organic acids which are reported as useful catalysts in the Biginelli reaction. The Biginelli reactions were successfully catalysed by tartaric, citric, and lactic acids. In these reactions, urea along with thiourea substrates that have been discovered to be suited [33, 34]. Ascorbic acid is a good catalyst but it takes a long time to react, and when thiourea is employed instead of urea, the related products are produced in lower yields.
Other organic acids which are tested for Biginelli reaction are Imidazole-1yl-acetic acid was used [35]. Use of this catalyst shows more than 85% yield. While using urea or thiourea as the starting material, there was no difference in yield. It might be used up to seven times before losing its potency.
Trifluoroacetic acid (TFA) was used as organic catalyst to synthesise Biginelli reaction with large number of aldehydes and urea or thiourea, gives Biginelli products in shorter reaction time with high yields [36].
Phenyl phosphonic acid in Biginelli reactions as a catalyst was used [37]. Electron-donating or electron-withdrawing properties of aromatic or aliphatic aldehydes, and heteroaromatic aldehydes were investigated as noble substrates. Use of urea shows 86–97% yield and 65–72% yield was achieved when thiourea was used. The Biginelli adducts in outstanding s (when urea was used, and in moderate yields [38]. Enantioselectivity of the Biginelli products was stated when SPINOL-phosphoric acid was used as catalyst [39] and more than 80% yield was achieved.
Stereoselectivity of Amino Acids Catalysts in Biginelli Reaction
Derivatives of Proline
In the Biginelli reactions, stereoselective catalysts such as l-proline and its derivatives are employed. This amino acid produces enamines, which are important intermediates to form Biginelli products.
When l-proline supported on silica was used, aromatic aldehydes show more 80% yield [40] but benzaldehyde and thiourea, gave low yields.
Other Amino Acids
l-amino acids supported on aluminium chloride (L-AAL/AlCl3), which were found to be robust and water-resistant, and acting as a Lewis acid or base in the reaction medium. This could be reused up to five times without losing its catalytic activity with more than 85%yields [41]. In the Biginelli reaction, glycine nitrate used as a catalyst which produced the preferred products in high yields (>72%), After ten reuse cycles, the catalyst was still effective [42]. When only 0.03 mol percent of glutamic acid was used in the Biginelli reaction, it showed good catalytic efficacy [43].
Polymers and Macrocycles
The Biginelli reaction has been stated to be catalysed by chitosan which have high molecular weight and are biodegradable as well as less toxic, and is reusable up to five times without losing its effectiveness. According to the scientists, the Knoevenagel condensation is involved in catalysis [44].
Calixarenes have been identified as effective catalysts by Silva and colleagues who discovered that adding a sulfonyl group with calixarenes enhanced the catalytic properties, gives high yields. Macrocyclic monomers were used in the same quantities as calixarenes resulted in lower performance, Calixarenes should be utilised as supramolecular catalysts [45].
The sulfonated-cyclodextrin were used under solvent-free conditions. The catalyst performs better with high yields (>71 percent) [46]. Cyclodextrin when combined with HCl formed the product after 12 hours, in a 48 percent yield [46]. When aromatic aldehydes were used instead of aliphatic aldehydes, excellent yields (>84 percent) were observed [47].
Enzymes and Biocatalysts
Preparation of biginelli adducts by using pineapple juice as a catalyst was recorded in 2011 [48]. Different lipases were used to catalyse the reaction [49]. The enzymes catalyst came from microorganisms, animals, and plants, and were purified. There are reports that using bovine serum albumin (BSA) and aldehydes in combination with ethanol generated the maximum Biginelli yields. The lipase catalyst can be used for two cycles only [49].
Biginelli Reactions with Other Organocatalysts
Organocatalysts such as Hydrazine derivatives were used as for the Biginelli reaction, contributed shorter reaction durations and results in higher yields of the target chemicals. (70–97%) where solvent employed were isopropyl alcohol or DMSO [50]. Ultrasound irradiation with water as the solvent provided more than 47 % yields using Thiamine hydrochloride (5mol%) as a catalyst [50]. The production of Biginelli adducts with ammonium trifluoroacetate as a catalyst was also reported [51].
Pentafluorophenyl ammonium triflate, a Bronsted acid, was as an operational catalyst in Biginelli reactions in elevated yields (>85%) in acetonitrile at room temperature. This approach worked for aliphatic aldehydes. In the absence of solvents and microwave irradiation with high yields, 11 Biginelli adducts were obtained [52].
In a Biginelli reaction, tetrabutylammonium bromide was also shown to serve as a catalyst [53] As a result, it catalysed the Biginelli reactions without solvents but with microwave irradiation.
In the synthesis of biginelli products, ammonium thiocyanate can also be utilised as a self-promoting reactant. Being a weak acid, it speeds up the process to produce Biginelli compounds in a solvent-free environment at 110°C. Increased yields, less reaction time, a solventless environment, simple workup, and the tolerability with a various functional groups are all advantages of this approach. Ammonium thiocyanate is a commonly available, less expensive, safer, and industrially acceptable substance that benefits both the economy and the environment in the current approach [54].
Sulfated polyborate is also used to catalyse the reaction in a solvent-free environment at 100°C. The catalyst has been verified as both a Bronsted and a Lewis acid catalyst which was made by dehydrating boric acid followed by sulfonation. The catalyst's recyclability has both economic and environmental benefits [55].
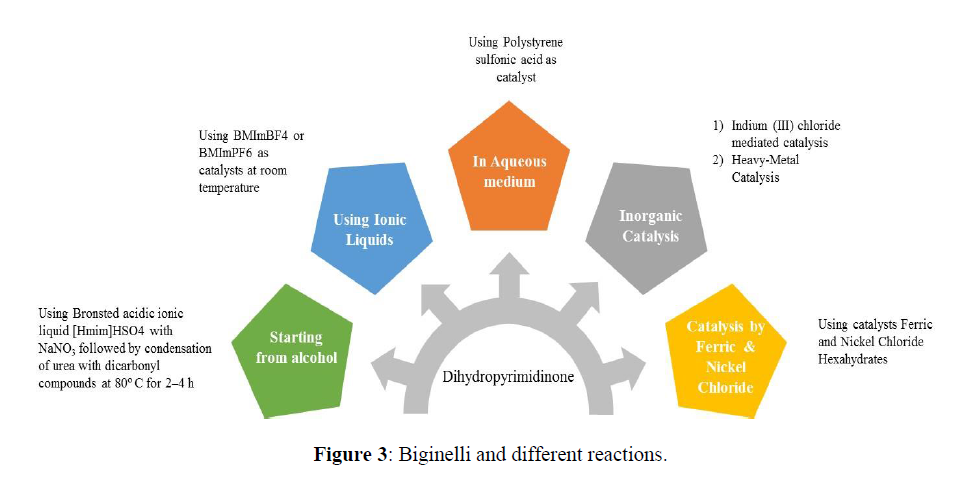
Biginelli and different reactions
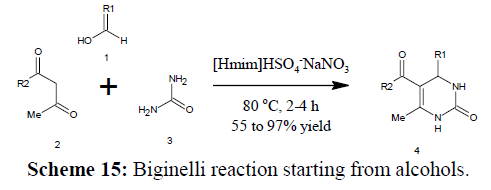
Biginelli reaction starting from alcohols
Biginelli reaction was reported using alcohols as starting material. The catalyst used is Bronsted acidic ionic liquid [Hmim]HSO4 which catalyses sequence oxidation of aromatic alcohols by using NaNO3 followed by condensation of urea with dicarbonyl compounds in the same vessel at 80ºC for 2–4 h to yield 3,4- dihydropyrimidin-2(1H)-ones in 55–97% yields (Scheme 15) [56].
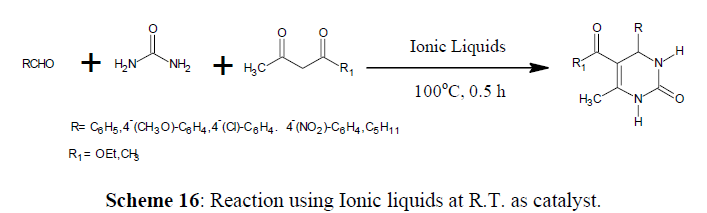
Biginelli Reaction Using Ionic liquids
The Ionic liquids at room temperature have a lot of potential as environmentally acceptable media for catalytic processes. The evidence provided that use of ionic liquids as catalysts are much effective [57]. There are reports of Biginelli reaction is performed under solvent-free condition at room temperature using 1-n-butyl-3-methylimidazolium tetrafluoroborate (BMImBF4) or hexafluorophosphorate (BMImPF6) [58] as catalysts (Scheme 16).
The following typical experimental procedure was followed
Aldehyde, β-dicarbonyl compound , urea (mole ratio 1:1:1.5) and BMImBF4 (0.05–0.2 mmol) when heated in round bottom flask at 100ºC for 30 minutes gives solid formed, filtered and washed using water and recrystallized with ethyl acetate, to achieve pure product.
The following are take away points of Biginelli reactions catalysed by ionic liquids
• No desirable product was detected when benzaldehyde, ethyl acetoacetate, and urea (mole rate 1:1:1.5) was heated at 100ºC for 30 minutes without ionic liquids, it indicates that a catalyst is important for the Biginelli reaction.
• When volume of Ionic liquid increases the conversion rate of reactants to products was increases.
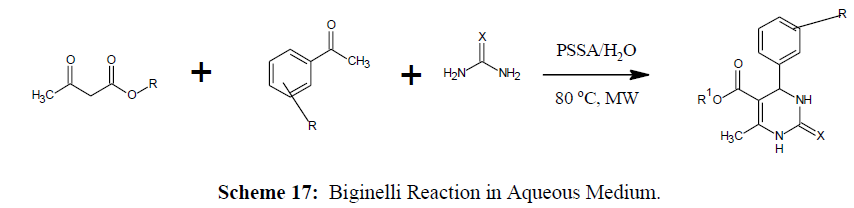
Biginelli Reaction in Aqueous Medium
Water is the cheapest and harmless to environment. Biginelli reaction in aqueous medium was reported for substituted 3,4-dihydropyimidin-2(1H)- ones synthesis by using Polystyrene sulfonic acid as catalyst [59] (Scheme 17).
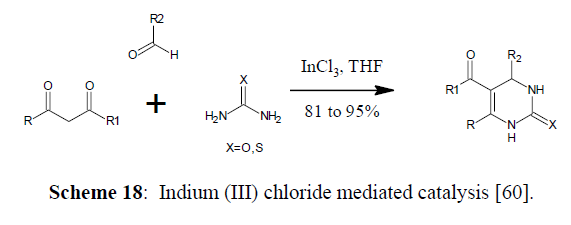
Use of Inorganic Catalysis for Biginelli Reaction
Indium (III) chloride mediated catalysis (Scheme 18,19)
Scheme 18: Indium (III) chloride mediated catalysis [60].
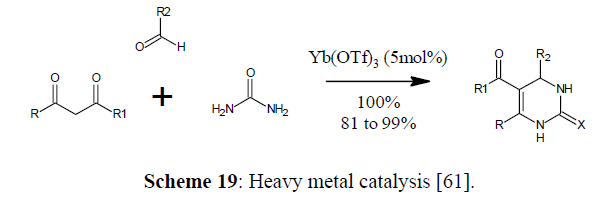
Scheme 19: Heavy metal catalysis [61].
Heavy-Metal Catalysis
Catalysis by Ferric and Nickel Chloride Hexahydrates
Biginelli reaction using catalysts Ferric [62] and Nickel Chloride Hexahydrates is also reported (Scheme 20) (Figure 3).
Advances in Biginelli reaction
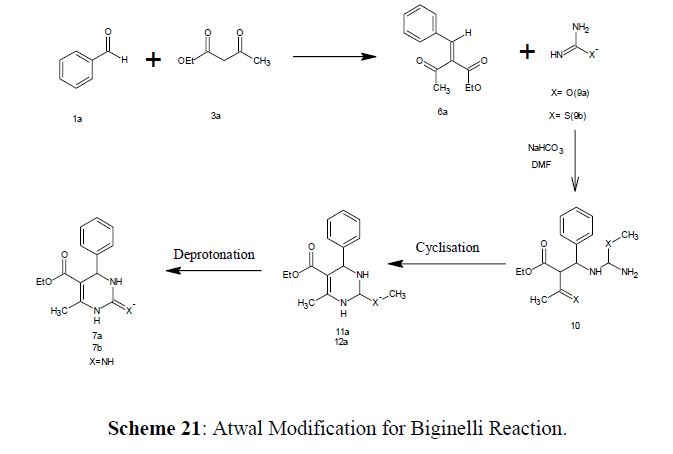
Atwal Modification (1987)
The classic Biginelli reaction is widely used in recent years but it is not trustworthy for the substrate aliphatic or aromatic aldehydes. K. Atwal and coworkers [63] published an authentic approach to Biginelli compounds in the middle of 1987.
Initially, an unsaturated ketoester (6a) undergoes condensation with a suitably safe urea (9a) or thiourea in the presence of sodium bicarbonate (9b). The reaction is thought to take place when a type 10 product of Michael addition is cyclized, yielding dihydropyrimidlnes 11a, 11b. The required Biginelli compounds 7a (urea) and 7b (thiourea) are generated in high overall yields after deprotection with HCl (for 11a) or trifluoroacetlc acid/ ethanethiol (for 11b) (Scheme 20).
Even though this method needs the earlier synthesis of unsaturated ketoesters (6a), it is a viable replacement to the conventional Biginelli condensation due to its reliability and broad applicability.
When guanidine or N, N-dimethyl guanidine undergoes condensation with 6a provides pyrimidines with 2-amino-substitution (Scheme 21).
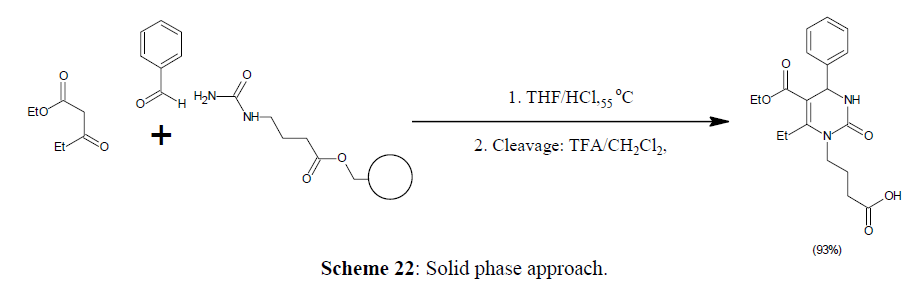
Solid phase approach
A new protocol for preparing DHPM libraries, polymer supported reagents can be used to for product purification and reaction workup [64]. Solid phase synthesis provides high product yields by using excess of reagents. Polymer supported Lewis acid in conjunction with polymer supported scavenging resins is used which remove excess of urea and permits for quick Biginelli synthesis as a simple strategy for purification.
In presence of a catalytic quantity of hydrochloric acid, γ-aminobutyric acid derived urea is connected with a Wang resin can be condensed with excess β-ketoester and aromatic aldehydes to yield the appropriate immobilised DHPMs. After cleavage the product from the resin using trifluoroacetic acid, the free DHPMs are produced in good yields and purity [64,65] (Scheme 22).
Pharmacological Significance
As previously mentioned, the DHPMs are the end-products of the Biginelli reaction, are extremely important in terms of their pharmacological applications. Kappe re-examined DHPM’s biological function as mitotic kinesin inhibitors, antagonists of α1a adrenergic receptor, as well as calcium channel modulators in 2000 [66].
The pharmacological activities of DHPMs has sparked interest in the Biginelli reaction because of the remarkable pharmacological characteristics correlated with this reaction are as follows
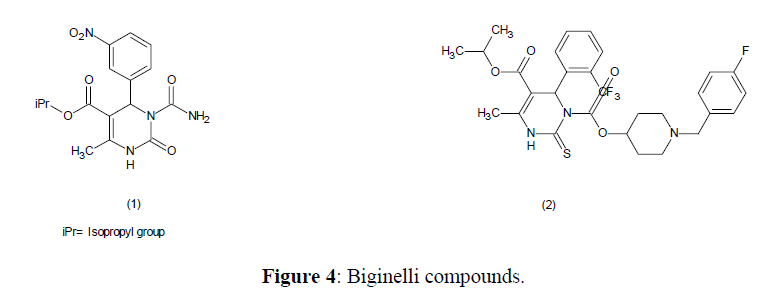
Antihypertensive Agents
There are reports that Biginelli compounds (1,2) are orally active antihypertensive agents (Figure 4).
The biological activity of DHPMs (3) containing a isopropyl, sec-butyl branched ester or an alkylthio group (e.g. SMe) was deemed to be ideal candidate for the activity. Compound (4) is a powerful calcium channel blocker mimicking dihydropyridine [67].
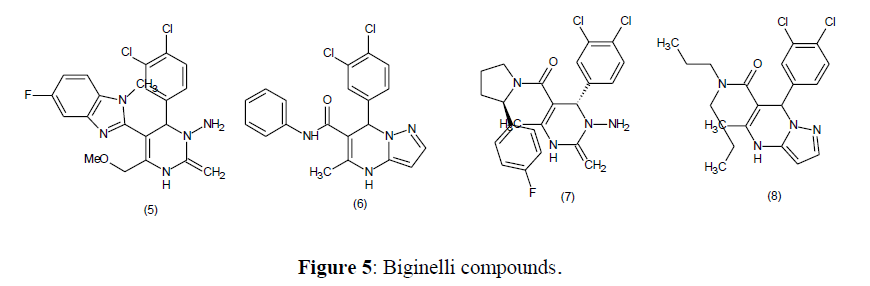
Potassium Channel Antagonists
The activity of potassium channel antagonists was demonstrated by anulation of the benzimidazole ring with Biginelli adducts and these types of agents are currently in preclinical development (5-8) [68] (Figure 5).
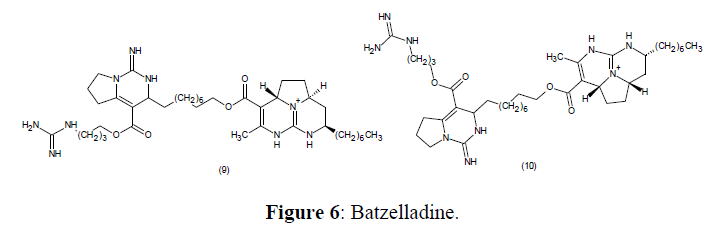
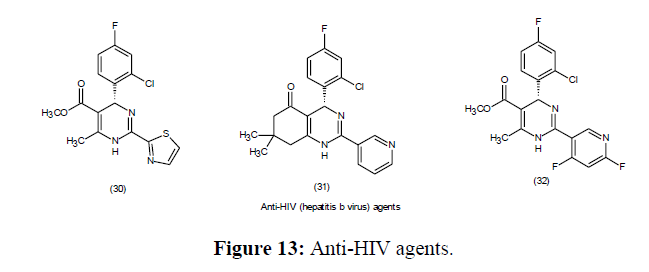
Anti-HIV Agents
The anti-HIV efficacy of Batzelladine A (9) with B (10) which are derivatives of DHPMs are obtained from the marine natural sources is significantly having low-molecular-weight which prevent HIV glycoprotein from binding to CD4 cells [69] (Figure 6).
Antitumor Activity
The human kinesin Eg5 protein is a potent therapeutic target for chemotherapeutics of cancer.
Monastrol (11) is earliest Biginelli product having outstanding activity against cancer and was discovered for being cell permeable molecule with a treatment on cancer cells involving the kinesin Eg5 inhibition [70]. Monastrol inhibits this enzyme's function, resulting in chromosome misalignment and bipolar spindle formation. There are also reports of furyl derivative (12) of dihydropyrimidine was more effective than monastrol [71] (Figure 7).
Anti-Malarials
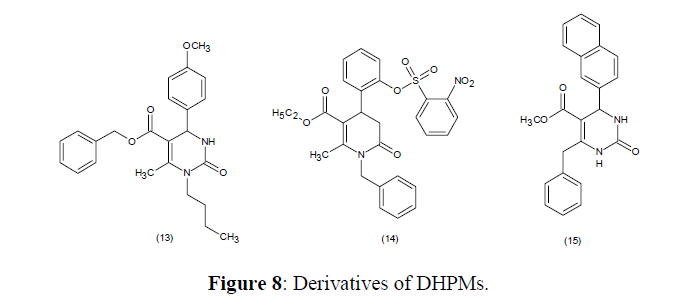
Pyridinone-amides derivatives of DHPMs (13-15) are reported as new class of Hsp70 modulators, which acts against P. falciparum [72] (Figure 8).
Anti-inflammatories
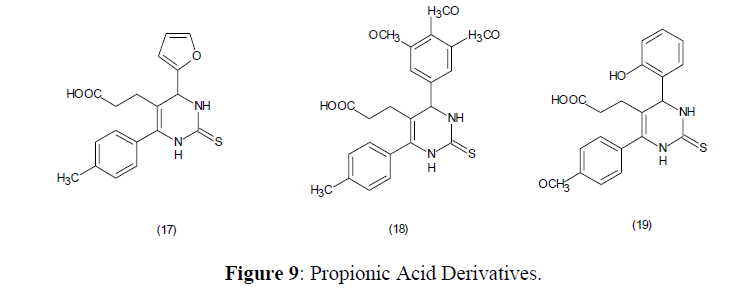
The 3-(4,6-disubstituted-2-thioxo-1,2,3,4-tetrahydropyrimidin-5-yl) propionic acid derivatives (17-19) showed anti-inflammatory effect in the rat paw technique [73] (Figure 9).
Synthesis of anti-inflammatory drugs using Biginelli reaction (Scheme 23)
Under microwave irradiation, three-component condensation of ethyl cyanoacetate, aldehydes, guanidine nitrate along with piperidine (2-3 drops) of was allowed to produce 2-amino dihydropyrimidinone derivatives were reported and the majority of the compounds were having antiinflammatory properties [74].
Anti-tubercular Activity
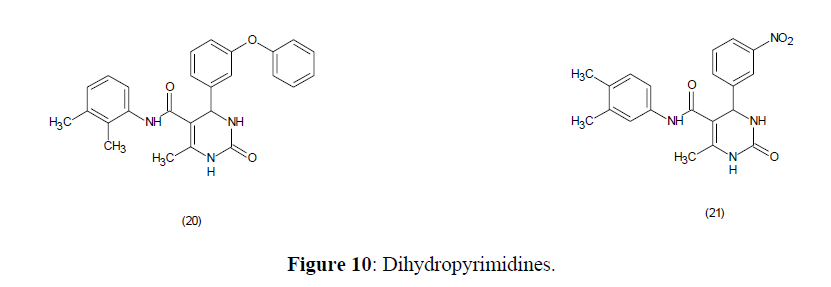
Dihydropyrimidines tested against Mycobacterium tuberculosis H37Rv strain showed anti tubercular activity (Figure 10).
The most active compounds, 6-methyl-2-oxo-1,2,3,4- tetrahydropyrimidine-5-carboxylate, found having more potency than isoniazid [75].
Anti-bacterial Activity
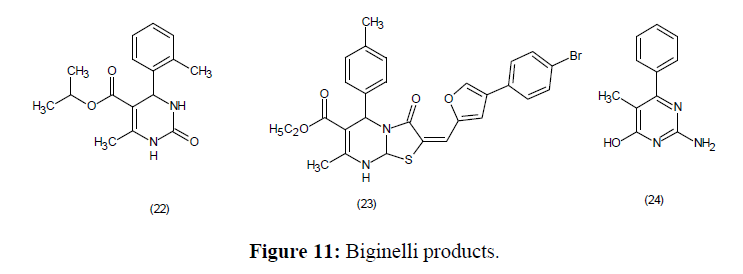
Antibacterial properties of ester, cyanide and some other substituted Biginelli products (22-24) have been identified (Figure 11).
Miscellaneous Activities [76-77]
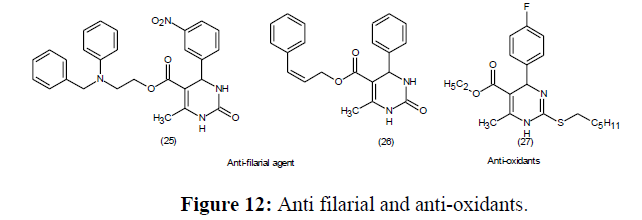
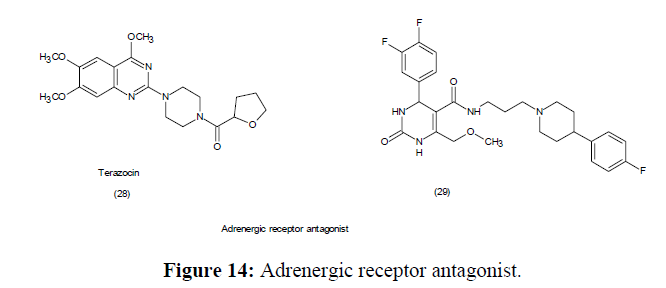
Since its discovery in 1893, the Biginelli dihydropyrimidine MCR reaction has progressed significantly. Mechanistic insights also led to logical modifications to the experiment protocols, allowing to form dihydropyrimidines in enantio selective complex natural products synthesis. The fascinating as well as diverse biological behaviour of dihydropyrimidines has been investigated by creating large libraries of compounds. The biological significance of new asymmetric approaches has been reported of the compounds belonging this class (Figure 12-14).
There are varied significant pharmacological activity for Biginelli adducts are reported like anti-cancer, anti-inflammatory, antibacterial, and antioxidant agents [78], Antimalarial [79], antitubercular [80], suppression of G protein-coupled receptor kinases (GRKs)/Rho-associated kinase isoform 1 (ROCK1) [81], heat shock protein 70 (Hsp70) [82] and A2B adenosine receptor antagonists.
Special applications of Biginelli reaction
Natural product synthesis
Natural products are essential in drug discovery and therapeutics which can only be found in trace quantities in marine/natural sources. Several alkaloids which contain the DHPM core are isolated from several marine sources in recent decades. Crambescidins, batzelladines, and monanchocidins are some of the alkaloids with a special fused tricyclic guanidinium core structure.
The crambescidin alkaloids have been shown to have cytotoxic properties against a variety of tumour cell lines [83], antifungal action in Candida albicans, antiviral action for herpes simplex virus (HSV), anti-HIV activity, and obstruction of HIV-1 envelope-mediated cell fusion have been reported [84].
The syntheses of alkaloids is done ‘tethered Biginelli condensation’ approach in which pyrrolidine derivative and an acetoacetate ester undergoes condensation in the presence of piperidinium acetate in dichloromethane to form cis- and trans-hydropyrrolopyrimidinone esters [85].
Materials chemistry
The Biginelli condensation reaction is widely used to produce heterocyclic systems for use in the improvement of functional materials such as sustainable polymers, dyes, adhesives, and other materials [86]. A few examples are briefly discussed in the following paragraphs.
Functional polymers
Because of the following factors, the Biginelli reaction is an extremely useful tool in polymer chemistry
(i) Increased efficiency and yield of the reaction confirms the quality of polymer.
(ii) When structure of three components of Biginelli reactants are varied, it results in variety of divercified products.
(iii) The tolerability of reaction towards the different reaction conditions and variety of functional groups to develop one-pot reactions.
(iv) Various kinds of reaction kinetics can be used to develop one-pot reactions
Recent research has established a list of Biginelli reaction applications in polymer chemistry, including polymer coupling, post-polymer modification, and the production of novel functional polymers [87].
Adhesives
In the presence of urea, the synthesis of poly DHPMs was announced through the Biginelli reaction mechanism of a bi-functional monomer which contains benzaldehyde as well as b-keto ester functionalities. These polycondensates have been discovered to be effective adhesives.
They tested the poly DHPMs' binding abilities with a variability of metal sheets, containing stainless steel, aluminium, and brass. Furthermore, the Biginelli polycondensates produced in situ act as glue to bond dissimilar metals simultaneously; where the Biginelli polycondensates appeared as very efficient, particularly on brass [88].
Fabric dyes
Ionic liquid employed as a solvent and N-methyl-2-pyrrolidonium hydrogen sulphate ([HNMP] + [HSO4]_ as a catalyst, a green methodology for the production of a novel family of DHPMs was reported [89]. The DHPM disperse dyes were used to dye hydrophobic fabrics like polyester and nylon using a high temperature–high pressure process (HTHP). Excellent fastness properties such as hostility to fading and running were discovered in the dyed textile fabrics. The colour strength of dyed polyester fabrics was also demonstrated to be excellent. The dihydropyrimidines derived from lawsone have dyeing qualities and high tinctorial strengths in this regard. These results are fascinating because they can permit colorists to run into ever-increasing eco-friendly constraints, create innovative effects across a variety of frameworks, and decrease processing expenses.
Biginelli reaction is one of important and significant reactions and products showed extensive variety of Pharmacological and other miscellaneous applications in variety of diverse area.
Conclusions
Since the discovery in 1893, the Biginelli dihydropyrimidine MCR reaction has advanced significantly. Mechanistic insights led the dihydropyrimidines to be produced in shorter reaction times and higher yields. Recyclability of the catalyst is the key feature of this reaction. Moreover, variety of substituents can be used. The biological activity of dihydropyrimidines has been studied. Various ecofriendly procedures for synthesis of DHPMs using aldehyde, ethyl acetoacetate and urea (3CR) by organocatalysts such as organic acids, amino acids, enzymes, polymers and macrocycles gives good yields. Biginelli reaction using ionic liquids, solid phase approach as well as using aqueous medium are the methods which are totally non-polluting, quantifying it as a green approach. As novel reactions are revealed and the biological significance of this class of molecules is investigated further, the Biginelli reaction will continue to advance.
Acknowledgement
The authors gratefully acknowledge Management and Principal of Mahatma Gandhi Vidyamandir’s Pharmacy College, Panchavati, Nashik for providing necessary facilities.
References
- Armstrong W, Andrew Combs P, Paul Tempest A et al., Acc Chem Res. 1996, 29(3): p. 123-131.
- Wender PA, Handy ST and Wright DL. Chem Ind. 1997, 765: p. 767-769.
- Gaich T and Baran PSJ. Org Chem. 2010, 75: p. 4657-4673.
- Zhu J and Bienayme H . 2005, p. 76.
- Ugi I and Domling A. Endeavour. 1994, 18: p. 115-122.
- Domling A and Ugi I. Angew Chem. 2000, 39: p. 3168.
- Dou GL, Shi CL and Shi DQ. J Comb Chem. 2008, 10: p. 810.
- Domling A and Ugi I. Angew Chem. 2000, p. 3168.
- Domling A. Chem Rev. 2006, 106(1): p. 17-89.
- Sanakausar R, Shaikh and Nazeruddin GM. J chem pharm res. 2014, 6(12): p. 505-534.
- Zhu J and Bienayme H. Hardcover, First edition, Wiley. 2005.
- https://www.analytik-jena.com/products/liquid-handling-automation/
- Biginelli P and Gazz. Chim Ital. 1893, 23: p. 360
- Sanakausar Shaikh Rand and Nazeruddin GM. J Chem Pharm. 2014, 6(12): p. 505-534.
- Dai lei Song, Wang Runxia, Chen Yongli et al., React. Kinet. 2008, 95(2): p. 385.
- Hu EH, Sidler DR and Dolling UH. J Org Chem. 1998, 63(10): p. 3454.
- Kappe CO, Tetrahedron. 1993, 49: p. 6937.
- Biginelli P. Gazz Chim Ital. 1893, 23: p. 360-413.
- CO Kappe. QSAR Comb Sci, 2003, 22(6): p. 630-645.
- Panda SS, Khanna P and Khanna L. Curr Org Chem. 2012, 16: p. 507-520.
- Heravi MM, Asadi S and Lashkariani BM. Mol Divers. 2013, 17: p. 389-407
- Kappe CO. QSAR Comb Sci. 2003, 22: p. 645.
- Trivedi AR, Bhuva VR, Dholariya BH et al., Bioorg Med Chem Lett. 2010, 20: p. 6100.
- Folkers K and Johnson TB. J Am Chem Soc. 1933, 55(9): p. 3784-3791.
- Sweet F and Fissekis JD. J Am Chem Soc. 1973, 95(26): p. 8741-8749.
- Folkers K, Harwood HJ and Johnson TB. J Am Chem Soc. 1932, 54(9): p. 3751-3758.
- Anjaneyulu B and Dharma GB. Int J Eng Res.2015, 3(6): p. 26-37.
- Kappe CO. J Org Chem. 1997, 62: p. 7201-7204.
- Folkers K and Johnson TB. J Am Chem Soc. 1933, 55: p. 3784-3791.
- Sweet F and Fissekis JD. J Am Chem Soc. 1973, 95(26): p. 8741-8749.
- Dalko PI and Moisan L. Angew Chem Int Ed. 2004, 43: p. 5138-5175.
- Amarante GW and Coelho F. Quim Nova. 2009, 32: p. 469-481.
- Suresh, Saini A, Kumar D et al., Green Chem Lett Rev. 2009, 2: p. 29-33
- Vasconcelos A, Oliveira PS, Ritter M et al., J Biochem Mol Toxicol. 2012, 26: p. 155-161.
- Kargar M, Hekmatshoar R, Mostashari A et al., Catal Commun. 2011, 15: p. 123-126.
- Karimi –Jaberi Z and Moaddeli MS. Org Chem. 2012, p. 1-4.
- Pandey J, Anand N and Tripathi RP. Tetrahedron. 2009, 65: p. 9350-9356.
- Sagar AD, Reddy SM, Pulle JS et al., ISRN Org Chem. 2011, 3: p. 649-654.
- Xu F, Huang D, Lin X et al., Org Biomol Chem. 2012, 10: p. 4467-4470
- Choghamarani GA and Zamani P. Chin Chem Lett. 2013, 9: p. 804-808.
- Karthikeyan P, Kumar SS, Arunrao AS et al., Res Chem Intermed. 2013, 39: p. 1335-1342.
- Sharma N, Sharma UK, Kumar R et al., RSC Adv. 2012, 2: p. 10648-10651.
- Abbasi E and Farhad H. Orient J Chem. 2013, 29: p. 731-733.
- Lal J, Gupta S and Agarwal DD. Catal Commun. 2012, 27: p. 38-43.
- da Silva DL, Fernandes SA, Sabino AA et al., Tetrahedron Lett. 2011, 52: p. 6328-6330
- Asghari S, Tajbakhsh M, Kenari JB et al., Chin Chem Lett. 2011, 22: p. 127-130.
- Zhou H, He M, Liu C et al., Lett. Org. Chem. 2006, 3: p. 225–227.
- Patil S, Jadhav SD and Mane SY. Int J Org Chem. 2011, 1: p. 125–131
- Sharma UK, Sharma N, Kumar R et al., Amino Acids. 2013, 44: p. 1031–1037.
- Suzuki I, Iwata Y and Takeda K. Tetrahedron Lett. 2008, 49: p. 3238–3241.
- Raju C, Uma R, Madhaiyan K et al., ISRN Org Chem. 2011, p. 1–5.
- Khaksar S, Vahdat SM and Moghaddamnejad RN. Monatsh Chem. 2012, 143: p. 1671–1674.
- Kadre T, Jetti SR, Bhatewara A et al., Arch Appl Sci Res. 2012, 4: p., 988–993.
- Khatri CK, Potadar SM and Chaturbhuj GU. Tetrahedron Letters. 2017, 58: p. 1778–1780.
- Khatri CK, Rekunge DS and Chaturbhuj GU. New J Chem. 2016, 40: p. 10412.
- Garima, Srivastava VP, Lal Dhar Yadav S. Tetrahedron Letters. 2010, 51(49): p. 6436-6438.
- Mathews CJ, Smith PJ and Welton T. Chem Commun. 2000, p. 1249.
- Papageorgiou N, Kalyanasundaram K and Gra¨tzel M. Inorg Chem. 1996, 35: p. 1168.
- Polshettiwar V and Varma RS. Tetrahedron Lett. 2007, 48: p. 7343.
- Brindaban CR. J Org Chem. 2000, 65: p. 6270.
- Ma Y, Wang L and Wang M. J Org Chem. 2000, 65: p. 3864.
- Lu J and Bai Y. 2002, p. 466-470.
- Reilly BCC and Atwal KS. Heterocycles. 1987, 26: p. 1185-1188.
- P. Wipf, A. Cunningham. Tetrahedron Letter, 1995, 36: p. 7819.
- Oliver Kappe C and Alexander Stadler. Acc Chem Res. 2000, p. 7.
- Kappe CO. J Eur Med Chem. 2000, 1043: p. 35.
- Zorkun S, Sarac S, Celebib S et al., Bioorg Med Chem. 2006, 14: p. 8582-8589.
- Chiang AN, Valderramos JC, Balachandran R et al., Bioorg Med Chem. 2009, 17: p. 1527-1533.
- Patil AD, Kumar NV, Kokke WC et al., J Org Chem. 1995, 60: p. 1182.
- Mayer TU, Kapoor TM, Haggarty SJ et al., Science. 1999, 286: p. 971–974.
- Agbaje OC, Fadeyi OO, Fadeyi SA et al., Bioorg Med Chem Lett. 2011, 21: p. 989-992.
- Chiang AN, Valderramos JC, Balachandran R et al., Bioorg Med Chem. 2009, 17: p. 1527-1531.
- Mokale SN, Shinde SS, Elgire RD et al., Bioorg Med Chem Lett., 2010, 20: p. 4424-4426.
- Jares-Erijman EA, Sakai R and Rinehart KL. J Org Chem. 1991, 56: p. 5712.
- Trivedi AR, Bhuva VR, Dholariya BH et al., Bioorg Med Chem Lett. 2010, 20: p. 6100-6102.
- Ismaili L, Nadaradjane A, Nicod L et al., Eur J Med Chem. 2008, 43: p. 1270-1275.
- Zhu X, Zhao G, Zhou X et al., Bioorg Med Chem Lett. 2010, 20: p. 299
- Fatima A, Terra BS, Neto LS et al., Elsevier. 2015, p. 317–337.
- Barrow JC, Nagarathnam D, Forray C et.al., J Med Chem. 2000, 43: p. 2703.
- Sehon CA, Hu E and Lee E. Med Chem. 2008, 51, 6631.
- Xin J, Chang L, Hou Z et al., Chem Eur J. 2008, 14: p. 3177.
- Wu YY, Chai Z, Liu XY et al., Eur J Org Chem. 2009, p. 904.
- Jares Erijman EA, Ingrum AL, Carney JR et al., J Org Chem. 1993, 58: p. 4805.
- Aron ZD and Overman LE. Chem Commun. 2004, p. 253.
- Zhao Y, Yu Y, Zhang Y et al., Polym Chem. 2015, 6: p. 4940.
- Nagarajaiah H, Mukhopadhyay A and Moorthy J. Tetrahedron Letters. 2016, 47: p. 5139-5147.
- Zhao Y, Wu H, Zhang Y et al., ACS Macro Lett. 2015, 4: p. 843.
- Dalko PI and Moisan L. Angew Chem Int Ed. 2004, 43: p. 5138–5175.
- Amarante GW and Coelho F. Quim Nova. 2009, 32: p. 469–481.