Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 6
Adsorption Behavior of Trypan Blue on Hydrotalcite
Sarra Hamouda1,2*, Salima Bouteraa1, Miloud Aissat1, Ismahene Mekki1, Abdellah Bahmani1 and Nourredine Bettahar1
1Laboratory of Inorganic Materials Chemistry and Application, University of Sciences and Technology of Oran - USTO BP. 1505 Oran, Algeria
2Center of Scientific Research and Technique in Physicochemical Analysis (CRAPC), Bou-Smail, Tipaza, Algeria
- *Corresponding Author:
- Sarra Hamouda
Laboratory of Inorganic Materials Chemistry and Application
University of Sciences and Technology of Oran - USTO BP. 1505 Oran, Algeria
Abstract
The current investigation reports an effective adsorption of anionic species on [Zn-Al-CO3] and [Mn-Al-CO3] hydrotalcite-like compounds, as novel, inexpensive available alternative to the commercial hydrotalcite. LDH [Zn-Al-CO3] and [Mn-Al-CO3] were synthesized by co-precipitation at constant pH with a molar ratio of 2:1. Their calcinations lead to the formation of mixed oxides exhibiting fine dispersion of metal cations and high surface area. The structural characterization of materials was carried out by X-ray diffraction, infrared spectroscopy (FTIR) and DTA/GT. The assay of the solutions was monitored by UV-Vis spectrometry. The removal of Trypan blue from aqueous solutions by adsorption onto hydrotalcite particles was investigated using batch rate experiments. The sorption process was also found to be influenced by contact time and solution pH. A comparative study of calcined and uncalcined LDH was performed. The adsorption kinetics study showed that the Pseudo-second-order kinetic model could be used to describe the adsorption of Trypan blue satisfactorily.
https://maviyolculuk.online/
https://mavitur.online/
https://marmaristeknekirala.com.tr
https://tekneturumarmaris.com.tr
https://bodrumteknekirala.com.tr
https://gocekteknekirala.com.tr
https://fethiyeteknekirala.com.tr
Keywords
LDH, Adsorption, Dye, Kinetic, Pollution
Introduction
The pollution of surface water and groundwater is one of the major concerns of the century due to industrial discharges [1,2]. It must be considered as the primary concern in the strategy of environmental preservation, and processing of water purification becomes then a very important environmental problem to solve. For this, we must provide means, technical as well as legal, sufficient to fight against this environmental disaster and allow the protection and improvement of the quality of all water resources with the main objective to achieve a good chemical and ecological status of aquatic environments.
Among the polluting activities, that may be mentioned tanneries, the textile and paper industries, which discharge effluents containing high concentrations of dyes [3]. Up to date, there are numerous methods such as chemical precipitation, electro coagulation, adsorption, reverse osmosis, and biological treatment have been developed for the removal of pollutant from wastewaters [4]. Among these potential separation methods mentioned above, adsorption is arguably regarded as a promising technique with the merits of simplicity, low cost, high efficiency, less production of sludge, and potential regeneration. Hydrotalcites (LDHs) are found increasing interest as adsorbents by virtue of their properties, which make them attractive materials for adsorbing anioniques species [5]. Their abundance in nature, low cost, high anion exchange capacity, good thermal stability, large surface area and regeneration ability make them suitable for wastewater purification [6].
Layered double hydroxides (LDHs) belong to the group of anionic clays, characterized by a stack of sheets positively charged of brucite-type 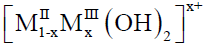 . The positive charge is compensated by the interlayer anions containing solvated species,
. The positive charge is compensated by the interlayer anions containing solvated species,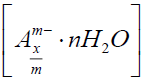 and a general formula of LDHs would be:
and a general formula of LDHs would be: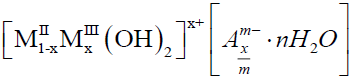 [7].
[7].
The aim of this work is to elucidate adsorption mechanisms of Trypan blue on [Zn–Al–CO3] and [Mn–Al–CO3] hydrotalcite and their calcined form, and assess a possibility of using this material for the removal of Trypan blue from aqueous solutions.
In this study, [Zn-Al-CO3] and [Mn-Al-CO3] were synthesized at a molar ratio of 2, these materials were then calcined at adequate temperatures. Interestingly, upon calcination it yields a solid solution of mixed metal oxides, which are used as potential environmentally-friendly catalysts for various organic transformations [8].
During Trypan blue adsorption experiments through these materials, various parameters such as the adsorption kinetics and the pH of the solution were examined.
Materials and Methods
Materials
Analytically pure ZnCl2⋅6H2O, MnCl2·4H2O, AlCl3·6H2O, NaOH, Na2CO3, HCl and Trypan blue (C34H28N6Na4O14S4) noted as TB were used without further purification and purchased from Biochem Chemopharma.
Sample preparation
[Zn-Al-CO3] and [Mn-Al-CO3] were obtained by co-precipitation method at a constant pH [9], starting from metal salt solutions with a mixture in suitable proportions, at a molar ratio 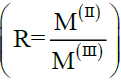 of 2 (where MII=ZnII or MnII, MIII= AlIII).
of 2 (where MII=ZnII or MnII, MIII= AlIII).
A mixed solution of 0.2 mol of ZnCI2⋅6H2O (or MnCl2) and 0.1 mol of AlCI3⋅6H2O in 100 ml of distilled water was added drop wise under vigorous stirring at room temperature to 100 ml of aqueous solution containing 0.2 mol of Na2CO3 and 0.1 mol of NaOH. During the co-precipitation process, the pH was maintained constant at 10 for [Zn-Al-CO3] and at 8 for [Mn-Al-CO3] by addition of 1 M NaOH solution. The precipitation was caused by the addition of the alkaline solution. The formed gel was transferred to a hermetic bottle and placed in an oven for its hydrothermal treatment. The synthesized product was then filtered and washed several times with distilled water, then dried and milled (denoted LDH).
Fractions of the obtained materials were calcined at 500°C for [Zn-Al-CO3] and 450°C for [Mn-Al-CO3] during 8 h (denoted CLDH). The obtained materials were characterized by powder X-ray diffraction (PXRD), FT-IR spectroscopy, and thermal (TGA/TDA) analysis.
Characterization
The diffractograms were carried out on powder with a diffractometer type X-ray Siemens D501 using CuKα radiation (λ=1.5418 Å). The FTIR spectra were collected on a Perkin Elmer spectrophotometer kind 16PC using KBr pellets. Thermogravimetric analyses (TGA/TDA) were performed with a TA Instruments SDT Q600 with a heating rate of 20°C/min from 50 to 800°C.
Adsorption studies
Batch adsorption experiments were conducted in order to evaluate the kinetic study and the effects of solution pH, by suspending 50 mg of each adsorbent in 20 ml of aqueous dye solutions with an initial concentration of 400 mg/l at room temperature and pH of solution. The kinetics study was investigated, using suspension of 250 mg of samples in 100 ml of dye solution, The suspensions were stirred at constant speed for time periods ranging from 5 to 180 min; a sample of 5 ml of dye solution was taken within a set time period.
The experiments were also done by varying the initial solution pH over a pH range of 4-10.
Each suspension was then centrifuged, and the supernatant was analyzed by UV/Visible spectrophotometer (Spectroscam 80D) at λmax= 607 nm. The adsorbed quantity was calculated by measuring the concentration of the solution before and after adsorption using the following equation:
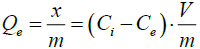 (1)
(1)
Where Qe (mg/g) is the adsorbed quantity at equilibrium (mg of dye/g of sorbent), Ci and Ce (mg/l) are the concentrations of BT dye at initial and equilibrium, respectively, m (g) is the mass of LDH and V (L) is the volume of the solution.
Results and Discussion
Characterization of samples
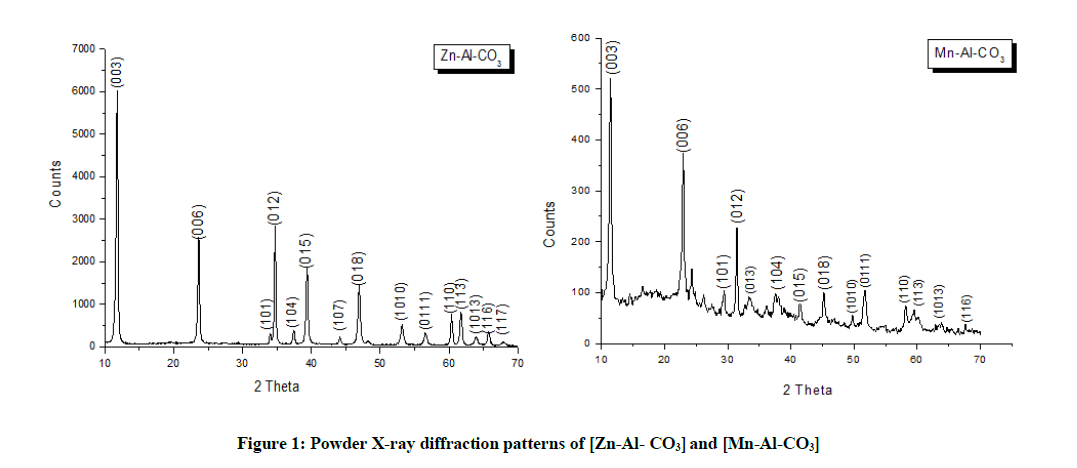
The X-ray diffraction patterns of the [Zn-Al-CO3] and [Mn-Al-CO3] are shown in Figure 1. All the diffraction peaks are in good agreement with layered structure [10-12]. They exhibit sharp and symmetric (003) and (006) reflections at low values of 2θ and less intense and asymmetrical reflections for (101), (012), (015), (018), (110) and (113) at high values of 2θ. The unit cell parameters for these samples were calculated, and the result is listed in Table 1 [13-17].
| Sample | d(003) (Å) | d(006) (Å) | d(110) (Å) | c(Å) | a(Å) |
|---|---|---|---|---|---|
| [Zn-Al-CO3] | 7.5 | 3.75 | 1.53 | 22.51 | 3.06 |
| [Mn-Al-CO3] | 7.74 | 3.87 | 1.58 | 23.25 | 3.16 |
Table 1: Lattice parameters of [Zn-Al-CO3] and [Mn-Al-CO3]
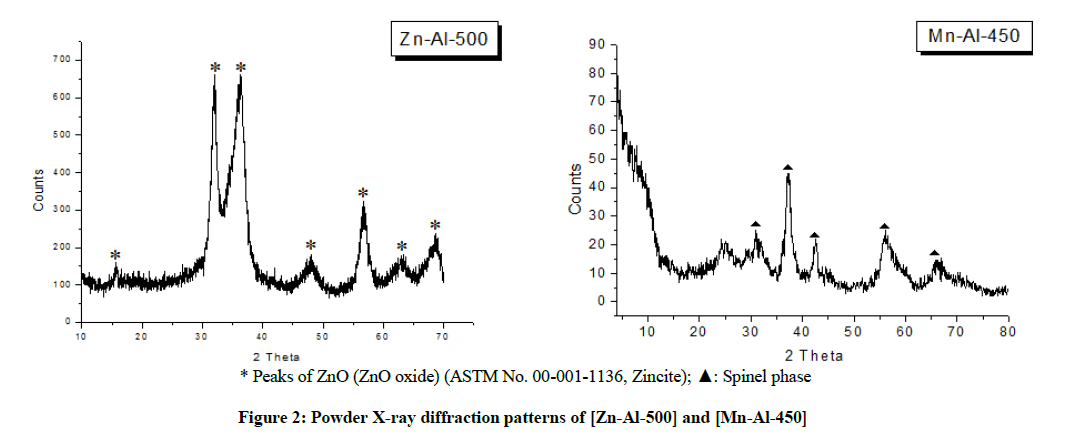
Powder X-ray diffraction pattern of the calcined samples showed remarkable changes after calcination. Figure 2 shows that the thermal treatment of [Zn-Al-CO3] at 500°C leads to a disappearance of the lamellar phase due to a dehydroxylation, releasing H2O and CO2 (dehydroxylation and decarbonatation). A comparison with the ASTM Standard: 00-001-1136 shows the appearance of an amorphous phase and zinc oxide [18,19]. Powder X-ray diffraction patterns of calcined sample of [Mn-Al-CO3] at 450°C (Figure 2) shows the transformation of the crystalline phase into an amorphous phase and the appearance of a new oxide identified as the spinel structure.
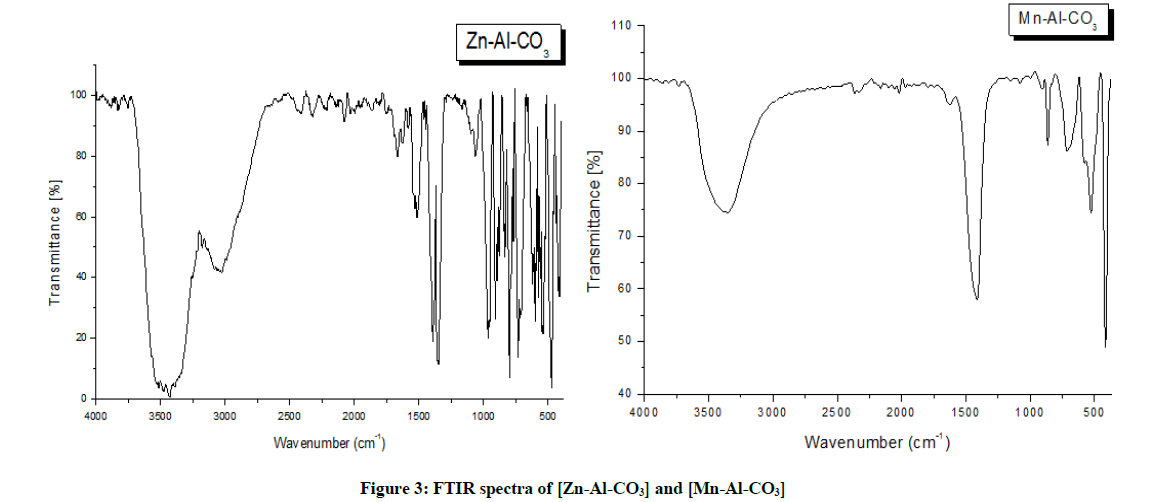
The FTIR spectra of the resultant hydrotalcite-type compounds are characteristic for this type of materials, as it is shown in Figure 3 [20,21]. The broad and strong absorption band in the range of 3600-3200 cm-1 is due to the O-H stretching vibration, both from the brucite like-layers and interlayer water molecules [13,16,17,22]. A weak shoulder around 3020 cm-1 is attributed to hydrogen bounding between water and carbonate anions in the interlayer galleries [13,21-23], in the case of [Zn-Al-CO3], while in the case of [Mn-Al-CO3] the band was not apparent in this position. Around 1600 cm-1 appears an absorption band associated with the deformation of the water molecules. The vibration band at 1500 cm-1 is assigned to the vibration mode of free carbonates. It can be observed for those samples, the appearance of another band around 1350 cm-1, attributed to the presence of carbonates. Vibration bands at low frequencies (below 1000 cm-1) are assigned to the metal-oxygen-metal bonds forming lamellar layers of each sample.
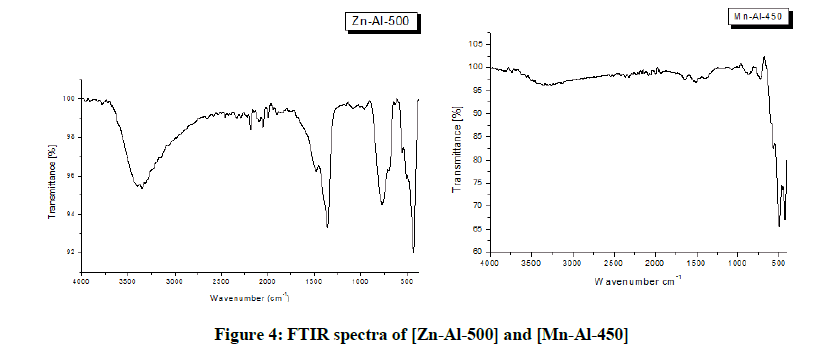
The infrared spectra of the calcined samples (Figure 4) show the relative reduction of the intensity of the characteristic absorption bands of water molecules in the range of 3600-3200 cm-1, and in the range from 700 cm-1 to 500 cm-1 an increase of those corresponding to the vibrations of the M-O bonds. Furthermore, it is observed that the band located at around 1370 cm-1 for both materials is still perceptible. However, the low intensity of this band, compared with that observed for the starting samples, suggests that carbonate ions were adsorbed to the grain surface of the calcined samples, the presence of that carbonate ions was due to contamination by atmospheric carbon dioxide [24,25].
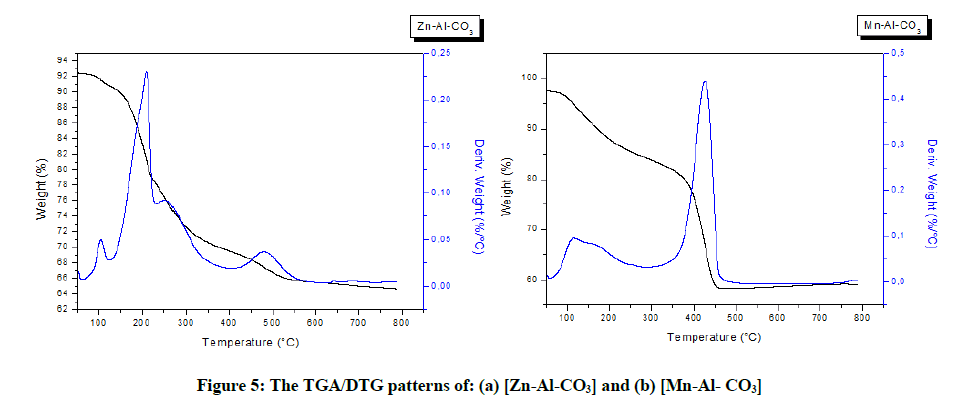
Thermoanalytical measurements were recorded to investigate the thermal decomposition behavior of [Zn-Al-CO3] and [Mn-Al-CO3] LDH during the thermal treatment (Figure 5). Each thermogram displays several weight losses well-differentiated as usually found in the literature [12]. For [Zn-Al-CO3], four weight loss steps are observed, with accompanying DTG peaks at 105°C, 210°C, 250°C and 480°C, the first weight loss that occurs in the range 50°C up to 125°C corresponds to the removal of physically adsorbed water. Heating samples at temperatures around 125-235°C leads to the loss of water molecules located in the interlamellar domain. A third weight loss was recorded in the range 235-405°C is associated with the decomposition of hydroxyl groups as water molecules, the fourth weight loss 405-600°C is associated with the decomposition of the interlayer carbonate anions as CO2, the total weight loss is 34.5%. For [Mn-Al-CO3], two main weight loss steps are obviously observed, with two accompanying DTG peaks at 115°C and 425°C in the first and the second stage, respectively. The first weight loss that occurs in the range 58-287°C should result from de removal of the adsorbed surface water and the interlayer water. The second one occurs in the range 287-500°C resulted from the dehydroxylation of [Mn-Al-CO3] layer and the decomposition of the interlayer CO3-2 anions. The total weight loss is 42%.
Effect of Contact Time on adsorption of Trypan blue
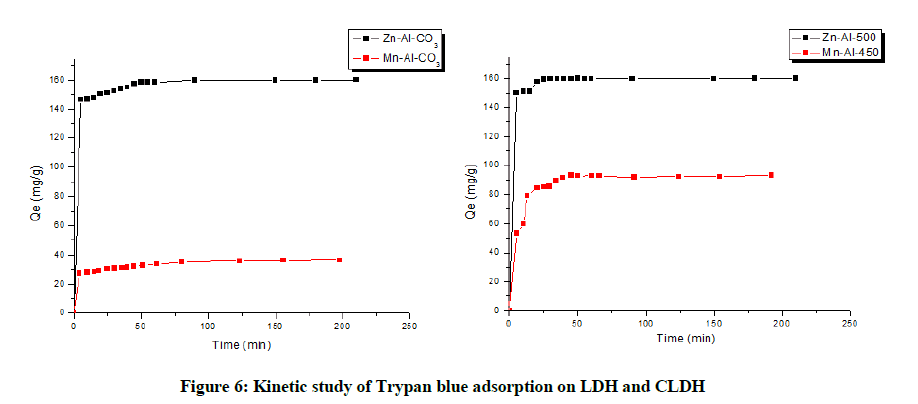
Plots of adsorption capacity versus contact time are shown in Fig.6, two types of phenomena were observed: Rapid adsorption followed by a slow adsorption: phenomenon probably due to the resistance to mass transfer within the grains of the clay particles. We explained that after the first step of adsorption (Rapid adsorption) Qe equilibrium reached and % Removal also became constant, so the optimal contact time was 60, 80, 25 and 40 minutes for Zn-Al-CO3, Mn-Al-CO3, Zn-Al-500 and Mn-Al-450 respectively. This optimal time period was the sufficient time for stabilization of this adsorbent and adsorbate dispersed solution (Table 2 and Figure 6).
| Contact time (min) | Qe (mg/g) | Removal (%) | |
|---|---|---|---|
| [Zn-Al-CO3] | 60 | 158.41 | 99 |
| [Mn-Al-CO3] | 80 | 35.08 | 26.2 |
| [Zn-Al-500] | 25 | 159.46 | 99.66 |
| [Mn-Al-450] | 40 | 91.49 | 25.34 |
Table 2: Optimum dye fixing time on LDH and CLDH
The kinetic models (Order of reactions)
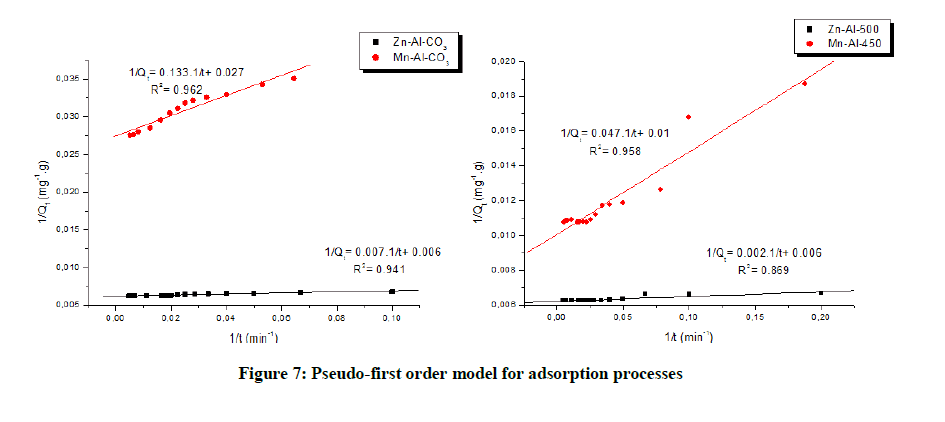
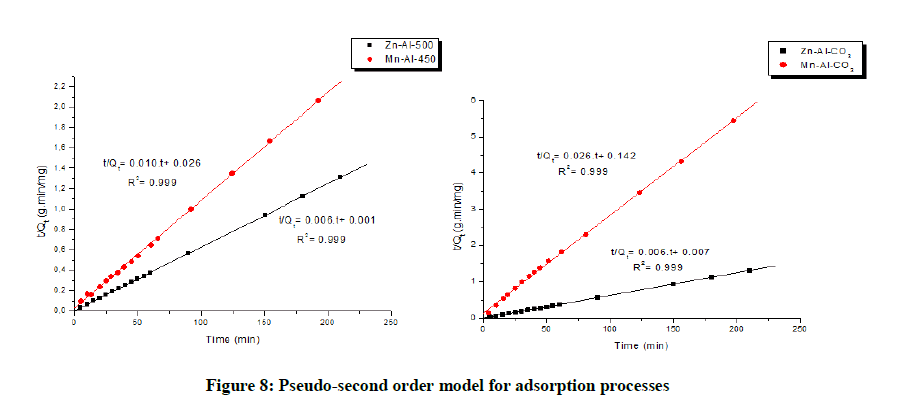
The pseudo first order and pseudo second order models were used to calculate the kinetic parameters (Table 3) [26,27]. The results showed that the correlation coefficient values of pseudo-second-order kinetic model were close to the unit and were higher than those obtained upon application of the pseudo-first order kinetic model. Suggesting that adsorption most probably followed a pseudo second order rate expression (Figures 7 and 8) [28].
| Pseudo-first ordermodel | Pseudo-second ordermodel | |||||
|---|---|---|---|---|---|---|
| Sample | K1 (min-1) | Qm (mg/g) | R2 | K2 (g/mg.mn) | Qm (mg/g) | R2 |
| Zn-Al-CO3 | 1.133 | 160.51 | 0.941 | 0.00531 | 161.03 | 0.999 |
| Mn-Al-CO3 | 4.871 | 36.4 | 0.962 | 0.00506 | 37.16 | 0.999 |
| Zn-Al-500 | 0.354 | 161.03 | 0.985 | 0.02217 | 160.51 | 0.999 |
| Mn-Al-450 | 4.401 | 98.81 | 0.989 | 0.00427 | 94.34 | 0.999 |
Table 3: Kinetic parameters of Trypan blue adsorption on the LDH and CLDH
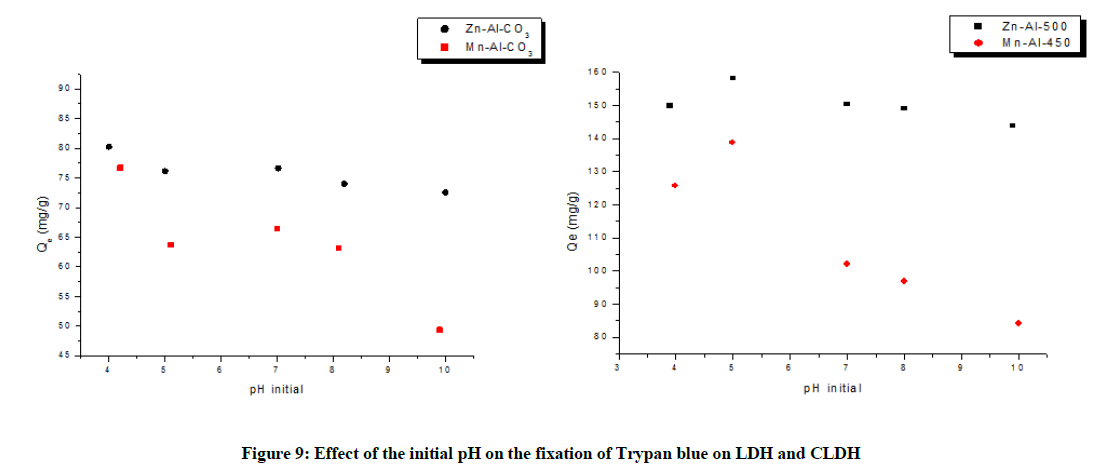
Influence of the pH
The adsorption capacity was slightly influenced by the pH (Figure 9). For [Zn-Al-CO3] and [Zn-Al-500], the study was performed at 4 ≤ pH ≤ 10 and illustrates the high stability of these samples in terms of solubility, showing the advantage of being used in the treatment of water in a wide range of pH [29].
However, for [Mn-Al-CO3] and [Mn-Al-450], the change of the adsorption capacity was significant. At high pH, the decrease of the adsorption may be due mainly to a competition between the anion of the dye and the carbonate ions (Table 4).
| Sample | pH opt | Qe (mg/g) | Removal (%) |
|---|---|---|---|
| [Zn-Al-CO3] | 7 | 80.27 | 50.17 |
| [Mn-Al-CO3] | 7 | 76.68 | 47.93 |
| [Zn-Al-500] | 5 | 158.15 | 98.84 |
| [Mn-Al-450] | 5 | 138.75 | 86.72 |
Table 4: Influence of pH on the adsorption capacity
Conclusion
It was confirmed that [Zn-Al-CO3] and [Mn-Al-CO3] hydrotalcites were successfully prepared using co-precipitation method. The results obtained show that the synthesized LDHs can be used as efficient adsorbents for the removal of the Trypan blue dye from aqueous solutions, and could have a potential to be utilized for wastewater treatment in terms of removal of hazardous material toxicant for most of aqueous organisms. The sorption process was also found to be influenced by contact time and solution pH. The adsorption behavior follows a pseudo-second-order reaction model.
Acknowledgement
This research was supported by Laboratory of Inorganic Materials Chemistry and Application, University of Sciences and Technology of Oran. We are also grateful for the characterization from the Center for Technical and Scientific Research in Physico-Chemical Analysis (CRAPC), Bou-Smail, Tipaza.
References
- H. Zaghouane-Boudiaf, M. Boutahala, L. Arab, Chem. Eng. Journal, 2012, 187, 142-149.
- A. Al-Kdasi, A. Idris, K. Saed, C.T. Guan, a review, Global Nest: the Int. J., 2004, 6, 222-230.
- Q. Wang, Z. Luan, N. Wei, J. Li, C. Liu, J. Hazard. Mater., 2009, 170, 690-698.
- D.J. Joo, W.S. Shin, J.H. Choi, S.J. Choi, M.C. Kim, M.H. Han, T.W. Ha, Y.H. Kim, Dyes Pigments, 2007, 73, 1, 59-64.
- W. Kagunya, M. Chibwe, W. Jones, Mol. Cryst. Liq. Cryst., 1994, 244, 155-160.
- Z. Q. Zhang, H. Y. Zeng, X. J. Liu, S. Xu, C. R. Chen, J. Z. Du, J. Taiwan Inst. Chem. Eng., 2016, 60, 361-368.
- H. Morioka, H. Tagaya, M. Karasu, J.I. Kadokawa, J. Mater. Res., 1998, 13, 4, 848-851.
- T. Baskaran, J. Christopher, V. Ganesh, A. Sakthivel, Catal. Commun., 2016, 74, 85-90.
- S. Velu, A. Ramani, B. M. Chanda, S. Sivasanker, J. Chem. Soc. Chem. Commun, 1997, 2107.
- L. Pesic, S. Salipurovic, V. Markovic, D. Vuselic, W. Kagunya, W. Jones, J. Mater. Chem., 1992, 2, 1069-1073.
- A. Bakhti, M. S. Ouali, Rev. sci de l'eau / J. Water Sci., 2007, 20, 241-250.
- L. Du, B. Qu, Z. Xu, Polym. Degrad. Stab., 2006, 91, 5, 995-1001.
- X. Wang, P. Wu, Z. Huang, N. Zhu, J. Wu, P. Li, Z. Dang, Appl. Clay Sci., 2014, 95, 95-103.
- E. M. Seftel, E. Popovici, M. Mertens, K. DeWitte, G. VanTendeloo, P. Cool, E.F. Vansant, Micropor. Mesopor. Mater., 2008, 113(1-3), 296-304.
- F. Barahuie, M. Z. Hussein, P. Arulselvan, S. Fakurazi, Z. Zainal, J. Solid State Chem., 2014, 217, 31-41.
- X. Wang, P. Wu, Y. Lu, Z. Huang, N. Zhu, C. Lin, Z. Dang, Sep. Purif. Technol., 2014, 132, 195-205.
- F. Z. Mahjoubi, A. Khalidi, M. Abdennouri, N. Barka, J. Taibah Univ. Sci., 2017, 11, 1, 90-100.
- S. Miyata, T. Kumura, Chem. Lett., 1973, 843-848.
- L. Hickey, J. T. Kloprogge, R. L. Frost, J. Mater. Sci., 2000, 35, 4347- 4355.
- J. M. Hernandez-Moreno, M. A. Ulibarri, J. L. Rendon, C. J. Serna, Phys. Chem. Miner., 1985, 12, 34.
- L. Cocheci, P. Barvinschi, R. Pode, E. Popovici, E. M. Seftel, Chem. Bull. "POLITEHNICA" Univ., 2010, 55(69), 1, 40-45.
- O. Saber, H. Tagaya, Mater. Chem. Phys., 2008, 108(2-3), 449-455.
- J. M. Fernandez, M. A. Ulibarri, F. M. Labajos, V. Rives, Mat. Chem., 1998, 8, 2507-2514.
- L. Lv, Y. Wang, M. Wei, J. Cheng, J. Hazard. Mater., 2008, 152, 1130-1137.
- F. Cavani, F. Trifiro, A. Vaccari, Catal. Today, 1991, 11, 173-301.
- A. Kumar, Master of Technology, National Institute of Technology, Rourkela, India, 2013.
- Z. Bouberka, A. Khenifi, H. Ait Mahamed, B. Haddou, N. Belkaid, N. Bettahar, Z. Derriche, J. Hazard. Mater., 2009, 162, 378–385.
- F.C. Wu, R.L. Tseng, R.S. Juang, Environ. Technol., 2001, 22, 721.
- M.M. Bouhent, Z. Derriche, R. Denoyel, V. Prevot, C. Forano, J. Solid State Chem., 2011, 184, 1016-1024.