Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 8
An Efficient Base Catalyzed Three Component One Pot Synthesis of Novel 2-Chloro/Marcapto-Quinoline Derivatives: A Potent Antimicrobial Agent
Devappa S Lamani1*, Venugopala Reddy KR1 and Bhojya Naik HS2
1Department of Chemistry Basaveshwar Science College, Bagalkot Rani Channama University, Karnataka, India
2Department of PG Studies and Research in Industrial Chemistry, School of Chemical Sciences, Kuvempu University, Shankaraghatta- 577451, Karnataka, India
- *Corresponding Author:
- Devappa S Lamani
Department of Chemistry Basaveshwar Science College
Bagalkot Rani Channama University
Karnataka, India
Abstract
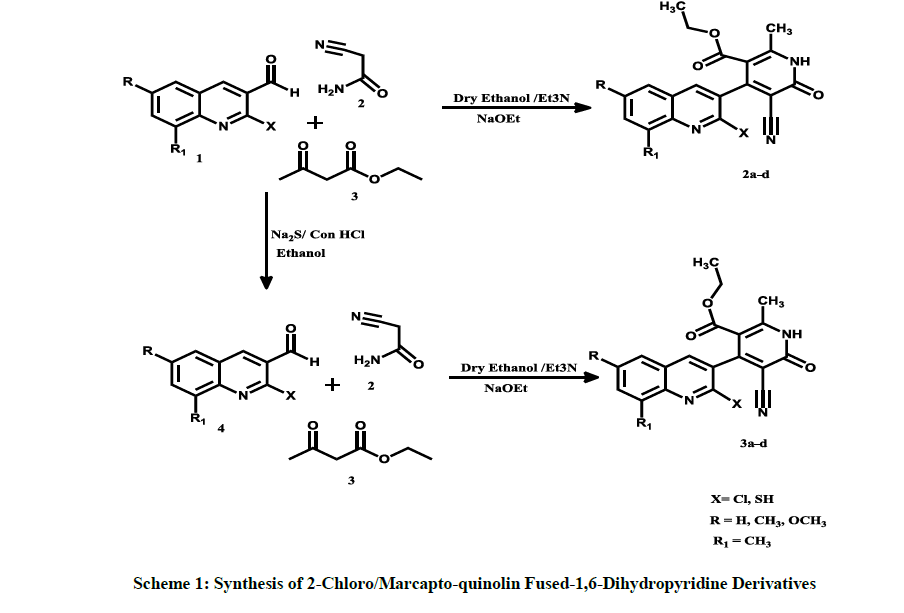
The present paper describes the simple and efficient protocol for the synthesis of 2-Chloro/Marcapto-quinolin fused-1,6-Dihydropyridine Derivatives (2a-d/3a-d) by (Et3N) and (NaOEt) catalyzed three-component one pot reaction of amine, aldehyde and ketones. The newly synthesized compounds were systematically characterized by Fourier-Transform Infrared Spectroscopy (FT-IR), Proton Nuclear Magnetic Resonance (1H-NMR), elemental analysis, and mass spectral analysis. The antibacterial potency of these compounds was tested against Gram-positive and Gram-negative bacteria. The sulfur containing quinoline compound 3a showed significant inhibitory effect on Staphylococcus aureus, Escherichia coli and Streptococcus pyogenes in consistent to halogen containing quinoline compound 2a.
Keywords
Chloro/Marcaptoquinoline, (Et3N), Michael reaction, (NaOEt), Antibacterial, One pot synthesis.
Introduction
It is well known that quinolines exhibit a wide range of biological activities [1] and are valuable reagents for the synthesis of nano- and mesostructures with enhanced electronic and photonic properties [2-4]. Due to wide range of applicability in medicinal, bioorganic, industrial as well as in the fields of synthetic organic chemistry, there has been an increasing interest in the development of efficient methodologies for the synthesis of quinolines. Among them, the Vilsmeier-Haack reagent has been proved to be a versatile reagent capable of executing a large variety of synthetic transformations. It finds application in formylation, cyclohaloaddition, and cycligation. The intramolecular cyclization potential of halomethyleniminium salts formed under the Vilsmeier condition by utility of quinoline as starting materials is used for the synthesis of different reported derivatives [5-11]. A promising strategy of the modern organic synthesis is performing a sequence of reactions without isolating intermediates (one-pot process). The importance of the Michael reaction for synthetic organic chemistry is well known [12,13]. It is successfully used in the synthesis of functionally substituted pyridine-2 chalcogenones [14], a number of which exhibit biological activity [15]. However, only few examples have been reported on the synthesis of heterocyclic compounds by the Michael reaction involving exchange of the methylene components [16].
The present work elaborates the scope of the Michael reaction with exchange of the methylene components to the synthesis of carbo and heterocycles. Here we report that aldehyde1, which was successfully used previously for preparing compounds with selective Juvenile activity [17], enters into condensation with cyanoacetamide 2 and ethyl acetoacetate 3 in the presence of easily accessible base catalyst. The reaction occurs at 80ºC ± 5ºC in ethanol and yields 2-Chloro/Marcapto- quinolin fused-1, 6-Dihydropyridine derivatives as a final product. The reaction apparently occurs via Michael reaction [18].
In continuation of our research work [18-23] towards the synthesis of potentially bioactive molecules by simple and practical approach. Herein, we report a rapid and efficient method for the synthesis of novel heterocyclic 2-Chloro/Marcapto-quinolin fused-1,6-dihydropyridine derivatives (Scheme 1) 2a-d/3a-d via one-pot three-component reaction with quantitative yield. The antibacterial potential of our synthesized compounds studied against Gram positive and Gram negative bacteria with six bacterial strains.
Chemistry
The chemistry of 2-cyanoacetamide, aldehyde and ethyl acetoacetate in presence of Et3N and NaOEt catalyst proceeded uneventfully and the product was isolated in quantitative yield after work up. It was observed that equimolar ratio of cyanoacetamide, aldehyde and, ethyl acetoacetate, respectively gave almost the quantitative yields. In a typical experiment, cyanoacetamide, Et3N and aldehyde were stirred in dry ethanol for 15-20 min, at room temperature followed by addition of diketones and NaOEt. The reaction mixture was stirred for an additional 3-4 h. The obtained precipitate was removed by filtration and the usual work-up gave the desired products in almost quantitative yields. We observed that addition of Et3N and NaOEt conditions gives better yield compared with other base such as pyridine, piperidine [17].
Our mechanistic investigations using spectral studies gave strong evidence for achievement of desired products. IR spectra of compounds (2a), and (3a) showed the carbonyl stretching frequency, at 1700-1710 cm-1, and bands observed in the region of 2210-2214 cm-1 and 3256-3252 cm-1 corresponding to (CN) and (-NH-) groups respectively [19,20]. The 1H-NMR spectra (3a) shows singlet appeared at δ: 10.01-10.05 and 12.04-12.06 ppm corresponding to (-SH-) and (-NH-) peaks. Further, the assigned structure was confirmed by its mass spectra with a molecular ion peak at m/z= 368 [M+H] for (2a) and 366 [M+H] for (3a) [23]. Hence, we synthesized eight more title compounds, which exhibited similar spectral data summarized in experimental section.
Antibacterial activity
The agar well diffusion method was used in order to determine the inhibition zones of synthesized compounds 2a and 3a. The compound 3a was found to be most active against almost all the microorganisms (Table 1). The MIC of compound 2a and 3a is summarized in (Table 2). The results obtained in this study indicate a considerable difference in antibacterial activity among the two newly synthesized compounds. The compound 3a sulfur containing quinolone exhibited significant antibacterial activity against pathogenic bacterial strains. Its bio-controlling potency was compared with the standard antibiotic ciprofloxacin. Generally, the Gram-positive bacteria is more susceptible having only an outer peptidoglycan layer which is not an effective permeability barrier [24] whereas the Gram negative bacteria possess an outer phospholipidic membrane carrying the structural lipopolysaccharide components. So the maximum inhibitory activity was observed in Gram-positive bacteria Staphylococcus aureus and Streptococcus pyogenes, Escherichia coli. In the case of Gram negative Pseudomonas aeruginosa and Klebsiella pneumoniae the zone of inhibitory activity was significant because of a multilayered phospholipidic membrane carrying the structural lipopolysaccharide components [25]. The sulfur containing quinoline compound 3a showed maximum inhibition zones as compared to that of halogen containing compound 2a. Thus, compound 3a is effective in controlling the growth of pathogenic strains to a considerable extent. The high activity of compound 3a as compared to that of compound 2a, suggested that it was solely responsible for antibacterial activity.
| Test Organisms | Inhibition Test organisms zone in diameter (mm) | ||
|---|---|---|---|
| Compound 2a | Compound 3a | Ciprofloxacin | |
| Pseudomonas Aeruginosa | 20.05 ± 1.27 | 19.50 ± 0.55 | 24.26 ± 1.10 |
| Klebsiella Pneumonia | 18.09 ± 0.90 | 16.33 ± 1.16 | 20.01 ± 1.25 |
| Escherichia coli | 29.40 ± 1.15 | 25.50 ± 1.13 | 30.55 ± 1.10 |
| Staphylococcus aureus | 28.13 ± 1.71 | 24.47 ± 1.16 | 29.00 ± 1.81 |
| Streptococcus pyogenes | 27.08 ± 1.13 | 21.13 ± 1.71 | 30.17 ± 1.48 |
| Bacillus subtilis | 22.50 ± 1.13 | 20.47 ± 1.16 | 26.40 ± 1.10 |
Values are mean ± S.E.M. of triplicates experiments. S: Streptomycin (10 g/disc). The values of negative control were subtracted from the values of samples and the corrected values are given.
Table 1: Antibacterial activity of the synthesized compounds (1 mg/well) against pathogenic bacterial strains
| Test organisms | Compound 2a | Compound 3a | Ciprofloxacin |
|---|---|---|---|
| Pseudomonas aeruginosa | 24 | 18.5 | 6.25 |
| Klebsiella pneumoniae | 32.5 | 22.5 | 12.5 |
| Escherichia coli | 13.5 | 11.5 | 6.5 |
| Staphylococcus aureus | 12.5 | 8.5 | 6.75 |
| Streptococcus pyogenes | 16 | 14.5 | 6.25 |
| Bacillus subtilis | 22.5 | 16.5 | 12.5 |
Table 2: The MIC values (10 g/disc) of synthesized compounds against the Microorganisms
Materials and Methods
All organic solvents used for the synthesis were of analytical grade. The TLC was performed on Baker-Flex silica gel 1B-F (1.55) plates using ethyl acetate and petroleum ether (1: 8). Melting points were determined on a Mel-Temp apparatus and were uncorrected. IR spectra were recorded in the matrix of KBr with Perkin-Elmer 1430 spectrometer. 1H-NMR spectra was recorded on Jeol spectrometer (400 MHz), and chemical shifts (δ) given in ppm relative to the TMS in CDCl3 solvent. Mass spectra were obtained on a JEOL-SX-102 instrument using fast atom bombardment (FAB positive). C, H and N analysis were performed at Cochin University, Sophisticated Test and Instrumentation Center, Kochi, Kerala, India. Column chromatography separations were obtained on silica gel (60-120 mesh).
Determination of antimicrobial activity
Microorganisms used
Test microorganism belongs to Gram-positive S. aureus-ATCC-29737, B. subtilis-NCIM- 2010 and S. pyogenes-NCIM-2608 and Gram-negative bacteria such as P. aeruginosa-ATCC-20852, K. pneumonia-MTCC-618 and Escherichia coli were obtained from National Chemical Laboratory (NCL), Pune, India.
Antimicrobial assay
The agar well diffusion method [26] was used for the assessment of antibacterial activity of the test samples. The media used was nutrient agar (Tryptone 10 g/l, yeast extract 5 g/l, sodium chloride 10 g/l, agar–agar 15 g/l, pH 7.2). 6 mm wells were created using a sterilized cork borer under aseptic conditions on the inoculated agar (108 Cfu/ml of bacteria) and 100 Ml (10 mg/ml) of test compounds were loaded to each wells. Antibacterial agent ciprofloxacin (10 mg/100 ml) was served as positive reference standard. Plates were then incubated at 37oC for 24 h. At the end of the incubation period, inhibition zones formed on the medium were evaluated in millimeters. The Minimal Inhibitory Concentrations (MICs) of the newly synthesized compounds 2a and 3a were determined by microdilution techniques in LB broth, according to Clinical and Laboratory Standards Institute (CLSI), USA guidelines.
General synthesis
Synthesis of ethyl 4-(2-chloroquinolin-3-yl)-5-cyano-2-methyl-6-oxo-1, 6-dihydro pyridine-3-carboxylate
The appropriate 2-cyanoacetamide (1.55 g 0.018 mol) and catalytic amount of Et3N was stirred in 50 ml ethanol under room temperature for 5 min, followed by addition of aldehyde (3.53 g 0.019 mol) for 30 min. After add catalytic amount of NaOEt than (2.31 ml 0.019 mol) ethyl acetoacetate continue the stirring with reflux for 80oC -85oC for additional 3-4 h. The product formed was collected by filtration the crude product was recrystallized from ethyl acetate and further purified by column chromatography on silica gel using ethyl acetate-pet ether as eluent (2: 8). Similarly, same procedure used for all the quinoline derivatives. This compound was obtained as yellowish solid in 83% yield; IR cm-1 (KBr) 3392 (NH), 2923 (Ar-CH), 2213 (CN), 1701 (C=0), 1379 (C=C), 1050, 666, 470; 1H-NMR (400 MHz, DMSO-d6) 1.10 (t, 3H, CH2-CH3), 2.42 (s, 3H, CH3), 3.89 (q, 2H, O-CH2), 10.02 (s, 1H, NH), 7.05-7.91 (4H, 5H, 6H, 7H, 8H, Ar-CH, Quinoline). FAB mass m/z=368(M+H). Anal. (%) for C19H14ClN3O2 (367.7), Calcd. C, 2.05; H, 3.84; N, 11.43; Found: C, 62.14; H, 3.74; N, 11.38.
Synthesis of ethyl 4-(2-chloro-6-methylquinolin-3-yl)-5-cyano-2-methyl-6-oxo-1,6-dihydropyridine-3-carboxylate
Brown solid in 83% yield; IR cm-1 (KBr) 3390 (NH), 2925 (Ar-CH), 2210 (CN), 1708 (C=0), 1374 (C=C), 1054, 664, 473; 1H-NMR (400 MHz, CDCl3) 1.09 (t, 3H, -CH2-CH3), 2.36 (s, 3H, CH3), 3.89 (q, 2H, O-CH2), 2.68 (s, 3H, CH3), 10.02 (s, 1H, NH), 7.05-7.91 (4H, 5H, 7H, 8H, Ar-CH, Quinoline), FAB mass m/z=382(M+H). Anal. (%) for C20H16ClN3O2 (381.81), Calcd. C, 62.91; H, 4.22; N, 11.01; Found: C, 62.85; H, 4.32; N, 11.12.
Synthesis of ethyl 4-(2-chloro-6-methoxyquinolin-3-yl)-5-cyano-2-methyl-6-oxo-1,6-dihydropyridine-3-carboxylate
Yellowish solid in 80% yield; IR cm-1 (KBr) 3390 (NH), 2925 (Ar-CH), 2210 (CN), 1706 (C=0), 1376 (C=C), 1054, 664, 473; 1H-NMR (400 MHz, DMSO) 1.09 (t, 3H, CH2-CH3), 2.35 (s, 3H, CH3), 3.93 (q, 2H, O-CH2), 2.73 (s, 3H, OCH3), 10.02 (s, 1H, NH), 7.05-7.92 (4H, 5H, 7H, 8H, Ar-CH, Quinoline), FAB mass m/z=398(M+H). Anal. (%) for C20H16ClN3O3 (397.8), Calcd. C, 60.38; H, 4.05; N, 10.56; Found: C, 60.26; H, 4.12; N, 10.58.
Synthesis of ethyl 4-(2-chloro-8-methylquinolin-3-yl)-5-cyano-2-methyl-6-oxo-1,6-dihydropyridine-3-carboxylate
Yellowish brown solid in 83% yield; IR cm-1 (KBr) 3392 (NH), 2923 (Ar-CH), 2210 (CN), 1708 (C=0), 1373 (C=C), 1054, 665, 473; 1H-NMR (400 MHz, DMSO) 1.09 (t, 3H, CH2-CH3), 2.36 (s, 3H, CH3), 3.89 (q, 2H, OCH2), 2.66 (s, 3H, CH3), 10.02 (s, 1H, NH), 7.04- 7.91 (4H, 5H, 6H, 7H, Ar-CH, Quinoline), FAB mass m/z=381(M+H). Anal. (%) for C20H16 ClN3 O2 (381.8), Calcd. C, 62.91; H, 4.22; N, 11.01; Found: C, 62.88; H, 4.33; N, 11.14.
Synthesis of ethyl 5-cyano-2-methyl-6-oxo-4-(2-sulfanylquinolin-3-yl)-1,6-dihydropyridine-3-carboxylate
Radish solid in 80% yield; IR cm-1 (KBr) 3392 (NH), 2922 (Ar-CH), 2214 (CN), 2566 (-SH), 1706 (C=0), 1381 (C=C), 1052, 668, 473; 1H-NMR (400 MHz, DMSO) 1.09 (t, 3H, CH2-CH3), 2.35 (s, 3H, CH3), 10.02 (s, 1H, NH), 12.02 (s, 1H, 1SH), 3.95 (q, 2H, OCH2), 7.05-7.93 (4H, 5H, 6H, 7H, 8H, Ar-CH, Quinoline), FAB mass m/z=366(M+H). Anal. (%) for C19H16N3O3S (365.4), Calcd. C, 62.45; H, 4.14; N, 11.50; S, 8.78. Found: C, 62.54; H, 4.21; N, 11.43; S, 8.69.
Synthesis of ethyl 5-cyano-2-methyl-4-(6-methyl-2-sulfanylquinolin-3-yl)-6-oxo-1,6-dihydropyridine-3-carboxylate
Yellowish solid in 83% yield; IR cm-1 (KBr) 3393 (NH), 2922 (Ar-CH), 2213 (CN), 2565 (-SH), 1706 (C=0), 1378 (C=C), 1052, 665, 473; 1H-NMR (400 MHz, DMSO) 1.12 (t, 3H, CH2-CH3), 2.35 (s, 3H, CH3), 10.01 (s, 1H, NH), 12.03, (s, 1H, SH), 3.93 (q, 2H, OCH2), 2.68 (s, 3H, CH3) 7.05-7.92 (4H, 5H, 7H, 8H, Ar-CH, Quinoline), FAB mass m/z=379(M+). Anal. (%) for C20H17N3O3S (379.4), Calcd. C, 63.31; H, 4.52; N, 11.07; S, 8.45. Found: C, 63.31; H, 4.52; N, 11.07; S, 8.54.
Synthesis of ethyl 5-cyano-4-(6-methoxy-2-sulfanylquinolin-3-yl)-2-methyl-6-oxo-1,6-dihydropyridine-3-carboxylate
Yellowish solid in 80% yield; IR cm-1 (KBr) 3393 (NH), 2922 (Ar-CH), 2213 (CN), 2565 (-SH), 1706 (C=0), 1378 (C=C), 1052, 665, 473; 1H-NMR (400 MHz, DMSO) 1.09 (t, 3H, CH2-CH3), 2.36 (s, 3H, CH3), 10.03 (s, 1H, NH), 12.05(s, 1H, SH), 3.96 (q, 2H, OCH2), 2.75 (s, 3H, OCH3) 7.05-7.93 (4H, 5H, 7H, 8H, Ar-CH, Quinoline), FAB mass m/z=396(M+H). Anal. for C20H17N3O4S (395.4), Calcd. C, 60.75; H, 4.33; N, 10.63; S, 8.11. Found: C, 60.82; H, 4.43; N, 10.53; S, 8.02.
Synthesis of ethyl 5-cyano-2-methyl-4-(8-methyl-2-sulfanylquinolin-3-yl)-6-oxo-1,6-dihydropyridine-3-carboxylate
Yellowish solid in 82% yield; IR cm-1 (KBr) 3393 (NH), 2923 (Ar-CH), 2213 (CN), 2565 (-SH), 1707 (C=0), 1378 (C=C), 1051, 665, 473; 1H-NMR (400 MHz, DMSO) 1.09 (t, 3H, CH2-CH3), 2.37 (s, 3H, CH3), 10.01 (s, 1H, NH), 12.03(s, 1H, SH), 3.93 (q, 2H, OCH2), 2.68 (s, 3H, CH3) 7.05-7.92 (4H, 5H, 6H, 7H, Ar-CH, Quinoline), FAB mass m/z=380(M+H). Anal. (%) for C20H17N3O2S (379.4), Calcd. C, 63.31; H, 4.52, N, 11.07; S, 8.45. Found: C, 63.31; H, 4.52; N, 11.07; S, 8.54.
Conclusion
We developed a versatile and useful access to different scaffold of biologically important 2-Chloro/Marcapto-quinolin fused-1,6-Dihydropyridine derivatives 2a-d/3a-d using base catalyzed one-pot three-component reaction. The efficiency of the employed methodology can be explained by the fact that used catalysts were easily accessible, easy work up gives high yield and fast reaction conversion, so that the reaction rate is increased. The evaluation of antibacterial activity of synthesized compounds proved sulfur containing quinoline compound 3a much effective against Staphylococcus aureus, Escherichia coli and Streptococcus pyogenes than those of halogen containing heterocycles 2a.
Acknowledgement
One of the authors D.S. Lamani is thankful to The Director, Sophisticated Instrumentation Facility Center, Indian Institute of Science, Bangalore, Karnataka University Dharwad for the spectral facilities. C, H and N analysis were performed at Cochin University, Sophisticated Test and Instrumentation Center, Cochi, Kerala, India. And also we thank to SC/ST cell Kuvempu University for awarding Junior Research Fellowship (JRF). We thank Dr. Gouthamchandra, Department of Biotechnology, Kuvempu University Shankarghatta, Karnataka for carryout Antibacterial Activity and valuable discussion.
References
- M.S. Chauhan, S.K. Srivastava, Curr. Med. Chem., 2001, 8, 1535.
- K. Mogilaiah, D.S. Chowdary, R.B. Rao, Ind. J. Chem., 2001, 40B, 43.
- Y.L. Chen, K.C. Fang, J.Y. Sheu, S.L. Hsu, C.C. Tzeng, J. Med. Chem., 2001, 44, 2374.
- A.A. Joshi, C.L. Viswanathan, J. Med Chem., 2006, 5, 105.
- G. Roma, M.D. Braccio, G. Grossi, F. Mattioli, M. Ghia, Eur. J. Med. Chem., 2000, 35, 1021.
- S.A. Jenekhe, L. Lu, M.M. Alam, Macromolecules., 2001, 34, 7315.
- B. Jiang, Y.C. Si, J. Org. Chem., 2002, 67, 9449.
- O. Gia, S. Mobilio, M. Palumbo, M. A. Pathak, Photochem. Photobiol. 1993, 57, 497.
- O. Gia, M.T. Conconi, E. Uriarte, Farmaco., 1997, 5, 289.
- T.R. Ravikumar Naik, H.S. Bhojya Naik, M. Raghavendra, S. Gopalkrishna Naik, Arkivoc 2006, 94.
- L.F. Tietze, Chem. Rev., 1996, 96, 115.
- V.D. Dyachenko, A.D. Dyachenko, A.N. Chernega, Russian J. Orga. Chem., 2004, 40, 406.
- V.D. Dyachenko, A.D. Dyachenko, A.N. Chernega, Translated from Zhurnal Organicheskoi Khimii., 2004, 40, 433.
- E.D. Bergman, D. Ginsburg, R. Pappo, Organic Reactions, Adams, R., Ed., New York: Wiley, 1959.
- . V.P. Litvinov, L.A. Rodinovskaya, Yu.A. Sharanin, A.M. Shestopalov, A. Senning, Sulfur Rep., 1992, 13, 1, V.D.Dyachenko, Doctoral (Chem.) Dissertation, Moscow,1998.
- V.P. Litvinov, S.G. Krivokolysko, V.D. Dyachenko, Khim. Geterotsikl. Soedin., 1999, 579.
- V.N. Borisov, Sovremennye problemy organicheskoi khimii (Current Problems in Organic Chemistry), K.A. Ogloblin, Ed., Leningrad: Leningr. Gos. Univ., 1975, 89.
- B.P. Nandeshwarappa, D.B. Arun Kumar, H.S. Bhojya Naik, K.M. Mahadevan, J. Sulfur. Chem. 2005, 26, 379.
- D.S. Lamani, K.R. Venugopala Reddy, H.S. Bhojya Naik, H.R. Prakash Naik,. L.R, Naik, J. Sulfur Chemistry.,2009,1-11.
- D.S. Lamani, K.R. Venugopala Reddy, H.S. Bhojya Naik,A.Savyasachi, H.R. Naik, J. Nucleosides., Nucleotides and Nucleic Acids., 2008, 27, 1210.
- D.S. Lamani, K.R. Venugopala Reddy, H.S. Bhojya Naik, H.R. Prakash Naik, A.M. Sridhar, J. Macromolec. Sci. Part A., 2008, 45, 864.
- D.S. Lamani K.R. Venugopala Reddy, H.S. Bhojya Naik A.B. Naik, H.R. Prakash Naik, Phosphorus, Sulfur Silicon, and Relat. Elem., 2008, 185, 11.
- H.R. Prakash Naik, H.S. Bhojya Naik, T.R.; Ravikumar Naik, H. Raja Naik, D.S. Lamani, T. Aravind, J. Sulfur Chem., 2008, 29, 592.
- S. Le Mee, A. Pierre, J. Markovits, G. Atassi, A. Jacquemin-Sablon, J. M. Saucier, Mol. Pharm., 1998, 53, 213-220.
- B.M. Baboir, New England J. Med., 298, 1978, 629.
- P.D. Duh, Y.Y. Tu, G.C. Yen, Wiss. Technol., 1999, 32, 269.