Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 6
Antioxidant, Anti-Inflammatory and Antinociceptive Activities of Essential Oils of Three Satureja Species (Satureja briquetti, Satureja atlantica and Satureja alpina) Growing Wild in the Middle Atlas Mountains of Morocco
Jennan S1*, Fouad R2, FarahA2, Bousta D3 and Mahjoubi F1
1Laboratory of Materials Engineering and Environment, Faculty of Sciences University Sidi Mohamed Ben Abdellah (USMBA), Fez, Morocco
2Laboratory of Applied Organic Chemistry, Sciences and Technologies Faculty, USMBA Fez, Morocco
3Laboratory of Neuroendocrinology and Nutritional and Climatic Environment, FSDM, USMBA-Fez, Morocco
- *Corresponding Author:
- Jennan S
Laboratory of Materials Engineering and Environment
Faculty of Sciences University Sidi Mohamed Ben Abdellah (USMBA), Fez, Morocco
Abstract
This article describes the biological activities of essential oils of three species of Satureja. The choice of plants and activities studied was motivated by the frequent traditional use of Satureja abriquetti, Satureja atlantica and Satureja alpina by indigenous populations for the efficacy and virtues they discovered. The aim of the experiments is to prove the antioxidant, anti-inflammatory and antinociceptive activities of the essential oils of S. briquetti, S. atlantica and S. alpina. The antioxidant activity was evaluated by three different methods namely the free radical scavenging, the Ferric Reducing Antioxidant Power and the phosphomolybdenum method. The Carrageenan induced paw oedema in rats assay was used to evaluate the anti-inflammatory activity of essential oils. In order to prove the antinociceptive activity, the plantar model was chosen. The three essential oils have interesting antioxidant powers, especially by the reductive and phosphomolybdenum methods where the activities of S. atlantica and S. alpina exceed that of S. briquetti. In order to evaluate the anti-inflammatory activities of essential oils, the dose used orally is 200 mg / kg. S. alpina represents the best anti-inflammatory profile with an inhibitory capacity of 91.4%. The essential oil of S. atlantica has a slightly higher antalgic power followed by S. briquetti and S. alpina respectively. The results obtained confirm the traditional therapeutic effects and the use of these plants by the autochthonous population of the Middle Atlas Mountains of Morocco.
Keywords
Antioxidant activity, Anti-inflammatory activity, Antinociceptive activity, Essential oils.
Introduction
The family Lamiaceae is known by the richness of its species in natural substances having aromatic and medicinal properties. The genus Satureja belongs to the Lamiaceae family and contains about 200 species of aromatic herbs and shrubs that are grown in the Middle East, Mediterranean region of Europe, Western Asia, North Africa, Canary Islands and South America. More than 30 species of this genus are distributed in the eastern regions of the Mediterranean [1].
The Moroccan populations use some Satureja to aromatize the tea as well as to flavor and preserve the butter. Panaceae of Arab medicine, they are indicated against chills, colds and rheumatism, in infusion, gargles or decoction. They are also used against liver diseases and are known for galactogenic, emmenagogue, vermifuge, diuretic, digestive and antiseptic virtues.
Known since the 15th century, Satureja's essential oils were honored in 1933 thanks to their antidiarrheal properties and their bactericidal properties in stomatology, their efficacy was attributed by Shultzik to the presence of phenols (Carvacrol, Thymol) [2]. Essential oils isolated from various species of Satureja have various biological properties. Most of them are known for their antibacterial and antifungal properties [1,3-5].
The antioxidant activity of essential oils and extracts of some species of Satureja has been studied [6,7]. The antalgic and anti-inflammatory effects of some species of Satureja have also been reported [8].
Oxygen, essential for life, is a free radical, it has two unpaired electrons. This explains its great reactivity. However, most of the oxidative reactions it is likely to cause spontaneously in a human organism are extremely slow. It is therefore not very toxic by itself. But under the action of ionizing radiations, UV rays, transition metals or during enzymatic reactions, many more reactive species are produced. The term "reactive species of oxygen" (ERO) includes oxygen free radicals, but also certain non-radical reactive oxygenated derivatives whose toxicity is significant [9]. The number of natural substances with proven antioxidant properties is in perpetual increase; this category of molecules counts representatives in most chemical classes of the vegetable kingdom: terpenes, alkaloids, coumarins, lignans, methylxanthines and polyphenols. The essential oils may have a direct anti-radical action, for example by the presence of certain compounds having conjugated double bonds (chamazulene) or phenolic functions (thymol, carvacrol) [10,11].
A large number of methods for measuring the antioxidant capacity have been developed; however, there is great variability between the studies and therefore a difficult correlation between the results. Because of the diversity of the reactive species and the different oxidative mechanisms involved in the organism, it is therefore difficult to determine a single universal method of measuring antioxidant capacity either in vitro or in vivo, hence the advantage of combining several methods [12].
Based on the traditional use of plants studied by the indigenous population especially against cold spells, the essential oils of Satureja briquetti, Satureja atlantica and Satureja alpina have been evaluated for some of their biological activities in this case, antioxidant, anti-inflammatory and analgesic activities.
These plants were chosen for at least two main reasons:
- the frequent traditional use of S. briquetti, S. atlantica and S. alpina by indigenous populations for the efficacy and virtues they have discovered;
- the use of these plants indistinctively by these populations because of the ignorance of their multiplicity and the resulting variable effects.
One of the objectives of this study is to verify the therapeutic effect of these plants and to identify the levels of the possible differentiations linked to their already established chemical profiles.
Materials and Methods
Plant material
Aerial parts of plant material of S. briquetti, S. atlantica and S. alpina were collected during the flowering stage (Mai 2011) on the middle atlas mountain of Sefrou in Morocco. The plant materials were identified by experimented botanist (INP 233, 269 and 268). Authenticated voucher specimens were deposited in the Herbarium of The National Institute of Medicinal and Aromatic Plants, Sidi Mohamed Ben Abdellah University, Fez, Morocco.
Isolation of the essential oil
The plants were subjected to hydrodistillation using a Clevenger-type system. In each test, three distillations of 100 g of the dried aerial parts of the aromatic plants in a 1 l flask topped by a column of 60 cm length connected to a refrigerant were carried out by boiling for three hours. The obtained oils were separated from water by decantation, dried over anhydrous sodium sulphate and kept in amber vials at 4°C until use. The oil yields were expressed as ml/100 g of plant material. The yields of the essential oils were 1.76%, 1.35% and 1.30% for S. briquetti, S. atlantica and S. alpina respectively.
Evaluation of the antioxidant activity
The DPPH method
The antioxidant potential of the essential oils was determined on the basis of their scavenging activity of the stable 1,1-diphenyl-2-picrylhydrazyl (DPPH) free radical. Briefly, 100 μl of various concentrations of the oil in methanol was added to 3 ml of a methanol solution of DPPH (1.01 x 10-2 M). The mixture was vigorously shaken and then allowed to stand at room temperature for 30 min in the dark. The absorbance of the mixture was measured at 514 nm. A mixture of 100 μl of methanol and 3 ml of DPPH solution was used as the control. The scavenging activity on the DPPH radical was expressed as inhibition percentage using the following equation:
% Inhibition = [(AB – AS)/AB] x 100
Where, AB: is the absorbance of the control reaction (containing all reagents except the test compound)
AS: is the absorbance of the test compound. Butylatedhydroxytoluene (BHT) was used as positive control. The tests were carried out in triplicate.
The oil concentration providing 50% inhibition (IC50) was calculated from the graph of inhibition percentage plotted against oil concentration (62.5, 125, 250, and 500 mg/l) [13].
Evaluation of total antioxidant capacity by Phosphomolybdenum method
The essay is based on the reduction of Mo (VI) to Mo (V) by the oil and subsequent formation of a green phosphate/Mo (V) complex at acid pH and determined by the method described by Prieto [14]. The oil (300 μl) was added to 3 ml of reagent solution (0, 6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate). The tubes were incubated at 95°C for 90 min. After the mixture had cooled to room temperature, the absorbance of the solution was measured at 695 nm against a blank. The antioxidant capacity of each sample expressed as ascorbic acid (A. A) equivalent using following linear equation established using ascorbic acid as standard:
[Y1 = 4.6098 X1 + 0.0071; R2 = 0.9963]
The antioxidant capacity of each sample expressed as BHT equivalent using following linear equation established using BHT as standard:
[Y2 = 2.2163 X2 + 0.0639; R2 = 0.9923]
Where, Y: is the absorbance at 695 nm
X: is the concentration as ascorbic acid or BHT equivalent (mg/ml)
The values are presented as the means of triplicate analysis.
Ferric Reducing Antioxidant Power (FRAP)
The reducing power of a product is associated with its antioxidant power. This test is realized according to the procedure of Oyaizu [15] based on the reduction of Fe3+present in the complex K3Fe(CN)6. The absorbance is determined at 700 nm. An increased absorbance indicates ferric reducing power capability of sample. In a reaction tube, 0.5 ml of oil solutions, standard or blank were mixed with 2.5 ml of sodium phosphate buffer (0.2 M; pH 6.6) and 2.5 ml of a solution of ferricyanure of potassium (1%). The mixture is brooded during 20 min in a thermostated bath at 50°C. After cooling, 2.5 ml of trichloroacetique acid (10%) are added to the mixture. Finally, 2.5 ml of FeCl3 (1%) was added to this solution. Absorbance of this mixture was measured at700 nm using a UV spectrophotometer.
Anti-inflammatory activity: Carrageenan induced paw oedema in rats assay
The method used was similar to that previously reported [16]. Rats were divided into five groups of five animals each. Group I served as the control group receiving normal saline. Groups II–IV was giving an oral administration of the essential oils at different concentrations. Animals in group V were given a Diclofenac (1%). 1 h later after oral administration, 0.1 ml of 1% carrageenan (in normal saline) was injected sub-cutaneously into the plantar aspect of the right hind paw of each rat. Measurements of paw size were performed immediately 3, 4, 5, and 6 h after injection of carrageenan. The oedema degree in the hind paw for each rat, and percentage inhibition for each group, were calculated according to the following formulae:
Oedema degree (E) = Vt - V0
% inhibition = (Ec – Et) / Ec
Where, V0: is the paw size before carrageenan injection, Vt: is the paw size after carrageenan injection at t h, Ec: is the mean oedema degree of the control group, Et is the mean oedema degree of the treated group.
Antinociceptive activity: Plantar test
The animals were randomly allocated into three groups of five each. Group I was assigned as control receiving normal saline. Group II received an oral administration of essential oils at dose of 200 mg/Kg. Group III received the standard drug i. e. Diclofenac 1%. 1 h after the oral administration, rats were placed on the plantar apparatus. Individually, the paw of each rat was placed over the radiant heat source (50°C) and the paw withdrawal from the heat was taken as the end point. The tolerance time (s) of each rat was determined [17].
Results
Determination of free radical scavenging activity by DPPH method
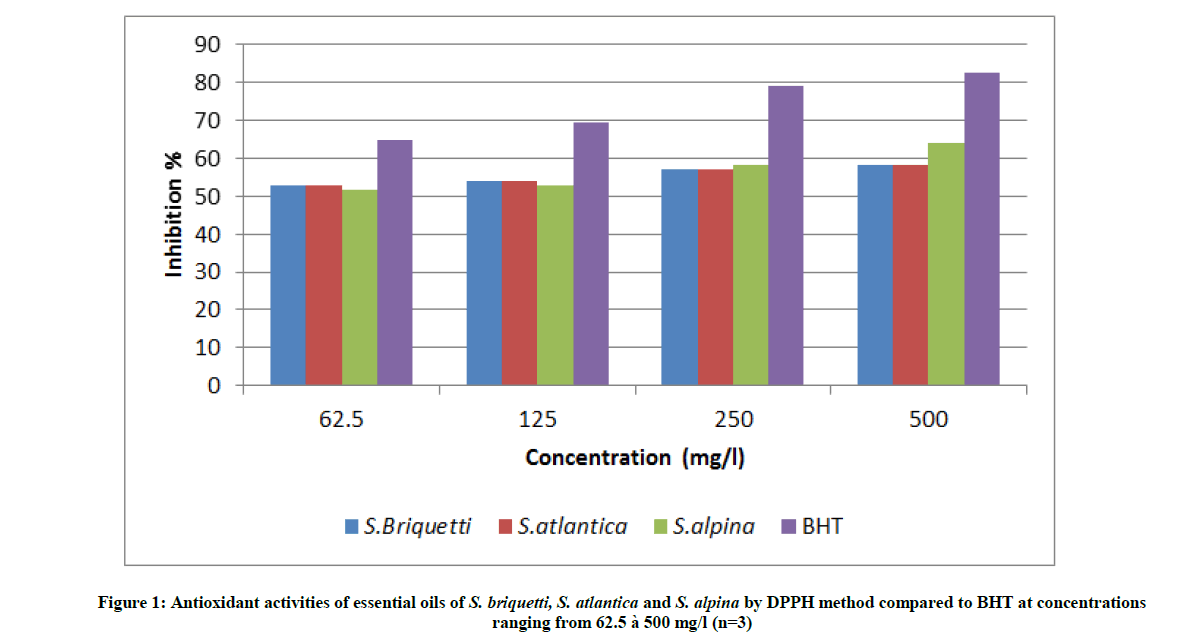
Antioxidant activity was analyzed using the DPPH free radical scavenging method. The results of the DPPH scavenging assay of the essential oils and the synthetic antioxidant (BHT) are shown in Figure 1. Assessed samples were able to reduce the stable violet DPPH radical to the yellow DPPH-H, reaching 50% of reduction with IC50 values ranging from 60 mg/l, for S. briquetti and S. atlantica, to 56 mg/l, for S. alpina. At low concentrations, the essential oils of S. briquetti and S. atlantica have similar anti-radical activity and exceed that of S. alpina. From a concentration of 250 mg/l, the antioxidant activity of S. alpina slightly exceeds that of the other two oils. The anti-radical power of the three essential oils is lower than the synthetic antioxidant (BHT).
The antioxidant is also evaluated by comparing the IC50 values shown in Table 1. The value of each IC50 expresses the essential oil concentration that reduces 50% of DPPH. The results confirm the important activity of S. alpina compared to those of S. briquetti and S. atlantica.
| Essential oil | IC50 (mg/l) |
|---|---|
| Satureja briquetti | 60 |
| Satureja atlantica | 60 |
| Satureja alpina | 56 |
| BHT | 50 |
Table 1: The IC50 values expressed in mg/l of the essential oils of Satureja briquetti, Satureja atlantica and Satureja alpina
Evaluation of total antioxidant capacity by Phosphomolybdenum method
Total antioxidant capacities of essential oils are expressed as number of equivalents of ascorbic acid and BHT (Table 2). The assay is based on the reduction of Mo (VI) to Mo (V) by the oil and subsequent formation of a green phosphate/Mo (V) complex at acid pH. The phosphomolybdenum method is quantitative since the antioxidant activity is expressed as the number of equivalents of ascorbic acid and BHT14. S. atlantica essential oil had a higher capacity than have the S. briquetti and S. alpina essential oils.
| Essential oil | Equivalent to ascorbic acid (mg/g of essential oil) | Equivalent to BHT (mg/g of essential oil) |
|---|---|---|
| Satureja briquetti | 115.68 ± 14.10 | 232.08 ± 29.33 |
| Satureja atlantica | 163.87 ± 10.57 | 332.30 ± 21.99 |
| Satureja alpina | 117.91 ± 0.79 | 236.84 ± 1.65 |
Table 2: Total antioxidant activities of essential oils of Satureja briquetti, Satureja atlantica and Satureja alpine (n=3)
Reducing power
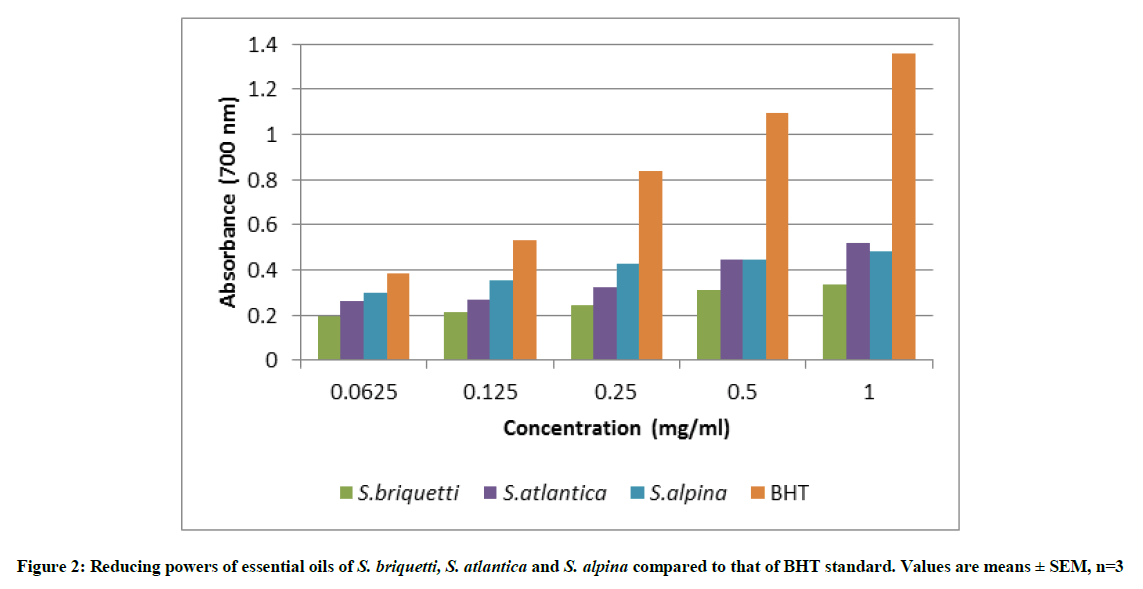
The reductive potential measures the ability of a sample to act as electron donor and, therefore, reacts with free radicals converting them to more stable products and thereby terminates radical chain reactions. Figure 2 shows the reducing powers of the essential oils and the BHT standard. The activities of the oils increased with increasing concentration. At concentrations ranging from 0.0625 to 0.25 mg/ml essential oil of S. alpina presents the most important reducing power followed by and S. atlantica and S. briquetti respectively. At a concentration of 1 mg/ml, the activity of S. atlantica exceeds that of S. alpina.
Anti-inflammatory activity
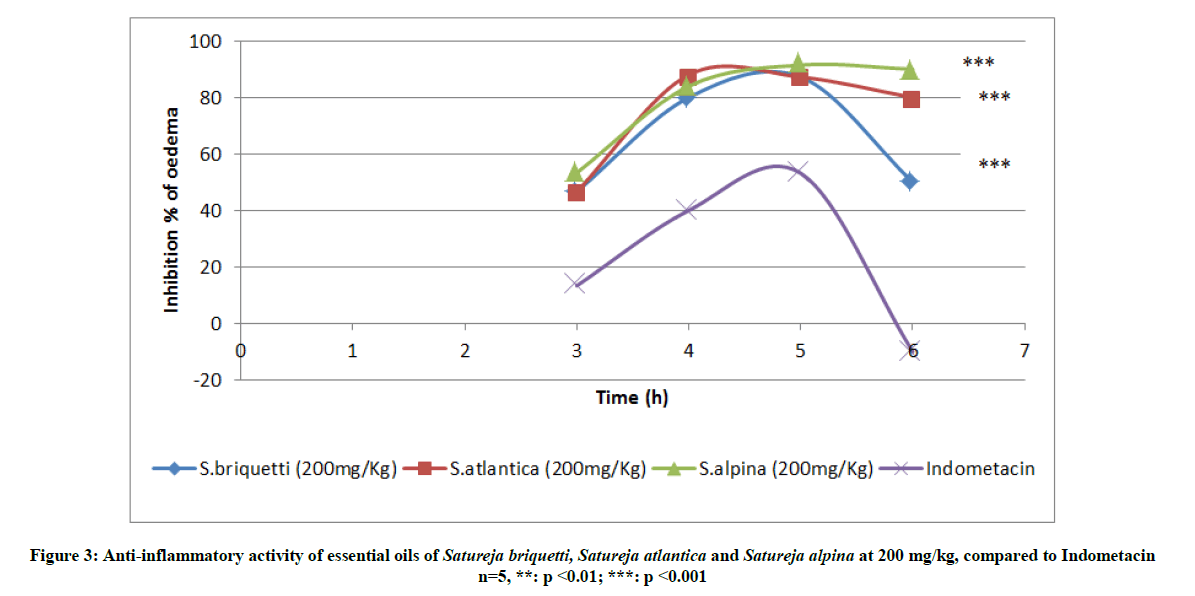
Injection of carrageenan into the hind paw induced a progressive edema reaching its maximum at 3 h. We noted a significant difference between anti-inflammatory essential oils activities and the indomethacin (Figure 3). 5 h after the injection of carrageenan, essential oils reach their maximum inhibition of edema. S. alpina represents the best anti-inflammatory profile with an inhibitory capacity of 91.4% at 5 h and which decreases slightly at 6 h to reach 90%. It is also noted that the effects of the essential oils of S. atlantica and S. briquetti are prolonged in time to reach after 6 h 80% and 50% of inhibition respectively.
Antinociceptive activity
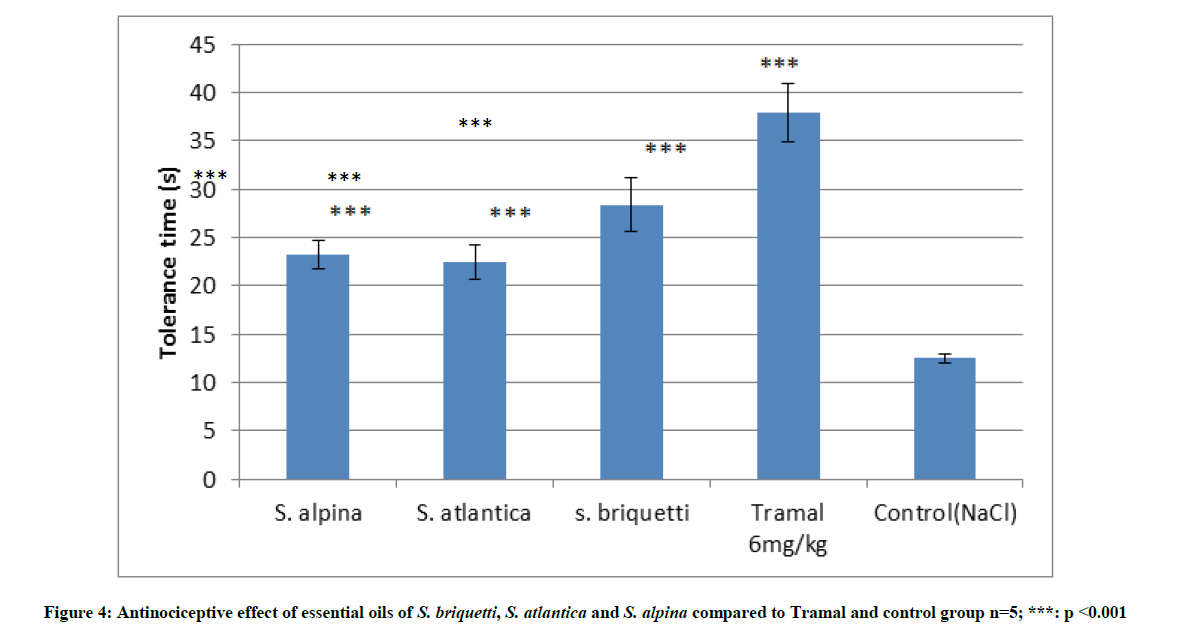
The evaluation of the antinociceptive activity of the essential oils of S. briquetti, S. atlantica and S. alpina was carried out on the basis of the Plantar model. The dose of 200 mg/kg was studied for its possible analgesic activity. The results are shown in Figure 4. It is noted that the tolerance times of the rats treated with essential oils are significantly greater than that of the control group which received only a physiological solution of NaCl. By comparing the essential oils between them, S. atlantica has a slightly higher analgesic power followed by S. briquetti and S. alpina respectively. Rats treated with the reference drug (Tramal) had a higher effect than rats treated with essential oils.
Discussion
Numerous studies have established the relationship between the chemical composition of the essential oil and its antioxidant activity. It has been reported that the antioxidant activity of essential oils is related to their chemical compositions, particularly by the presence of compounds containing a hydroxyl function [18-20]. More an oil is rich in terpene alcohols and phenolic compounds, the more its antioxidant power is important [21,22]. In our knowledge, no studies on the antioxidant activity of S. briquetti, S. atlantica and S. alpina have been carried out. On the other hand, the antioxidant activity of various species of the genus Satureja has been subject of several studies [7,23-26]. Ozturk et al. has demonstrated the very important antioxidant activity of S. thymbra from Turkey as well as that of its major compounds thymol and carvacrol [27]. The antioxidant power of the essential oil of S. cuneifolia was also explained by the high content of carvacro [13]. It has been reported that essential oils of the genus Satureja are rich in isopropanoids such as thymol, carvacrol, β-caryophyllene, γ-terpinene, p-cymene and linalool which are known for their potential antioxidants [1]. Inflammation is a complex process, which is frequently associated with pain and involves several events, such as increased vascular permeability, increased migration of granulocytes and mononuclear cells [28]. Several experimental protocols of inflammation and pain are used to assess the potency of drugs. In this study, and in order to evaluate the anti-inflammatory activities of the essential oils of S. briquetti, S. atlantica and S. alpina, the Carrageenan induced paw oedema in rats model was used [17]. Carrageenan is a pro-inflammatory that acts in 2 phases. The first phase or early phase corresponds to the secretion of histamine and serotonin. The second phase or late phase corresponds to a secretion of bradykinin and prostaglandin [29]. In the Carrageenan induced paw oedema in rats model, the early phase reached its peak at 180 minutes [30]. In this study, essential oils showed anti-inflammatory effects in both phases but activity was more pronounced during the late phase of inflammation. These results suggest that the essential oils tested act in the late phase involving arachidonic acid metabolites probably by the inhibition of cyclooxygenase [31].
Several studies have highlighted the anti-inflammatory and analgesic activities of certain essential oils [32-35]. On the other hand, few studies on the anti-inflammatory and antalgic effects of Satureja species have been consulted [8,36]. The main constituents of essential oils known for their antiinflammatory effects are monoterpenols, methyl-ether phenols, esters, aldehydes and especially sesquiterpenes [37]. It has been reported that monoterpenes can also be responsible for anti-inflammatory activity by acting on several inflammatory mediators [38].
Conclusion
The various oils studied were evaluated for their biological properties; in this case the antioxidant, anti-inflammatory and analgesic activities. The results obtained from the evaluation of the antioxidant activities show that all the oils are endowed with a very important antioxidant power. The evaluation of the anti-inflammatory activity of essential oils of S. briquetti, S. atlantica and S. alpina showed a strong anti-inflammatory potential of the different oils exceeding that of the reference anti-inflammatory drug.
References
- S. Momtaz, M. Abdollahi, Int. J. Pharmacol., 2010, 6, 346-353.
- J. Bellakhdar, M. Ait-Igri, M. Rombourg. Al biruniya, Rev. Mar. Pharm., 1986, 4, 67.
- F. Oke, A. Belma, S. Ozturk, Food Chem., 2009, 112, 874-879.
- H. Baydar, O. Sadiç, G. Ozkan, Food Control., 2004, 15, 169-172.
- M. Skocibusic, N. Bezic, V. Dunkic, Food Chem., 2006, 96, 20-28.
- G. Ozkan, B. Simsek, H. Kuleasan, Journal of food engineering., 2007, 79, 1391-1396.
- J.D. Dorman, R. Hiltunen. Food Chemistry, 2004, 88, 193 – 199.
- V. Hajhashemi, A. Ghannadi, S.K. Pezeshkian, J. Ethnopharmacol., 2002, 82, 83- 87.
- A. Favier. L’actualité chimique, 2003, 108-115.
- D.L. McKay, J.B. Blumberg, Phytother. Res., 2006, 20, 519-530.
- B. Bozin, N. Mimica-Dukic, N. Simin, G. Anackov, J. Agric. Food Chem., 2006, 54, 1822-1828.
- E. Niki, Free Radic. Biol. Med., 2010, 49, 503-515.
- J. Kubola, S. Siriamornpun, Food Chem., 2008, 110, 881- 890.
- P. Prieto, M. Pineda, M. Aguilar, Anal. Biochem., 1999, 269, 337-341.
- M. Oyaizu, Japan. J. Nutr., 1986, 44, 307-315.
- C.A. Winter, E.A. Risley, G.W. Nuss, Exp. Biol. Med., 1962, 111, 544-547.
- K. Hargreaves, R. Dubner, F. Brown, C. Flores, J. Joris, Pain., 1989, 32, 77-88.
- S. Bouhdid, S.N. Skali, M. Idaomar, A. Zhiri, D. Baudoux, M. Amensour, J. Abrini, Afr. J. Biotechnol., 2008, 7, 1563-1570.
- I. Gulçin, I.G. Sat, S. Beydemir, M. Elmastas, O.I. Kufrevioglu, Food Chem., 2004, 87, 393-400.
- S.S. Chun, D.A. Vattem, Y.T. Lin, K. Shetty, Process Biochemistry., 2005, 40, 809-816.
- S.A. Fayed, Research Journal of Agriculture and Biological Sciences., 2009, 5, 740-747.
- S.R. Zhuang, S.L. Chen, J.H. Tsai, C.C. Huang, T.C. Wu, W.S. Liu, H.C. Tseng, H.S. Lee, M.C. Huang, G.T. Shane, C.H. Yang, Y.C. Shen, Y.Y. Yan, C.K. Wang. Phytother. Res., 2009, 23, 785-790.
- A. Giweli, M.A. Džamić, M. Soković, S.M. Ristić, P.D. Marin, Molecules., 2012, 17, 4836-4850.
- S. Bagheri, H. Ahmadvand, A. Khosrowbeygi. Asian Pac J Trop Biomed, 2013, 3, 22-27.
- D. Bukvički, D. Stojković, M. Soković, Meat Science., 2014, 96, 1355-1360.
- L. Cherrat, L. Espina, M. Bakkali, Innov. Food Sci. Emerg. Technol., 2014, 22, 221-229.
- M. Ozturk, Food Chem., 2012, 134, 48-54.
- S. Silbernagl, F. Lang. Atlas de Poche de Physiopatologie, 1st (Edn.), Gallimard (Edi.), France, Flammarion, 2000.
- R. Vinegar, W. Schreiber, R. Hugo, J. Pharmacol. Exp. Ther., 1969, 166, 96-103.
- O.A. Olajide, M.J. Makinde, S.O. Awe, J. Ethnopharmacol., 1999, 66, 113-117.
- B. Tepe, M. Sokmen, H.A. Akpulat, Food Chem., 2006, 95, 200-204.
- J.S. Raut, S.M. Karuppayil, Industrial Crops and Products., 2014, 62, 250-264.
- S. Afoulous, H. Ferhout, E.G. Raoelison, Food Chem. Toxicol., 2013, 56, 352-362.
- M.R. Loizzo, F. Menichini, F. Conforti, Food Chem., 2009, 117, 174-180.
- J. Koudou, A.A. Abena, P. Ngaissona, Fitoterapia., 2005, 76, 700-703.
- S. Aydin, Y. Oztiirk, Phytother. Res., 1996, 10, 342-344.
- D. Pénoël, R. Jollois, P. Franchomme, J. Mars. L’aromathérapie exactement, Roger Jollois (Edi.), 1999, 446.
- J. Silva, W. Abebe, S.M. Sousa, J. Ethnopharmacol., 2003, 89, 277-283.