Research Article - Der Pharma Chemica ( 2019) Volume 11, Issue 1
Calcined Tungstophosphoric Acid: An Efficient Catalyst for One-pot Three-component Betti Bases Synthesis at Mild Reaction Condition
Abdulrahman I Alharthi*Abdulrahman I Alharthi, Department of Chemistry, College of Science and Humanities, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj 11942, Saudi Arabia, Email: saalharthi@iau.edu.sa
Abstract
An efficient one-pot three-component Betti bases synthesis procedure has been developed. In the current study, substituted aldehydes β-napthol and aniline have been used in the presence of calcined tungstophosphoric acid as a heterogeneous catalyst with dichloromethane as a solvent at room temperature. Calcined (X) form of tungstophosphoric acid was compared with non calcined (Y) in terms of product yield and reaction time. The former exhibited better performance as compared to the latter. The study concluded that calcined tungstophosphoric acid acts as an efficient environmentally friendly catalyst for the one-pot three-component Betti bases synthesis under mild condition.
Keywords
Tungstophosphoric acid, Betti bases, Calcination, Catalyst
Introduction
Betti reaction is one of the important multicomponent syntheses which were developed in the early twentieth century by Mario Betti [1]. The reaction is characteristically an amino alkylation reaction with carbon-carbon and carbon-nitrogen bond formation simultaneously [2]. A typical Betti reaction is a modified Mannich reaction, where 2-napthol, benzaldehyde and amine derivatives react together to provide 1-(α-aminoalkyl-2-napthols, commonly called Betti bases [3]. Recently, more attention has been given to Betti bases as seen in numerous biological [4,5], catalytic [6,7] and synthetic applications [8,9].
Classical Betti bases were initially prepared by reacting ammonia or urea, an aldehyde and β-napthol in alcoholic potassium hydroxide solution in two steps taking several hours (9-36 h) [9]. Later, one-pot three component methods were adopted to overcome the demerits of the classical method by selecting different reacting species [10-13]. Unfortunately, such methods also suffered from shortcomings including side reactions, long reaction time and low yields.
Recently, several green and convenient techniques have been also developed to synthesize Betti bases [14,15]. These were also reported in the preparation of 1-amidoalkyl-2-napthol by the condensation of aryl aldehydes, β-napthol and acetonitrile or amide in the presence of Bronsted or Lewis acid catalysts such as Ce (SO4)2 [16], K5CoW12O40.3H2O [17] and SiO2-FeCl3 [18].
Tungstophosphoric acid (H3PW12O40), a class of heteropoly acid (HPA), having Bronsted acid properties has recently gained recognition in organic synthesis [19,20]. Tungstophosphoric acid has high thermal stability and high solubility in polar solvents [21]. It was also reported that tungstophosphoric acid proved to be stronger than common mineral acids (HCl, H2SO4, HNO3) and upon calcination (up to 400ºC) develop super acid sites [22,23].
In the present investigation, synthesis of Betti bases has been reported by using calcined tungstophosphoric acid as a catalyst. Further, both the physical forms (calcined, non-calcined) of tungstophosphoric acid performance were evaluated in terms of yield, time of reaction and amount of catalysts.
Materials and Methods
All the materials and chemicals were procured from Sigma Aldrich. Fourier Transform Infrared (FTIR) spectra for the synthesized compounds were taken by thermo scientific iD5 ATR diamond Nicolet iS 5FTIR spectrometer. Proton Nuclear Magnetic Resonance (1H-NMR) and Carbon-13 Nuclear Magnetic Resonance (13C-NMR) spectra of compounds were recorded with BRUKER-PLUS (400 MHz). Calcination of catalyst (tungstophosphoric acid) was performed using thermocraft incorporated tube furnace.
Tungstophosphoric acid calcination
An accurately weighed tungstophosphoric acid was taken in crucible and placed in a tube furnace around 400ºC up to 4 h.
General procedure for the synthesis of betti bases (4a-13a) via one-pot multicomponent reaction using tungstophosphoric acid
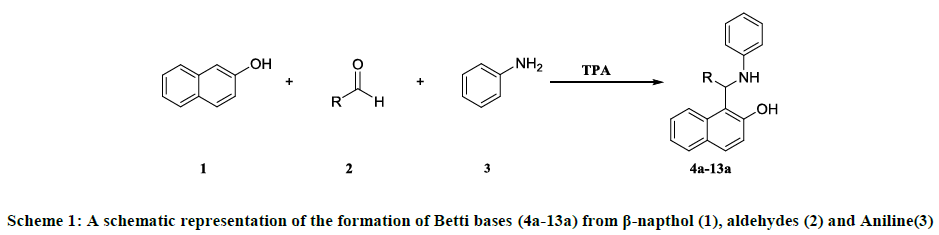
A mixture of aldehydes (2 mmol), β-napthol (2 mmol) and aniline (2.5 mmol) was stirred in dichloromethane (DCM) in presence of varying mol% of calcined as well as non-calcined tungstophosphoric acid at room temperature. The resulting product was obtained by recrystallization of ethanol.
A general Scheme 1 for the Betti bases is shown in the figure below.
Spectral data of few selected compounds
1-((phenylamino)(2,4,6-trimethoxyphenyl)methyl)naphthalene-2-ol (5a)
FTIR (cm-1, ATR); 3388 (NH), 3200 (OH), 1602 (C=C, Ar); 1H-NMR(DMSO) δ (ppm)=10.2 (1H, s, OH), 8.93 (1H, s, Ar-H), 7.07-7.77 (9H, m, Ar-H), 6.50 (1H, br, NH), 5.76 (1H, s, CH), 3.45-3.82 (9H, s, OCH3); 13C-NMR (DMSO) δ (ppm)=172.33, 165.60, 156.82, 130.12, 130.04, 129.47, 123.09, 122.43, 100.78, 92.47, 57.88, 57.48.
1-(indolin-2-yl)(phenylamino) methyl)naphthalene-2-ol (6a)
FTIR (cm-1, ATR); 3371 (NH), 3220 (OH), 1622 (C=C, Ar); 1H-NMR (DMSO) δ (ppm)=9.93 (1H, s, NH, indole), 9.39 (1H, s, OH), 8.29 (1H, d, Ar-H, J=10.2 Hz), 8.10-8.08 (1H, d, CH, J=7.6 Hz), 8.80 (1H, s, NH), 7.17-7.75 (16H, m, Ar-H); 13C-NMR (DMSO) δ (ppm)=168.41, 163.65, 155.80, 130.16, 130.08, 129.44, 124.01, 124.33, 100.98, 91.50, 56.40, 54.22.
1-((phenylamino)(1H-pyrrol-2-yl)methyl) napthalen-2-ol (7a)
FTIR (cm-1, ATR); 3346 (NH), 3204 (OH), 1621 (C=C, Ar); 1H-NMR (DMSO) δ (ppm)=11.76 (1H, s, NH, pyrole) 10.21 (1H, s, OH), 9.71 (1H, s, Ar-H), 9.48 (1H, br, NH), 6.98-7.77 (10H, m, Ar-H), 6.21-6.71 (3H, m, pyrole) 5.01 (1H, s, CH); 13C-NMR (DMSO) δ (ppm)=155.73, 152.62, 150.99, 149.06, 135.05, 131.04, 129.75, 129.65, 129.26, 128.18, 127.99, 126.56, 126.44, 126.37, 124.30, 123.09, 121.20, 119.06, 116.90, 116.08, 114.32, 110.16, 109.09.
1-(1-(phenylamino)pentyl)napthalen-2- ol (12a)
FTIR (cm-1, ATR); 3392 (NH), 3208 (OH), 1598 (C=C, Ar); 1H-NMR (DMSO) δ (ppm)=8.79(1H, br, NH), 7.17-8.20 (10H, m, Ar-H), 3.12-3.10 (1H, d, CH, J=7.6 Hz), 2.95-2.93(2H, t, CH2, J=8.4 Hz), 1.81-1.77(2H, t, CH2, J=14.8. Hz), 0.85(1H, s, CH3); 13C-NMR (DMSO) δ (ppm)=155.26, 145.49, 137.03, 128.58, 122.57, 121.89, 119.53, 60.22, 22.72, 19.02, 14.56.
Conclusion
In conclusion, in the current work, an easy technique has been reported to synthesize one-pot three-component reaction for Betti bases synthesis using calcined tungstophosphoric acid in dichloromethane as a solvent simply at room temperature. The calcined version of tungstophosphoric acid was found more effective for the product formation in the limited time of frame with high yield. Moreover, heat treated tungstophosphoric acid can be used for different condensation reactions.
Acknowledgment
I am very grateful to Prince Sattam Bin Abdulaziz University for providing facilities for successful completion of this research work.
References
- C. Cardellicchio, M.A.M. Capozzi, F. Naso, Tetrahedron Asymm., 2010, 21, 507-517.
- R. Teimuri-Mofrad, M. Gholamhosseini Nazari, S. Esmati, A. Shahrisa, Res. Chem. Intermed., 2017, 43, 6845-6861.
- I. Szatma´ri, F. Fulo¨p, Tetrahedron., 2013, 69, 1255-1278.
- A.Y. Shen, C.T. Tsai, C.L. Chen, Eur. J. Med. Chem., 1999, 34, 877-879.
- H.R. Shaterian, H. Yarahmadi, Tetrahedron Lett., 2008, 49, 1297-1300.
- J. Lu, X. Xu, C. Wang, J. He, Y. Hu, H. Hu, Tetrahedron Lett., 2002, 43, 8367-8369.
- I. Szatmari, A. Heteny, L. Lazar, F. Fulop, J. Heterocycl. Chem., 2004, 41, 367-373.
- R.S. Hamid, Y. Hossein, G. Majid, Turk. J. Chem., 2009, 33, 449-457.
- M. Jie, G. Gan, C. Chen, L. Jie, G. Jian, L. Yi, P. Dongsheng, RSC Adv., 2017, 7, 13868-13875.
- G. Hong, S. Jing, Y. Chao-Guo, Chinese Chemical Lett., 2015, 26, 353-356.
- C. Cimarelli, D. Fratoni, A. Mazzanti, G. Palmieri, Tetrahedron Asymm., 2011, 22, 591-596.
- I. Szatmari, F. Fulop, Curr. Org. Synth., 2004, 1, 155-165.
- J. Lu, X. Xu, C. Wang, Tetrahedron Lett., 2002, 43, 8367-8369.
- R. Csutortoki, I. Szatma ri, F. Fulop, Curr. Org. Synth., 2013, 10, 564-583.
- A. Kumar, M.K. Gupta, M. Kumar, Tetrahedron Lett., 2010, 51, 1582-1584.
- N.P. Selvam, P.T. Perumal, Tetrahedron Lett., 2006, 47, 7481-7483.
- M. Wang, S. Zhi-Guo, L. Yan, Org. Prep. Proce. Int., 2011, 43, 484-488.
- R.S. Hamid, H. Asghar, G. Majid, ARKIVOC, 2009, 59-67.
- V.K. Ivan, Chem. Rev., 1998, 98, 171-198.
- M. Misono, Catal. Rev. Sci. Eng., 1988, 30, 339-340.
- K.R. Jaspreet, K. Gagandeep, Tetrahedron lett., 2012, 53, 646-649.
- K. Nowińska, R. Fiedorow, J. Adamiec, J. Chem. Soc. Faraday Trans., 1991, 87, 749-753.
- T. Okuhara, T. Nishimura, H. Watanabe, M. Misono, J. Mol. Catal., 1992, 74, 247-256.
- L.L. Wei, L.W. Li, Y.L. Qiu, The Scientific World Journal, 2013, 2013, 1-4.
- N. Lingaiah, M. Baseeruddin, Satyender, K. Srinivas, Cataly. Commun., 2007, 8, 1729-1734.
- M.K. Mohammad, R.K. Ahmad, M. Hassan, Syn. lett., 2006, 6, 916-920.
- A. Dorehgiraee, H. Khabazzadeh, K. Saidi, ARKIVOC, 2009, 7, 303-310.