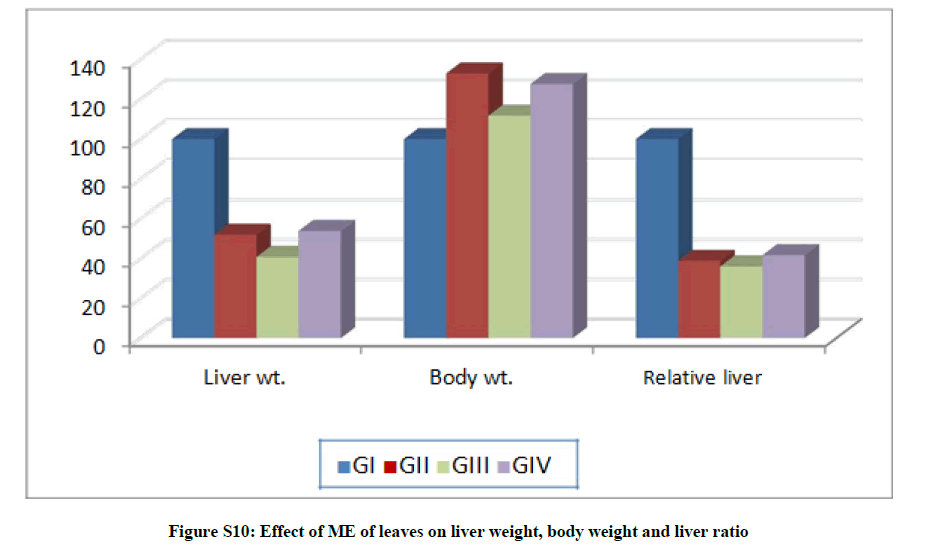Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 10
Chemical Composition and Liver Protection of the Methanol Extract of Fortunella margarita (Lour.) Swingle Leaves
Magdy MD Mohammed1*, Nabaweya A Ibrahim1, Seham S El-Hawary2, Sanaa A Ali3, Maha Z Rizk3 and Eman AW El-Abd1
1Department of Pharmacognosy, Pharmaceutical and Drug Industries Research Division, National Research Centre, Dokki-12622, Cairo, Egypt
2Department of Pharmacognosy, Faculty of Pharmacy, Cairo University, Giza, Egypt
3Department of Therapeutic Chemistry, Pharmaceutical and Drug Industries Research Division, National Research Centre, Dokki-12622, Cairo, Egypt
- *Corresponding Author:
- Magdy MD Mohammed
Department of Pharmacognosy
Pharmaceutical and Drug Industries Research Division
National Research Centre
Dokki-12622, Cairo, Egypt
Abstract
Liquid chromatography-mass spectrometric (LC/MS) analysis of the leave’s methanol extract of Fortunella margarita (Lour.) Swingle led to the identification without isolation of four main compounds. Extensive chromatographic purification revealed the isolation of acacetin 7-neohesperidoside, acacetin 7-rutinoside and 4-(β-D-glucopyranosyloxy) benzoic acid. Proton Nuclear Magnetic Resonance (1H-NMR) and Carbon-13 Nuclear Magnetic resonance (13C-NMR) along with HR-ESI/MS were used to elucidate the chemical structure of the isolated compounds. Liver protection against DMN and/or CCl4-induced toxicity was evaluated for the ME of F. margarita, and the obtained results showed that the ME decreased the elevated liver enzymes to the normal level, indicating the protection of structural integrity of hepatocytic cell membrane or regeneration of damaged liver cells.
Keywords
Fortunella margarita (Lour.) Swingle, Rutaceae, Acacetin glycosides, Hepatoprotective, DMN, CCl4.
Introduction
Fortunella margarita Lour. Swingle (Synonym: Citrus japonica Thunb.) belongs to the family Rutaceae, and known as Nagami or oval kumquat, its origin back to Asia (Southeastern region) i.e., China, and it can be cultivated elsewhere as in Egypt. Folkloric medicinal uses of F. margarita recorded a wide range of activity such as anti-fevers, anti-hepatitis and anti-asthmatic [1], and anti-inflammatory [2]. Leaf decoction of Fortunella used to treat coughs (i.e., whooping coughs), headache, viral hepatitis, diabetes, fever, etc. [3]. Moreover, F. margarita as well as other species used to prepare fruit salad, marmalade and food preservative [4].
Phytochemical investigation of Fortunella species revealed the presence of some phytochemicals i.e., ascorbic acid, carotenoids [5], flavonoids [6,7], and essential oils [4]. In addition, Chen et al. [8] stated that the polymethoxylated flavonoid ´natsudaidain` isolated from F. margarita is more potent inhibitor of tumor cell growth than quercetin and apigenin [9].
Furthermore, Nogata et al. [10] reported that F. margarita fruit extract has anti-inflammatory effect by inhibiting cyclooxygenase and lipooxygenase. The antioxidant potential of carotenoids isolated from F. margarita was evaluated by Olson [11].
The presented study was conducted to discover the phenolic content of the ME of F. margarita leaves, and study its hepatoprotective activity which documented by a histopathological supplementation.
Materials and Methods
Plant materials
F. margarita (Lour.) Swingle leaves were collected from the Green Farm, 171 Cairo-Alexandria Desert Rood, Wadi El-Natroun, El-Beheira Governorate, Egypt, during the flowering stage August 2006. The plant was kindly authenticated by Mrs. Therese Labib, Consultant of Plant Taxonomy at the Ministry of Agriculture at EL-Orman Botanical Garden, a voucher specimen (No. 000120FC 04-09-06-25) was kept at the herbarium of EL-Orman Botanical Garden. The leaves were air dried at r.t., powdered and kept for further processing.
Extraction procedure
Air dried powdered leaves (500 g) were extracted using Soxhlet apparatus with solvents of increasing polarities pet. ether, CH2Cl2, and MeOH till exhaustion. The obtained MeOH extract ME (15 g) was applied to column chromatography (100 × 2 cm i.d., 530 g). Elution was successively carried out starting with ethyl acetate (100%) and increasing polarity with methanol stepwise till 100% methanol. 150 fractions (50 ml, each) were successively collected, monitored by TLC using n-BuOH–AcOH–H2O (4: 1: 5, upper layer), and development was carried out using ferric chloride reagent (FeCl3). Fractions were further chromatographed and similar fractions combined together to afford finally seven main fractions (I-VII) of overlapped brown coloured spots, further purification over Sephadex LH-20 led to the isolation of three main phenolic compounds; compound (1) 10 mg from fractions (VI and VII), compound (2) 22 mg from fractions (IV and V), and compound (3) 48 mg from fractions (I-III).
General experimental procedures
Nuclear Magnetic Resonance (NMR) Spectra were obtained using a pulse sequence supplied from JEOL 500 MHz. Chemical shifts were given in values (ppm) relative to Trimethylsilane (TMS) as an internal reference for both carbon and proton. LC/MS and HR-ESI/MS: as reported [12,13]. All used solvents were of AR grade. Kiesel gel 60 F254 (Merck) was used for analytical TLC. Silica gel for column chromatography (70-230 mesh, Merck, ASTM), and Sephadex LH-20 (Pharmacia, Uppsala, Sweden).
In vivo hepatoprotective study
Chemicals and biochemical kits
Chemicals used in the present study were of high analytical grade, products of Sigma (USA), Merck (Germany) and BDH (England). Kits used for the quantitative determination of different biochemical parameters were purchased from Bio-diagnostic Company.
Animals
Male Wister albino rats weighting (120-150 g) used for DMN-liver adenoma, and male Wister albino mice weighting (20-30 g) used for CCl4-induced toxicity, were supplied from the animal house of National Research Centre (Dokki, Giza, Egypt). Animals were kept for two weeks to accommodate on laboratory (environmental and nutritional) conditions, given food and water all over the period of the experiment.
Ethics
Appropriate anesthetic and sacrifice procedures were followed ensuring that animals didn't suffer at any stage of the experiments. Anesthetic procedures complied with the legal ethical guidelines approved by the Ethical Committee of the Federal Legislation and National Institutes of Health Guidelines in USA and were approved by the ethical committee of the National Research Centre in Egypt with registration number (13-014) for hepatoprotective study.
Hepatoprotective assay for liver adenoma by DMN
Experimental design
24 male albino rats were divided into four groups:
G-1: Normal control rats (without any treatment).
G-2: Dimethyl nitrosamine (DMN) treated group.
G-3: DMN group co-administrated with methanolic extract (ME) of F. margarita.
G-4: ME of F. margarita leaves.
Samples induction
Dried ME of F. margarita leaves (100 mg/kg body weight) was given orally for four weeks. Hepatic fibrosis was induced by intraperitoneal injections of DMN dissolved in saline in doses of 10 mg/kg body weight. The injections were given on the first three consecutive days of each week over a period of four weeks. Control animals were also received an equal volume of 0.15 mol/l NaCl without DMN. The injections were given without anesthesia [14].
Hepatoprotective assay for liver toxicity by CCl4
Experimental design
32 male albino mice were divided into four groups (each of eight mice).
G-I: Normal healthy control (no treatment).
G-II: Normal healthy mice orally administrated with ME twice a week for 7 weeks.
G-III: IP injected with CCl4.
G-IV: IP injected with CCl4 and orally treated with ME twice a week for 7 weeks.
Samples induction
Dried ME of F. margarita leaves (500 mg/kg body weight) was given orally twice a week for 7 weeks, liver damage in mice was induced by intraperitoneal injection of CCl4 (200 mg/kg body weight), twice a week for 7 weeks.
Biochemical parameters
Estimation of γ-glutamyl transferase activity (γ-GT) as reported [15,16], aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities [17-19], alkaline phosphatase (ALP) activity [20,21], albumin level total protein [22,23], glucose level using Stanbio Laboratory Diagnostic kits (USA), total cholesterol (TC) [24], and triglycerides levels [25].
Statistical analysis
All data were expressed as mean ± S.D. of eight rats in each group. Statistical analysis was carried out by one-way analysis of variance (ANOVA), CoStat software computer program.
Histopathological analysis
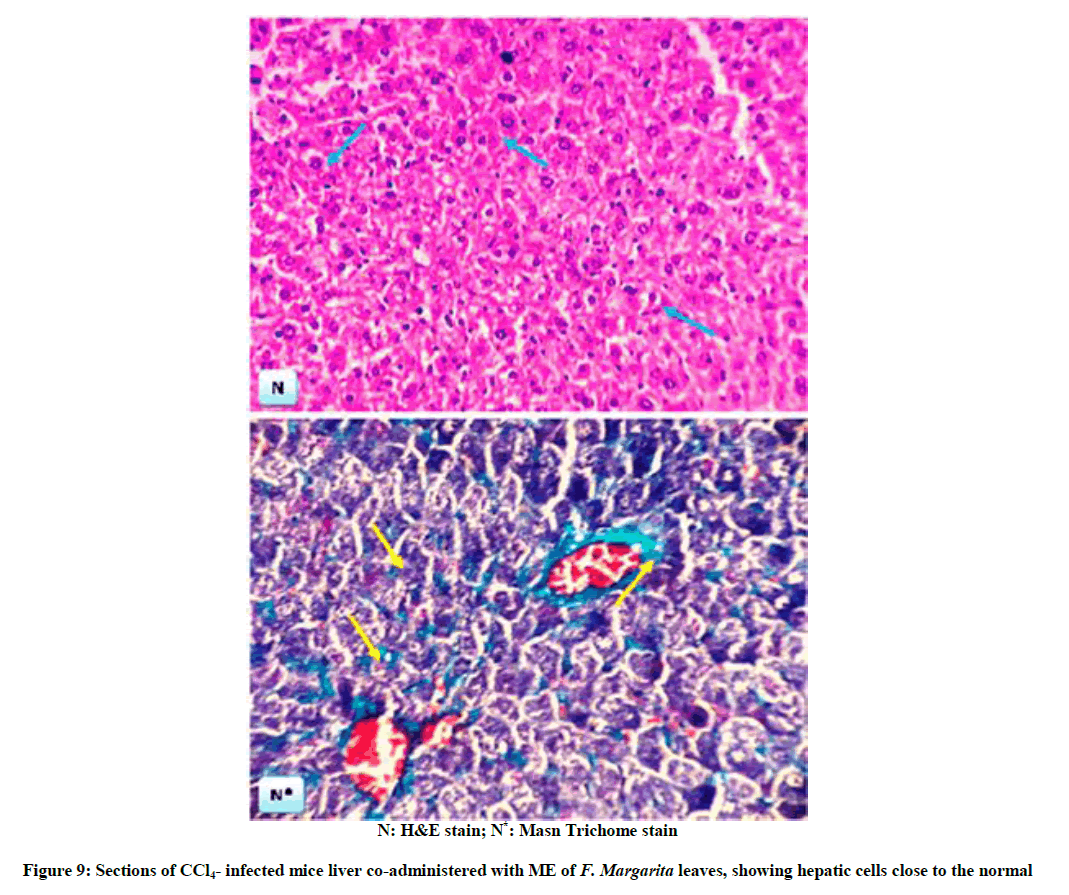
Treated groups with maximum activity were chosen (one animal each) for this purpose. The animals were sacrificed and the abdomen was cut open to remove the liver. Representative slices from liver were taken from the eviscerated animals and fixed in 10% formalin. Paraffin embedded sections were taken after fixation. Sections (5 μm thick) were stained with haemoatoxylin and eosin (H&E). Masson’s Trichrome was used for detecting the collagen deposition in the hepatic tissue for CCl4-induced toxicity. The stained sections were studied by light microscope for demonstration of pathological changes [26].
Results and Discussion
Chemical composition
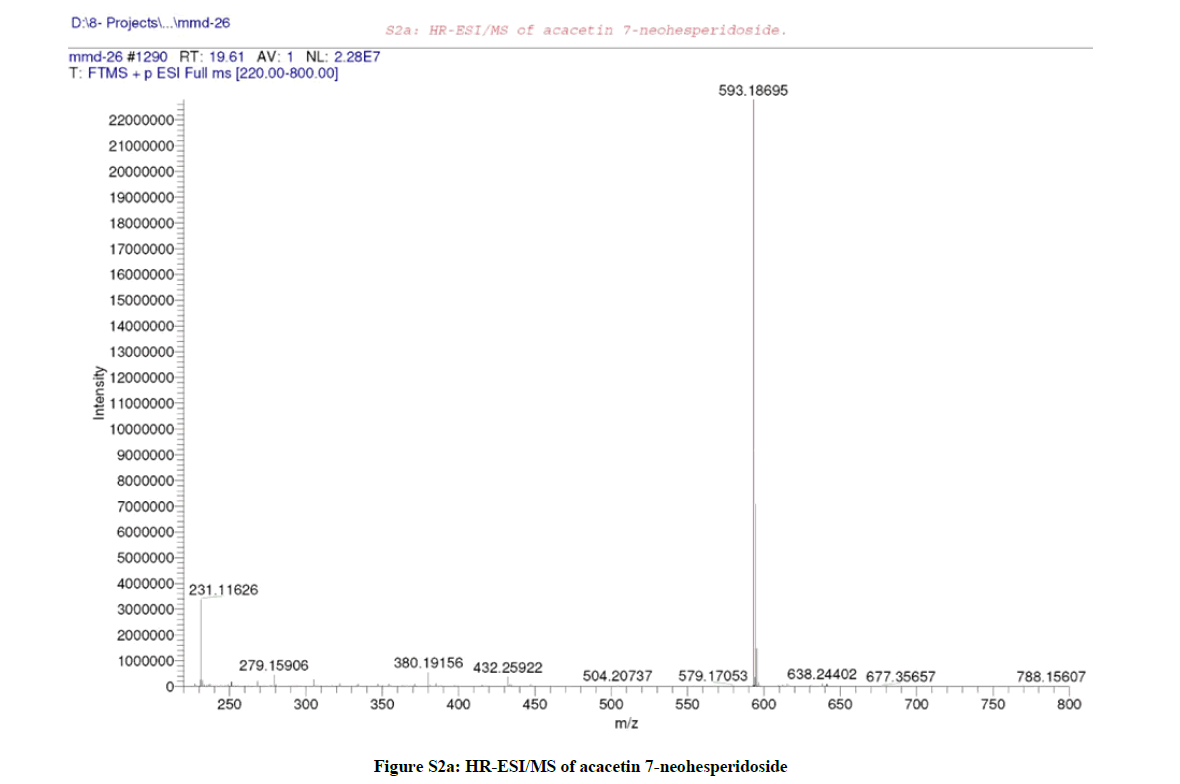
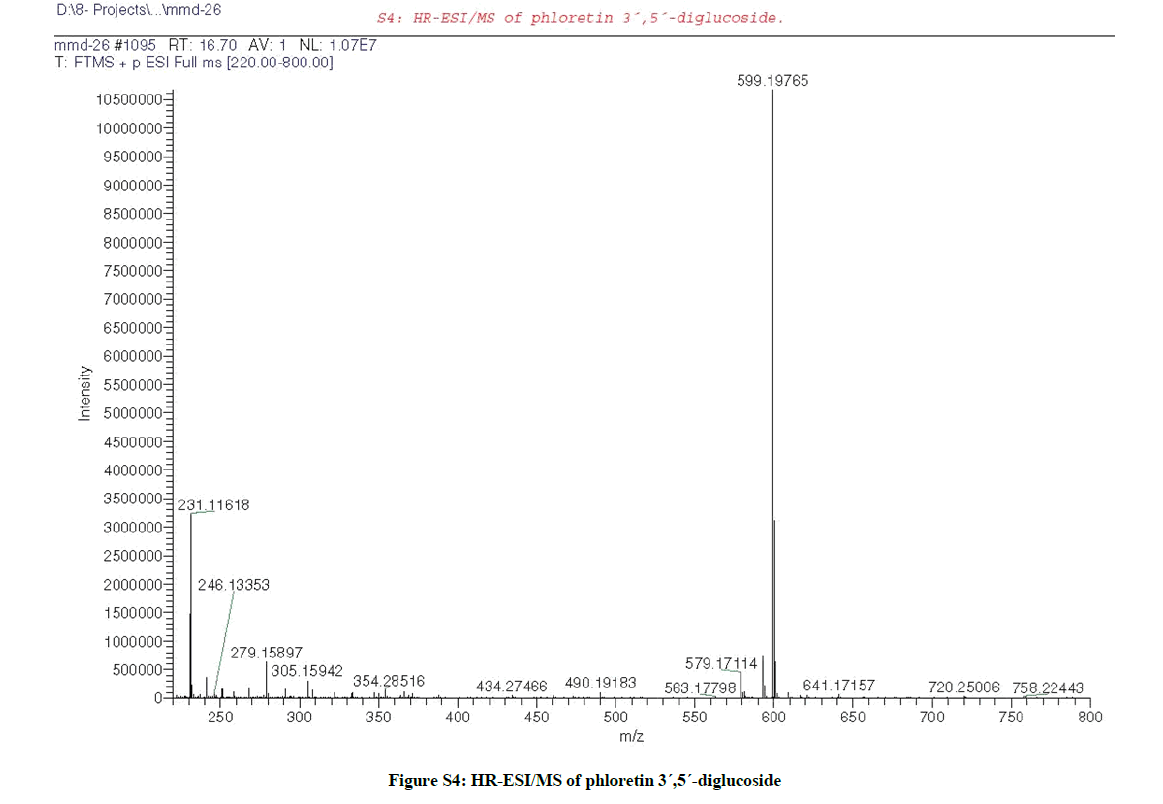
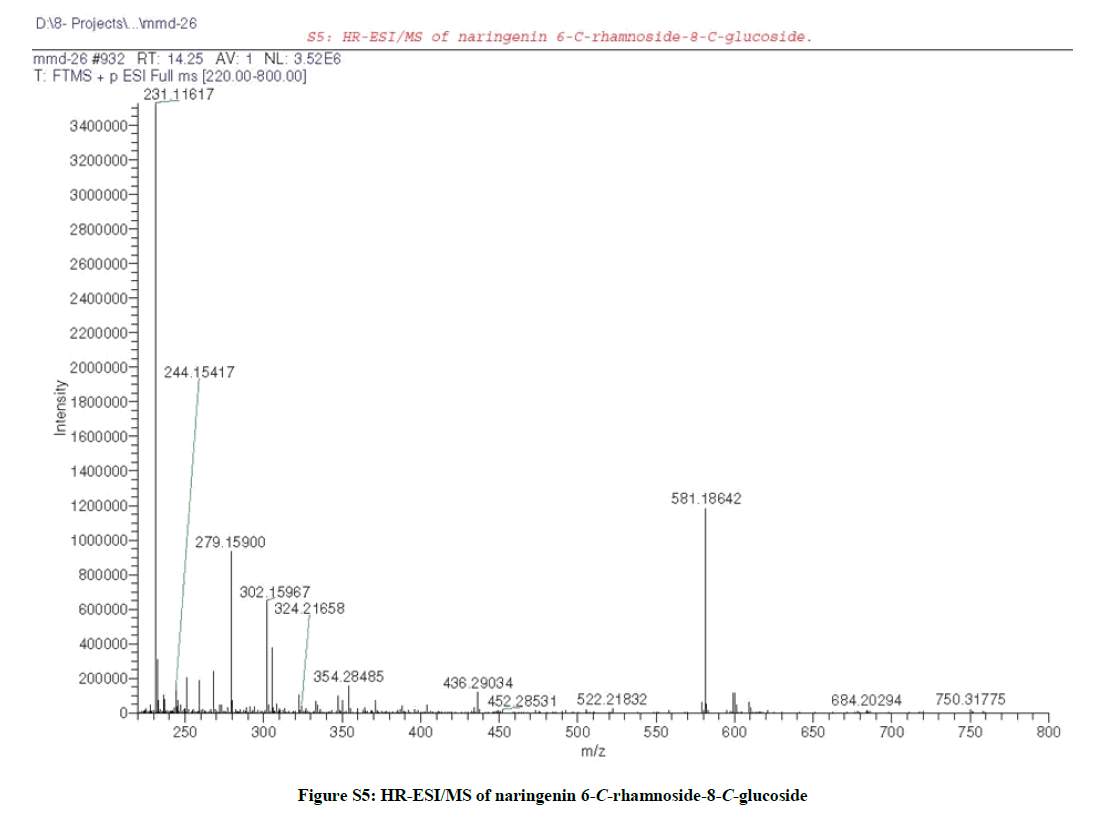
LC/MS analysis (S1) [12] of the leaves ME of F. margarita led to the identification without isolation of four flavonoids (Table S1); acacetin 7- O-neohesperidoside (Figures S2a,b) at Rt 19.61 with m/z 593.1869 corresponding to [M+H]+ (calcd 593.1870 for C28H33O14) [27], acacetin 7- rutinoside (Figures S3a,b) at Rt 20.93 with m/z 593.1869 corresponding to [M+H]+ (calcd 593.1870 for C28H33O14) [6], along with, phloretin 3´,5´-diglucoside (Figure S4) at Rt 16.70 with m/z 599.1976 corresponding to [M+H]+ (calcd 599.1976 for C27H35O15) [7], and naringenin 6-C-rhamnoside- 8-C-glucoside (Figure S5) at Rt 14.25 with m/z 581.1864 corresponding to [M+H]+ (calcd 581.1870 for C27H33O14) [6].
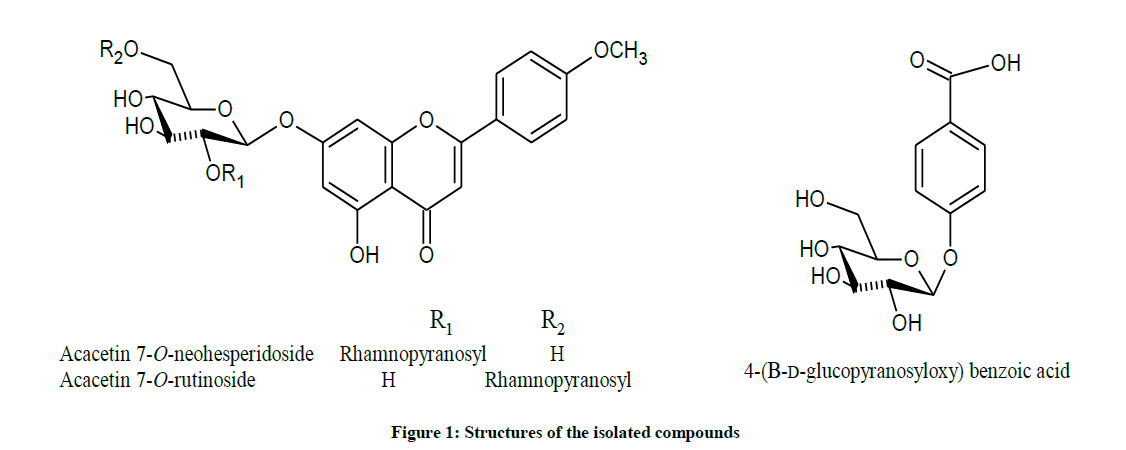
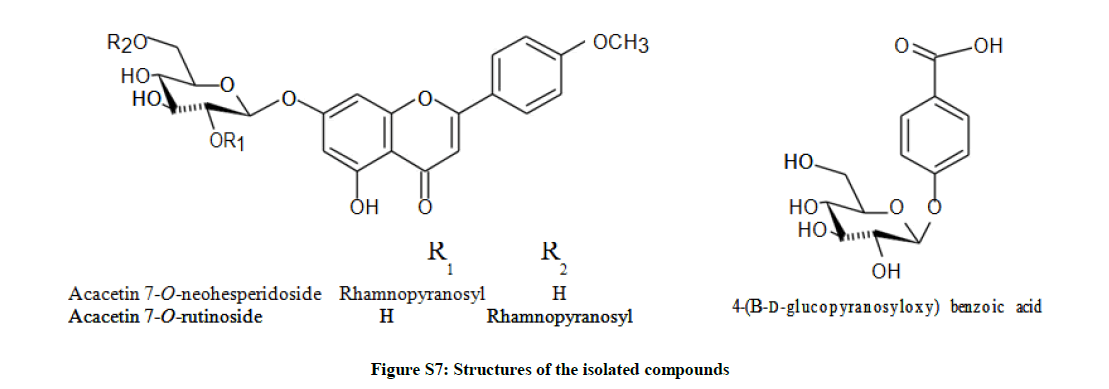
Extensive chromatographic purification resulted in the isolation of; acacetin 7-neohesperidoside (S2a-d), acacetin 7-rutinoside (Figures S3a-c) and 4-(β-D-glucopyranosyloxy) benzoic acid (Figures S6a-c) [28], and their structures were elucidated by One-dimensional Nuclear Magnetic Resonance(1D-NMR) and HR-ESI, and formulated as shown (Figures 1 and Table S1).
Hepatoprotective Assay
Against DMN-induced liver adenoma
DMN is a member of nitroso compounds, which is introduced in some food products and processed meat [29]. Moreover, it can be formed via nitrosation of amines present in food and drugs [30]. DMN is carcinogenic to spleen, lungs, liver and kidney [31] its metabolism led to the formation of methanol and formaldehyde, together with reactive oxygen species (ROS) i.e., superoxide anion (O2-), hydrogen peroxide (H2O2) and hydroxyl radicals (OH·) that damage liver cells, in addition, alkylating intermediates may also formed and react with nucleic acids and proteins leading to oxidative stress [32].
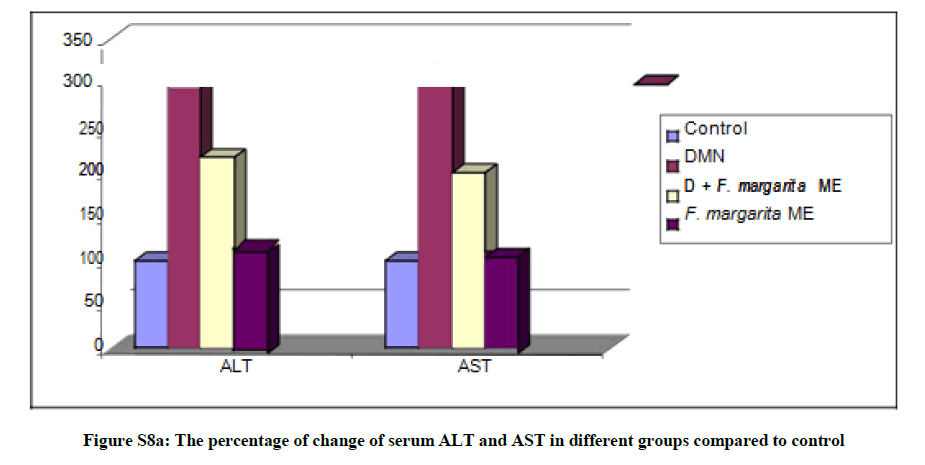
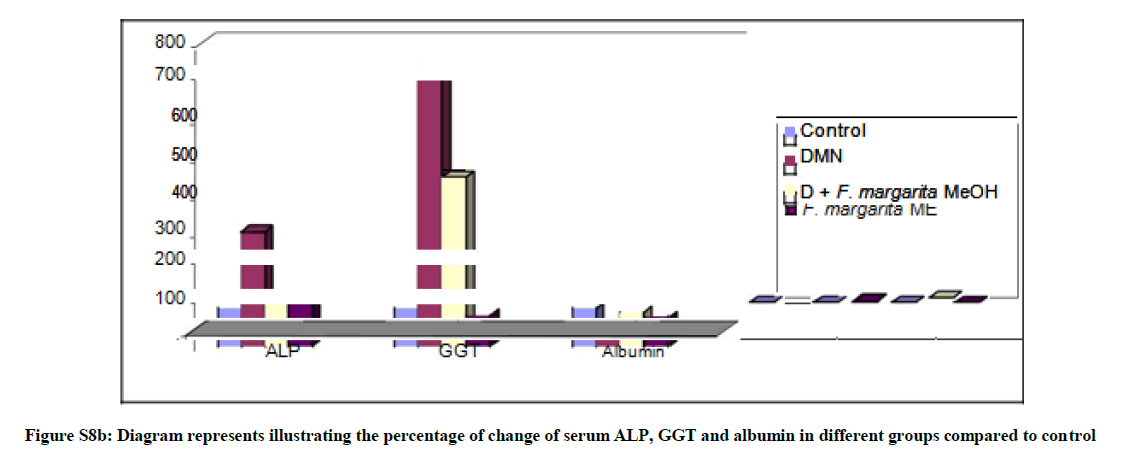
In the present study, the hepatocellular oxidative damage caused by DMN led to a marked elevation (Table S2and Figures S8a,b) in the levels of liver function enzymes; ALT, AST, γ-GT and ALP, with concomitant decrease in the level of serum albumin in DMN treated rats (G-2) compared to the control groups (G-1, P<0.0001). Our findings were compatible with several studies reported similar alterations of these markers during DMN administration [33-35].
Briefly, oral administration of the ME of F. margarita leaves (G-4) resulted in significant reduction of the serum ALT, AST, γ-GT and ALP. Moreover, its administration as prophylactic agent to rats treated with DMN (G-3) restored the level of serum albumin to its normal level (P<0.0001), these observations suggested that F. margarita ME tend to prevent liver damage by maintaining the integrity of the plasma membrane, thereby suppressing the leakage of enzymes through membranes, exhibiting hepatoprotective activity. This might be the reason for the restoration in the activities of the marker enzymes during administration of these extracts.
These biochemical findings were supported by histopathological observations (Figures 2-5), which accounted for the potent hepatoprotective action of the ME of F. margarita leaves.
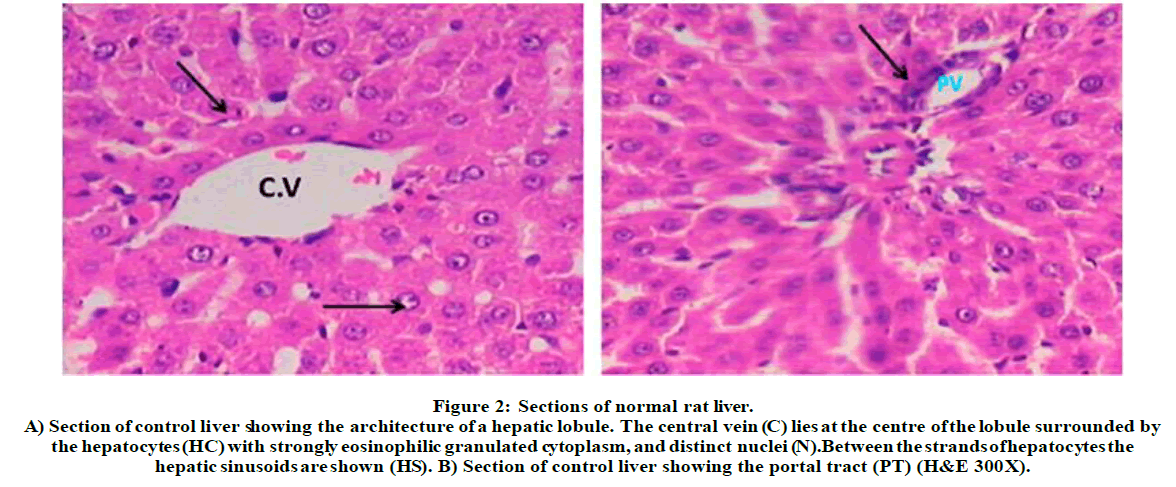
Figure 2: Sections of normal rat liver.
A) Section of control liver showing the architecture of a hepatic lobule. The central vein (C) lies at the centre of the lobule surrounded by the hepatocytes (HC) with strongly eosinophilic granulated cytoplasm, and distinct nuclei (N). Between the strands of hepatocytes the hepatic sinusoids are shown (HS). B) Section of control liver showing the portal tract (PT) (H&E 300X).
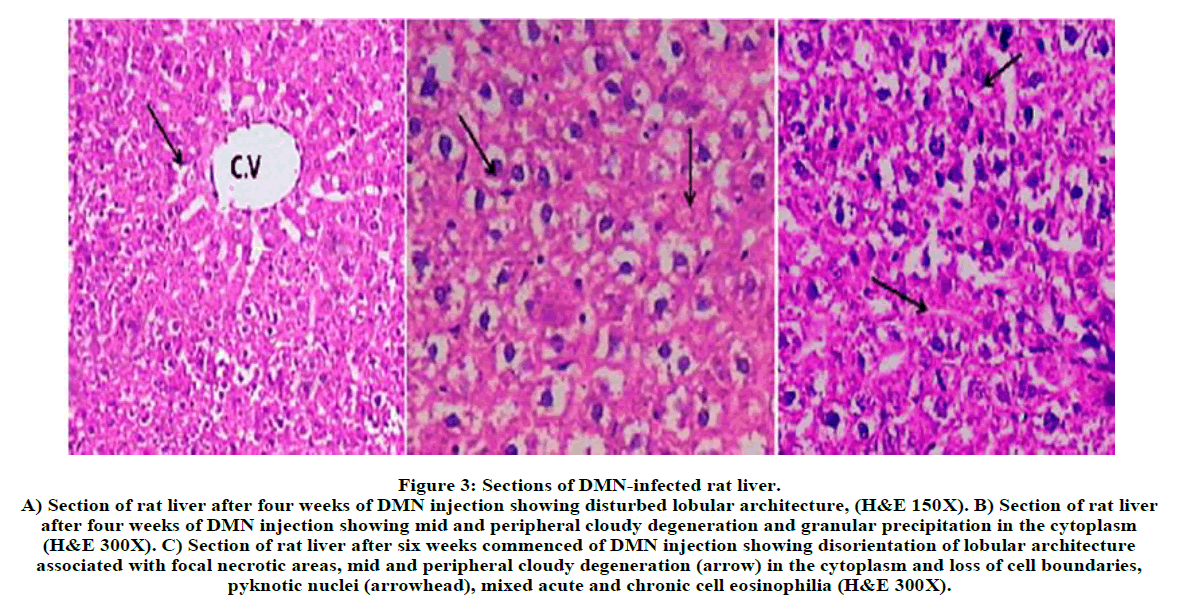
Figure 3: Sections of DMN-infected rat liver.
A) Section of rat liver after four weeks of DMN injection showing disturbed lobular architecture, (H&E 150X). B) Section of rat liver after four weeks of DMN injection showing mid and peripheral cloudy degeneration and granular precipitation in the cytoplasm (H&E 300X). C) Section of rat liver after six weeks commenced of DMN injection showing disorientation of lobular architecture associated with focal necrotic areas, mid and peripheral cloudy degeneration (arrow) in the cytoplasm and loss of cell boundaries, pyknotic nuclei (arrowhead), mixed acute and chronic cell eosinophilia (H&E 300X).
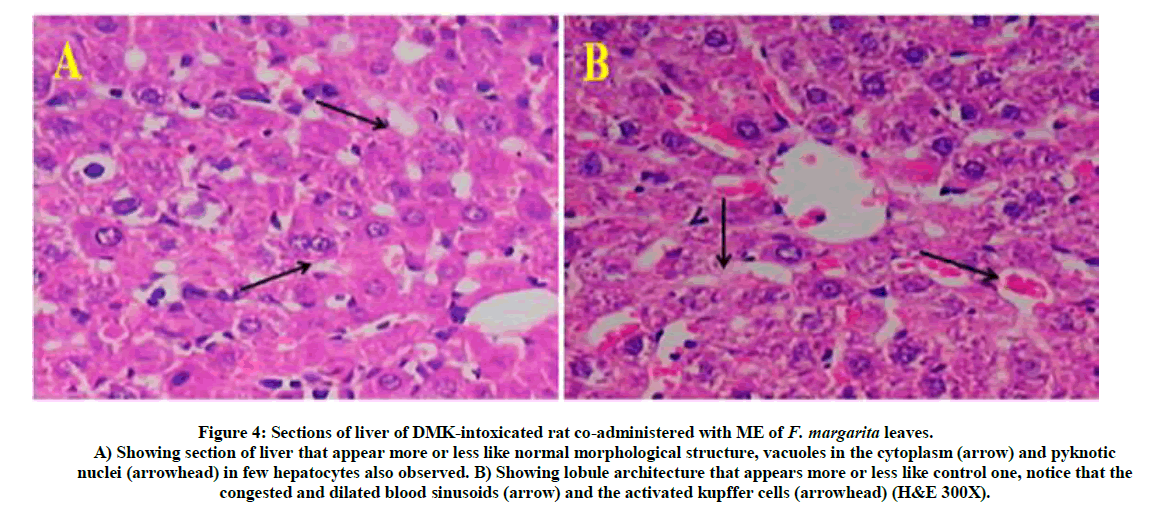
Figure 4: Sections of liver of DMK-intoxicated rat co-administered with ME of F. margarita leaves.
A) Showing section of liver that appear more or less like normal morphological structure, vacuoles in the cytoplasm (arrow) and pyknotic nuclei (arrowhead) in few hepatocytes also observed. B) Showing lobule architecture that appears more or less like control one, notice that the congested and dilated blood sinusoids (arrow) and the activated kupffer cells (arrowhead) (H&E 300X).
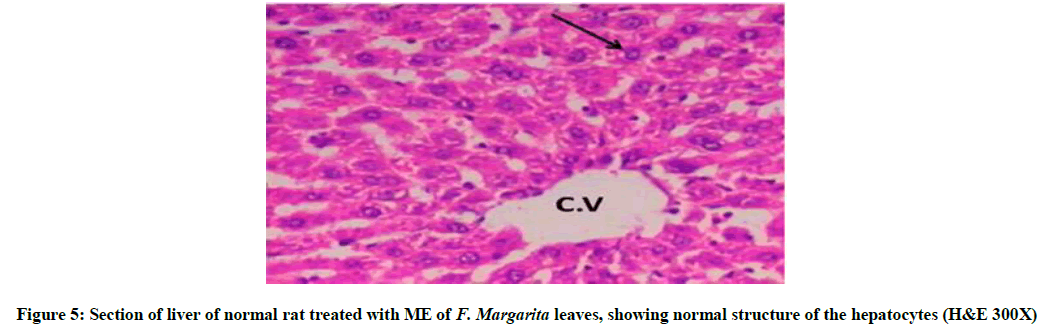
Briefly, Figure 5, represent liver section of normal rat treated with ME of F. margarita leaves and revealed normal structure of the hepatocytes. Histopathological patterns appeared in (Figure 3) revealed liver-injury of rats treated with DMN for four weeks, indicating disturbed lobular architecture, mid and peripheral cloudy degeneration and granular precipitation in the cytoplasm. Furthermore, six weeks of DMN injection showed disorientation of lobular architecture associated with focal necrotic areas, mid and peripheral cloudy degeneration in the cytoplasm and loss of cell boundaries, pyknotic nuclei, mixed acute and chronic cell eosinophilia. However, Figure 4, represent liver-injury of DMN-intoxicated rat co-administered with ME of F. margarita leaves, showing more or less like normal morphological structure, vacuoles in the cytoplasm and pyknotic nuclei in few hepatocytes also observed, in addition, lobule architecture appears more or less like control one (Figure 2), notice that the congested and dilated blood sinusoids and the activated kupffer cells.
Against CCl4-induced liver toxicity
Carbon tetrachloride (CCl4) a well-established potent hepatotoxic agent, which is capable of induce cirrhosis and hepatic carcinoma selectivity. It is bio-changed into trichloromethyl (•CCl3) free radical via cytochrome (P-450), then attached to cellular proteins to form trichloromethyl peroxyl (•OOCCl3) free radical in presence of oxygen [36], which is in turn, destroys Ca2+ homeostasis, peroxidises lipids, and finally damage liver [37-39]. Moreover, the generated reactive oxygen species (ROS) can affect the antioxidant defense mechanisms [40]. Uncounted reports illustrated the harmful effects upon CCl4-induced toxicity and the resulted elevation in liver enzymes.
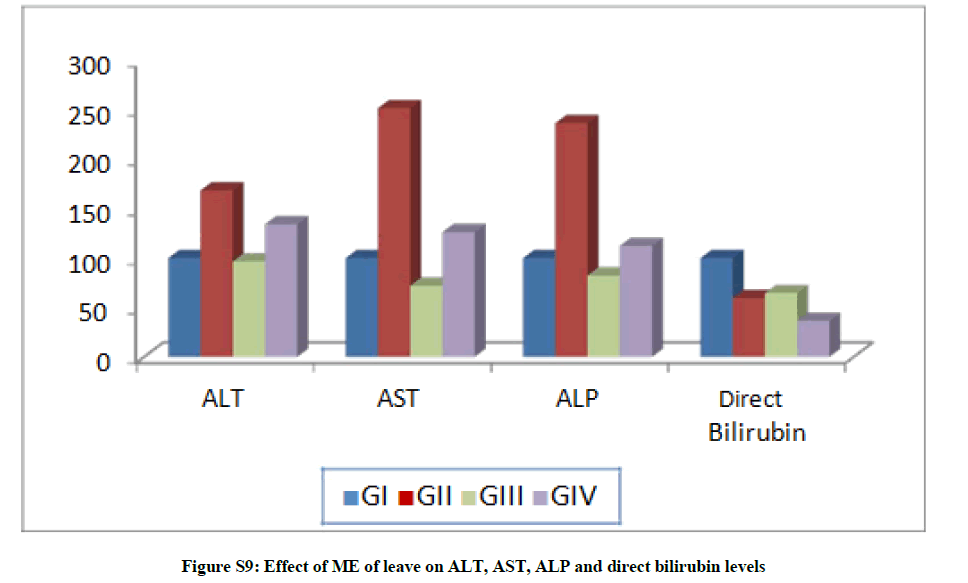
Treatment with ME of F. margarita leaves led to significant increases in the ALT level for G-II and G-IV by 68.52 and 34.21%, respectively, while it decreased by 3.33% in G-III. Moreover, AST level was increased significantly by 151.9 and 26.29% for G-II and G-IV, and decreased by 27.98% for G-III. Furthermore, ALP level increased by 136.78% for G-II and decreased by 17.63% for G-III. In addition, the level of direct bilirubin recorded decrease in all treated groups (Table S3 and Figure S7).
Liver weight observations revealed decreases by 48 and 46.13% for G-II and G-IV, respectively. Moreover, increases in body weight were recorded in treated groups G-II and G-IV by 32.91 and 27.59%, respectively. Liver ratio showed significant decreases in treated groups G-II and G-IV by 61.12 and 58.32%, respectively, (Table S4 and Figures S8a,8b).
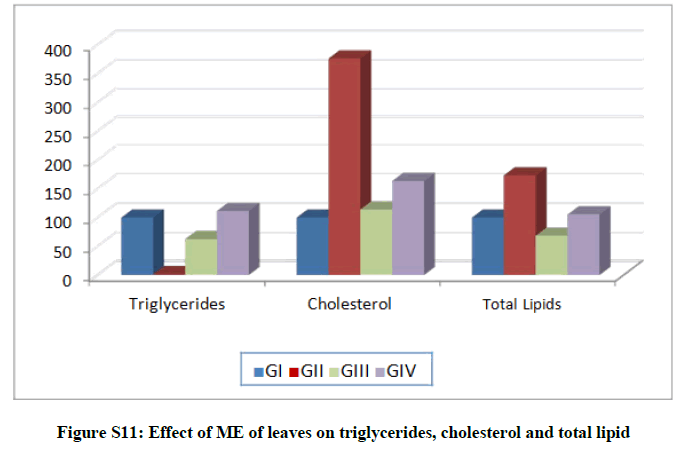
Triglycerides level recorded increases in both treated groups G-II and G-IV by 56.22 and 11.50% respectively, and decreased in G-III by 37.47%. In addition, significant increases were recorded in cholesterol levels in treated groups (GII, G-III, and G-IV) by 276.16, 13.98 and 63.59%, respectively. Finally, total lipids increased in case of G-II and G-IV by 73.3 and 5.37%, respectively, and decreased for G-III by 31.66% (Table S5 and Figure S11).
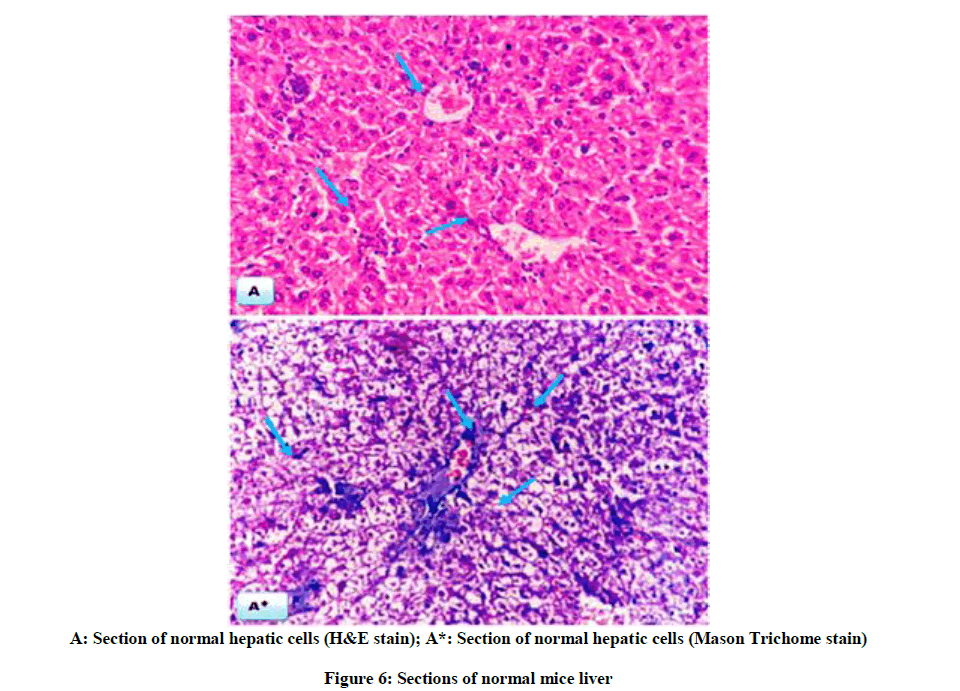
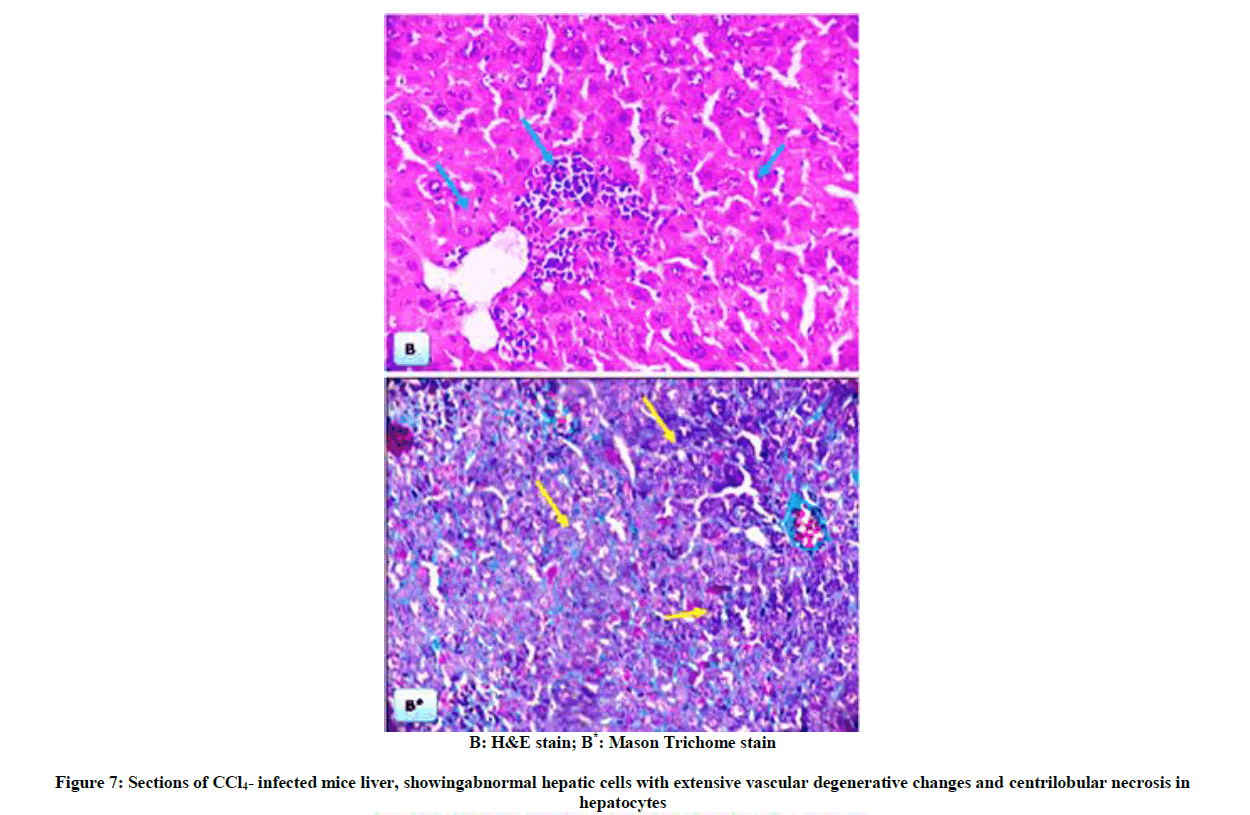
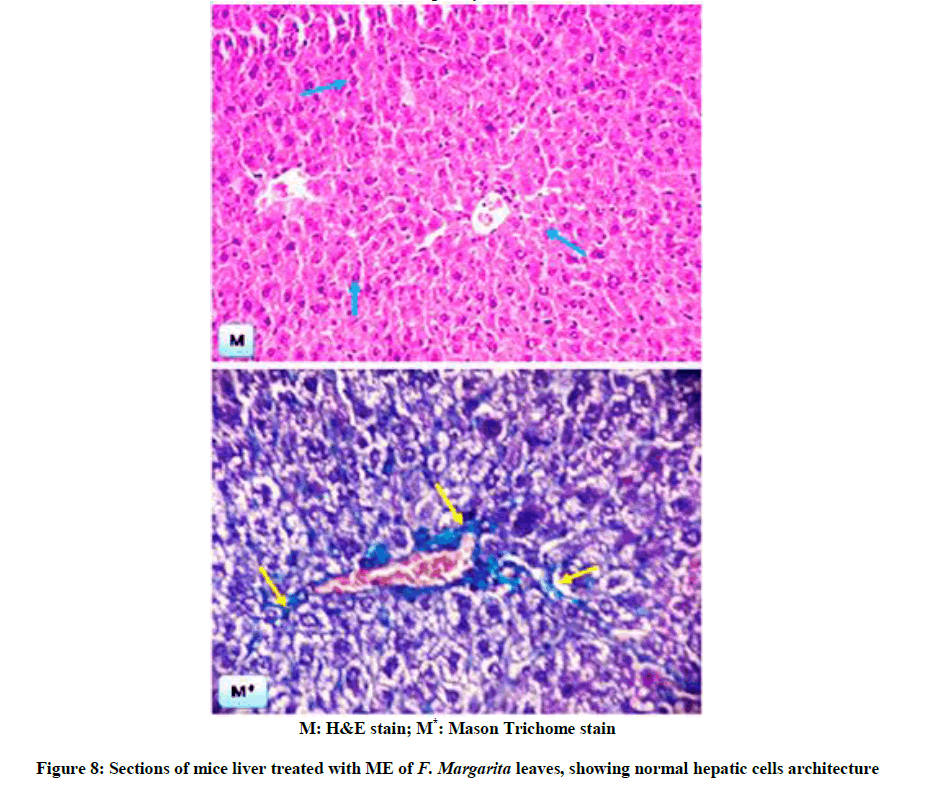
The above biochemical records were assured by histological architectures (Figures 6-9), briefly, Figure 6, and represent liver sections of control mice, with normal histological structure of hepatic lobules and central vein (CV). Histological patterns appeared in Figure 7 represent liver sections for mice administered with CCl4 for 7 weeks, and revealed extensive fibrosis in the periportal areas, massive vascular degeneration and the dilated blood sinusoids are filled with red cells (arrow). Moreover, Figure 8, no changes in hepatic cells architecture were recorded for mice treated with ME, which accounted for its safety. In addition, co-administration of ME to CCl4-intoxicated mice showing spontaneous regression of fibrosis in the periportal zone and hepatic cells closed to the control (Figure 9).
In our study, a significant restoration was observed for the liver enzymes upon administration of ME of F. margarita leaves, the elevated serum levels of total lipids, cholesterol and triglycerides upon CCl4-toxicity were significantly restored to normal on administration of ME of F. margarita leaves.
The resulted hepatoprotective action induced by ME of F. margarita leaves against DMN-induced liver adenoma and/or against CCl4-induced liver toxicity, can be accounted mainly on its phenolic content ´the isolated compounds 1-3.
Numerous reports confirmed the potential effect of phenolics as hepatoprotective agents, and its correlation with their antioxidant activity that able to scavenge the ROS formed from DMN and/or CCl4 metabolism [41,42].
Sun et al. [43] confirmed the synergistic antioxidant potential of the isolated phenolics from F. margarita leaves. Moreover, Barreca et al. [44] studied the antioxidant activity of F. japonica fruit juice, and attributed this activity to the isolated flavonoids i.e., apigenin and acacetin glycosides. Furthermore, Salama et al. [45] reported the hepatoprotective action of acacetin glycoside isolated from Onopordum alexandrium. In addition, Cho et al. [46] reported that acacetin suppresses TLR4 signaling and promotes autophagic flux for GalN/LPS-induced liver injury. Furthermore, Guimarães et al. [47] stated that plant phenolics synergistically are responsible for the antihyperlipidemic effect, restoring enzymatic activities, and ameliorating the toxic effects induced on liver, and Mariod et al. [48] confirmed these effects with the antioxidant potential of phenolics.
Acknowledgement
The authors are grateful for the financial support by National Research Centre, Cairo, Egypt.
References
- A.E. Khaleel, S.H. El-Gayed, J. Nolte, R. Fobbe, Al-Azhar J. Pharmaceut. Sci., 2001, 28, 258-266.
- C.C. Lin, P.F. Hung, S.C. Ho, Food Chem., 2008, 109, 95-103.
- H.D. Neuwinger, African traditional medicine: a dictionary of plant use and applications, Stuttgart, Germany, Medpharm Scientific Publisher, 2000.
- N.A. Ibrahim, S.S. El-Hawary, M.M.D. Mohammed, M.A. Farid, N.A.M. Abdel Wahed, M.A. Ali, E.A.W. El-Abd, J. Appl. Pharmaceut. Sci., 2015, 5, 6-12.
- D.E. Breithaupt, A. Bamedi, J. Agric. Food Chem. 2001, 49, 2064-2070.
- E.S. Sadek, P. Dimitris, D.P. Makris, Plant Foods for Hum. Nutr., 2009, 64, 297-302.
- K. Ogawa, A. Kawasaki, M. Omura, T. Yoshida, Y. Ikoma, M. Yano, Phytochem. 2001, 57, 737-742.
- J. Chen, A.M. Montanari, W.W. Widmer, J. Agric. Food Chem.,1997, 45, 364-368.
- C. Kandaswami, E. Perkin, D.S. Soloniuk, G. Drzewiecki, E.Jr. Middleton, Cancer Lett.,1991, 56, 147-152.
- Y. Nogata, K. IchinYoza, K. IchiKusmoto, N. Kohyama, K. Sekiya, H. Ohta, J. Agric. Food Chem. 1996, 44, 725-729.
- J.A. Olson, J. Nutr., 1996, 126, 1208-1212.
- M.M.D. Mohammed, M. Spiteller, N.A. Ibrahim, K.M. Mohamed, Rec. Nat. Prod., 2016, 10, 385-391.
- M.M.D. Mohammed, L.P. Christensen, P.L. Colla, Nat. Prod. Res., 2014, 28, 221-229.
- J. George, Clin. Chim. Acta, 2003, 335, 39-47.
- J.P. Persijn, W. van der Slik, J. Clin. Chem. Clin. Biochem., 1976, 14, 421-427.
- G. Szasz, Clin. Chem., 1969, 15, 124-136.
- D.S. Young, Annals Clin. Biochem., 1997, 34, 579-581.
- H.V. Bergmeyer, M. Horder, R. Rej, J. Clin. Chem. Clin. Biochem., 1985, 24, 497-503.
- S. Rietman, S. Frankle, Am. J. Clin. Pathol., 1957, 28, 56-63.
- J.A. Demetriou, D.A. Drewes, J.B. Gin, Enzymes in Clinical Chemistry, Principles and Technics 2nd (Edn.), R.J.Henry, D.C. Cannon, J.W. Winkelman (Edi.), New York: Harper & Row Hagerstown, 1974.
- A.L. Babson, S.J. Greely, C.M. Coleman, G.E. Philips, Clin. Chem., 1966, 12, 482-490.
- B.T. Dumas, W.A. Watson, H.G. Biggs, Clin. Chim. Acta, 1971, 31, 87-96.
- F.L. Rodkey, Clin. Chem., 1965, 11, 478-487.
- P. Roeschlau, E. Bernt, W.J. Gruber, Clin. Chem. Biochem.,1974, 12, 403-407.
- P. Fossati, L. Prencipe, Clin. Chem.,1982, 28, 2077-2080.
- C. Hirsch, C.S. Zouain, J.B. Alves, A.M. Goes, Parasitol.,1997, 115, 21-28.
- S.M. Khafagy, N. Sabri, N.A. El-Salam, A. El-Din, Planta Med., 1979, 35, 186-187.
- U. Dirks, K. Herrmann, Phytochem., 1984, 23, 1811-1812.
- M.M.D. Mohammed, Nat. Prod. Chem. Res., 2016, 4, 228.
- H.M. Schuller, Life Sci., 2007, 80, 2274-2280.
- T. Suzuki, T. Itoh, M. Hayashi, Y. Nishikawa, S. Ikezaki, F. Furukawa, M. Takahashi, T. Sofuni, Environ. Mol. Mutagen., 1996, 28, 348-353.
- K. Pradeep, C.V. Mohan, K. Gobianand, S. Karthikeyan, Chem. Biol. Inter., 2007, 167, 12-18.
- H.S. Lee, K.H. Jung, I.S. Park, S.W. Kwon, D.H. Lee, S.S. Hong, Digest. Dis. Sci., 2009, 54, 782-788.
- E.O. Farombi, S. Shrotriya, H.K. Na, S.H. Kim, Y.J. Surh, Food Chem. Toxicol., 2008, 46, 1279-1287.
- E.O. Farombi, S Shrotriya, Y.J. Surh, Life Sci., 2009, 84, 149-155.
- K.G. Nevin, L. Vijayamma, Toxicol. Pharmacol., 2005,20, 471-477.
- H. De Groot, T. Noll, Biochem. Pharmacol.,1986, 35, 15-19.
- G.A. Clawson, Pathol. Immunopathol. Res.,1989, 8, 104-112.
- S. Azri, H.P. Mata, L.L. Reid, A.J. Gandolfi, K. Brendel, Toxicol. Appl. Pharmacol., 1992, 112, 81-86.
- M.M.D. Mohammed, N.A. Ibrahim, S.A. Ali, M.A. Hamed, N.S. El-Rigal, J. Food Biochem., 2018, 42.
- C. Nichita, G. Neagu, V. Vulturescu, R. Albulescu, V. Vulturescu, N. Badea, M. Giurginca, A. Meghea, Mol. Cryst. Liq. Cryst., 2010, 523, 228-235.
- N.A. Ibrahim, H.R. El-Seedi, M.M.D. Mohammed, Nat. Prod. Res., 2007, 21, 857-866.
- J. Sun, Y. Huang, G. Sun, X. Sun, M. Qin, D. Zhao, 2012, 37, 1958-1962.
- D. Barreca, E. Bellocco, C. Caristi, U. Leuzzi, G. Gattuso, ACS Symposium Series, 2013, 1129, 103-119.
- M.M. Salama, S.M. Ezzat, A.A. Sleem, J. Natur. Sci. C., 2011, 66, 251-259.
- H.I. Cho, J.H. Park, H.S. Choi, J.H. Kwak, D.U. Lee, S.K. Lee, S.M. Lee, J. Nat. Prod., 2014, 77, 2497-2503.
- P.R. Guimarães, A.M.P. Galvão, C.M. Batista, G.S. Azevedo, R.D. Oliveira, R.P. Lamounier, N. Freire, A.M.D. Barros, E. Sakurai, J.P. Oliveira, E.C. Vieira, J.I. Alvarez-Leite, Brazilian J. Med. Biol. Res., 2000, 33, 1027-1036.
- A.A. Mariod, B. Matthaus, I.H. Hussein, Inter. J. Food Sci. Technol., 2008, 43, 921-926.
Supplementary Material
Complete structure elucidation of the isolated compounds (1-3)
Compound 1
It was isolated as white amorphous powder (10 mg), with molecular formula C28H32O14 (+ve) as elucidated by HRESIMS (Figure S2a), which recorded a quasi-molecular ion peak appeared at m/z 593.1869 corresponding to [M+H]+ (calcd 593.1870 for C28H33O14).
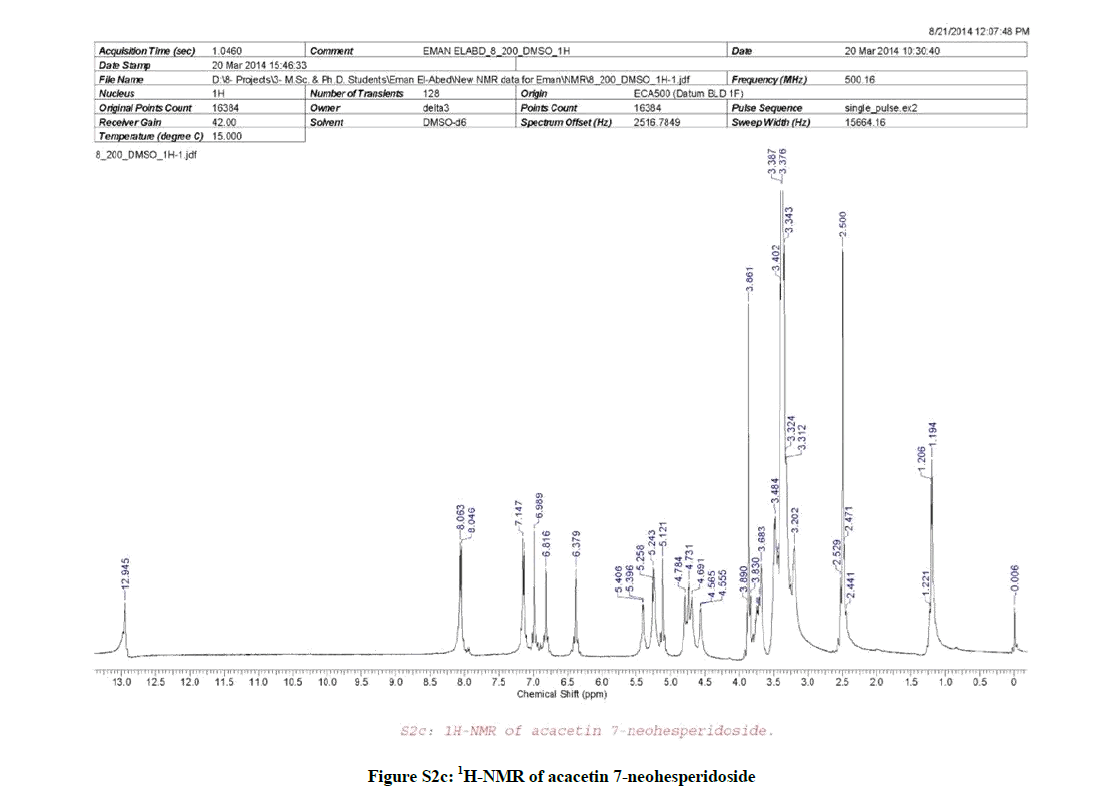
1H-NMR (Figure S2c, DMSO-d6) spectrum revealed the presence of a sharp singlet signal at δH 6.99 (1H, s, H-3) characteristic for the flavone type, two sets of doublet signals appeared at δH 8.05 (2H, d, J=8.5 Hz, H-2´ & 6´) and at δH7.14 (2H, d, J= 8.5 Hz, H-3´ & 5´) characteristic for the AA´BB´ system (ring-B) of flavonoids. Moreover, two sets of broad singlet signals appeared at δH 6.82 (1H, brs, H-8) and δH 6.38 (1H, brs, H-6), a sharp singlet signal appeared at δH 3.86 (3H, s) characteristic for a phenolic methoxyl group was observed. Furthermore, two anomeric proton signals appeared at δH 5.40 (1H, d, J= 5 Hz, H-1´´) characteristic for a β-sugar moiety, which confirmed to be glucose from the 13C-NMR spectrum (Figure S2d), and at δH 5.12 (1H, brs, H-1´´´) characteristic for rhamnose sugar moiety and confirmed by the doublet methyl signal appeared at δH 1.20 (3H, d, J= 6 Hz, rhamnose H3C-6´´´), a down field shift signal appeared at δH 4.73 (1H, brm, H-2´´) suggested a (1→2) interlinkage, multiplet signals appeared at δH 3.20-3.89 (9H, m, H-3´´, 4´´, 5´´, 6´´, 2´´´, 3´´´, 4´´´, 5´´´), and finally a broad singlet signal appeared at δH 12.95 (1H, brs) characteristic for the 5-OH of the flavonoid.
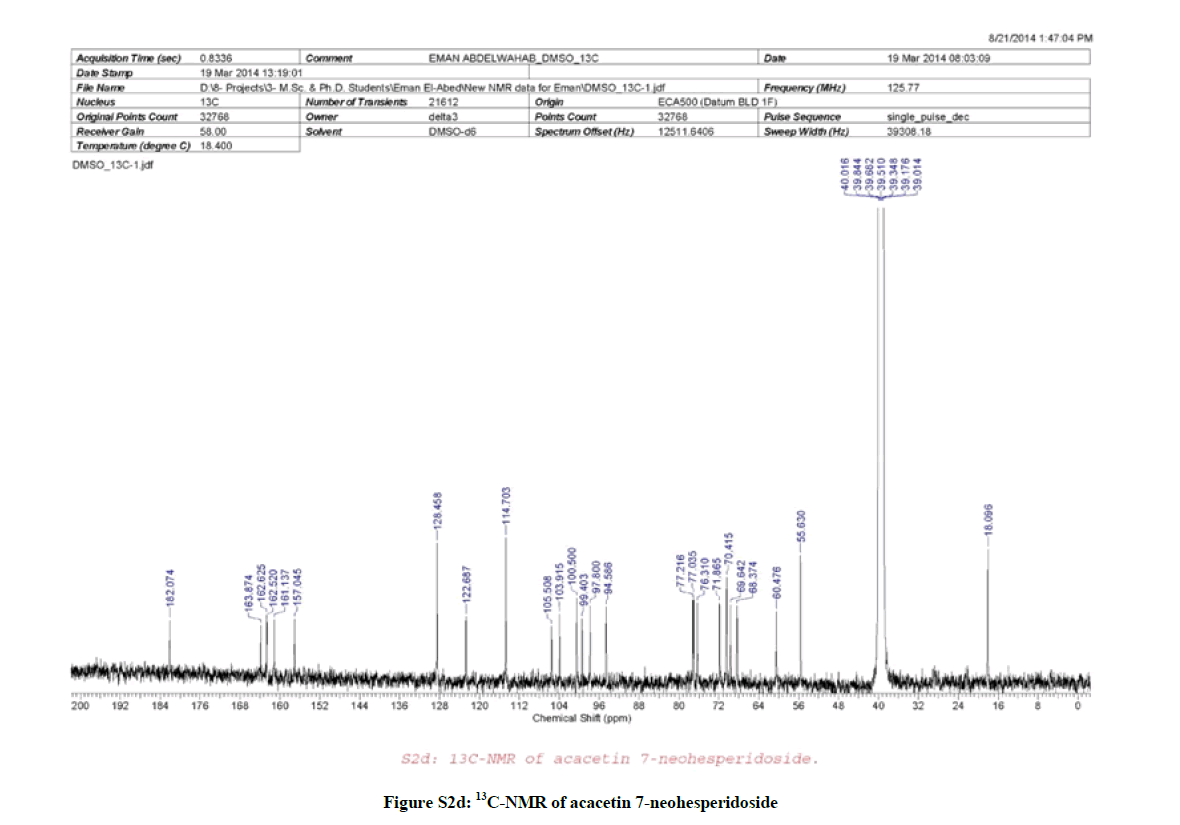
13C-NMR (Figure S2d, DMSO-d6) spectrum; δC 163.87 (C-2), 103.92 (C-3), 182.07 (C-4), 162.52 (C-5), 99.40 (C-6), 162.63 (C-7), 94.59 (C-8), 157.05 (C-9), 105.51 (C-10), 122.69 (C-1´), 128.46 (C-2´), 114.70 (C-3´), 161.14 (C-4´), 114.70 (C-5´), 128.46 (C-6´), 100.50 (C-1´´), 77.04 (C-2´´), 76.31 (C-3´´), 70.48 (C-4´´), 77.22 (C-5´´), 60.47 (C-6´´), 97.80 (C-1´´´), 70.42 (C-2´´´), 69.64 (C-3´´´), 71.87 (C-4´´´), 68.37 (C-5´´´), 18.10 (C-6´´´), 55.63 (OCH3).
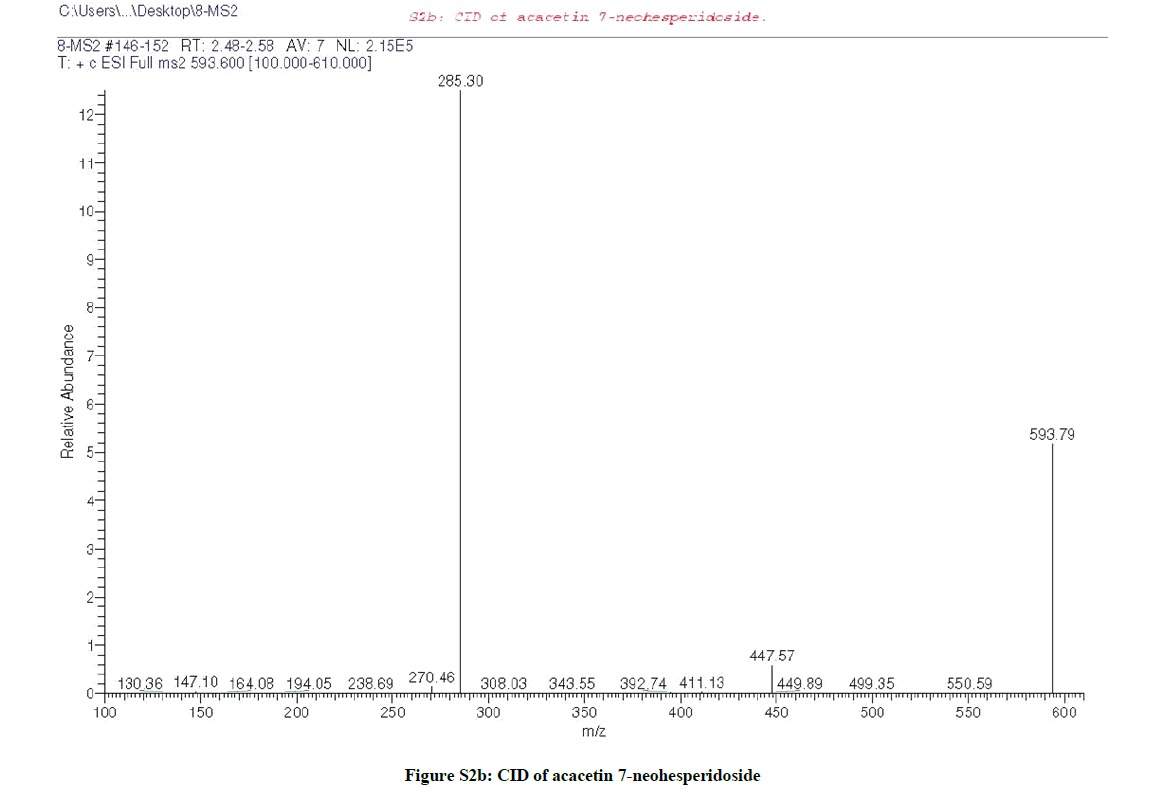
LC/MS analysis (Figures S1 and S2a,b) suggested that compound (1) may be acacetin 7-O-neohesperidoside or acacetin 7-O-rutinoside, however, the down field shift of the broad signal at δH 4.73 (1H, brm, H-2´´) and at δC 77.04 (C-2´´) confirmed the sugar’s interlinkage to be (1→2) (neohesperidoside), further ESI-MS/MS (Figure S2b) confirmed the neohesperidoside form.
Hence, and according to the above mentioned data and by comparing with literature, compound (1) was established as formulated in Figure (S7) and assigned to acacetin 7-O-neohesperidoside [1].
Compound 2
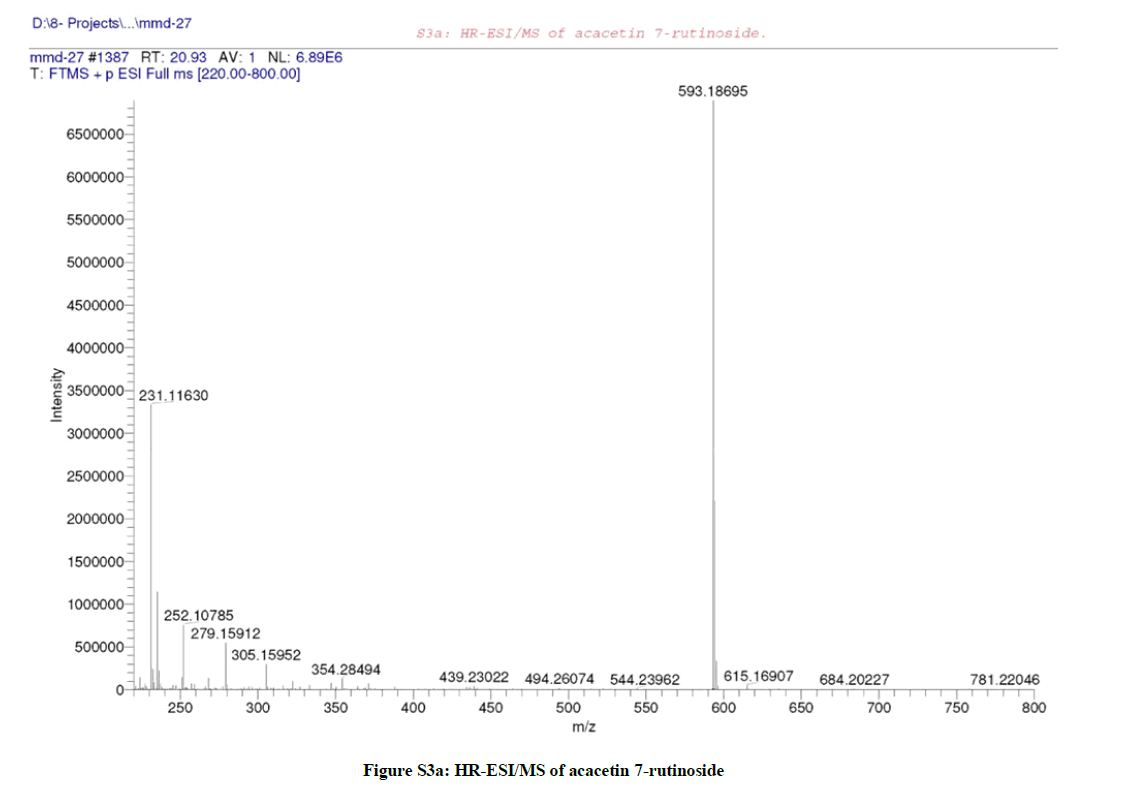
It was isolated as white amorphous powder (22 mg), with molecular formula C28H32O14 (+ve) as elucidated by HRESIMS (Figure S3a), which recorded a quasi-molecular ion peak appeared at m/z 593.1869 corresponding to [M+H]+ (calcd 593.1870 for C28H33O14).
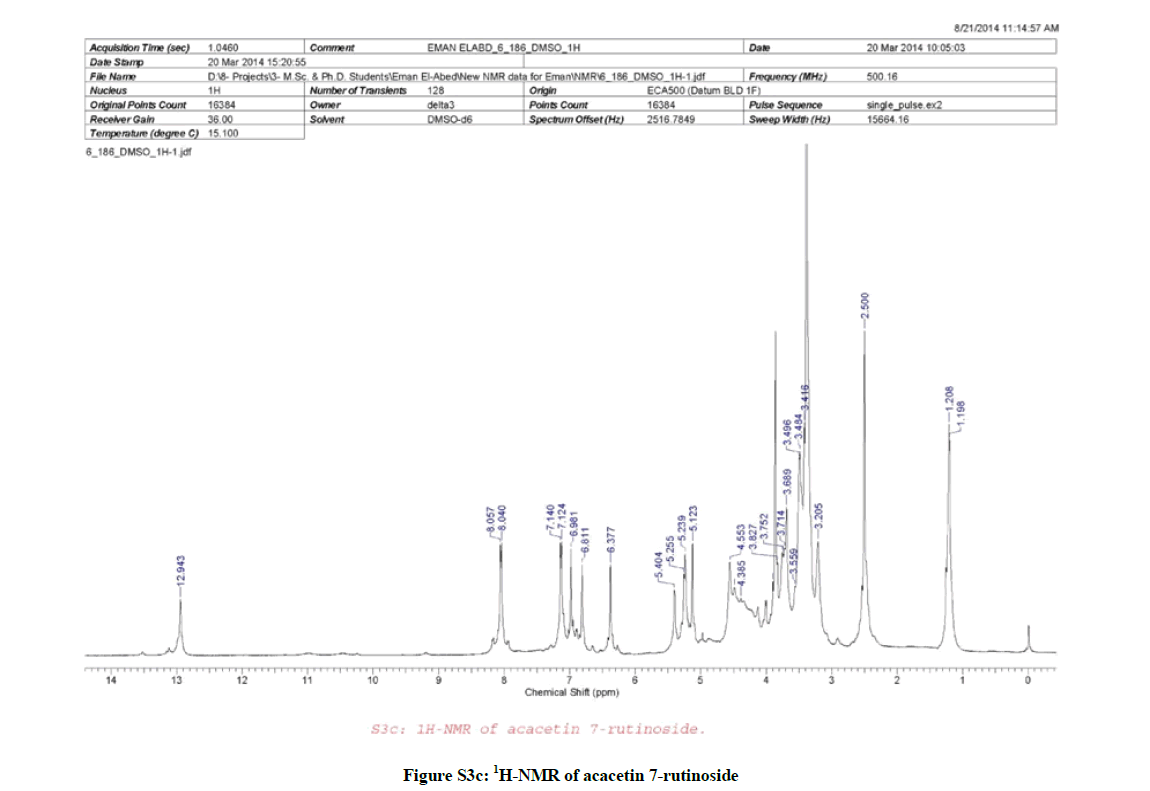
1H-NMR (Figure S3c, DMSO-d6) spectrum of compound (2) was similar to that of compound (1) with a slight differences, which can be summarizes as follow, a sharp singlet signal at δH 6.98 (1H, s, H-3) characteristic for the flavone type, two sets of doublet signals appeared at δH 8.04 (2H, d, J=8.5 Hz, H-2´ & 6´) and at δH 7.13 (2H, d, J=8.5 Hz, H-3´ & 5´) characteristic for the AA´BB´ system, in addition, two sets of broad singlet signals appeared at δH 6.81 (1H, brs, H-8) and δH 6.38 (1H, brs, H-6), a sharp phenolic methoxyl appeared at δH 3.86 (3H, s). Furthermore, two anomeric proton signals appeared at δH 5.39 (1H, d, J=5 Hz, H-1´´) characteristic for a β-sugar moiety, and at δH 5.12 (1H, brs, H-1´´´) characteristic for rhamnose sugar as confirmed by the presence of a doublet methyl signal appeared at δH 1.19 (3H, d, J=5 Hz, rhamnose H3C-6´´´), at δH 3.20-4.38 (10H, m, H-2´´, H-3´´, 4´´, 5´´, 6´´, 2´´´, 3´´´, 4´´´, 5´´´) for the rest sugar protons, and finally a broad singlet signal appeared at δH 12.95 (1H, brs) characteristic for the 5-OH of the flavonoid.
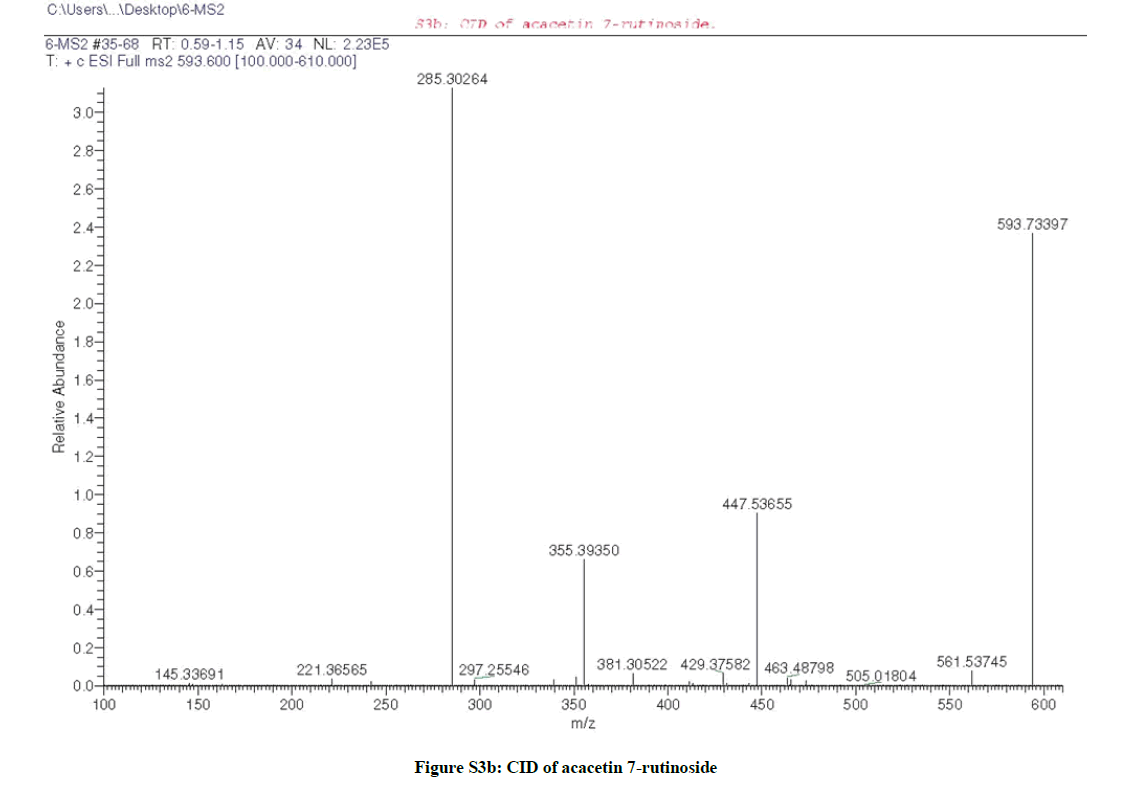
Due to the similarity between the 1H-NMR spectra (Figures S2c and S3c) of both compounds (1 & 2), it was suggested that compound (2) is the isomeric form of compound (1), which was confirmed by the same molecular ion mass with different retention time as appeared in the total ion chromatogram of the LC/MS (Figures S2a and S3a) of both compounds. Furthermore, the CID/ESI-MS/MS fragmentation (Figure S3b) of compound (2) revealed the difference between the two compounds (1 & 2) as the mass fragmentations corresponding to acacetin 7-O-rutnioside [2].
These findings unambiguously established the structure of compound (2) as formulated in Figure (S7) and assigned to acacetin 7-O-rutinoside [2].
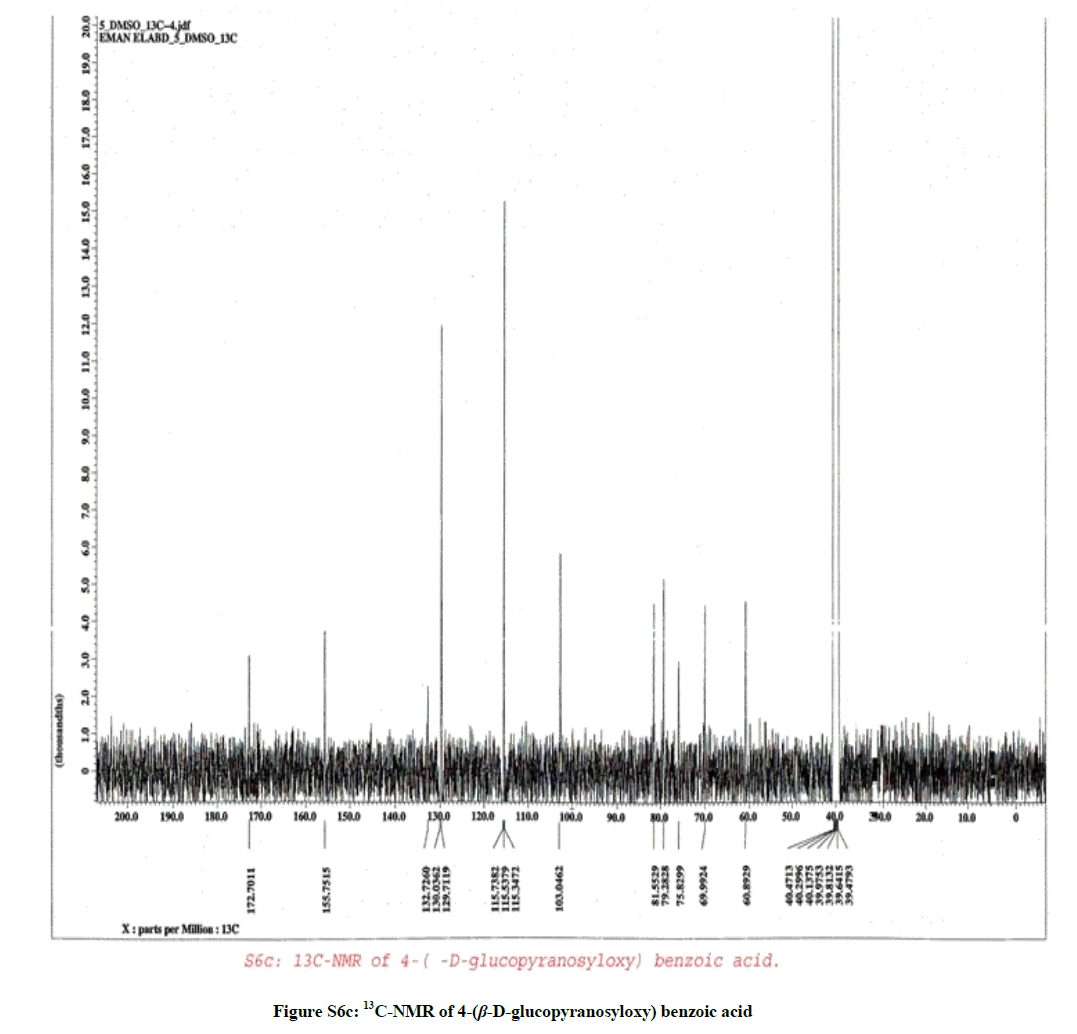
Compound 3
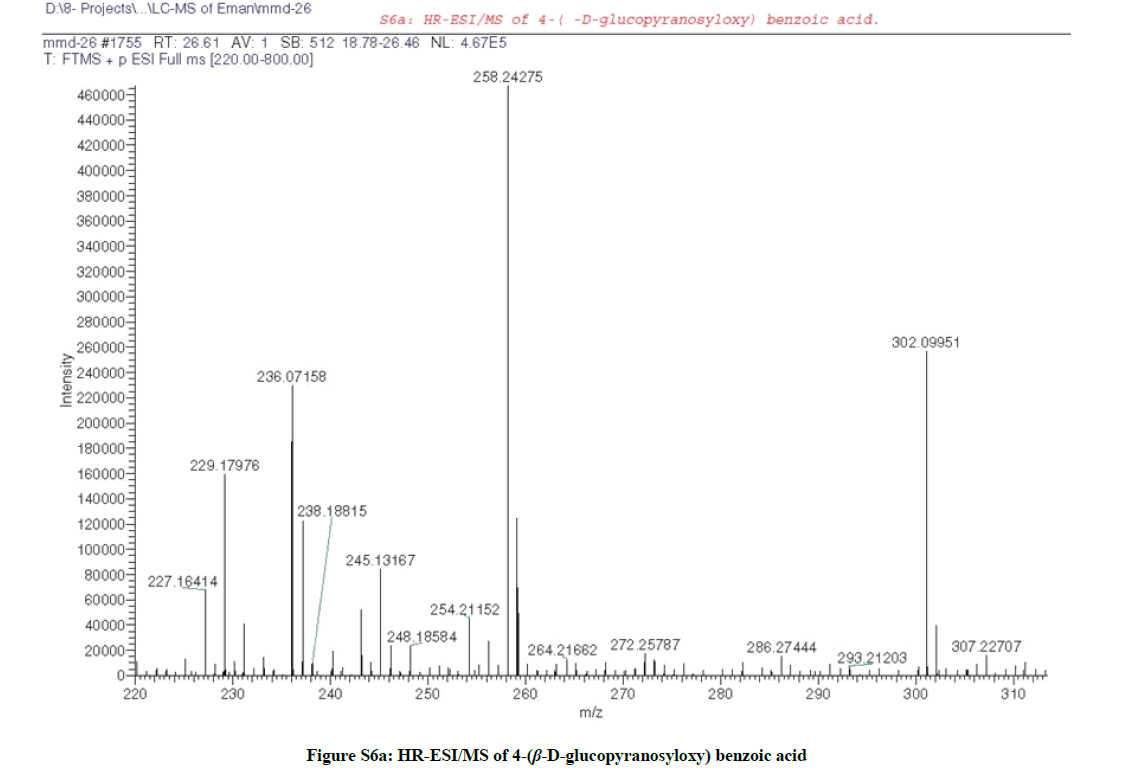
It was isolated as amorphous powder (48 mg), with molecular formula C13H16O8 as elucidated by HRESIMS (+ve) (Figure S6a), which revealed the presence of a quasi-molecular ion peak at m/z 302.0995 [M+2H]+ (calcd 302.0996 for C13H18O8).
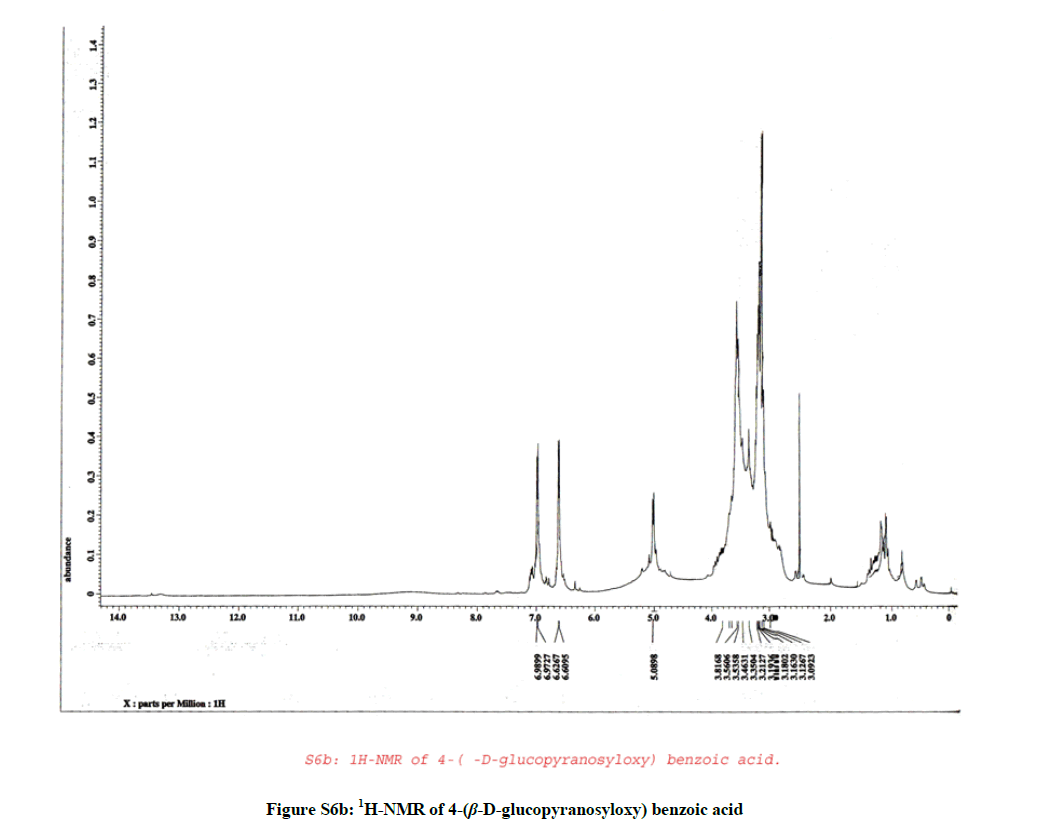
1H-NMR (Figure S6b, DMSO-d6) spectrum of compound (3) revealed the presence of two sets of doublet signals appeared at δH 6.98 (2H, d, J=8.6 Hz, H-2 & 6) and at δH 6.62 (2H, d, J=8.6 Hz, H-3 & 5) characteristic for the AA´BB´ system. Furthermore, the shielded doublet appeared at δH 5.00 (1H, d, J=7.5 Hz, H-1´) corresponding to the anomeric proton of a β-configuration for the sugar moiety, finally at δH 3.20-3.81 (6H, m, H-2´, 3´, 4´, 5´, 6´).
13C-NMR (Figure S6c, DMSO-d6) spectrum of compound (3) showed the characteristic signal for a phenolic carboxylic carbonyl group at δC 172.70 (COOH), δC 155.75 (C-4), δC 132.73 (C-1), δC 129.71 (C-2 & C-6), δC 115.53 (C-3 & C-5), δC 103.05 (C-1´), δC 81.55 (C-5´), δC 79.28 (C-3´), δC 75.83 (C-2´), δC 69.99 (C-4´) and 60.89 (C-6´).
According to the above mentioned data compound (3) was structurally elucidated as formulated in Figure (S7) and assigned to 4-(β-D-glucopyranosyloxy) benzoic acid [3,4].
| No. | leaves | Rt | [M+H]+ | Main fragments | U. V. |
|---|---|---|---|---|---|
| 1 | + | 14.25 | 581 | 581, 231, 244, 279, 302, 581 | 328, 292 |
| 2 | + | 16.7 | 599 | 599, 231, 246, 279, 579 | 240, 285 |
| 3 | + | 19.67 | 593 | 593, 231, 279, 380 | 235, 270, 330 |
| 4 | + | 20.96 | 593 | 593, 231, 244, 568 | 235, 265, 325 |
Table S1: Compounds identified by LC/MS of the ME of F. margarita leaves
| Groups | Control | DMN | DMN + F. margarita ME | F. margarita ME |
|---|---|---|---|---|
| Parameters | Group-1 | Group-2 | Group-3 | Group-4 |
| ALT | 21.62 ± 1.25 | 64.50 ± 1.00a | 47.50 ± 2.17a* | 25.03 ± 2.79n |
| AST | 16.47 ± 0.71 | 51.90 ± 2.65a | 33.23 ± 1.70a* | 18.37 ± 2.92n |
| γ-GT | 8.13 ± 2.70 | 59.16 ± 5.75a | 36.73 ± 1.16a | 6.80 ± 1.83n |
| ALP | 38.66 ± 6.02 | 118.56 ± 5.00a | 9.00 ± 43.00b* | 44.83 ± 0.76b |
| Albumin | 6.85 ± 0.18 | 3.14 ± 0.22a | 6.24 ± 0.089n* | 5.36 ± 0.069a |
Data are presented as mean ± SD; AST, ALT, GGT, ALP are expressed in U/L. Albumin is expressed in g/dl; aP ≤ 0.0001, bP ≤ 0.05, n: non-significance as compared with normal group; *P ≤0.0001, compared with DMN group
Table S2: The prophylactic effect of the ME of F. margarita leaves on the levels of ALT, AST, ALP, GGT and Albumin in DMN treated rats in serum
| Groups | ALT | AST | ALP | Direct Bilirubin | ANOVA (P ≤) |
|---|---|---|---|---|---|
| GI (Control) | 154.13 ± 19.62efgh | 135.21 ± 15.70e | 37.55 ± 11.76ef | 11.31 ± 2.84a | 0.0001 |
| GII (MeOH) | 259.75 ± 31.90a | 340.60 ± 87.3a | 88.91 ± 11.48a | 6.73 ± 1.46cd | 0.0001 |
| GIII (CCl4) | 148.99 ± 13.69fghi | 97.38 ± 14.28f | 30.93 ± 5.15fgh | 7.31 ± 1.34c | 0.0001 |
| GIV(CCl4+MeOH) | 206.86 ± 21.04bc | 170.76 ± 23.39bcd | 42.19 ± 5.47de | 4.13 ± 1.65e | 0.001 |
Unshared letters between groups are significance value at p<0.0001
Table S3: Effect ME of leaves on ALT, AST, ALP and direct bilirubin levels
| Groups | Liver wt. | Body wt. | Relative liver | ANOVA |
|---|---|---|---|---|
| GI (Control) | 3.75 ± 0.46a | 25.37 ± 2.82ef | 15.02 ± 2.96a | 0.0001 |
| GII (MeOH) | 1.95 ± 0.21efg | 33.72 ± 2.67a | 5.84 ± 0.89ef | 0.0001 |
| GIII (CCl4) | 1.53 ± 0.17h | 28.34 ± 2.49cd | 5.42 ± 0.55f | 0.0001 |
| GIV(CCl4+MeOH) | 2.02 ± 0.15def | 32.37 ± 2.01ab | 6.26 ± 0.67def | 0.0001 |
Unshared letters between groups are significance value at p<0.0001
Table S4: Effect of ME of leaves on liver weight, body weight and liver ratio
| Groups | Triglycerides | Cholesterol | Total Lipids | ANOVA (P≤) |
|---|---|---|---|---|
| GI (Control) | 478.39 ± 79.17bcd | 97.88 ± 17.44g | 422.03 ± 80.76bcd | 0.0001 |
| GII (MeOH) | 747.34 ± 57.38a | 368.19 ± 41.9a | 731.38 ± 74.49a | 0.0001 |
| GIII (CCl4) | 299.13 ± 39.77hi | 111.56 ± 18.04fg | 288.40 ± 62.46fg | 0.0001 |
| GIV(CCl4+MeOH) | 533.39 ± 46.07b | 160.12 ± 35.08cd | 444.71 ± 82.83bc | 0.0001 |
Unshared letters between groups are significance value at p<0.0001
Table S5: Effect of ME of leaves on triglycerides, cholesterol and total lipids