Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 7
Cobalt (Ii), Nickel (Ii), Copper (Ii) and Zinc (Ii) Complexes of New N, N-Bis (Thiophen-2-Ylmethylene) Benzene-1, 2-Diamine: Synthesis, Spectroscopic and Antibacterial Studies
Sanjeev A1, Bhaskar S1, Nasreen SK1, Naresh Reddy N1, Karunakar D2, Ramesh P1,3 and Muralidhar Reddy P1*
1Department of Chemistry, Nizam College, Osmania University, Hyderabad, India
2Department of Chemistry, Tara Government College (Autonomous), Sangareddy, TS, India
3Department of Chemistry, S. R. & B. G. N. R. Government College, Khammam, India
- *Corresponding Author:
- Muralidhar Reddy P
Department of Chemistry
Nizam College
Osmania University
Hyderabad, India
Abstract
Cobalt(II), Nickel(II), Copper(II) and Zinc(II) complexes with N,N-bis(thiophen-2-ylmethylene)benzene-1,2-diamine ligand have been synthesized and characterized by UV-Visible, Infrared, mass, electronic and Proton Nuclear Magnetic Resonance (1H-NMR) spectral data. Spectral data suggest that Co(II), Cu(II) and Ni(II) complexes with octahedral geometry and the Zn(II) complex with a tetrahedral structure. The antibacterial activity of all the ligands and complexes against gram-positive bacteria (Bacillus subtilis)/gram–negative bacteria (Escherichia Coli A+ and E. Coli strain Top-10) was compared with the activity of existing commercial antibacterial compound like ampicillin. All metal complexes found to be more active than respective ligand.
Keywords
Octohedral Geometry, Ampicillin.
Introduction
Complexes of transition metal ions with multidentate organic ligands [1] have been the subject of intensive research because they not only have interesting spectral and magnetic properties, but they also possess a diverse spectrum of biological activities [2-5] and metal-ligand (M-L) interactions has traditionally played an introductory role in several branches of chemistry and beyond [6], and continues to be key in various research areas from bioinorganic chemistry to molecular framework materials to small molecule catalysis [7,8]. Schiff bases have known to be useful M-L, with important roles in biology including antibacterial [9-20], antifungal, anticancer, antioxidant, anti-inflammatory, antispasmodics [21], antimalarial, and antiviral activity, and applications in chemistry, such as polymerization [22,23], reduction [24], oxidation [25,26], hydrolysis [12,27] aldol reaction, Henry reaction, epoxidation, hydrosilylation, Diels–Alder reaction and in asymmetric catalysis [28].
The chemistry of co-ordination compounds containing metal-nitrogen [29] bonds has very interesting area in recent years. This is partly due to the extraordinary properties of many of the complexes with nitrogen ligands act a challenge to interpretation. Several N4 and N2O2 donor [30-34] M(II) Schiff base compounds were reported as catalysts in the reduction of organic substrates having nitro, olefinic, acetylenic, and aldehyde groups under mild reaction conditions and as well observed appreciable biological activities [10,12-16,18-20,24,26,27,31-45]. In this study, we have successfully synthesized the metal complexes of Co(II), Ni(II), Cu(II) and Zn(II) with N,N-bis(thiophen-2-ylmethylene)benzene-1,2- diamine ligand. Both ligand and its metal complexes were screened for antibacterial activity.
Material and Methods
Materials
All the solvents, amines, aldehydes, and metal salts used in this study were of analytical grade. The solvents were distilled by standard procedures before use. The water used in the study was essentially double distilled water. The solvents were distilled and stored over molecular sieves. The purity of the ligands was checked by Thin Layer Chromatography (TLC) using Merck 60F254 silica gel plates.
Physical Measurements
The MPs of all the ligands were determined on a Buchi- 510 melting point apparatus. UV-Visible spectra were recorded with Shimadzu UV- 2140 A, a UV-Visible double beam spectrophotometer with matched quartz cells of path length 1 cm at Osmania University, Hyderabad. The Infrared (IR) spectra were recorded in KBr pellets on Perkin Elmer-283 spectrophotometer at Osmania University, Hyderabad. The scanning rate was 6 min in the range of 4000-200 cm-1. 1H-NMR spectra were recorded in CDCl3 or Dimethyl sulfoxide (DMSO)-d6 on a Bruker AVAVCE III 400 MHz -NMR spectrometer using TMS as the internal standard at the SAIF, Central Facilities For Research And Development, Osmania University, Hyderabad.
Synthesis of ligand
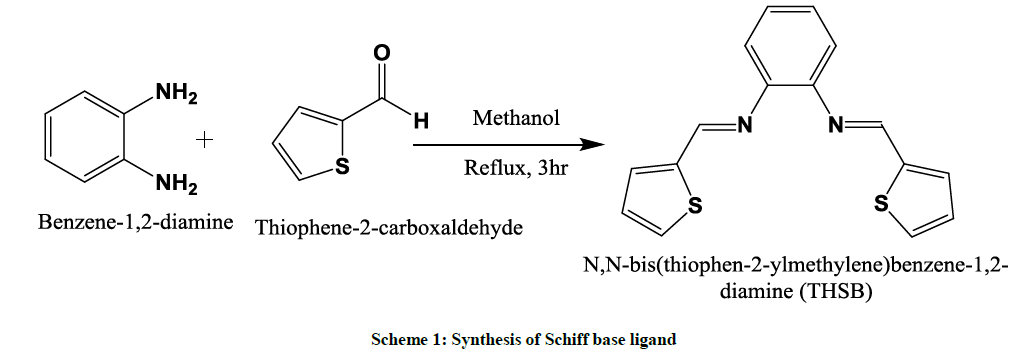
The Schiff base N,N-bis(thiophen-2-ylmethylene)benzene-1,2-diamine (THSB) was synthesized by the reaction of Orthophenylenediamine (4.62 mmol, 0.5 g ), Thiophene-2-carboxaldehyde (9.24 mmol, 1.036 g) in methanol (15 ml). The solution was stirred for 3 hours. The reaction was monitored by TLC, using (3:7) ethyl acetate: hexane mixture was eluent. The mustered yellow precipitate obtained was filtered, and was recrystallized from alcohol (Scheme 1).
Preparation of metal complexes
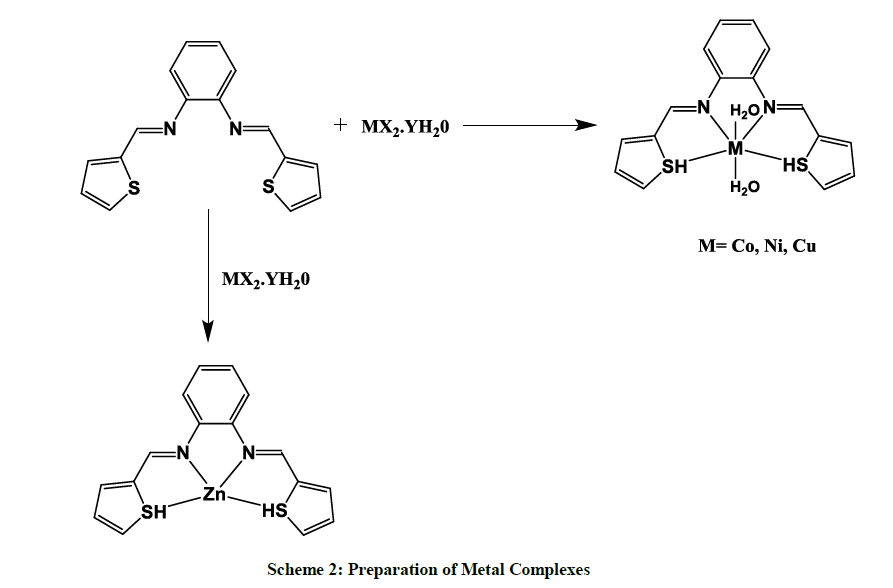
The Schiff base THSB (0.67 mmol 0.2 g) was dissolved in methanol (15 ml) and was added to a stirred solution of metal(II) chloride hexahydrate (0.67 mmol 0.159 g, dissolved in methanol-20 ml). The resulting mixture was reflux for 4 h, the mixture was filtered and the precipitation of products was washed with methanol and dried at room temperature for 24 h. a Blue color [Co(THSB)(H2O)2] complex, a Mustered yellow color [Ni(THSB)(H2O)2] complex, a green color [Cu(THSB)(H2O)2] and Cream color [Zn(THSB)](H2O)2 complex was obtained (Scheme 2).
Results and Discussion
IR spectroscopy
The IR spectrum of Schiff base ligand (L) exhibits characteristic band at 1560 cm-1 supports the formation of azomethine (-HC=N) [18- 20,45,46]. The formation of Schiff base ligand was identified by the disappearance of peaks at 1680-1695 for –CHO and 3400 cm-1 for –NH2 of precursors. The infrared spectra of all Schiff base metal complexes show medium intensity band due to C-N appeared at 1611–1636 cm-1 was also found to be shifted towards higher side about 1600-1620 cm-1 compared to ligand spectrum and supports the coordination of imino nitrogen (-HC=N) of ligand to the metal ion. In the IR spectrum of Schiff base and metal complexes, coordination of nitrogen to metal is further supported by the presence of weak intensity band in the region 620-653 cm-1 and in addition a weak intensity band appeared at 437-447 supports the (M-O) stretching frequency. The presence of coordinated water in the complexes is indicated by a broad band in the region 3200-3560 cm-1 and two weaker bands in the region 840 and 740-758 cm-1 due to ν(–OH) rocking and wagging mode of vibrations, respectively [18-20,45,46].
Electronic spectra
Electronic absorption spectra of the Schiff base and its Cu(II), Ni(II), Co(II), Zn(II) complexes were recorded in methanol solution in the range of 200-800 nm region. The absorption bands in the range of 270-290 nm are assigned to benzene π–π transition. In the UV-absorption spectra of all the metal complexes azomethine chromophore π–π transitions are located at 300-330 nm and the charge transfer LMCT bands are appeared in the range of 380-440 nm. Cobalt complexes generally exhibits two d–d transition bands assigned to 4T1g(F)→4T2g(F)(ν1) and 4T1g(F)→4T1g(P)(ν3) transitions, suggesting octahedral geometry for Co(II) complexes [13,33,34,38]. Nickel complexes exhibits three d–d transition bands which are signed to 3A2g(F)→3T2g(F)(ν1); 3A2g(F)→3T1g(F)(ν2) and 3A2g(F)→3T1g(P)(ν3) transitions, respectively. This indicates octahedral geometry for Ni(II) complexes [24,33,34]. The electronic spectra of copper (II) complexes exhibit broad d-d absorption bands in the region 400-770 nm indicative of a distorted octahedral system with weak apical coordination of solvent/anion molecules [12,33,34]. Zn(II) complex does not exhibit d–d electronic transition due to the completely filled d-orbital. Four coordinate Zinc (II) complexes, in general, would have tetrahedral geometry.
1H-NMR Spectral data
The Proton Nuclear Magnetic Resonance (1H-NMR) spectra of Schiff base ligand (L) gives signal corresponding to azomethine (-CH=N) proton in the range of 7.50 ppm suggesting the condensation of aromatic aldehyde with primary amine. All the aromatic protons appeared in both the ligands appeared as multiplets in the range of 7.3-7.7 ppm.
Mass Spectral analysis
The proposed structures of all the complexes were further confirmed by comparing their molecular formula with m/z values. The m/z values are THSB(297) [Co(THSB)(H2O)2] (391), [Cu(THSB)(H2O)2] (396), [Ni(THSB)(H2O)2](391), [Zn(THSB)](H2O)2] (398). This data is in good agreement with respective molecular formulae of metal complexes.
Anti bacterial activity
In the present study, the new ligand and its complexes with Co(II), Ni(II), Cu(II) and Zn(II) have been assayed for their antibacterial activity and the results are presented.
Antibacterial testing by paper disc method
The antibacterial activity of the compounds was determined by the paper disc method. The compounds were dissolved in DMSO and tried at a concentration of 100 μg / ml. The Whatman filter paper discs (6 mm diameter) with different compounds were placed, aseptically on seed nutrient agar plates (prepared from the readymade dehydrated media) (Figure 1) with different bacteria and incubated for 72 h at 38°C. At the end of the incubation period, the diameter of the growth inhibition zones (Table 1) was measured. Ampicillin was taken as standard against the bacteria and solvent was used as negative control (hence the study is made independent of the solvent used).
| Compound | Bacillus Subtilis Cm | Escherichia Coli A+ Cm | Top-10 Cm |
|---|---|---|---|
| Ampicillin | 0.5 | 0 | 0.2 |
| THSB-L | 0.7 | 0.4 | 0.6 |
| THSB-Co | 0.9 | 0.6 | 0.8 |
| THSB-Ni | 0.8 | 0.5 | 0.7 |
| THSB-Cu | 1.3 | 0.8 | 0.9 |
| THSB-Zn | 0.6 | 0.4 | 0.5 |
Table 1: Zone of inhibition for ligand & metal complexes
From the preliminary screening of compounds only Cu(II) complex was very active against all three strains of bacteria. According to the Tweedy’s chelation theory, chelation noticeably decreases the polarity of the metal ion due to partial sharing of its positive charge with donor groups and probable Π-electron delocalization over the whole chelate ring. This chelation possibly will enhance the lipophilic nature of the central metal atom, which then favors its penetration through the lipid layers of cell membrane. This compound was further tested at different dilutions to determine their minimum inhibitory concentration (MIC) [12,16,17,47-51].
Conclusion
In summary we have accomplished the facile synthesis of four complexes of the Schiff base N,N-bis(thiophen-2-ylmethylene)benzene-1,2- diamine by non-template method and characterized them by, IR, UV–Vis, and EPR. The ligand acts as a tetra dentate ligand. The physicochemical and spectral data reveal an octahedral geometry for the Co(II), Cu(II) and Ni(II) complexes, a tetrahedral structure for zinc(II) complex. The preliminary screening of compounds Co(II) and Cu(II) complexes were very active against all three strains of bacteria.
Acknowledgement
Authors thank the University Grants Commission (UGC), New Delhi for financial support under UGC-BSR Research Start-Up Grant (UGC Lr. No. F.30-322/2016 (BSR) dated 16.02.2017) and Nizam College, Osmania University for providing the necessary facilities for carrying this work.
References
- P.G. Cozzi, Chem. Soc. Rev., 2004, 33, 410-421.
- N. Al-Awadi, M.S. El-Ezaby, H. Abu-Soûd, Inorganica Chimica Acta., 1982, 67, 131-138.
- A. Arbuse, M. Font, M.A. Martínez, X. Fontrodona, M.J. Prieto, V. Moreno, X. Sala, A. Llobet, Inorg. Chem., 2009, 48, 11098-11107.
- J.R. Morrow, K.A. Kolasa, Inorganica Chimica Acta., 1992, 195, 245-248.
- A. Sigel, H. Sigel, R.K. Sigel, Nickel and its surprising impact in nature: metal ions in life sciences, John Wiley & Sons, 2007.
- T. Marino, T. Mineva, N. Russo, E. Sicilia, M. Toscano, Potential energy surfaces of metal-ligand interactions obtained using density functional theory, in: Metal-Ligand Interactions in Chemistry, Physics and Biology, Springer, 2000, 207-226.
- D.A. Atwood, M.J. Harvey, Chem. Rev., 2001, 101, 37-52.
- V.C. Gibson, S.K. Spitzmesser, Chemi. Rev., 2003, 103, 283-316.
- M.T. Kaczmarek, R. Jastrząb, E. Hołderna-Kędzia, W. Radecka-Paryzek, Inorganica Chimica Acta., 2009, 362, 3127-3133.
- E.R. Krishna, P.M. Reddy, M. Sarangapani, G. Hanmanthu, B. Geeta, K.S. Rani, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2012, 97, 189-196.
- P.M. Reddy, Y.-P. Ho, K. Shanker, R. Rohini, V. Ravinder, Eur. J. Med. Chem., 2009, 44, 2621-2625.
- P.M. Reddy, A.V.S.S. Prasad, V. Ravinder, Trans. Metal Chem., 2007, 32, 507-513.
- P.M. Reddy, A.V.S.S. Prasad, K. Shanker, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2007, 68, 1000-1006.
- P.M. Reddy, R. Rohini, E.R. Krishna, A.R. Hu, V. Ravinder, Int. J. Mol. Sci., 2012, 13, 4982-4992.
- P.M. Reddy, K. Shanker, R. Rohini, V. Ravinder, Int. J. Chem. Tech. Res., 2009, 1, 367-372.
- R. Rohini, P.M. Reddy, K. Shanker, A.R. Hu, V. Ravinder, Eur. J. Med. Chem., 2010, 45, 1200-1205.
- R. Rohini, P.M. Reddy, K. Shanker, K. Kanthaiah, V. Ravinder, A. Hu, Arch Pharm Res., 2011, 34, 1077-1084.
- K. Shanker, R. Rohini, V. Ravinder, R.M. Reddy, Y.P. Ho, Spectrochim Acta A Mol Biomol Spectrosc., 2009, 73, 205-211.
- K. Shanker, R. Rohini, K. Shravankumar, P.M. Reddy, Y.P. Ho, V. Ravinder, J. Indian Chem. Soc., 2009, 86, 153-161.
- K. Shanker, P.M. Reddy, R. Rohini, Y.P. Ho, V. Ravinder, J. Coord. Chem., 2009, 62, 3040-3049.
- M.J. Hearn, M.H. Cynamon, J. Antimicrob. Chemother., 2004, 53, 185-191.
- K. Gupta, A.K. Sutar, Coord. Chem. Rev., 2008, 252, 1420-1450.
- K. Gupta, A.K. Sutar, C.-C. Lin, Coord. Chem. Rev., 2009, 253, 1926-1946.
- P.M. Reddy, A.V.S.S. Prasad, R. Rohini, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2008, 70, 704-712.
- M. Matsumoto, K. Kuroda, Tetrahedron Lett., 1981, 22, 4437-4440.
- P.M. Reddy, A.V.S.S. Prasad, C.K. Reddy, V. Ravinder, Trans. Metal Chem., 2008, 33, 251-258.
- P.M. Reddy, K. Shanker, V. Srinivas, E.R. Krishna, R. Rohini, G. Srikanth, A. Hu, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2015, 139, 43-48.
- T. Katsuki, Chem. Soc. Rev., 2004, 33, 437-444.
- S. Adsule, V. Barve, D. Chen, F. Ahmed, Q.P. Dou, S. Padhye, F.H. Sarkar, J. Med. Chem., 2006, 49, 7242-7246.
- E. Kim, E.E. Chufán, K. Kamaraj, K.D. Karlin, Chem. Rev., 2004, 104, 1077-1134.
- M. Ashok, A.V.S.S. Prasad, P.M. Reddy, V. Ravinder, J. App. Spectrosc., 2008, 75, 872-879.
- M. Ashok, A.V.S.S. Prasad, P.M. Reddy, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2009, 72, 204-208.
- G. Budige, M.R. Puchakayala, S.R. Kongara, A. Hu, R. Vadde, Chem. Pharm. Bull. (Tokyo), 2011, 59, 166-171.
- B. Geeta, K. Shravankumar, P.M. Reddy, E. Ravikrishna, M. Sarangapani, K.K. Reddy, V. Ravinder, Spectrochimica Acta Part a-Molecular and Biomolecular Spectroscopy., 2010, 77, 911-915.
- A. Prasad, P.M. Reddy, K. Shanker, R. Rohini, V. Ravinder, Coloration Technology., 2009, 125, 284-287.
- P.U. Rani, P.M. Reddy, M. Sarangapani, V. Ravinder, J. Indian Chem. Soc., 2007, 84, 122-129.
- P.U. Rani, P.M. Reddy, K. Shanker, V. Ravinder, Trans. Metal Chem., 2008, 33, 153-160.
- M.B. Reddy, K. Shanker, P.U. Rani, R. Rohini, C.K. Reddy, V. Ravinder, J. Indian Chem. Soc., 2007, 84, 971-977.
- P.M. Reddy, M. Ashok, P.U. Rani, V. Raninder*, Int. J. Chem. Sci.,, 2007, 5, 953-959.
- P.M. Reddy, M. Ashok, P.U. Rani, V. Ravinder, Mat. Sci. Res. Ind.,, 2006, 3, 251-255
- P.M. Reddy, M. Ashok, P.U. Rani, V. Ravinder, Int. J. Chem. Sci.,, 2007, 5, 489-495.
- P.M. Reddy, C.K. Reddy, K.S. Rani, M.B. Reddy, V. Rivinder, J. Indian Chem. Soc.,2007, 84, 733-738.
- P.M. Reddy, K. Shanker, P.U. Rani, B.K. Rao, V. Ravinder, J. Indian Chem. Soc., 2007, 84, 411-417.
- P.M. Reddy, K. Shanker, R. Rohini, M. Sarangapani, V. Ravinder, Spectrochim Acta A Mol Biomol Spectrosc., 2008, 70, 1231-1237.
- K. Shanker, M. Ashok, P.M. Reddy, R. Rohini, V. Ravinder, Int. J. Chem. Tech. Res., 2009, 1, 777-783.
- P.M. Reddy, W.Y. Hsu, J.F. Hu, Y.P. Ho, J. Am. Soc. Mass Spectrom., 2010, 21, 421-424.
- R. Rohini, P.M. Reddy, K. Shanker, A.R. Hu, V. Ravinder, J. Braz. Chem. Soc., 2010, 21, 897-904.
- R. Rohini, P.M. Reddy, K. Shanker, V. Ravinder, Acta Chimica Slovenica, 2009, 56, 900-907.
- R. Rohini, K. Shanker, P.M. Reddy, Y.-P. Ho, V. Ravinder, Eur. J. Med. Chem., 2009, 44, 3330-3339.
- R. Rohini, K. Shanker, P.M. Reddy, V. Ravinder, J. Braz. Chem. Soc., 2010, 21, 49-U36.
- R. Rohini, K. Shanker, P.M. Reddy, V.C. Sekhar, V. Ravinder, Archiv Der Pharmazie., 2009, 342, 533-540.