Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 7
Comparative Study on the Nitric Oxide Scavenging and Hemolytic Activity of Fresh Leaf Juice and Dry Leaf Extracts of Clinacanthus Nutans (Burm. F) Lindau
Akowuah GA1, Chin JH2, Yeong SW1, Quah SY1 and Ahmad M3*
1Faculty of Pharmaceutical Sciences, UCSI University, No. 1, Jalan Menara Gading, Kuala Lumpur, Malaysia
2Faculty of Medicine, Department of Biochemistry, MAHSA University, Bandar Saujana Putra Campus, Jalan SP2, Bandar Saujana Putra, 42610 Jenjarom, Kuala Langat, Selangor, Malaysia
3Faculty of Pharmaceutical Sciences, Universiti Sains Malaysia, 11800 Pulau Pinang, Malaysia
- *Corresponding Author:
- Ahmad M
Faculty of Pharmaceutical Sciences
Universiti Sains Malaysia
11800 Pulau Pinang, Malaysia
Abstract
The purpose of the present study was to compare nitric oxide scavenging and hemolytic activities of dry leaf extracts and fresh leaf juice of Clinacanthus nutans (Burm.f) Lindau. Methanol and cold aqueous extracts of the dry leaf were prepared by macerating in the respective solvent at room temperature. Hot aqueous extract was prepared by mixing the leaf powder in distilled water and the concoction was heated on a hot water bath set at 100ºC. Fresh leaves juice was prepared by direct grinding of fresh plant leaves without adding any solvent. In vitro radical scavenging was determined by using Griess reagent and hemolytic activity was assayed on a healthy human erythrocyte. All the extracts demonstrated dose-dependent nitric oxide radical scavenging activity comparable to that of the positive reference compound, quercetin. The extracts showed no hemolytic activity at the concentration of 0.5-2.0 mg/ml. Fresh leaf juice and dry leaf extracts of C. nutans showed nitric oxide radical scavenging potency and non-hemolytic.
Keywords
Clinacanthus nutans, Leaf extracts, Leaf juice, Hemolytic activity, Nitric oxide scavenging.
Introduction
Clinacanthus nutans (Burm. f) Lindau belongs to the family of Acanthaceae and native to tropical Asia countries where it is cultivated for its medicinal properties. The leaves are used to treat insect and snake bite, burns, skin ulcers, shingles, and hives [1]. Anti-proliferative effect of the leaves of C. nutans extracts on cultured human cancer cell lines has been reported [1]. The leaves of C. nutans contain sterols (stigmasterol and β-sitosterol) and C-glycosyl flavones (vitexin, isovitexin, shaftoside, isomollupentin-7-O-β-gluropyranoside, orientin and isoorientin) [2,3].
Herbal medicines are used in various forms such as whole herbs, teas, capsules and tablets. Temperature and solvent are important parameters which determined the extraction yield of botanical samples. The constituents in botanical extracts also vary with the extraction methods, temperature and solvents. The selection of solvent for phytochemical extraction depends on the polarity of the compounds being extracted and molecular weight [4,5]. Ideally, the extraction solvent should possess low toxicity, preservative action and do not cause the extracted compounds to dissociate or form complexes during the extraction.
Preparations from plants need to be screened for possible hemolytic effect on red cell membrane. Membranes of human erythrocytes have variable stability and can be hemolysed by reactive oxygen species and lead to diseases such as sickle cell anemia and thalassemia [6,7]. The radical scavenging and hemolytic activities of fresh leaf and dry leaf extracts may differ due to the drying method. In this study, nitric oxide scavenging and hemolytic activities of fresh leaf juice and dry leaf extracts of C. nutans were investigated and compared.
Materials and Methods
Chemicals, reagents and solvents
Cold distilled water at 27 ± 1ºC was obtained from Ultrapure System (Elga Labwater, UK). Aluminium chloride, sodium nitrite, quercetin, and (+)-catechin hydrate were supplied by Sigma-Aldrich (St. Louis, MO, USA). All other chemicals and solvents were analytical grade.
Plant material and ATR-FTIR fingerprints
Approximately 6.5 kg fresh leaves of C. nutans was collected from a private commercialised herbal park known as Yik Poh Ling Herbal Farm, Persatuan Pangkaji Herbal Tradisional, Pantai, Seremban, Negeri Sembilan. Voucher specimen was prepared and identified at Institute of Bioscience, University Putra Malaysia in July 2013: C. nutans (SK 2208/13). Five hundred grams of fresh leaves of each plant were kept for juice processing. The remaining leaves were washed thoroughly under tap water and air-dried at 37ºC for 7 days. The weight of the dried leaves was measured and grounded to powder using a blender (Waring, USA). The processed leaves or leave powder was kept in an air-tight bottle for further use in subsequent process for extraction.
Preparation of dried leaves extracts and fresh leave juice
Methanol extracts of dried leaves: The methanol extract of C. nutans was prepared by weighing 75 g of dry pulverized leaves and macerated with 300 ml distilled water in a conical flask at room temperature for two days. After maceration, the extract was filtered. The extraction was repeated under the same condition until colourless extract was obtained. The extracts were filtered and concentrated under vacuum at below 40°C and then freeze-dried. The yield of extraction was calculated, and the extract was stored in desiccators until further used for study.
Hot aqueous extracts: Hot aqueous extracts of C. nutans was prepared by weighing 75 g of dry pulverized leaves and macerated with 300 ml distilled water in a conical flask. The mixture was placed in hot water bath set at 100ºC for 30 min. Then, the mixture was left to cool to room temperature and filtered after 24 h. The extraction was repeated under the same condition until colourless extract was obtained. The extracts were filtered and then freeze-dried. The yield of extraction was calculated, and the extract was stored in desiccators until further used for study.
Cold aqueous extracts: Cold Aqueous Extracts of C. nutans was prepared by weighing 75 g of dry pulverized leaves were weighed and macerated using 300 ml of cold distilled water in a conical flask. The solution was filtered after 24 h and concentrated and the concentrated extract was freeze-dried. The freeze-dried extract was stored in air tight bottle for further use.
Fresh leaves juice: Fresh leaves juice of C. nutans was prepared by direct grinding (without adding any solvent) of 100 g fresh plant leaves of each. The leaf juice was collected in an air tight clean bottle and freeze-dried. The extract was stored in air tight bottle for further use while the remaining juice was kept in refrigerator and used within 24 h.
Total flavonoid content
The total flavonoid content (TFC) in C. nutans preparation was quantified using aluminium chloride colorimetric assay by a spectrophotometer according to the method of Pekal and Pyrzyniska [8]. Absorbance was measured at 510 nm by spectrophotometer and distilled water was used as blank. Catechin (20-100 mg/l) was used as the standard and the TFC of each preparation was expressed as mg catechin equivalent (CAE) per gram of dry weight of sample. The experiments were performed in triplicates.
Nitric oxide radical scavenging activity
Nitric oxide radical scavenging activity was determined by using Griess reagent based on the method of Tsikas [9]. The concentrations of the samples used were 50, 100, 200, and 400 μg/ml. Absorbance was measured at 545 nm and quercetin was used as positive reference compound. The experiments were performed in triplicates. The percentage radical scavenging activity was calculated relative to the control by using the formula:
Nitric oxide scavenged (%)=[(Acontrol – Asample)/Acontrol] × 100
where Acontrol = absorbance of sample and Asample control=absorbance in the presence of the samples of extracts or standards.
Hemolytic activity
Hemolytic activity was assayed on human erythrocytes (B positive blood group) based on the method of Zohra and Fawzia [10]. The samples were prepared in the phosphate saline buffer at the concentration of 0.5, 1.0, 2.0, and 4.0 mg/ml. Absorbance was measured at 540 nm. Phosphate buffer saline was used for negative control (0% hemolysis) and triton X-100 (0.1%) was used for positive control (100% haemolysis). Percentage hemolytic activity was calculated relative to the positive control by using the formula:
Hemolytic activity (%)=(Asample/Apositive control) × 100
where Asampl =absorbance of sample and Apositive control=absorbance of the positive control.
ATR-FTIR fingerprinting of leaf samples
Attenuated Total Reflectance-Fourier Transform Infrared (ATR-FTIR) spectra of the dried leaf and fresh leaf were collected with Nicolet™ iS5 FTIR spectrometer (Thermo scientific, USA). The instrument is equipped with iD5 ATR accessory featuring a top plate diamond crystal with a fixed angel of incidence of 42°. The qualitative ATR-FTIR spectroscopy analysis of the dry leaves powder and fresh juice was conducted in the mid-IR range of 4000–400 cm-1. A background scan of air was done prior to each scanning of the working standards. All spectra were recorded at 4 cm-1 resolution with average of 16 scans per spectrum. The diamond crystal of the ATR accessory was cleaned with 70% ethanol between scanning.
Data analysis
All data are presented as mean (μ) ± standard error mean (SEM) and performed with statistical Package for social Sciences (SPSS 15). Data were analysed by one-way ANOVA, followed by Tukey’s test. P<0.05 was considered as significant.
Results and Discussion
Extraction yield
The yield of C. nutans crude leaves preparations were significantly different (p<0.05) with respect to the solvent used for the extraction. The yields of the extraction decreased in the following order: cold aqueous extract (6.87%), methanol extract (5.56%), hot aqueous extracts (3.60%), and juice (0.55%). The high yield of the methanol extract indicates high solubility of the constituents of the leaf in the solvent. The higher yield of the cold aqueous extract as compared to hot aqueous extract can be ascribed to lose in heat labile constituents. Continuous exposure of plant extracts to higher temperature can lead the destruction of heat labile constituents [11]. Thus, boiling infusion process in preparing hot aqueous extract could lead to low yield than cold aqueous extraction [4].
The lower yield in the juice of the fresh leaf was probably due to the direct grinding and squeezing of the fresh leaf which may not be efficient extraction process as compared to chemical solvent extraction method. Although, mechanical forces allowed leaves to breakdown and release the liquid soluble secondary metabolites, solvent extraction methods have higher efficiency by using aqueous and methanol as extraction solvents [4]. The findings of the present study are in accordance with the study conducted by Chan et al. [12] which showed that the yields of fresh juices of Magnoliae officinalis and other medicinal found in South-East Asia were lower than those of other extraction solvents such as water or ethanol.
Total flavonoid content (TFC)
The TFC method used aluminium chloride (AlCl3) solution that is selective only for flavonols and flavones and with the presence of NaNO2 in alkaline medium it also showed specificity for catechins [8]. The electropositive aluminum ion (Al3+), attracts the atomic nuclei of the aromatic ring in the flavonoids to create a charge-transfer resonance hybrid. This hybrid, which is stable in aqueous medium, reacts with sodium nitrite (NaNO2) in an alkaline medium to form a pink coloured complex that can be measured using the spectrophotometer at 510 nm.
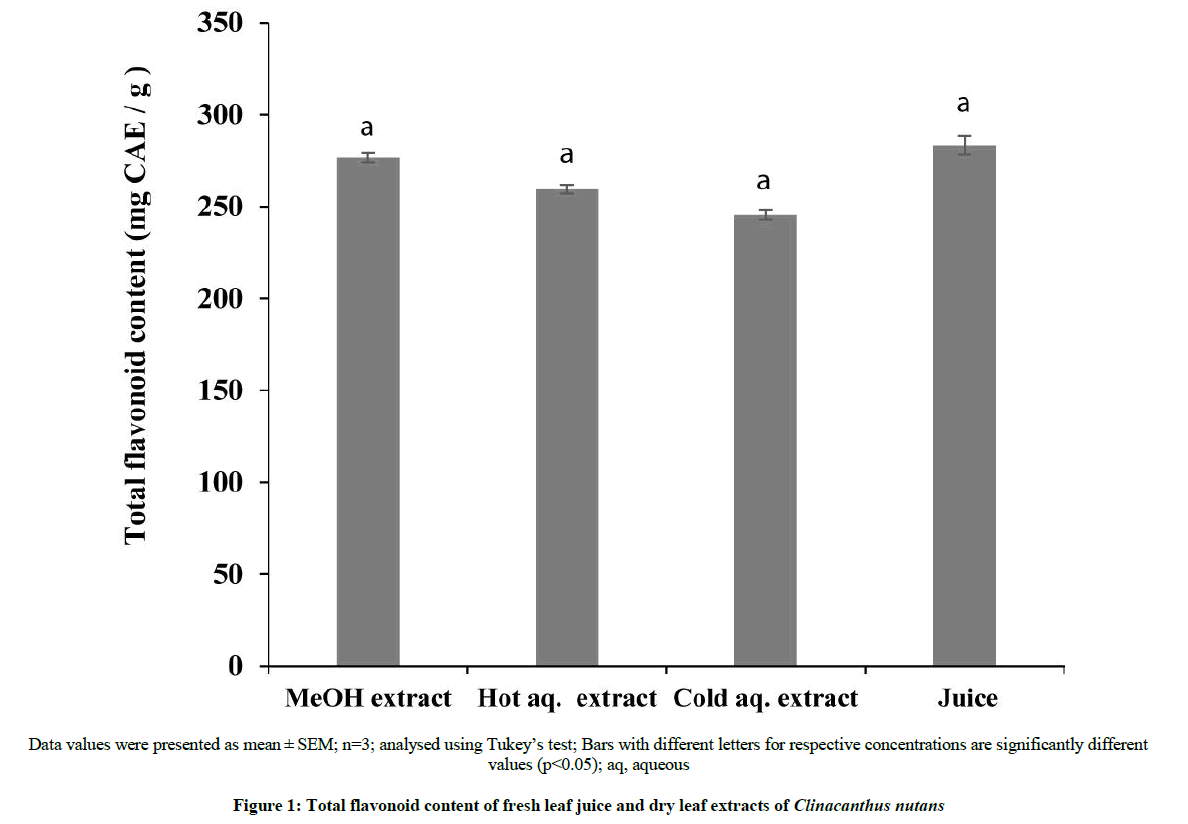
The leaf preparations of C. nutans were found to contain flavonoids (Figure 1). TFC of the four preparations were significantly different (p<0.05). The juice contained the highest flavonoid content (283.33 ± 5.11 mg CAE/g of sample) followed by methanol extract (276.67 ± 2.50 CAE/g of sample), hot aqueous extract (259.52 ± 2.35 mg CAE/g of sample) and cold aqueous extract (245.48 ± 2.48 mg CAE /g of sample). The higher flavonoid content in hot aqueous extract of C. nutans than its cold extract suggests thermostability of the flavonoid groups (including glycosides) present in the leaf. The thermally stable flavonoids were extracted by the hot aqueous solvent.
The flavonoid content in botanical extracts has been reported to vary with conditions such as temperature, pressure and period of exposure [13]. Flavonoids which contain polyhydroxy groups and their glycosides have improved solubility in water and thermally stable [14]. The differences between the flavonoid content of the fresh leaf and dry leaf preparations could be ascribed to drying method and the efficiency of the extraction process. Vaidya et al. [15] found differences in flavonoids content between fresh and dry leaf extracts of sixteen Scutellaria species.
Nitric oxide radical scavenging activity
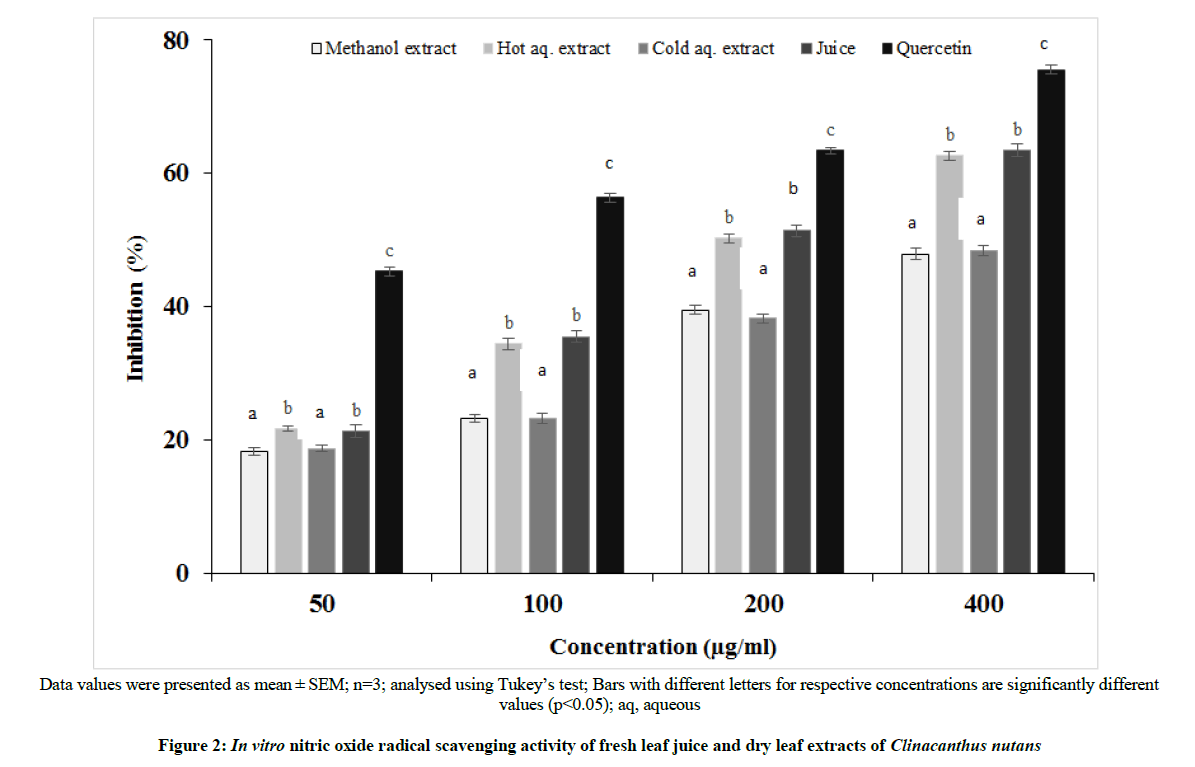
Nitric oxide reacts with oxygen to form nitrogen dioxide which can initiate auto-oxidation of fatty acid in lipid membranes leading to membrane damage [16]. The result of the nitric oxide radical scavenging activity is shown in Figure 2. The scavenging activity of the four preparations was significantly different (p<0.05). The dry leaf extracts and fresh leaf juice decreased the content of nitrite generated from the in vitro decomposition of sodium nitroprusside. The positive reference compound, quercetin, demonstrated the highest inhibition of the nitric oxide radical followed by the, juice, methanol extract, hot aqueous extract, and the cold aqueous extract. The degree of nitric oxide radical inhibition of the preparations was dose-dependent.
Data values were presented as mean ± SEM; n=3; analysed using Tukey’s test; Bars with different letters for respective concentrations are significantly different values (p<0.05); aq, aqueous
Figure 2: In vitro nitric oxide radical scavenging activity of fresh leaf juice and dry leaf extracts of Clinacanthus nutans
The presence of flavonoids compounds in the C. nutans leaf preparations contributed to its nitric oxide radical scavenging activity. Flavonoids are effective nitric oxide radical scavengers due to the presence of the phenolic hydroxyl groups which are the main active group that scavenge and inhibit free radicals [17]. The nitric oxide radical scavenging activity of the juice and the hot aqueous dried leaf extract observed in this study was comparable to the value of the aqueous extract reported by Yong et al. [1]. The juice or extracts from the fresh leaf of C. nutans was not included in their study. The results of the present study revealed that the juice or extracts prepared from leaves of C. nutans may inhibit generation of nitric oxide radicals and prevent oxidative stress ailments.
Hemolytic activity
The present study showed that the leaves are rich source of flavonoids and possess nitric oxide radical scavenger activity. Nevertheless, the leaves of C. nutans were screened for possible hemolytic effect on red cell membrane. In this study, the phosphate buffer saline was used for 0% hemolysis while Triton X-100 (0.1%) was considered as 100% haemolytic activity. The results showed that the leaf possess no hemolytic activity at concentrations of 0.5 to 2.0 mg/ml/ At concentration of 4 mg/ml, percentage hemolysis for juice, methanol, hot aqueous and cold aqueous extracts were 0.002%, 0.004%, 0.003%, and 0.004% which was not significant (p>0.001) when compared to the positive control. This shows that the leaf of C. nutans is non-hemolytic. In vitro hemolysis activities have been used by numerous scientists for toxicological evaluation of plants and their reports show that herbal medicinal products could prevent oxidative damage in intact red blood cells membranes [18-20].
ATR-FTIR fingerprints of leaf samples
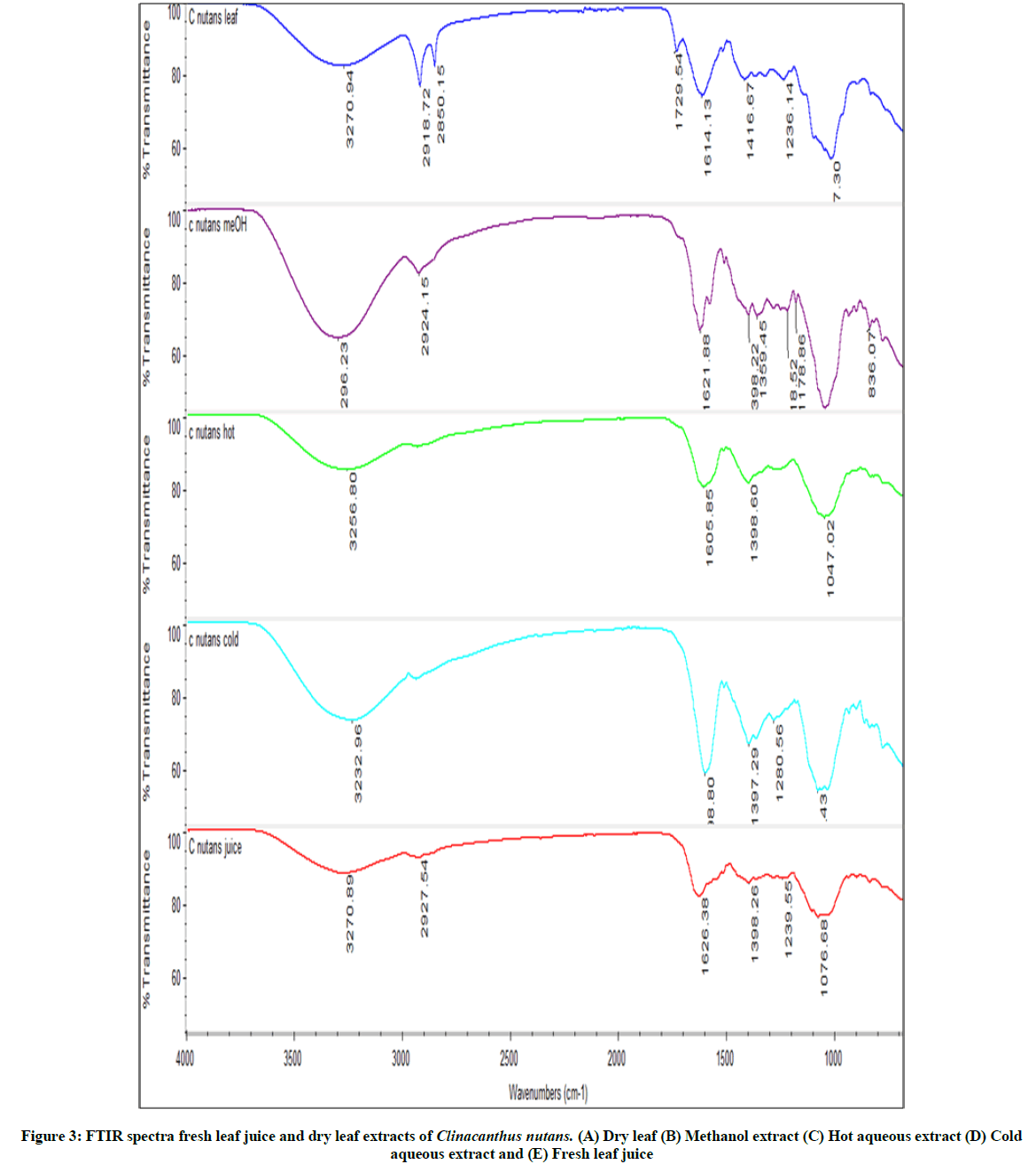
FTIR fingerprints of C. nutans dry and fresh leaves preparations were similar (Figure 3). Qualitative FTIR analysis of the extract confirmed the presence of hydroxyl and carbonyl group as functional group of chemical constituents in the extract. The FTIR fingerprints showed that the constituents of all the preparations contain hydroxyl and carboxyl group. The broad and strong intensity band situated in the range 3296-3232 cm-1 is attributed to hydroxyl (–OH) stretching. The strong peak at 1729-1598 cm-1 is due to a carbonyl (C=O) stretching in carboxy. The peak observed at 1280-1017 cm-1 was assigned to the ester (C-O) stretching of alcohols and carboxylic acids. The presence of hydroxyl, and carbonyl peaks suggest that the compounds containing hydroxyl and carbonyl as functional group were present in the dried leaves and all preparations. Phenolic and flavonoids compounds C. nutans leaves (including (C-glycosyl flavones) contain hydroxyl and carbonyls as functional groups. also have hydroxyl as functional group.
There are concerns with the use of herbal medicines such as non-standardized products, potential toxicities and adverse effects, adulteration, contamination and potential herb-drug interactions [21-23]. Fingerprinting of herbal plant materials are used for identification and quality control to ensure that consistent material is used to produce herbal medicine and supplements are free from adulteration and contamination. Quality of herbal medicines has direct impact on their safety and efficacy [12].
Conclusion
In conclusion, fresh leaf juice and dry leaf extracts of C. nutans are rich in flavonoids. The fresh leaf juice showed the highest flavonoid content as compared to the dry leaf extracts. The leaf demonstrated the nitric oxide radical scavenging activity hence could prevent oxidative damage of living membranes by providing health-promoting benefits. They leaf is non-hemolytic thus suitable to be used in herbal medicinal products. The preparations from the fresh and dried leaves showed similar FTIR fingerprints.
Acknowledgement
The authors are grateful for the financial supports from CERVIE, UCSI, University research funding (Proj-In-FPS-004) and Universiti Sains Malaysia Research Grant Scheme (304/PFARMASI/6331040) to carry out the work successfully.
References
- S.K. Al Deeb, I. Hamdan, S.M. Al Najjar, Talanta., 2004, 64, 695-702.
- Y.K. Yong, J.J Tan, M.S. Hui, H.S. Chiong, Evid. Based Complement. Alternat. Med., 2003, 13, 1-8.
- A. Ghasemzadeh, A. Nasiri, H.Z. Jaafar, A. Baghdadi, I, Ahmad, Molecules., 2014, 19(11), 17632-17648.
- S. Sakdarat, A. Shuyprom, G.K. Waterman, Thai J. Phtopharm., 2008, 13(2), 12.
- M. Liza, R.A. Rahman, A. Rahmat, I. Zaidul, Food Bioprod. Process., 2010, 88, 319-326.
- S.F. Tu, R.H. Liu, Y.B. Cheng, Y.M. Hsu, Y.C. Du, M. El-Shazly, Molecules., 2014, 9(12), 20382-20390.
- F.H. Ko, G. Hsiao, Y.H. KuO, Free Radic. Biol. Med., 1997, 22, 215-222.
- J.A. Manthey, K. Grohmann, N. Guthrie, Curr. Med. Chem., 2001, 8, 135-153.
- A. Pekal, K. Pyrzynska, Food Anal. Methods, 2014, 7, 1776-1782.
- D. Tsikas, D., 2007, J. Chromatogr., 2007, 851(1-2), 51-70.
- M. Zohra, A. Fawzia, J. Pharma Sci. Res., 2014, 495-450.
- L. Chutima, T. Phaechamud, K. Yodkhum, P. Wetwitayaklung, Res. J. Pharm. Biol. Chem. Sci., 2012, 3(1), 725-739.
- L.W. Chan, E.L. Cheah, C.L. Saw, W. Weng, P.W. Heng, Chin. Med., 2008, 3(15), 15.
- N.S. Yaacob, N.N. Kamal, M.N. Norazmi, BMC Complement. Altern. Med., 2014, 14, 252.
- I.M. Nor Qhairul, M. Fadzelly, Int. Food Res. J., 2013, 20, 307-312.
- B.N. Vaidya. T.A. Brearley, N. Joshee, J. Med. Active Plants, 2014, 2(3), 42-59.
- J.R. Steinert, T. Chernova, I.D. Forsythe, The Neuroscientist, 2010, 16, 435-452.
- L.A. Garcia, E. Guillamon, A. Villares A. Mauricio, J.A. Rostagno, Inflamm. Res., 2009, 58, 537-552.
- M.A. Ebrahimzadeh, S.F. Nabavi, S.M. Nabavi, B. Eslami, Cent. Eur. J. Biol., 2010, 5, 338-345.
- S. Nabavi, M. Ebrahimzadeh M, Nabavi, B. Eslami, A. Dehpour, Eur. Rev. Med. Pharmacol. Sci., 2011, 15, 157-164.
- C.G. Zou, N.S. Agar, G.L. Jones, Life Sci., 2000, 69, 75-86.
- V.M. Ghandhi, K.M. Cherian, Toxicol. In Vitro., 2000, 14(6), 513-516.
- S. Deng, B.J. West, C.J. Jensen, Adv J. Food Sci Technol., 2011, 3(2), 155-159.