Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 4
Design and Synthesis of Medicinally Privileged Heterocycles and their Antimicrobial Activity
Mohd. Shahnawaz Khan1* and Daya Saran Seth2
1Department of Chemistry, JK. Lakshmipat University Jaipur (Raj)-302026, India
2Department of Chemistry, School of Chemical Sciences, St. John's College, Agra (UP)-282002, India
- *Corresponding Author:
- Mohd. Shahnawaz Khan
Department of Chemistry
JK. Lakshmipat University Jaipur (Raj)-302026, India
Abstract
In the present synthesis work, we were synthesized viable bio active nitrogen and oxygen atom containing heterocyclic compounds like pyrazole, pyrazolone, coumarin, azo coumarin, and naphthyl coumarin. A metal catalyst free reaction to afford the targeted compounds (1-7) in good yields. All three types of coumarins were synthesized in one step ecofriendly approach under solvent free condition. The beauty of this work easily available primary material, gentle conditions, smooth operation and economical cost. The synthesized compound structures were confirmed by advance spectroscopic techniques and elemental analysis. Besides, compounds also evaluated for their in vitro antimicrobial activity.
Keywords
Synthesis, Coumarins, Pyrazole, Pyrazolone, Antimicrobial activity
Introduction
In various literatures have been found that the morbidity, mortality and costs related to the treatment of infectious diseases have been increased by antimicrobial resistance. The cumbersome challenge for researcher and scientist that particularly multiple resistance in bacterial strains that have spread widely [1]. The development of drug resistance infective agents reveals new threats to society which are increased antibiotic use. Indeed, it is an urgent need for a discovery of new class of antimicrobial agents having new and diverse structures from those of existing agents to treat multiple resistant infections [2-4]. Heterocyclic chemistry is one of the most valuable distinctive source of novel compounds. The compounds having heterocyclic ring system continue to attract academia and researchers due to their diverse biological activities [5]. Natural and synthetic organic molecules which possess coumarin, pyrazole and pyrazolone nucleus have significant pharmacological properties many literatures reported the relevant and recent accomplishment in the chemical and biological activities of coumarin, pyrazole and pyrazolone derivatives in drug discovery [6-24]. The various biological activities reported such as antimicrobial [6] antibacterial [7], antifungal [8], antioxidant [9] anti-inflammatory [15,20], analgesic agents [20].
Indeed, our interest to design and synthesis of coumarin, pyrazole and pyrazolone derivatives which have been shown remarkable antimicrobial activity.
Materials and Methods
All chemicals used for synthesis of compounds were obtained from Rankem, CDH and Spectrochem Pvt. Ltd. (India). Silica-gel-coated Aluminium plates (Merck) used to find the purity of synthesized compounds with different solvent systems using iodine vapors as spot detecting agent. Melting points were determined in open capillaries and are uncorrected. The IR spectra recorded on Perkin Elmer Spectrum RX1, FTIR spectrophotometer using KBr discs. Proton spectra were recorded in Bruker DRX-300 (300 MHz) by using Deuterated Dimethyl Sulfoxide (DMSO-d6) as solvent and chemical shift (δ) is reported in ppm using Tetramethylsilane (TMS) as an internal standard. Elemental analysis was performed on Elementor Vario EL III [24].
The targeted compounds were synthesized in following steps
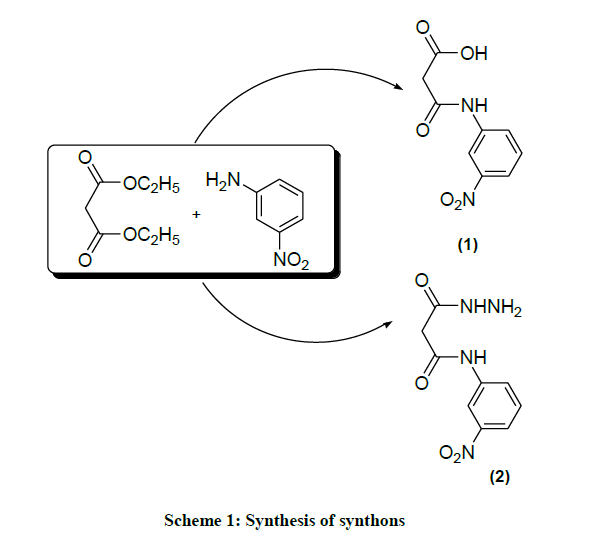
3-(3-nitrophenylamino)-3-oxopropanoic acid (1): The m-nitro aniline (0.05 mol, 6.9 g), diethyl malonate (0.05 mol) was added and gentle reflux for 2.5 h and the mixture cool it at RT, added 10 ml ethanol and kept this solution for overnight and filtered. The filtrate was mixed with 20% sodium carbonate solution (20 ml) and the dry steam was blown into a mixture for 1 h. This was filtered off and the filtrate was acidified with conc. HCl till white precipitate. It was filtered and washes with double distilled water and recrystallized from absolute ethanol [24]. White powder; Yield: 71.11%; M.p. 120-122°C; FTIR (KBr, cm-1): 737 (C-H bending), 1351 (C-NO2 str.), 1433 (CH2 asym. def.), 1487 (phenyl ring str.), 1594 (-CONH- str.), 2729 (O-H str.), 2823 (C-H str. aliphatic.), 3356 (N-H str.); 1H-NMR (DMSO-d6, δ ppm): 3.63 (s, 2H), 7.60-7.95 (m, 4H), 8.63 (s, 1H), 10.65 (s, 1H). Anal. Calcd. for C9H8O5N2 (224.0): C, 48.21; H, 3.57; N, 12.50; Found: C, 48.30; H, 3.55; N, 12.53.
3-(3-nitrophenylamino)-3-oxopropane hydrazide (2): The m-nitro aniline (0.05 mol, 6.9 g), diethyl malonate (0.05 mol) was added and gentle refluxed for 2.5 h and the mixture was cooled at RT and 10 ml absolute ethanol was added to the filtrate after the solution was concentrated, cool it at room temperature (RT), further added 20 ml ethanol and hydrazine hydrate (0.05 mol, 99%) and refluxed for 45-50 min on the steam bath. The white crystalline solid was separated at ambient temperature filtered and recrystallized from absolute ethanol [24]. White crystal; Yield: 85.96%; M.p 152-154°C; FTIR (KBr, cm-1) 741 (C-H bending), 1012 (C-N str. & ArC-H bending), 1511 (-N-N- str.), 1595 (-CONH- str.), 1629 (C=C str.), 2810 (C-H str.), 3478 (N-H str.); 1H-NMR (DMSO-d6, δ ppm): 3.54 (s, 2H), 4.31 (s, 2H), 5.80 (s, 1H), 7.80-8.56 (m, 4H), 9.21 (s, 1H). Aanal. Calcd. for C9H10O4N4 (238.0): C, 45.38; H, 4.20; N, 23.53; Found: C, 45.40; H, 4.18; N, 23.55.
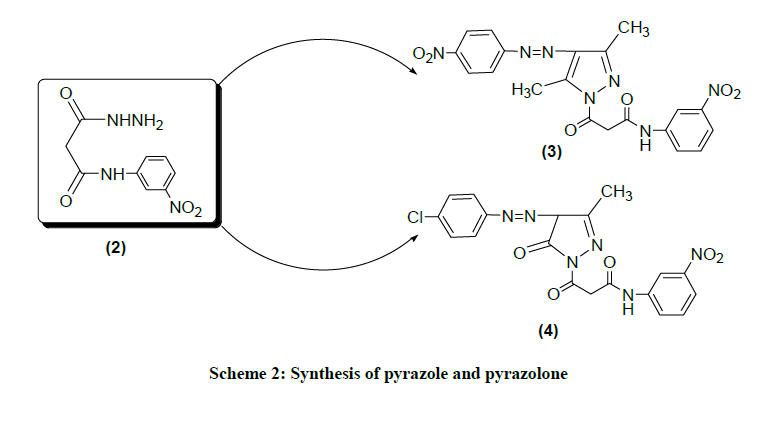
1-(3-nitroanilinomalonyl)-3,5-dimethyl-4-(4-nitrophenylazo) pyrazole (3): The mixture of compound (2) (0.001 mole, 0.23 g), 2,3-diketo-3- (4-nitrophenylazo)pentane (0.001 mol, 0.25 g) was placed in round bottom flask, 20 ml of ethanol and 3-5 drops of glacial acetic acid were added and gentle refluxed it for 4-5 h. The completion of reaction monitored by TLC. A mixture cooled at ambient room temperature and solid product separated, filtered and was recrystallize from methanol and absolute ethanol. Dark yellow powder; Yield: 78.26%; M.p. 214-216°C.; FTIR (KBr, cm-1) 1029 (C-N str. & ArC-H bend.), 1260 (-N-N- str.), 1335 (C-NO2 str.), 1520 (-N=N- str.), 1596 (CONH str.), 1671 (C=O str.), 2827 (C-H str.), 3436 (N-H str.); 1H NMR (DMSO-d6, δ ppm): 2.80 (s, 3H), 2.71 (s, 3H), 3.32 (s, 2H), 7.71-8.29 (m, 8H), 8.63 (s, 1H). Anal. Calcd. for C20H17O6N7 (331.0): C, 53.21; H, 3.77; N, 21.73; Found: C, 53.25; H, 3.75; N, 21.33.
1-(3-nitroanilinomalonyl)-3-methyl-4-(4-chlorophenylazo) pyrazolin-5-one (4): The mixture of compound (2) (0.001 mol, 0.23 g), ethyl-2,3- dioxobutyrate-2-(4-chlorophenylazo)hydrazone (0.001 mol, 0.27 g) was placed in round bottom flask 20 ml of ethanol and 5 drops of glacial acetic acid was added and gentle refluxed it for 3-4 h. The completion of reaction monitored by TLC. The mixture cooled at ambient room temperature and solid product separated, filtered and was recrystallize from methanol and absolute ethanol. Dark yellow powder; Yield: 73.33%; M.p. 186-189°C.; FTIR (KBr, cm-1) 810 (C-Cl str.), 1029 (C-N str. & ArC-H bend.), 1252 (-N-N- str.), 1350 (C-NO2 str.), 1487 (-N=N- str.), 1593 (-CONH- str.), 1668 (C=O str.), 2806 (C-H str.), 3423 (N-H str.); 1H-NMR (DMSO-d6, δ ppm): 1.06 (s, 3H), 2.26 (s, 1H), 3.37 (s, 2H), 7.48-7.95 (m, 8H), 8.59 (s, 1H). Anal. Calcd. for C19H15O5N6Cl (442.5): C, 51.53; H, 3.39; N, 18.98; Found: C, 51.52; H, 3.40; N, 18.98.
General procedure for the synthesis of various coumarins
The mixture of compound (1) (0.001 mol) and substituted aldehyde, azo aldehyde & hydroxyl naphthaldehyde (0.001 mol) were placed in a round bottom flask and 3 drops of pyridine used as a catalyst this mixture was heated in an oil bath at 110-115°C for 4 h. A solid mixture first melted and solidified, later a solid mass was boiling with 15 ml absolute ethanol and filtered hot and recrystallize by absolute ethanol.
8-bromo-6-chlorocoumarin-3-carboxy-(3'-nitro)phenylamide (5): Yellow powder; Yield: 60.46%; M.p. 200-201°C.; FTIR: (KBr, cm-1) 698 (C-Cl str.), 760 (C-Br str.), 1255 (NO2 str.), 1356 (C-NO2 str.), 1590 (-CONH- str.), 1720 (-CO-O- & C=O str.), 3015 (C-H aromatic str.), 3450 (N-H str.); 1H-NMR (DMSO-d6, δ ppm): 7.25-8.76 (m, 6H), 8.10 (s, 1H), 8.26 (s, 1H). Analysis for C16H8O5N2ClBr (423.4): C, 45.44; H, 1.89; N, 6.63; Found: C, 45.46; H, 1.88; N, 6.60.
2-hydroxynaphthylcoumarin-3-carboxy-(3'-nitro)phenylamide (6): White powder; Yield: 65.71%; M.p. >300°C; FTIR (KBr, cm-1) 1231 (NO2 str.), 1351 (C-NO2 str.), 1592 (-CONH- str.), 1707 (-CO-O- & C=O str.), 3020 (C-H aromatic str.), 3464 (N-H str.); 1H NMR (DMSO-d6, δ ppm): 6.95-8.70 (m, 10H), 8.75 (s, 1H), 8.90 (s, 1H). Anal. Calcd. for C20H12O5N2 (360.0): C, 66.67; H, 3.33; N,7.78; Found: C, 66.64; H, 3.30; N, 7.75.
6-(3'-methoxy)phenylazocoumarin-3-carboxy-(3'-nitro)phenyl amide (7): Light yellow powder; Yield: 77.27%; M.p. >300°C; FTIR: (KBr, cm-1) 1252 (NO2 str.), 1351 (C-NO2 str.), 1460 (-N=N- str.), 1591 (-CONH-str.), 1711 (-CO-O- & C=O str.), 2819 (O-CH3 str.), 3010 (C-H aromatic str.), 3455 (N-H str.); 1H-NMR (DMSO-d6, δ ppm): 3.83 (s, 3H), 7.21-8.85 (m, 11H), 8.53 (s, 1H), 8.33 (s, 1H). Anal. Calcd. for C23H16O6N4 (444.0): C, 62.16; H, 3.60; N, 12.61; Found: C, 62.15; H, 3.55; N, 12.60.
Antibacterial activity
The agar well diffusion method was applied for evaluation of antimicrobial activity of all targeted compounds. A broth culture kept overnight and the bacterial strains were adjusted 108 colony forming units (Colony Forming Unit per ml) and 100 μl of diluted inoculum was spread over the Petri plates containing 25 ml of nutrient agar media. The eight wells of 8 mm diameter were made on each plate using a sterile cork borer. The synthesized compounds were dissolved in DMSO and the wells were filled with 100 μl of the tested compound having a concentration 4 mg/ml. The petri plates were incubated at 30°C for 48-72 h. The antibacterial activity was evaluated by measuring an inhibition zone of bacteria surrounding the wells after 48 h and 72 h. A standard drug Streptomycin (4.0 mg/ml) used as positive control of bacteria. Dimethyl Sulfoxide was used as negative control and did not show any inhibition zone and the experiments were performed in triplicates [21,23].
Antifungal activity
The agar well diffusion method was used for the evaluation of antifungal activity of the compounds. A broth culture kept overnight of the fungal strains were adjusted 108 colony forming units (Colony Forming Unit per ml) and 100 μl of diluted inoculum was spread over the Petri plates containing 25 ml of Sabouraud's dextrose agar media (pH 5.6). The eight wells of 8 mm diameter were made on each plate using a sterile cork borer. The synthesized compounds were dissolved in dimethyl sulfoxide (DMSO) and the wells were filled with 100 μl of the tested compound having a concentration 4 mg/ml. The petri plates were incubated at 30°C for 48-72 h. The antifungal activity was evaluated by measuring an inhibition zone of fungi surrounding the wells after 48 h and 72 h. A standard drug Fluconazole (4 mg/ml) served as positive antifungal control. DMSO used a negative control which did not shown any inhibition zone and the experiments were performed in triplicates [21,23].
Result and Discussion
Chemistry
This unique work contained the designed, synthesis and spectral identification of malonamic acid, malonamic acid hydrazide, coumarins, pyrazole and pyrazolone compounds. All synthesized compounds are stable solids and dissolve in DMSO at room temperature. The final structure of targeted compounds was established by analytical and advance spectroscopic techniques. In first step to synthesize the synthons i.e. malanomic acid and malanomic acid hydrazide by condensation of diethyl malonate and m-nitroaniline in absence of catalyst. The reactants were gentle refluxed and it was concentrated at room temperature. The filtrate was mixed with sodium carbonate solution and the steam was blown through it. On cooling, some dianilide got separated. This was filtered off and the filtrate was acidified with conc. HCl to afford white precipitate N-(3-nitro)phenyl malonamic acid (1) it was filtered and recrystallize by absolute ethanol. The compound (1) showed important IR absorption bands at 1594 cm-1 a band, at 2729 cm-1 a band at 3355 cm-1 which refer (-CONH-) group, (O-H) group and (N-H) group, respectively. The 1H-NMR spectra of compound showed a signal of amide proton a singlet at 8.63 ppm and for acid proton a singlet at 10.65 ppm. The compound (2) malonamic acid hydrazide obtained by the reaction of above filtrate with hydrazine hydrate (99%). IR spectra showed significant aborption bands at 1595 cm-1, a band at 1511 cm-1 and a band at 3478 cm-1 which refer to (CONH) group, (-N-N-) group, (N-H) group, respectively. The proton spectra of compound (2) showed a signal singlet at 5.80 ppm to (CONH) amide proton, signal siglet at 4.31 ppm NH2 proton given, signal of (CONH-Ar) group proton singlet at 9.21 ppm. The chemical reactions were shown in Scheme 1. In second step to obtain the target heterocyclic compounds like pyrazole and pyrazolone by condensation of compound (2) and 2,3-diketo-3-(4- nitrophenylazo)pentane and ethyl-2,3-dioxobutyrate-2-(4-chlorophenylazo) hydrazone respectively. Compounds (3) and (4) were shown new IR streching of azo group (-N=N-) at 1520-1487 cm-1 and new band streching of (-N-N-) group at 1260-1252 cm-1, respectively. The proton spectra of compound (3) was shown singlet signal at 2.80 ppm & 2.71 ppm for two methyl group of pyrazole ring and compound (4) was shown singlet signal at 1.06 ppm of one methyl group of pyrazolone ring. The chemical reactions were shown in Scheme 2. In third step to synthesize different type of coumarins by direct condensation reaction of a malonamic acid (1) with 3-bromo-5-chloro salicylaldehyde, 2-hydroxy-1-napthaldehyde and 2-hydroxy-5-(3-methoxy)phenylazobenzaldehyde in presence of 3 drops of pyridine as catalyst to afford halogenated coumarin (5) benzocoumarin (6) and azocoumarin (7), respectively. The IR spectra of compounds (5), (6) & (7) give important absorption bands at 1720 cm-1, 1707 cm-1 and 1711 cm-1 (-CO-O- & C=O) carbonyl-lactone group of coumarin ring, respectively. Similarly other band appear at 1590 cm-1, 1592 cm-1 and 1591 cm-1 due to (-CONH-) group. The proton spectra of halogenated coumarin appeared in the region of 7.25-8.76 ppm for aromatic protons, 8.10 ppm for amide proton, singlet signal at 8.26 ppm for C4 proton of coumarin ring and the proton spectra of napthylcoumarin showed the characterstic signals at 8.70-6.95 ppm for aromatic ring protons, signal at 8.75 ppm for amide proton, 8.90 ppm signal for C4 proton of coumarin ring. The 1H-NMR spectra of showed the characterstic signals of proton in compound (7) signal at 8.85-7.21 ppm for aromatic ring proton, signal at 3.83 ppm for methoxy proton, signal at 8.53 ppm for secondary amide proton, signal at 8.33 ppm for C4 proton of coumarin ring. The chemical reaction summarized in Scheme 3. The green synthesis of targeted coumarins via knoevenagel condensation reacton to affords coumarin derivatives in excellent yield.
Biology
Antibacterial activity
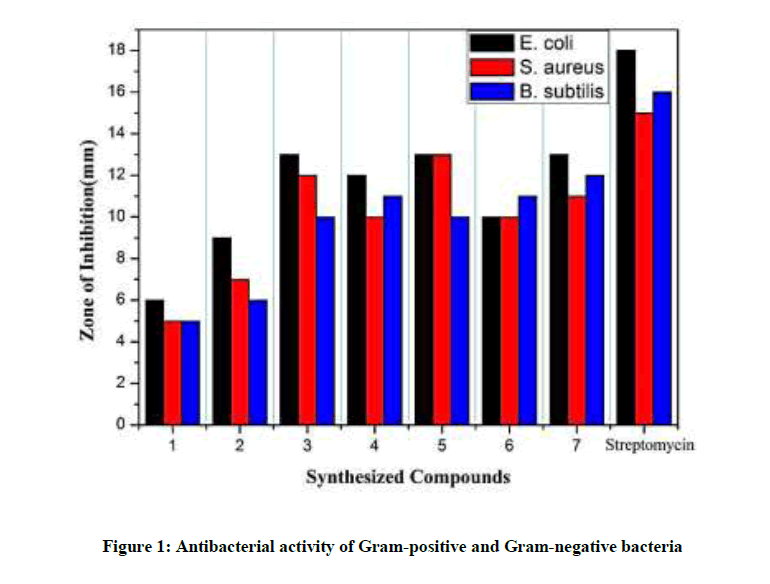
The targeted compounds (1-7) were screened for their in vitro antibacterial activity against three bacteria, Bacillus subtilis and Staphylococcus aureus representing the Gram-(+) bacteria and Escherichia coli representing the Gram-(-) bacteria (Table 1) by agar well diffusion method and streptomycin used as standard drug [21,23]. All the synthesized compounds exhibit moderate to good antibacterial activity against the Gram-(+) as well as the Gram-(-) bacteria. The screened results disclose that the compounds (3), (5) and (7) showed 13 mm inhibition zone against E. coli and a zone of inhibition of 12 mm, 13 mm and 11 mm respectively against S. aureus. Compound (4) and (6) containing pyrazolone and napthyl coumarin ring was found to be showing very moderate antibacterial activity against all three bacterial strains. All tested compounds (1-7) revealed remarkable antibacterial activity against E. coli. All seven compounds were not found potent as the standard drug streptomycin against B. subtilis, S. aureus and E. coli bacteria. Therefore, we concluded that all seven compounds are more effective against the Gram-(-) bacteria than the Gram-(+) bacteria (Figure 1).
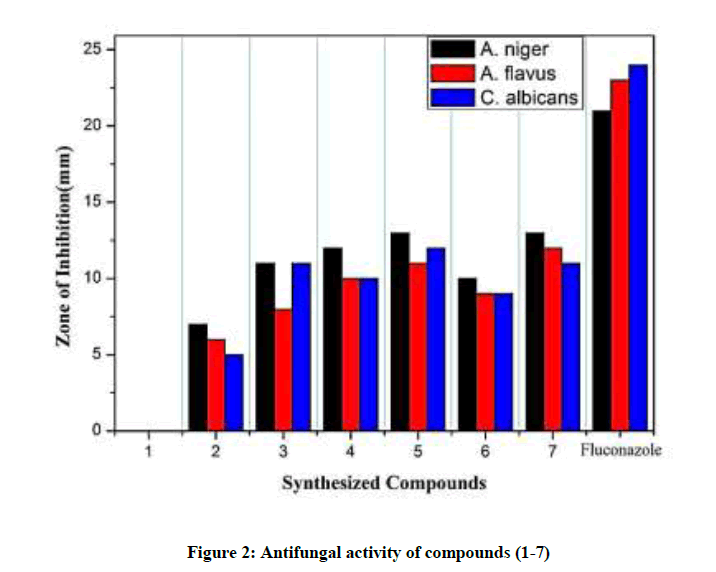
| Compounds code | Antibacterial activity | Antifungal activity | ||||
|---|---|---|---|---|---|---|
| E. coli | S. aureus | B. subtilis | A. niger | A. flavus | C. albicans | |
| 1 | 6 | 5 | 5 | 0 | 0 | 0 |
| 2 | 9 | 7 | 6 | 7 | 6 | 5 |
| 3 | 13 | 12 | 10 | 11 | 8 | 11 |
| 4 | 12 | 10 | 11 | 12 | 10 | 10 |
| 5 | 13 | 13 | 10 | 13 | 11 | 12 |
| 6 | 10 | 10 | 11 | 10 | 9 | 9 |
| 7 | 13 | 11 | 12 | 13 | 12 | 11 |
| Streptomycin | 18 | 15 | 16 | NA | NA | NA |
| Fluconazole | NA | NA | NA | 21 | 23 | 24 |
| Control (DMSO) | 0 | 0 | 0 | 0 | 0 | 0 |
**Zone of inhibition in (mm)
Table 1: In vitro antimicrobial activities of compounds (1-7)
Antifungal activity
The synthesized compounds were screened for in vitro antifungal activity against three pathogen i.e. Aspergillus niger, Aspergillus flavus and Candida albicans by agar well diffusion method (Table 1). Fluconazole used as standard drug and the tested compounds (1-7) showed moderate to good antifungal activity. The compound (1) did not show antifungal activity against all three pathogenic fungal strains. The synthesized compounds 4 mg/ml was found to be as effective, with a zone of inhibition from 05 mm to 13 mm against A. niger, A. flavus and C. albicans. The screened compounds (4, 5 & 7) showed remarkable zone of inhibition against A. niger and C. Albicans (Figure 2).
Conclusion
Our aim to develop novel, multifaceted and cost-effective method for the synthesis of bioactive compounds. This efficient synthetic methodology very useful to get significant bioactive heterocyclic compounds. During chemical reactions very less amount of catalyst, reagents and solvents used. The synthesized compounds yield was also remarkable. Besides, the beauty of this synthesis for the preparation of various coumarins, we followed the greener synthesis method (i.e. Metal catalyst and solvent free reaction) and got excellent yields of desired compounds without generation of environmentally harmful solid or liquid waste. The designed compounds screened for their antimicrobial activity and results revealed that all organic compounds showed significant antibacterial activity against Gram-negative bacteria and slightly moderate against Gram-positive bacteria. The screened compounds (1-7) showed moderate to good antifungal activity, but the compound (1) did not show antifungal activity against all fungal strains. While the compounds (4, 5 & 7) were found to possess good antifungal activity. The detailed molecular modelling and chemical modifications studies are in progress in our laboratory.
Acknowledgement
We are gratefully acknowledged to Central Drug Research Institute, Lucknow for spectral and microanalysis. We are grateful to Microbiology Department, Gajra Raja Medical College, Gwalior (MP) for providing antimicrobial activity. We are thankful to JK. Lakshmipat University, Jaipur for providing platform for research work.
References
- P.M. Hawkey, A.M. Jones, J. Antimicrob. Chemother., 2009, 64(1), i3-10.
- L. Popiolek, K. Paruch, P. Patrejko, A. Biernausiuk, M. Wujee, J. Iran. Chem. Soc., 2016, 13(10), 1945-1951.
- R.C. Moellering, Int. J. Antimicrob. Agents., 2010, 37(2), 2-9.
- V. Padmavathi, P. Thriveni, G.S. Reddy, D. Deepti, Eur. J. Med. Chem., 2008, 43(5), 917-924.
- S. Singhal, S. Arora, S. Agarwal, R. Sharma, N. Singhal, World. J. Pharm. Pharm. Sci., 2013, 2, 4661-4681.
- M. Cacic, M. Trkovnik, F. Cacic, E. Has-Schon, Molecules., 2006, 11(2), 134-147.
- S.M. de Souza, F.D. Monache, A.Z. Smania Jr., Z. Naturforsch., 2005, 60c, 693-700.
- S. Sardaria, Y. Morib, K. Horitab, R.G. Micetichac, S. Nishibeb, M. Daneshtalaba, Bioorg. Med. Chem., 1999, 7(9), 1933-1940.
- A. Basile, S. Sorbo, V. Spadaro, M. Bruno, A. Maggio, N. Faraone, S. Rosselli, Molecules., 2009, 14(3), 939-952.
- M. Mladenović, N. Vuković, S. Sukdolak, S. Solujić, Molecules., 2010, 15(6), 4294-4308.
- E.M. Sharshira, N.M. Mahrous Hamada, Molecule., 2012, 17, 4962-4971.
- P. Khloya, P. Kumar, A. Mittal, N.K. Aggarwal, P.K. Sharma, Org. Med. Chem. Lett., 2013, 3(1), 9.
- A. Tanitame, Y. Oyamada, J. Med. Chem., 2004, 47, 3693-3696.
- M. Agrawal, P.K. Sonar, S.K. Saraf, Med. Chem. Res., 2012, 21, 3376-3381.
- R.S. Kumara, I.A. Arif, A. Ahamed, A. Idhayadhulla, Saudi J. Biol. Sci., 2016, 23(5), 614-620.
- H.F. Rizk, S.A. Ibrahim, M.A. El-Borai, Arabian. J. Chem., 2017, 10(2), S3303-S3309.
- H.B. Mehta, P.K. Patel, B.C. Dixit, R.B. Dixit, Arabian. J. Chem., 2017, 10(2), S1901-S1912.
- M. Amir, S.A. Javed, M.Z. Hassan, Ind. J. Chem., 2013, 52B, 1493-1499.
- M.S. Karthieyan, B.S. Holla, N.S. Kumari, Eur. J. Med. Chem., 2007, 42(1), 30-36.
- M. Amir, H. Kumar, S.A. Khan, Med. Chem. Lett., 2008, 18(3), 918-922.
- C. Perez, M. Pauli, P. Bazevque, Acta. Biol. Med. Exp., 1990, 15, 113-115.
- J.M. Andrews, J. Antimicrob. Chemother., 2001, 48(1), 5-16.
- P. Khloya, P. Kumar, A. Mittal, N.K. Aggarwal, P.K. Sharma, Org. Med. Chem. Lett., 2013, 3, 9.
- M.S. Khan, D.S. Seth, Conf. Synth. Org. Chem., 2016, 1-30.