Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 4
Dolutegravir, Second Generation Integrase Inhibitor: A New Hope for HIV Patients
Geeta Yadav1* and Piyush Kumar2
1Department of Pharmaceutical Sciences, Manav Bharti University, Solan, India
2National Institute of Pharmaceutical Education and Research (NIPER), Raebareli-229010, Uttar Pradesh, India
- *Corresponding Author:
- Geeta Yadav
Department of Pharmaceutical Sciences
Manav Bharti University
Solan, India
Abstract
Undeterred efforts have been made and will be made in future to make it possible for HIV-infected individuals to achieve the goals of virologic suppression and one more addition to this rigorous exercise is dolutegravir drug. It is a recent integrase inhibitor approved by the United States of Food and Drug Administration (USFDA) for use in the treatment-naïve and treatment-experienced HIV-infected patients. This article has reviewed all the aspects of drug including the structural and functional analyses, in vitro activity, pharmacokinetics, drug-drug interactions, MOA, metabolism, excretion, dosing/adverse effects and resistance profile. Dolutegravir is a potent and well tolerated antiretroviral agent that may play an important role in the treatment of patients harboring resistance to other antiretrovirals. Some new combinations of drug with other antiretrovirals are also in pipeline which may hope to increase the immunologic response of the HIV patients.
Keywords
Dolutegravir, Antiretroviral, Integrase inhibitor, HIV
Introduction
With the use of antiretroviral of high potency, tolerability, and resistance profiles, lifespan time has increased for HIV patients but even after so much advancement in therapy, they are struggling with an unknown fear of death [1,2]. Therefore, the need for new antiretroviral agents still continues to be substantial even after more than 20 years into the era of antiretroviral therapy, which have better tolerability profile, higher barriers to resistance, and less drug-drug interactions. These principles have been inspiring the scientists all over the world to develop new agents that are mainly focused on novel therapeutic targets. The development of drugs targeting on critical steps in the life cycle of HIV-1 are drug classes that include HIV-1 reverse-transcriptase inhibitors (Both nucleoside analogues and non-nucleoside inhibitors), HIV-1 protease inhibitors, and HIV-1 entry inhibitors (Fusion inhibitors and CCR5 antagonists). The newest approved class of drug in HIV treatment is the Integrase Inhibitor (INI).
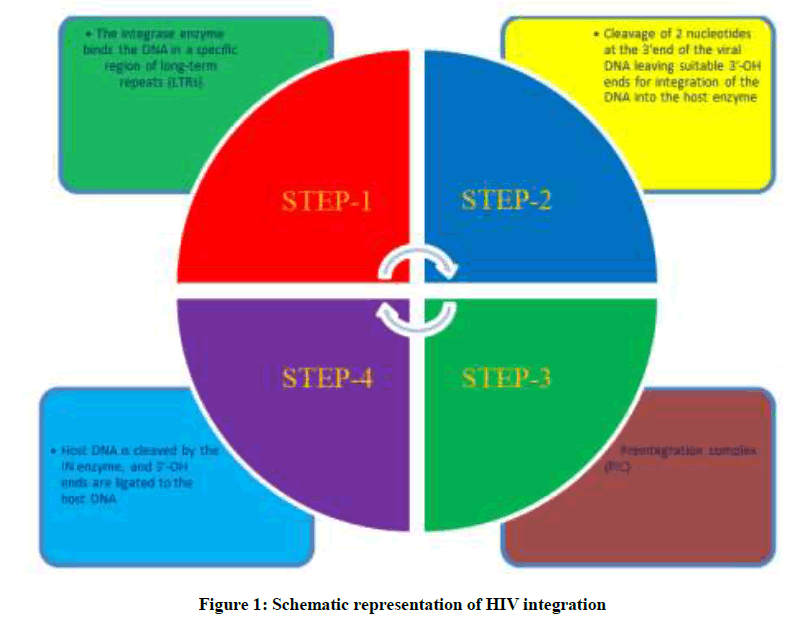
Retroviral DNA Integration with the host DNA is a crucial step in the life cycle of human immunodeficiency virus (HIV) [3], as shown in Figure 1. This integration process is a multistep process initiated by the viral integrase (IN) enzyme to catalyze the insertion of the viral DNA into the host genome. The process of HIV-1 integration occurs through 3 essential steps: formation of the preintegration viral DNA complex, 3’ processing and strand transfer [4]. HIV IN binds at specific sequences in the long terminal repeats (LTRs) of the viral DNA. This DNA binding process takes place in the cytoplasm. Next step is cutting of GT dinucleotides from the 3’ termini of the linear cDNA through a process called 3’ processing. After this step next step is translocation of processed viral DNA into the nucleus, where IN inserts the viral DNA into the host chromosome by a process called strand transfer which is a part of the preintegration complex [4-6].
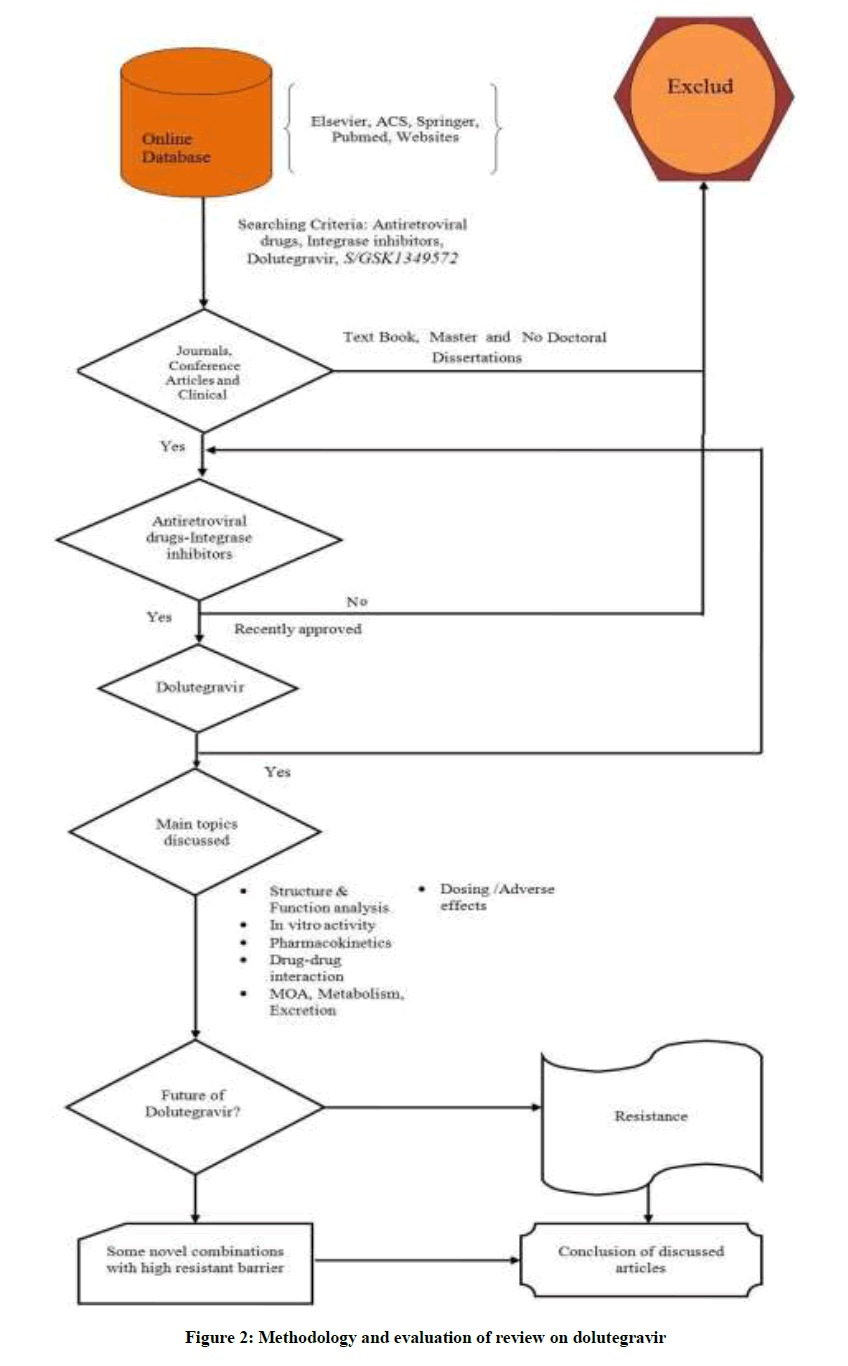
INIs represent a class of drug used in the treatment of HIV infected individuals, blocking HIV genome transfer and integration into the host cell DNA [7]. In this category, first drug which got FDA approval was Raltegravir (RAL) which have been found to be highly convincing in the treatment of antiretroviral- naive and experienced subjects and one more recent drug is Elvitegravir (EVG) [8-12]. But these first-generation INIs also suffer from some severe drawbacks like they share some common resistance pathways and that was witnessed during clinical studies of RAL, subjects were found to have virus with 1 of 3 signature mutational pathways like N155H, Q148H/K/R, or Y143C/H/R, in the integrase enzyme gene [13]. So, in these circumstances continuing RAL treatment may lead to the evaluation of some secondary mutations (Y143 or Q148 pathways) [14]. In addition to this, EVG does not have any activity against RAL-resistant isolates and same case is with RAL [14-16]. Therefore, there is a need for new INIs with high genetic barrier to resistance as well as high anti- HIV activity. So, recent addition for fulfilling all these basic requirements is Dolutegravir (DTG). This review article aims to cover all the aspects related to the dolutegravir which will help the scientists, academicians and common men to satisfy their knowledge pangs, like in vitro activity, pharmacokinetics, drug-drug interactions, MOA, metabolism, excretion, dosing/ adverse effects and resistance profile of dolutegravir as shown in Figure 2, which exemplify methodology and evaluation of dolutegravir with the help of different information sources.
DTG discovered by a Shionogi and GlaxoSmithKline research collaboration, is a second generation novel HIV-1 integrase strand transfer inhibitor having activity against INI resistant viruses. In addition to it, also have favorable pharmacokinetic properties [17-19]. It is generally recommended for use in combination with other antiretroviral agents for the treatment of patients infected with HIV. It is marketed as a small, yellow, 50 mg tablet and there is no specific condition with its consumption (without any regard to time and food).
There are excellent reviews and research papers recently published which sum up the side effects and other clinical phase studies of DTG in combination with other drugs [20,21].
Different aspects of DTG drug
Structural and functional analyses of DTG
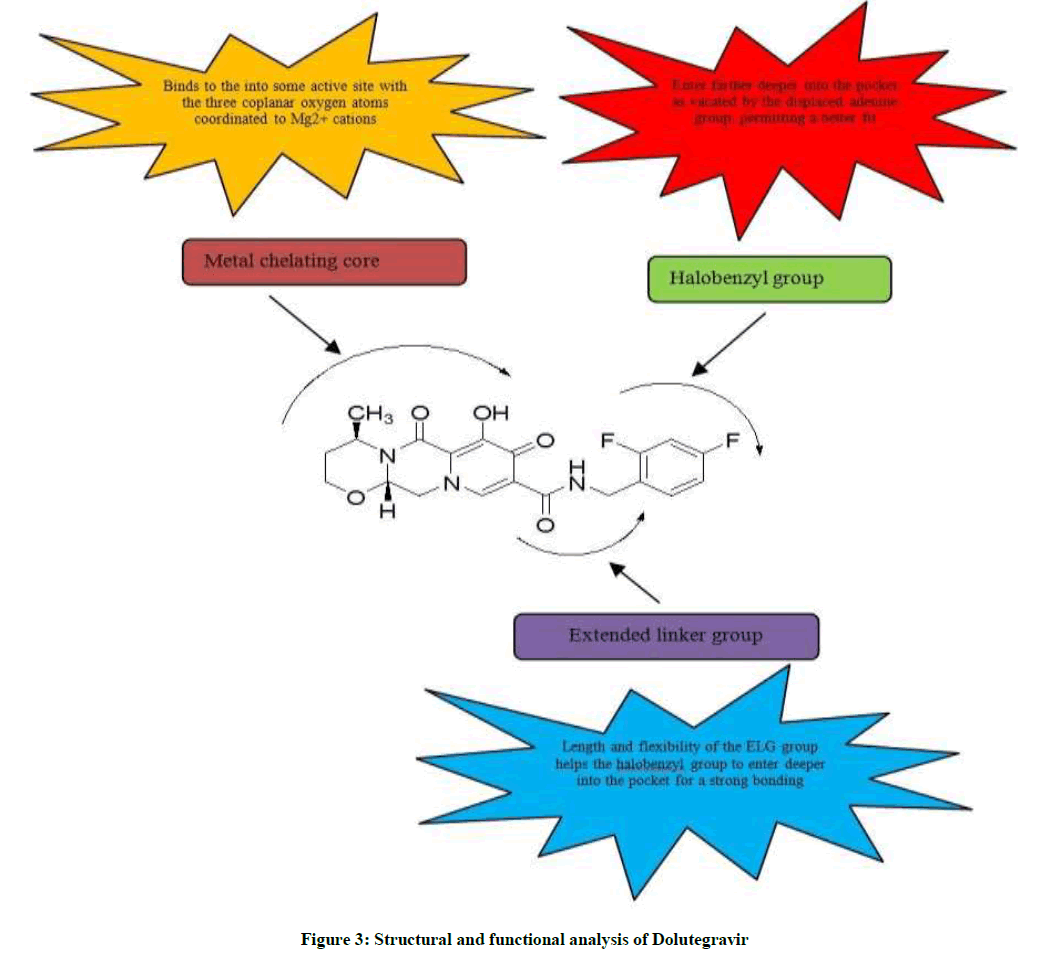
Dolutegravir (DTG, S/GSK1349572) effectively inhibits HIV-1 IN variants which are resistant to the first-generation INIs. Increased potency of DTG is mainly attributable to its structure which shares almost the same space within the IN active site as other INIs and also make strong interactions with the β4-α2 loop of the catalytic core domain. Dolutegravir molecule has been divided into three main structural parts like tricyclic metal-chelating core, difluorophenyl ring and linker group as shown in Figure 3. Tricyclic metal-chelating core binds to the into some active site with the three coplanar oxygen atoms coordinated to Mg2+ cations The extended linker region of DTG allows it to enter farther deeper into the pocket as vacated by the displaced viral DNA base, to make more compatible bonding with the viral DNA [19].
In vitro activity
Dolutegravir has shown potent in vitro activity against many INI-resistant mutants and also against wild-type HIV-1. It has shown potent in vitro activity against HIV-1, with mean EC50 values of 0.5 nM to 2.1 nM, IC50 of 2.7 nM and an IC50 of 2.0 nM in Peripheral Blood Mononuclear Cells (PBMC) and MT-4 cells. It also shows activity against HIV-2 virus with EC50 of 0.09 nM to 0.61 nM in PBMC assays. In variety of cell types cellular toxicity value is in the micromolar range which indicates that the antiviral effect of S/GSK1349572 is not due to cytotoxicity. S/GSK1349572 shows potency against all integrase- resistant single mutants with an FC as high as 3.6-fold. No virus with high resistance was observed in the presence of 32 nM or higher concentrations of S/GSK1349572. In vitro experimental studies witnessed no toxicity when used in combination with other anti-retrovirals, but a synergistic effect was observed with nevirapine, efavirenz, abacavir, stavudine, lopinavir, amprenavir and enfuvirtide drugs and an additive effect when only used in combination with maraviroc. No effect on efficacy was also observed on exposure to the adefovir and ribavirin [22].
Pharmacokinetics
Dolutegravir has a favourable pharmacokinetic profile without requirement of boosters and its terminal half-life is approximately 13-15 h [23,24]. AUC0-24 h and Cmax values are slightly less than the dose in the range of 2-50 mg following single and multiple doses. One notable change is the non-linearness in Cmax and AUC with the increase in dose, so, a twice-daily 50 mg regimen has been evaluated in the phase 3 clinical trial instead of a once-daily 100 mg dose [24-26]. 1.6 μg/ml is the reported geometric mean steady-state concentration at the end of the dosing interval (Ctau) for a 50 mg dose, which was found to be approximately 25-fold higher than the protein-adjusted IC90 (0.064 μg/ml). A monotherapy study of, 10 days of dolutegravir 50 mg daily dose in integrase inhibitor naïve HIV-1-infected subjects demonstrated a reduction in HIV-1 RNA. This reduction was sustained for 4 days after discontinuation of dolutegravir only because of plasma concentrations which remained above the protein adjusted IC90. Variability in exposure was minimum like 50 mg dosing achieved a geometric mean Cmax of 3.34 mg/ml (16% coefficient of variation), an AUC0-24 h of 43.4 mg.h/ml (20% coefficient of variation), a t1/2 of 12.0 h (22% coefficient of variation) and a C24 h of 0.83 mg/ml (26% coefficient of variation) [24-26]. According to reports, a pediatric granule formulation of dolutegravir is currently under development phase and preliminary data investigation also reported the increased exposure of granules when mixed in purified water in comparison to the tablet formulation [25].
Drug-drug interactions
Dolutegravir pharmacokinetic study evaluated the effect of food and found that its absorption is modestly increased with food according to fat content [26]. Fat content affects the absorption of dolutegravir as noticed by the increased median Tmax from 2 h to 5 h for low, moderate and high-fat meals respectively. Whereas dolutegravir AUC increases from 33 to 66% when administered with low-fat (300 kcal, 7% fat), moderate fat (600 kcal, 30% fat) and high fat food (870 kcal, 53% fat), respectively [24,26]. But these changes are not expected to affect safety or efficacy, So, dolutegravir can be prescribed without any regard to food. Dolutegravir causes drug-drug interactions with integrase inhibitors and some other drugs which are shown in Table 1.
| S. No. | Interacting drug class | Interacting drug | Effect on dolutegravir |
|---|---|---|---|
| 1 | Antiretrovirals NRTIs | Tenofovir | No significant effect observed [27] |
| 2 | Antiretrovirals NNRTIs | Efavirenz | DTG AUC, Cmax, and Cmin decreased 57, 39, and 75% [28] |
| Etravirine | DTG AUC, Cmax, and Cmin decreased 70.6, 51.6, and 87.9%. [29] ETR/DRV/r administration results in 25, 11.8, and37.1% decreases in DTG AUC, Cmax, and Cmin |
||
| ETR/LPV/r administration results in 11, 7, and 28% increases in DTG AUC, Cmax, and Cmin [29] | |||
| 3 | Antiretrovirals PIs | Darunavir/r | DTG AUC, Cmax, and Cmin decreased 22, 11, and 38% [30] |
| Atazanavir | DTG AUC, Cmax, and Cmin increased 91, 50, and 180% [31] | ||
| Lopinavir/r | No significant effect observed [30] | ||
| Fosamprenavir | DTG AUC, Cmax, and Cmin decreased 35, 24, and 49% [32] | ||
| Tipranavir | DTG AUC, Cmax, and Cmin decreased 59, 46, and 76% [28] | ||
| 4 | Antituberculosis drugs | Rifampin | DTG AUC and Cmin increased 33 and 22% with DTG 50 mg b.i.d.+ rifampin 600 mg q.d. compared with DTG 50 mg daily [33] |
| Rifabutin | DTG AUC and Cmin decreased 5 and 30%, Cmax increased 15% [34] | ||
| 5 | Acid-reducing agents- PPIs/H2 RA | Omeprazole | No significant effect observed [35] |
| Antacids | DTG AUC, Cmax, and Cmin decreased 73.6, 72.4, and 74.4% [35] |
DTG-Dolutegravir;
ETR-Etravirine;
EVG-Elvitegravir;
LPV-Lopinavir;
NNRTI-Non-nucleoside reverse transcriptase inhibitor-NRTI, Nucleos(t)ide reverse transcriptase inhibitor;
PI-Protease Inhibitor;
PPI-Proton pump inhibitor;
r-Ritonavir;
RAL-Raltegravir
Table 1: Dolutegravir (DTG) drug interaction with integrase inhibitors and other category drugs
Mechanism of action
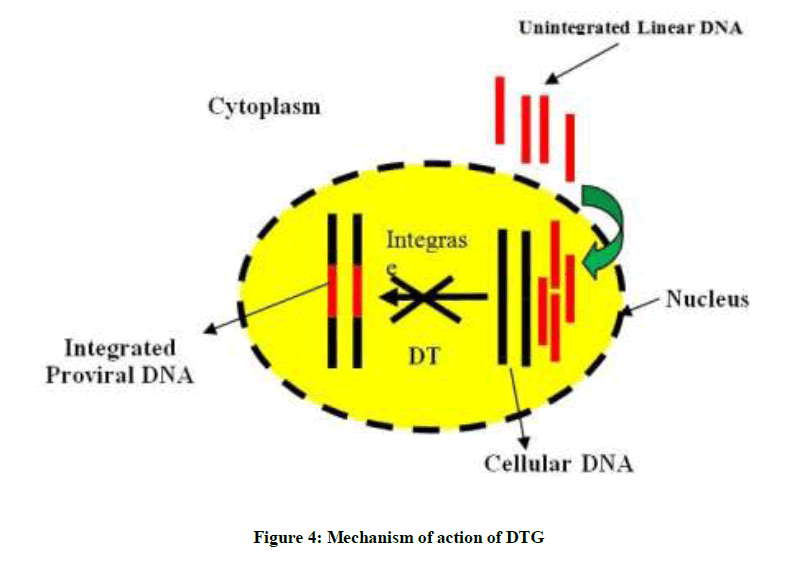
Dolutegravir inhibits HIV integrase enzyme by binding to its specific amino acids in the active site and block the strand transfer step which results in no formation of integrated proviral DNA, which is essential for the HIV replication cycle as demonstrated in Figure 4. In this process, the integrase inhibitor chelate with the two Mg2+ ions in the integrase catalytic active site, unable the integrase enzyme to complete the strand transfer [23]. Inhibition of the integrase strand transfer reaction by DTG is confirmed by an accumulation of 2-long terminal repeat (2-LTR) circles in treated cells at less DTG concentrations in comparison to that cause cell toxicity [36,37].
Metabolism/Excretion
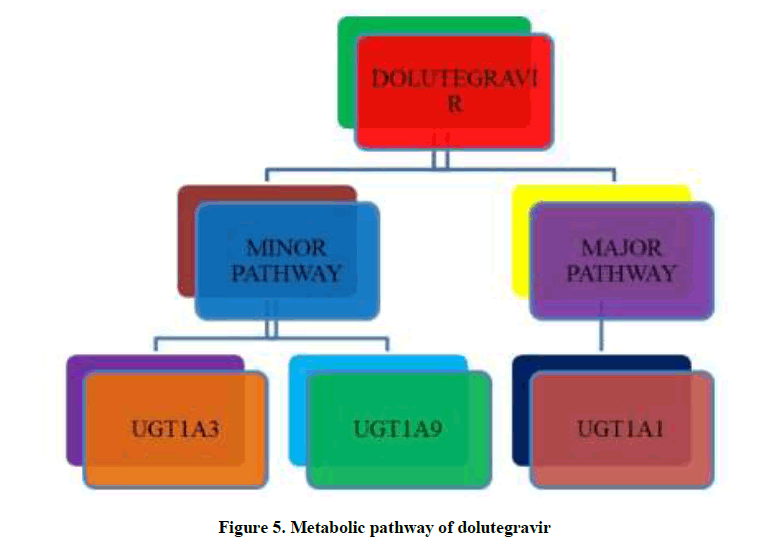
Dolutegravir metabolism occurs through CYP3A4 (UGT1A1 glucuronidation) a major pathway while UGT1A3 and UGT1A9 are only minor pathways, which is catalysed by UDP-glucuronosyl transferase (UGT) 1A1 enzyme as shown in Figure 5. In vitro studies reported that it is not a cytochrome P450 inducer and neither an inhibitor. However, dolutegravir is an OCT2 inhibitor [23-38]. Dolutegravir is also a substrate of UGT1A3, UGT1A9, BCRP, and P-gp [39]. It is the predominant circulating compound in plasma and the renal elimination of unchanged drug is extremely low (< 1% of the dose). Approximately 53% of a dose is recovered completely as DTG in feces and nearly about 31% is excreted in the urine primarily as DTG-glucuronide and other minor metabolites [40].
Dose/Adverse effects
Dolutegravir tablets are usually taken unboosted, orally and without regard to food [41-48]. Different dose combination studies with other drugs are reported to be performed to find the best combination with high resistance barrier as shown in Table 2. The most common adverse effects reported to be associated with dolutegravir Phase III SPRING-2 trial were nausea, headache, diarrhea, nasophryngitis and also a slight increase in creatinine level due to inhibition of creatinine secretion. However, it has no effect on glomerular filtration rate [49,50]. Some common drug-related adverse events were also notified during Phase III VIKING-3 trial in treatment-experienced subjects were diarrhea, nausea, and headache [50].
| S. No. | Phase study | Patients | Dolutegravir vs other drug combinations |
|---|---|---|---|
| 1 | Phase III SPRING-2 Study | Treatment naïve | Dolutegravir 50 mg once daily versus raltegravir 400 mg twice daily, each in combination with either tenofovir DF/emtricitabine (Truvada) or abacavir/lamivudine (Epzicom) [42] |
| 2 | Phase III SINGLE Study | Treatment naïve | Dolutegravir 50 mg in combination with abacavir/lamivudine (Epzicom) once daily versus tenofovir DF/emtricitabine/ efavirenz (Atripla) once daily [43] |
| 3 | Phase III SAILING Study | Treatment experienced, integrase inhibitor-naïve | Dolutegravir 50 mg once daily versus raltegravir 400 mg twice daily, each in combination with background therapy [44] |
| 4 | Phase III VIKING-3 Study | Treatment-experienced with previous or current failure on raltegravir or elvitegravir | Open-label dolutegravir 50 mg twice daily with current failing background regimen for 7 days, then with an optimized background regimen [45] |
| 5 | Phase III VIKING-4 Study | Treatment-experienced with virus resistant to raltegravir and/ or elvitegravir at screening | Dolutegravir 50 mg twice daily versus placebo, each in combination with current failing background regimen for 7 days, then with open-label dolutegravir 50 mg twice daily in combination with an optimized background regimen for both arms [46] |
| 6 | Combination under study | A fixed-dose combination (FDC) tablet (dolutegravir 50 mg abacavir 600 mg/lamivudine 300 mg) and a dolutegravir pediatric granule [47,48] |
Table 2: Dolutegravir (DTG) drug combinations in different phases of the clinical trials
Resistance
DTG has been found to show a higher genetic barrier to resistance than raltegravir and elvitegravir [51,52]. As such resistance mutations associated with dolutegravir have not yet been identified but viruses containing G140S, E138K, R148H, R263K, and G140S/Q148HRK mutations have been found to show some level of resistance to dolutegravir [52,41]. Raltegravir-resistant virus carries a number of mutations, among which mutation at position Q148 had more reduced susceptibility to dolutegravir [53]. In vitro selection studies reported R263K mutation which commonly emerges in integrase in the presence of dolutegravir. R263K confers low-level resistance against dolutegravir and diminishes HIV DNA integration and viral fitness and no secondary mutation H51Y and E138K has been shown to compensate for the defects associated with the R263K primary resistance mutation against dolutegravir. All secondary mutations have a modest effect on resistance against this drug [54,55].
Future of DTG
ViiV Healthcare has requested US regulatory for the approval of a new single-tablet regimen (STR) which is a combination of dolutegravir, lamivudine and abacavir. According to the company reports, a European regulatory application has also been submitted. In the aforementioned trials this combination worked well as separate pills. So, In case of approval, this new formulation will give the first single-pill, once-daily regimen that does not contain tenofovir/emtricitabine and but could be beneficial especially for people suffering from kidney disease or osteoporosis. In inter science Conference on Antimicrobial Agents and Chemotherapy (ICAAC), presenting results also proved the statistical superiority of dolutegravir over darunavir/ritonavir. Based on these findings the researchers conclude that dolutegravir may provide a potent and well-tolerated single pill as a new option for first-line HIV treatment [56].
Conclusion
HIV-1 integrase is a unique target for antiretroviral therapy. Dolutegravir, a once-daily HIV strand integrase inhibitor currently approved for HIV-1 infected patients, provides equivalent antiviral efficacy and better tolerability in comparison to the already approved antiretroviral drugs. Efforts are ongoing for the approval of new Single-tablet Regimen (STR) containing dolutegravir, abacavir and lamivudine and also it would reduce the number of pills required for effective antiretroviral treatment. Because of its unique mechanism of action, demonstrated virologic activity, resistance profile and tolerability, it is a significant advancement in HIV-1 therapeutics which will help HIV patients in long run.
Acknowledgments
One of the authors acknowledges Manav Bharti University Solan for the support and opportunity provided to the author.
References
- E. Serrao, S. Odde, K. Ramkumar, N. Neamati, Retrovir., 2009, 6, 1-25.
- T. Hawkins, Antivir. Res., 2010, 85, 201-209.
- H.M. Sakai, J. Kawamura, S. Sakuragi, R. Sakuragi, A. Shibata, N. Ishimoto, S. Ono, Ueda, A. Adachi, J. Virol., 1993, 67, 1169-1174.
- E. Asante-Appiah, A.M. Skalka, Antiviral Res., 1997, 36, 139-156.
- A. Engelman, K. Mizuuchi, R. Craigie, Cell., 1991, 67, 1211-1221.
- D. Esposito, R. Craigie, Adv. Virus Res., 1999, 52, 319-333.
- W.G. Powderly, J. Antimicrob. Chemother., 2010, 65, 2485-2458.
- M. Markowitz, B.Y. Nguyen, E. Gotuzzo, F. Mendo, W. Ratanasuwan, C. Kovacs, H. Teppler, J. Acquir. Immune. Defic. Syndr., 2007, 46, 125-133.
- R.T. Steigbigel, D.A. Cooper, P.N. Kumar, J.E. Eron, M. Schechter, M. Markowitz, H.N. Teppler, Engl. J. Med., 2008, 359, 339-354.
- J.L. Mbisa, S.A. Martin, P.A. Cane, Infect. Drug Resist., 2011, 4, 65-76.
- E. DeJesus, D. Berger, M. Markowitz, C. Cohen, T. Hawkins, P. Ruane, B.P. Kearney, J. Acquir. Immune Defic. Syndr., 2006, 43, 1-5.
- A.R. Zolopa, D.S. Berger, H. Lampiris, L. Zhong, S.L. Chuck, J.V. Enejosa, A.K. Cheng, J. Infect. Dis., 2010, 201, 814-822.
- D.A. Cooper, R.T. Steigbigel, J.M. Gatell, J.K. Rockstroh, C. Katlama, P. Yeni, B.Y. Nguyen, N. Engl. J. Med., 2008, 359, 355-365.
- E. DeJesus, C. Cohen, R. Elion, 4th IAS Conference, Sydney, Australia, 2007, 22-25.
- J. Marinello, C. Marchand, B.T. Mott, A. Bain, C.J. Thomas, Y. Pommier, Biochem., 2008, 47, 9345-9354.
- C. Garrido, J. Villacian, N. Zahonero, Antimicrob. Agents Chemother., 2012, 56, 2873-2878.
- A. Sato, M. Kobayashi, T. Yoshinaga, T. Fujiwara, M. Underwood, B. Johns, Poster presented at the 5th IAS Conference, Cape Town, South Africa, 19-22 July 2009.
- M. Underwood, B. Johns, A. Sato, T. Fujiwara, W. Spreen, Poster presented at the 5th IAS Conference, Cape Town, South Africa, 19-22 July 2009.
- S. Hare, S.J. Smith, M. Métifiot, A. Jaxa-Chamiec, Y. Pommier, S.H. Hughes, P. Cherepanov, Mol. Pharmacol., 2011, 80(4), 565-572.
- C. Hoffmann, T. Welz, M. Sabranski, M. Kolb, E. Wolf, H.J. Stellbrink, C. Wyen, HIV Med., 2017, 18(1), 56-63.
- P.E. Sax, E. DeJesus, G. Crofoot, D. Ward, P. Benson, R. Dretler, K. White, The Lancet HIV., 2017, 4(4), 154-160.
- M. Kobayashi, T. Yoshinaga, T. Seki, C. Wakasa-Morimoto, K.W. Brown, R. Ferris, T. Fujiwara, Antimicrob. Agents Chemother., 2011, 55(2), 813-821.
- S. Min, I. Song, J. Borland, S. Chen, Y. Lou, T. Fujiwara, S.C. Piscitelli, Antimicrob. Agents Chemother., 2010, 54, 254-258.
- S. Min, L. Sloan, E. DeJesus, T. Hawkins, L. McCurdy, I. Song, J. Lalezari, AIDS., 2011, 25, 1737-1745.
- P. Patel, I. Song, J. Borland, 19th Conference on Retroviruses and Opportunistic Infections, Seattle, WA, March 5-8, 2012.
- I. Song, J. Borland, S. Chen, P. Patel, T. Wajima, A. Peppercorn, S.C. Piscitelli, Antimicrob. Agents Chemother., 2012, 56(3), 1627-1629.
- I. Song, S.S. Min, J. Borland, Y. Lou, S. Chen, P. Patel, T. Ischibashi, S.C. Piscitelli, J. Acquir. Immune Defic. Syndr., 2010, 55, 365-367.
- I. Song, J. Borland, Y. Lou, 12th International Workshop on Clinical Pharmacology of HIV Therapy; Miami, FL, April 13-15, 2011.
- I. Song, J. Borland, S. Min, P. Patel, T. Ischibashi, S.C. Piscitelli, Antimicrob. Agents Chemother., 2011, 55, 3517-3521.
- I. Song, S.S. Min, J. Borland, P. Patel, T. Ischibashi, S.C. Piscitelli, J. Clin. Pharmacol., 2011, 51, 237-242.
- I. Song, J. Borland, S. Chen, P. Patel, T. Ischibashi, S.C. Piscitelli, Br. J. Clin. Pharmacol., 2011, 72, 103-108.
- I. Song, J. Borland, S. Chen, 51st Interscience Conference on Antimicrobial Agents and Chemotherapy; Chicago, IL, September 17-20, 2011.
- K.E. Dooley, E. Purdy, P. Sayre, 19th Conference on Retroviruses and Opportunistic Infections; Seattle, WA, March 5-8, 2012.
- K.E. Dooley, P. Sayre, J. Borland, 13th International Workshop on Clinical Pharmacology of HIV Therapy; Barcelona, Spain, April 16-18, 2012.
- P. Patel, I. Song, J. Borland, J. Antimicrob. Chemother., 2011, 66, 1567-1572.
- T. Bar-Magen, R.D. Sloan, V.H. Faltenbacher, D.A. Donahue, B.D. Kuhl, M. Oliveira, H. Xu, M.A.Wainberg, Retrovirol., 2009, 6, 103.
- R.D. Sloan, M.A. Wainberg, Retrovirol., 2011, 8, 52.
- I. Song, J. Borland, S. Chen, 13th International Workshop on Clinical Pharmacology of HIV Therapy; Barcelona, Spain, March 16-18, 2012.
- ViiV Healthcare ULC. Tivicay (dolutegravir) Prescribing Information. Research Triangle Park, NC August, 2013.
- S. Castellino, L. Moss, D. Wagner, J. Borland, I. Song, S. Chen, Y. Lou, S. Min, I. Goljer, A. Culp, S.C. Piscitelli, P.M. Savina, Antimicrob. Agents Chemother., 2013, 57(8), 3536-3546.
- P.K. Quashie, R.D. Sloan, M.A. Wainberg, BMC Med., 2012, 10, 34.
- I. Song, J. Borland, S. Chen, Antimicrob. Agents Chemother., 2012, 56(3), 1627-1629.
- ViiV Healthcare. A Randomized, Double Blind Study of the Safety and Efficacy of GSK1349572 50 mg Once Daily to Raltegravir 400 mg Twice Daily Both Administered With Fixed-dose Dual Nucleoside Reverse Transcriptase Inhibitor Therapy Over 96 Weeks in HIV-1 Infected Antiretroviral Therapy Naive Adult Subjects. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on October 14, 2010. NLM Identifier: NCT01227824. Last accessed June 20, 2017.
- ViiV Healthcare. A Randomized, Double-Blind Study of the Safety and Efficacy of GSK1349572 Plus Abacavir/Lamivudine Fixed-Dose Combination Therapy Administered Once Daily Compared to Atripla Over 96 Weeks in HIV-1 Infected Antiretroviral Therapy Naive Adult Subjects. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on December 16, 2010. NLM Identifier: NCT01263015. Last accessed June 20, 2017.
- ViiV Healthcare. A Randomized, Double-blind Study of the Safety and Efficacy of GSK1349572 50 mg Once Daily Versus Raltegravir 400 mg Twice Daily, Both Administered With an Investigator-selected Background Regimen Over 48 Weeks in HIV-1 Infected, Integrase Inhibitor-naive, Antiretroviral-Experienced Adults. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on October 21, 2010. NLM Identifier: NCT01231516. Last accessed June 20, 2017.
- ViiV Healthcare. A Phase III Study to Demonstrate the Antiviral Activity and Safety of Dolutegravir in HIV-1 Infected Adult Subjects with Treatment Failure on an Integrase Inhibitor Containing Regimen. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on March 31, 2011. NLM Identifier: NCT01328041. Last accessed June 20, 2017.
- ViiV Healthcare. A Phase III Randomized, Double-blind Trial Investigating the Activity of Dolutegravir 50 mg BID vs Placebo Over 7 Days in HIV-1-infected Subjects With RAL/ELV Resistance, Followed by an Open-label Phase With an Optimized Background Regimen. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on March 29, 2012. NLM Identifier: NCT01568892. Last accessed June 2, 2017.
- ViiV Healthcare. A Randomized, Open-Label, Single-Dose, 3-Period, Crossover Evaluation of the Relative Bioavailability of Two Experimental Fixed-Dose Combination Tablet Formulations of Dolutegravir 50 mg/Abacavir 600 mg/Lamivudine 300 mg Compared to Co-Administered Dolutegravir 50 mg and EPZICOM™ (Abacavir 600 mg/Lamivudine 300 mg) Tablets in Healthy Adult Subjects. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on May 26, 2011. NLM Identifier: NCT01366547. Last accessed June 21, 2017.
- F. Raffi, A. Rachlis, H.J. Stellbrink, 19th International AIDS Conference; Washington DC, July 22-27, 2012.
- F. Raffi, A. Rachlis, H.J. Stellbrink, 19th International AIDS Conference; July 22-27, 2012.
- S. Walmsley, A. Antela, N. Clumeck, 52nd Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), San Francisco, CA, September 9-12, 2012.
- M.W. Tang, R.W. Shafer, Drugs. 2012, 72(9), 1-25.
- M.R. Underwood, B.A. Johns, A. Sato, J.N. Martin, S.G. Deeks, T. Fujiwara, J AIDS., 2012, 61(3), 297-301.
- T. Mesplède, P.K. Quashie, N. Osman, Y. Han, D.N. Singhroy, Y. Lie, C.J. Petropoulos, W. Huang, M.A. Wainberg, Retrovirol., 2013, 10, 22.
- P.K. Quashie, T. Mesplède, Y.S. Han, M. Oliveira, D. Singhroy, T. Fujiwara, M. Underwood, M.A. Wainberg, J. Virol., 2012, 86(5), 2696-705.
- M. Boyd, D. Cooper, The Lancet Infect. Dis., 2013, 13(11), 908-909.