Mini Review - Der Pharma Chemica ( 2022) Volume 14, Issue 1
Drug Repurposing - An Opportunity in Drug Research
Tanya Gupta1 and Dipankar Nath2*2Department of Chemistry, Shifa Pharmacist Institute, Silchar, Assam, India
Dipankar Nath, Department of Chemistry, Shifa Pharmacist Institute, Silchar, Assam, India, Email: dipankarnath0@gmail.com
Received: 21-Dec-2021, Manuscript No. dpc-22-40350; Accepted Date: Dec 23, 2021 ; Editor assigned: 23-Dec-2021, Pre QC No. dpc-22-40350 (PQ); Reviewed: 08-Jan-2022, QC No. dpc-22-40350; Revised: 14-Jan-2022, Manuscript No. dpc-22-40350 (R); Published: 26-Jan-2022, DOI: 10.4172/0975-413X.14.1.1-9
Abstract
Drug Repurposing opportunities arise from potential observations, discussions, and collaborations, including the development of different meaningful platforms for the identification of drugs and their potential targets and allow the accessing of compounds. Drug Repurposing is an approach that uses the existing approved/failed drug for novel clinical use. This approach has the advantage that the compounds have already undergone different drug discovery phases and have sufficient information related to safety, efficacy, formulation, dose, and potential toxicity. As a result, this strategy is gaining considerable attention in the area of drug research because of its cost-effective and time-saving capabilities. In recent years many pharmaceuticals are utilizing this approach for their drug discovery and development program, in fact in the Covid-19 era, its utility increased by many folds.
Keywords
Repurposing; Drug; Clinical trial; Computational approach
Introduction
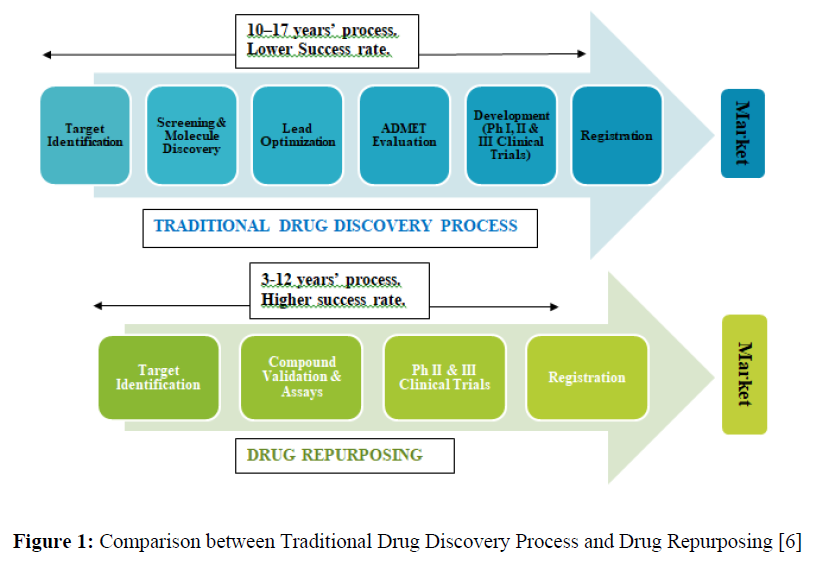
Drug development is a very challenging, complex, long-running, and expensive process with a very low success rate. Lately, a significant decline has been observed in the number of drugs approved for clinical use. The high failure rate and high investments of the traditional drug delivery system surged us the way to look for an alternative. The most suitable one that emerged in this context was ‘Drug Repurposing’. Drug repurposing has become a popular strategy in recent years. It can be compared to drug recycling [1]. It is an alternative drug development strategy whereby already approved drugs for treatment or management of some diseases and/or whose targets have already been located, are explored and exploited [2]. The goal of repurposing drugs is to find ways to make use of already approved drugs or those that did not complete clinical trials to find new uses or indications [3]. This strategy differs from the traditional drug development strategies in that it is efficient, economical, and riskless [4]. This makes drug repurposing one of the most active areas in pharmacology and pharmaceutical chemistry in the last few decades [5] (Figure 1).
Drug repurposing
Drug repurposing’ is an efficacious, successful, and innovative way to look for and bring about new targets or new indications of any drug that has already been approved by the FDA and that already exists or survives in the market [7]. It is a novel strategy to discover new therapeutic uses for already known drugs, including approved, discontinued, shelved, and experimental drugs, by exploiting them [8].
It is a process of recognition and spotting of new medical uses, outside the scope of the original pharmacological indication, for already approved/ existing drugs.
Drug repurposing is a 3-12 year process with reduced safety and pharmacokinetic uncertainty [9].
It is considered to be an excellent alternative over the de novo or traditional drug development process which is relatively a slower, time-consuming, costly, and a risk involved process.
Brief history on repurposing
History has observed numerous drug compounds that have been repurposed either opportunistic or accidental. Despite being far from new this strategy has attained appreciable momentum in the past few years; about one-third of the approvals in recent years count for drug repurposing, and at present around 25% of the annual revenue for the pharmaceutical industries is generated by repurposed drugs [10].
The most ancient example of drug repurposing could be acetylsalicylic acid. It was originally marketed as an analgesic but was repositioned in the 1980s and consequently, it evolved as an antiplatelet aggregation drug (at low doses only). This indication is still applying to prevent cardiovascular events [11].
Sildenafil is another megahit in the history of drug repurposing. Sildenafil which is a phosphodiesterase type 5 (PDE5) inhibitor was brought to the market for the treatment of cardiovascular disorders like coronary artery disease (CAD), hypertension, and angina pectoris. It was realized during the trials that the patients taking sildenafil exhibited marked penile erection. In 1988, it was repurposed and the outcome was that it was introduced in the market as an approved drug for erectile dysfunction [8].
Likewise, the next big blockbuster example is amantadine; it was introduced as an antiviral drug to treat influenza infections. Later, after a few years, it was successfully repurposed & it emerged as one of the drugs for Parkinson’s disease (PD). Another supernova is thalidomide. Initially, the drug was indicated for the treatment of motion sickness in pregnant women. But the drug was taken back from the market as it emerged out to be a teratogen. Its use was found to induce a range of birth defects, commonly malformations of the limbs. Later in 2006, the drug was repurposed and successfully re-approved for the treatment of multiple myeloma [8]. Similarly, there are several other such examples of drugs being repurposed in history and a few of them are mentioned below[table 1].
| Sl. No | Repurposed drug | Structure | Targeted Use | Repurposed Use |
|---|---|---|---|---|
| 1 | Bupropion | 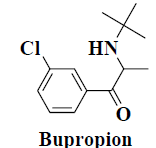 |
Depression | Smoking cessation |
| 2 | Fluoxetine | 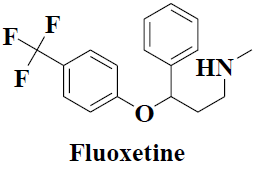 |
Depression | Premenstrual dysphoria |
| 3 | Isoniazid | 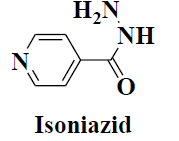 |
Tuberculosis | Certain types of tumors |
| 4 | Methotrexate | 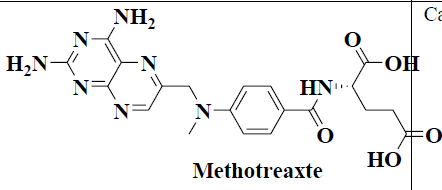 |
Cancer | Psoriasis, Rheumatoid Arthritis |
| 5 | Minoxidil | 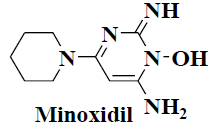 |
Hypertension | Alopecia |
| 6 | Orlistat | 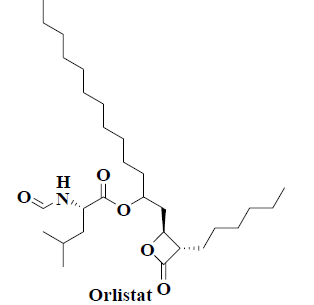 |
Obesity | Cancer |
| 7 | Propranolol |  |
Hypertension | Migraine |
| 8 | Retinoic acid |  |
Acne | Acute leukaemia |
| 9 | Ropinirole |  |
Parkinsonism | Restless leg syndrome |
| 10 | Simvastatin |  |
Cardiovascular disorders | Lung cancer |
| 11 | Topiramate | 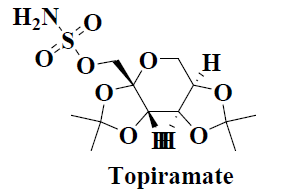 |
Fungal infections | Inflammatory bowel disease |
| 12 | Valsartan | 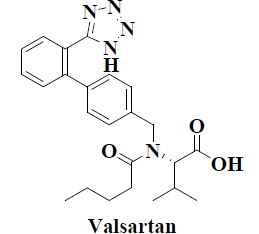 |
Cardiovascular disorders | Alzheimer’s disease |
| 13 | Tamoxifen | 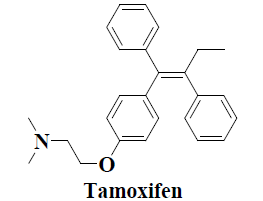 |
Anticancer(Breast cancer) | Systemic lupus erythematosus |
| 14 | Aspirin | 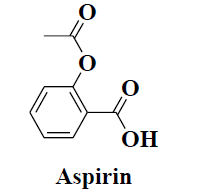 |
Analgesic and antipyretic. | Anti-platelet |
| 15 | Sildenafil |  |
Coronary artery disease | Erectile dysfunction |
Advantages of drug repurposing
Repurposing is preferred as a drug development strategy as it is efficient, economical, and riskless. The various advantages associated with repurposing are as follows:
1. Repurposing simplifies the regulatory procedures for introducing a previously approved drug on the market [11].
2. It takes into account previously known data, especially the drug’s safety and toxicity, which makes the initial phases of development of a repurposed drug considerably faster and therefore cheaper [10,11].
3. It increases the chances of introducing the drug in the market [15].
4. The drug does not undergo the initial stages of drug development as in the case of the traditional drug development process which takes up to nine years but instead goes directly to later phases i.e., the preclinical testing and clinical trials, thus reduces risk, time, and costs [9, 16].
5. There is a depletion of the risks associated with failures in the early stages of traditional drug development [13], this brings down the cost of the overall process.
6. Due to known pharmacokinetics and pharmacodynamics (PK-PD) profile and other data (efficacy, safety, and toxicity) related to the already approved drugs, further processing becomes easier [7,16].
7. It offers affordable, cheap, and faster treatment.
8. It is a technique that requires fewer efforts when compared to the primitive drug development process.
Categories of drugs suitable for repurposing [17,18].
A. Drugs whose mode of action is similar to more than one disease.
B. Drugs that fail to prove clinical efficacy (during the clinical trials) for a disease it was being processed for.
C. Drugs that have been shelved for some reasons.
D. Drugs for which the patents are going to expire.
E. Drugs whose generic formulations have already been marketed.
F. Drugs from academic institutions and public sector labs were never commercialized.
G. Drugs that were discontinued for commercial or safety reasons.
H. Drugs available in developing markets but not commercialized in the larger developed markets.
I. Drugs with incremental new indications (also called line extension drugs).
J. Drugs that are already in clinical development demonstrated polypharmacology.
Challenges faced by the strategy of repurposing
1. One of the main challenges faced by this strategy is that repurposed drugs are imparted with poor protection of intellectual property [11]. Acquiring a secondary patent reduces the chances of recovering the Research and Development (R&D) costs by the marketing of the drug [19]. Decreased possibilities of return on investment discourage companies from developing the repurposed drugs.
2. Accessibility of data and/or the availability of the compound may be an issue for the repurposing of drugs. Although this is not a major issue as several data banks available these days. Still, there is a certain category of data (for example, clinical trials) whose public access is limited [20].
3. New targeted drug discovery is considered more advantageous over drug repurposing.
4. Dose dependency is another challenge faced by this technique of repurposing. A drug’s dose varies with the disease for which it is being used, so after repurposing the appropriate dose should be studied through clinical trials [12].
Considerations/ prerequisites for repurposing of drug
a. To identify the appropriate compound or drug.
b. To make use of the resources in an appropriate and justified manner.
c. Not to fail in bringing back the already failed drugs into the clinical trials [19].
Steps or stages
In drug repurposing, we opt for the identification and discovery of drug candidates for new uses, inspection of mechanism or signaling pathway involved in drug or disease and eventually proving the effectiveness of the drug in phase 2 and 3 clinical trials [7]. Drug repurposing consists of four major steps v.i.z,
(i) Selection of target compound;
(ii) Clinical trial (phase 2 and 3);
(iii) NDA (New Drug Application) and
(iv) PMS (Post Marketing Surveillance).
Selection of target compound: Selection or identification of lead candidates is the most vital step of repurposing a drug. The selection of the target compound is based upon its pharmacological actions. Pharmacological actions apart from the principal or primary pharmacological action should be taken into consideration and the compound should be selected for repurposing. This step involves more advanced and systematic approaches required for the generation of the latest hypothesis in drug repurposing [7]. Compounds with multi-mechanisms or acting on multiple receptors or other mediators or having multiple binding sites may be considered good targets for repurposing. This works well for repurposing drugs for few rare diseases or for ailments for which there is the least number of treatment options.
Clinical trial (phase 2 and 3): Once the target compound has been successfully selected, we proceed with the clinical trials of the same. Since the compounds selected for repurposing are already approved or existing ones, we do not necessarily need to go for the phase 1 trial as for the approved drugs the question of safety does not arise. For the phase 2 trial patient’s groups with the target disease [21] or for the condition for which the drug is to be repurposed are selected and the effectiveness of the compound in that condition is evaluated. Next, in the phase 3 trial larger patient population with the target disease is selected and the therapeutic benefits or any new indication other than the original pharmacological uses are confirmed [22].
NDA (New Drug Application): Next, through NDA (New Drug Application) the drug sponsors or people carrying out the research formally propose the FDA or DGCI for the re-approval of the drug considering the new indications for sale and marketing [23] after successful phase 3 trial.
PMS (Post Marketing Surveillance): This involves safety surveillance (Pharmacovigilance) after the drug receives permission to be sold in the market. The safety surveillance is intended to recognize any rare or long-term adverse effects over a bigger population and longer period, which was impractical during the phase1-3 clinical trials. Harmful effects discovered in the PMS may result in the withdrawal of the drug from the market [22]. In the case of a repurposed drug, its effectiveness or success considering the new indication is also assessed.
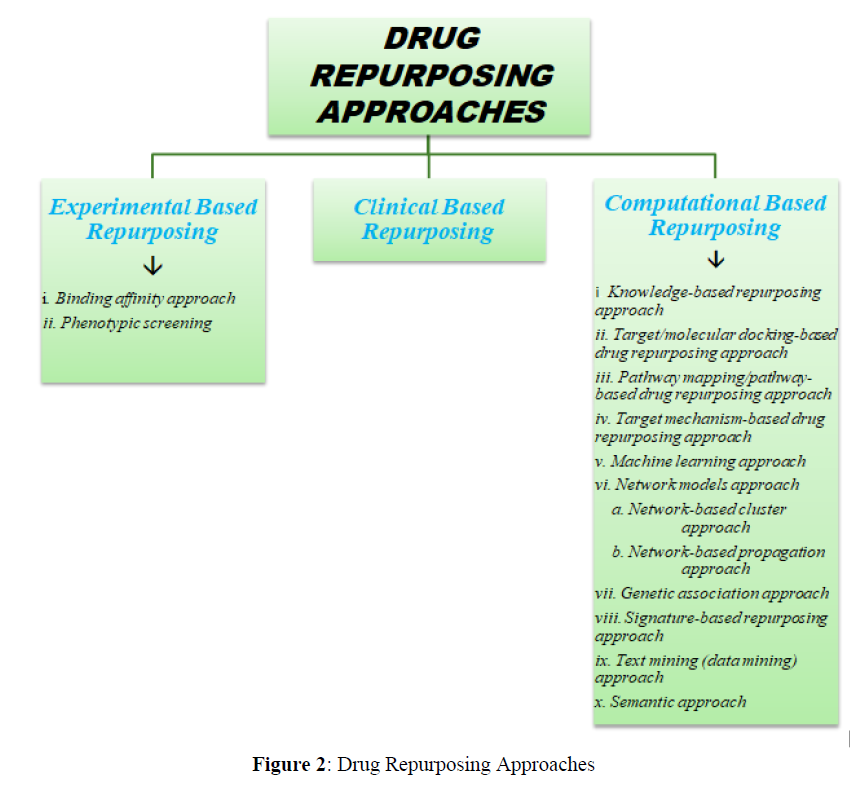
Drug Repurposing Approaches
Drugs can be repurposed in multiple ways, a spread of approaches have been developed which include experimental-based approaches, clinical based approaches, computational approaches, and mixed approaches (Figure 2).
Experimental Based Repurposing
The Experimental strategies in drug repurposing include the following
Binding Affinity Approach: To find all possible binding targets of drug candidates, techniques like chromatography and mass spectrometry are being used. Conjointly alternative qualitative methods of analysis may also be employed for the identification of target molecules.
Phenotypic Screening: Phenotypic Screening is amongst the well-known documented ways of identifying systemic or biological effects of drugs or medicinal products. In this approach, either using cell-based screens {high throughput screening (HTS)} or even whole organisms are employed to screen target drug candidates [24]. Significant cell-based assays that are carried out in drug discovery and repurposing are cell viability assay, signaling pathways assay, and disease-related mechanistic assay, are important cell-based assays that are being performed in drug discovery and repurposing [25]. Selection of wave length (λ max).
Clinical Based Repurposing
Clinical trials play a significant role in drug repurposing. It may be possible that drugs that have been recalled from the market may be repurposed for other indications. These trials have assisted in the repurposing of many drugs. Some examples of drugs repurposed by this method include Apomorphine which was initially indicated for Parkinson’s disease but was later repurposed for erectile dysfunction, dapoxetine which was at first indicated for analgesia and depression and was then repurposed for hypertension.
Computational Based Repurposing
Knowledge-based repurposing approach
The basis of this approach lies in already available data related to those of ligands and receptors. Different models have evolved and advanced to search out the targets that have not been explored/ discovered yet. This is done to discover the novel biomarkers, pathophysiology, and receptors for various diseases [7]. Repurposing of drugs by this approach could be based on mechanism, pathway, receptor, etc. [26].
Target/molecular docking-based drug repurposing approach
It is a known in-silico structure-based standard method widely used in drug discovery molecular docking. Identification of novel compounds of therapeutic interest, prediction of ligand-target interactions at a molecular level, and other such information can be availed by employing docking techniques. High-Throughput Screening (HTS) is considered the gold standard for discovering biologically active compounds [27]. This screening does not require any input of information related to any biological or pharmacological properties. In this approach, the targets are linked with the pathophysiology behind the disease and therefore the process of drug discovery is built. The entire screening process utilizes very little time for completion.
Pathway mapping/pathway-based drug repurposing approach
For prediction of the relatedness between disease and drugs information such as protein-protein interaction, cell signaling, and metabolic pathways can be important and useful. This approach involves the identification of a specific/selected pathway that is related to different diseases and thereby finding out drugs to target that particular pathway for curing of diseases. The pathways concerned with the specific disease can be outlined from the data available in the central database of patients and these pathways are the basis for the repurposing of drugs [28].
Target mechanism-based drug repurposing approach
This approach assists in predicting the novel mechanism of action of drugs after gathering information from signaling pathways, interaction networks of various proteins, and conjointly by knowledge obtained from omics. This leads to the feasibility of drug repurposing for existing drugs. Additionally, this will contribute to precision medicine [7]. This approach not only locates different pathophysiological mechanisms related to the diseases or drugs but also provides for the identification of those mechanisms related to the treatment of drugs to respective diseases [13,28].
Machine learning approach
Different Machine learning (ML) approaches including Deep learning (DL), Gradient boosted machine with trees (GBM), Random forest (RF), Support vector machine (SVM), logistic regression with elastic net regularization (EN), Deep neural networks (DNN) are the commonly used methods of drug repurposing [29].
Network models approach
Networks and their analysis can provide valuable information about drug mechanism, indications, and how drug targets work and thus enlighten the therapeutic efficacies and potentials of those drugs [30]. Network-based approaches are well-established methods and are widely accepted and employed in drug repositioning as it helps in discovering interactions and relationships of biological and biomedical entities due to its ability to combine data from multiple data sources. From different databases available the data regarding the varied interaction patterns are obtained and are interpreted computationally. The interaction patterns considered here could be protein-protein, drug-disease, disease-gene, drug-target, drug-drug, disease-disease, etc., there are two types of network-based approaches are available: network-based cluster approaches and network-based propagation approaches.
• Network-based cluster approach: By this approach various novel drug-disease relationships or drug-target relationships can be discovered. This approach uses cluster algorithms as per the topology structures of networks to find several modules (also known as sub networks, groups, or cliques). Few difficulties such as overlapping of clusters cannot be detected by CLIQUE, OPTICS, DBSCAN, and STING, etc. [29]. To overcome this, scientists have used the k-means-based network cluster algorithm [4].
• Network-based propagation approach: This is another significant approach under network-based strategies. Studies have suggested that these methods stand out well in detecting disease-targets, disease-genes, and disease-drug relationships. They can be classified into two types: local and global approach. The global approach has many advantages over Local propagation approaches and currently used by many researchers to achieve outstanding performance [4,29].
Genetic association approach
Genetic alterations in the whole genome can be assessed using Genome-Wide Association Studies (GWAS). Repurposing of several drugs can be done by identifying novel targets which contribute to specific diseases. This can be done with the help of data obtained from the GWAS as it contributes to specific diseases and provides the pathophysiology of various diseases. However, the data available from GWAS fails to provide the exact pathophysiological mechanism. At the same time, the available data is considered to be inappropriate due to the gene variants.
Signature-based repurposing approach
Genes can be expressed to find off-target mechanisms and also novel pathophysiological mechanisms related to various diseases. This is the basis of this approach. Identification of the distinctive mechanism of action of drugs is the primary advantage of this approach. Signature-based repurposing involves methods that use the databases for the prediction of the mechanism (molecular- and/or genetic-level mechanism) of action of drugs.
Text mining (data mining) approach
Text mining (TM) techniques have been increasingly emerging to the latest understandings from scientific literature which largely contributes to the identification of relationships between biological concepts or biological entities. A massive amount of information regarding genes, drugs, and diseases is stored in different databases. The data procured from these databases conclusively help in the prediction of new indications of existing drugs via a text mining approach. Text mining tools suggest a huge number of biological entity relationships which helps researchers in verifying their experimental results. Generally, text mining includes four steps [31] which are:
1. Information retrieval (IR),
2. Entity recognition (NER),
3. Information extraction (IE), and
4. Knowledge discovery.
Semantic approach
In recent years, a semantics-based approach has been applied to drug repurposing. In this technique data from different sources is combined which assists in the prediction of therapeutic potency and novel indications for already approved drugs [30].Semantic inference includes techniques like topic modeling which makes use of different databases for the repurposing of existing drugs [7]. It differentiates between already known direct drug-target associations and random drug-target associations with high precision and also it identifies indirect drug-target associations [30].The working system of this method mainly includes three steps, they are;
1. Initially, the semantic network is set up by procuring information from the medical databases concerning biological entity relationships.
2. Then, the addition of prior information obtained in the above step aids in the construction of semantics networks based on existing ontology networks.
3. Finally, for the prediction of novel relationships in the semantic network, mining algorithms are designed.
Conclusion
De novo drug development or the traditional system of drug development is time-consuming, high investment, and the risk-prone process has paved the way to look for an alternative strategy for drug development. Overcoming all these drawbacks, ‘Drug repurposing' has drawn the attention of all the pharmaceutical industries and R&D sectors which offers faster and cheaper ways for bringing new drugs into the market. The repurposing of drugs for a therapeutic indication other than the one for which they were initially marketed is a growing trend. This approach presents lower R&D costs, higher success rates, lower investment risk, and reduces research time. Repurposing is a drug discovery method that could help treat patients and improve their quality of life by bringing new therapeutic options to market. However, this approach must be considered as an add-on rather than an alternative to the search for novel drugs.
References
- Gargi Dey. Journal of Medical Science and Research.
- Dueñas-González A, García-López P, Herrera LA et al., Molecular cancer. 2008, 7(1): p. 82.
- Marise Khalil and Katie Taylor. Pharmacists Can Play Key Role in Drug Repurposing.
- Xue H, Li J, Xie H et al., Int J Biol Sci. 2018, 14(10): p. 1232.
- Blessy Mariyam Babu. Int J Res Pharm. 2020, 11(3).
- Chakraborty A and Trivedi V. Austin J Biotechnol Bioeng. 2015, 2: p. 3-5.
- Dhir N, Jain A, Mahendru D et al., Drug Repurposing and Orphan Disease Therapeutics. 2020.
- Talevi A and Bellera CL. Expert Opin Drug Discov. 2020, 15(4): p. 397- 401.
- Oliveira EAM, Lang KL. J Appl Pharm Sci. 2018, 8(08): p. 157-65.
- Naylor DM. Drug Discovery. 2015, (16)2: p. 57-72.
- Jourdan JP, Bureau R, Rochais C et al., J Pharm Pharmacol. 2020, 72(9): 1145-1151.
- Ko Y. Applied Sciences. 2020, 10(15): p. 5076.
- Rudrapal M, Khairnar SJ and Jadhav AG. IntechOpen. 2020.
- Lee HM and Kim Y. Schizophr Res Treatment. 2016, 6378137.
- Thayer A. Drug Repurposing. 2012, 90(40).
- Jin G and Wong ST. Drug discovery today. 2014, 19(5): p. 637-44.
- Padhy BM and Gupta YK. J Postgrad Med. 2011, 57(2): p. 153.
- Naylor DM. Drug Discovery. 2015, 16(2): p. 57-72.
- Gargi Dey. J Med Sci Res. 2019, 7(2).
- Saini M, Parihar N, Soni SL et al., Asian j pharm res dev. 2020, 8(4): p. 194-212.
- Van Norman GA. JACC Basic Transl Sci. 2019, 4(3): p. 428-37.
- Dipti S Pawar. WJPLS. 2016, 2(1): p. 93-99.
- https://www.fda.gov/drugs/types-applications/new-drug-application-nda
- Aulner N, Danckaert A, Ihm J et al., Trends Parasitol. 2019, 35(7): p.559-570.
- Zheng W, Thorne N and McKew JC. Drug Discov Today. 2013, 18(21-22): p. 1067-73.
- Pushpakom S, Iorio F, Eyers PA et al., Nat Rev Drug Discov. 2019, 18(1): p. 41-58.
- Pinzi L and Rastelli G. Int J Mol Sci. 2019, 20(18): p. 4331.
- Park K. Transl Clin Pharmacol. 2019, 27(2): p.59-63.
- GNS HS, Saraswathy GR, Murahari M et al., Biomed Pharmacother. 2019, 110: p. 700-16.
- Jarada TN, Rokne JG and Alhajj R. J Cheminform. 2020, 12(1): p. 1-23.
- Zhu F, Patumcharoenpol P, Zhang C et al., J Biomed Inform. 2013, 46(2): p. 200-11.
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref