Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 1
Effect of Ivermectin on the Postpartum Dynamic of Gastrointestinal Strongyles Infection in Traditionally Managed Ewes
Ahmed Hadef1,2* and Youcef Hadef3,42Department of Veterinary Sciences, University Chadli Ben Djedid, El Tarf, PB 73, 36000, ELTarf, Algeria
3Department of Pharmacy, Laboratory of Analytical Chemistry, Faculty of Medecine, University Badji Mokhtar, PB 205, 23000, Annaba, Algeria
4Laboratory for the Development and Control of Pharmaceutical and Hospital, Faculty of Medecine, University Badji Mokhtar, PB 205, 23000, Annaba, Algeria
Ahmed Hadef, Institute of Veterinary Sciences, University of Frères Mentouri Constantine, El Khroub, 25160, Constantine, Algeria,
Abstract
The effect of ivermectin on the dynamic of egg shedding of gastrointestinal strongyles was studied in post-parturient ewes (n=12) from traditionally managed farm located in a north eastern province of Algeria. Fecal samples were collected during the postpartum period to monitor the worm egg count (WEC) before and 3, 22 and 34 days after subcutaneous administration of 0.2 mg/kg of ivermectin. Furthermore, the pretreatment level of ewe’s fat reserve was assessed by the scoring of body condition (BCS) and weighing of body weight (BW) to investigate its effect on ivermectin efficacy. The injection of ivermectin to low infected ewes caused a remove of egg shedding (0 eggs) and a perfect efficacy (100%) in some ewes during the first month post-treatment, mainly on day 3 corresponding to a theoretical time of ivermectin maximal plasmatic concentration. However, on day 34, ewes had markedly shed worm eggs in feces (P<0.05) expressing the diminution of ivermectin efficacy about 87%. This resumption of eggs excretion was significantly and closely correlated to pre-treatment BCS (r=-0.91, p=0.01). The present study has revealed a possible presence of strongyle resistant against ivermectin during the first week post-treatment from low infected ewes which displaying a postpartum energy deficit (poor body fat reserve), and the necessity to take in account a probable inconspicuous development of this resistance following the arbitrary use of this avermectin in traditionally managed flocks.
Keywords
BCS, Ewes, Ivermectin, Postpartum, Strongyle
Introduction
In Algeria, nematode control in sheep is commonly based on the use of albendazole and ivermectin [1] that seems to have a good efficacy in Algerian farms condition [2]. Helminthic resistance to these latter drugs was not detected during first studies in Algerian semi-arid area [2]. The appearance of nematodes resistant against anthelmintic in ovine was firstly reported in tropical countries such as described in South Africa [3]. The climatic conditions favor to the maintenance of the digestive cycle and the frequent use of anthelmintics were considered as major elements favorable for the development of resistance [4].
In Algeria, resistance to anthelminthic was firstly reported in a pilot farm [5]. From data published in 2007 [6], strongyles, mainly Teladorsagia, Trichostrongylus, Marshallagia and Nematodirus, resistant against benzimidazoles (five farms) and ivermectin (one farm) were detected in sheep during a study conducted in eastern Algeria in pilot farms showing low infection level. On the other hand, another study in Algerian semiarid area using fecal egg count reduction (FECR) method has revealed high efficiency of ivermectin (>95%) at the dose of 0.2 mg/kg body weight and a global efficiency of albendazole at 5 mg/kg was 94.2% with a some resistance of Marshallagia marshalli to this latter drug [1]. From previously studies [1,6], it was suggested that FECR tests using quantitative coproscopic method based on McMaster technique were not always consistent due to the low-level of infection. Otherwise, the modified Wisconsin technique was estimated as more adapted to low level infection context to determine fecal worm egg counts [7] and it was used in USA to study parasite resistance in US Cattle in 19 states [8].
The efficacy of anthelminthic is dependent to some factors [9] such as those related to the drug (pharmacokinetic) or to the individual status (immunity, physiological stage). The relationship between anthelminthic's pharmacodynamic and its clinical efficacy has been suggested in a study conducted in sheep treated with ivermectin [10]. This Macrocyclic Lactone (ML) characterized by their endectocidal activity was detected in ewe’s plasma of Senegalese Peulh breed between 30 min and 20 days and their time to reach peak plasma concentration (Tmax) and mean residence time (MRT) were 2.75 ± 2.21 days and 5.84 ± 2.80 days, respectively [11]. The physiological stage was determined as a factor influencing plasma kinetic of ivermectin. This drug had been a higher mean residence time (MRT) values in pregnant ewes (8.8 ± 1.4 days) than those observed in nonpregnant ewes (5.3 ± 1.9 days) when it is administered subcutaneously [12]. The implication of anthelminthic resistance in relation to ewe’s reproductive stage was discussed in study conducted in ewes experimentally infected by benzimidazole resistant strains of Ostertagia circumcincta and Trichostrongylus colubriformis during the postpartum period [13]. This stage was recognized as a period of immunity relaxation and rise of strongyles egg shedding [14].
The aim of this study was to explore the dynamic of ivermectin effect against gastrointestinal strongyles by determination of egg shedding reduction rate using modified Wisconsin technique, in traditionally managed ewes during their critical period of postpartum and its relation to pretreatment body fat reserve status.
Materials and Methods
Animal and sampling
The study was carried out in private farm located in littoral province in the north east of Algeria. Ewes (n=12) bred under traditional and semi extensive management system were monitored during their postpartum period following autumn and winter lambing seasons in regard to their infestation by gastrointestinal strongyles by bimonthly assessment of worm egg counts. This latter test was performed on fecal samples (3 g) using the modified Wisconsin technique based on Sheather’s solution flotation [7].
Pretreatment fat reserve status
Before treatment, the body weight of studied ewes was weighed and the amount of their body fat reserve was assessed using the scoring of body condition. This subjective evaluation was done according to the method described by Suiter [15] using a scale of 1 to 5 for emaciated and excessively fat animals, respectively.
Treatment
Due to traditional management system characterizing the study farm, deworming was indiscriminately used and it was based in a part on the regular use of ivermectin. This treatment was given by subcutaneous injection at the recommended dose (0.2 mg/kg body weight) using a commercial formulation of ivermectin 1%.
The efficacy rate (ER) of ivermectin was calculated in 7 ewes using formula inspired to that proposed by World Association for the Advancement of Veterinary Parasitology (WAAVP) and reported by Coles et al. [16].
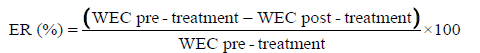
However, in the present study, the pretreatment WEC was established 13 days before treatment and the post-treatment fecal WECs were determined early on days 3 after administration. This day correspond to the time when the drug theoretically reaches its peak plasma concentration [11,17] and also to ivermectin elimination half-life [17,18]. Furthermore, WECs were recorded on days 22 corresponding to time of drug excretion from fat tissue and on day 34 corresponding to period when ivermectin could be still detectable [18]. These times, 22 and 34 days post-treatment, were comparable respectively to the 3 weeks and the 4 weeks of ivermectin remanence reported by some authors [19,20].
Statistical analysis
Data analysis was performed by using SPSS Statistics 17.0 for Windows Statistical Software. The "t" test of Student was used to compare means of fecal worm egg counts (WEC) recorded according to the time of treatment. The Pearson correlation “r” was calculated to analyze the relationship between egg shedding dynamics and indicators of body fat reserve, body condition score and body weight.
Result and Discussion
Evolution of egg shedding during pre and post treatment periods
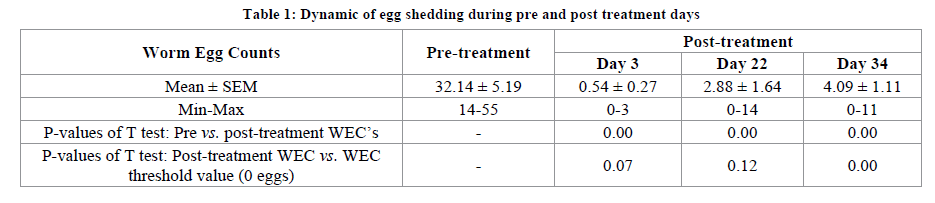
The determination of fecal worm egg counts in ewes lambed during autumn and winter allowed the assessment of the dynamic of strongyles egg shedding during the postpartum period before and after treatment. Pretreated ewes revealed a low level infection varies from 14 to 55 eggs per fecal sample of 3 g (Table 1) showing the necessity of deworming in some females compared to other. This result confirmed the presence of immunity relaxation in postparturient ewes probably due to nutritional stress leading to an increase of the egg shedding in some cases [14]. Furthermore, it demonstrated the arbitrary use of anthelminthic in the present traditionally managed farm and justified our choice of Wisconsin modified technique adapted to the present condition of low infection [7].
The subcutaneous administration of 0.2 mg/kg of ivermectin caused a clear reduction of egg shedding (Table 1) ensuring a total suppression of infection (0 eggs) from day 3 to day 34 post-treatment in some ewes. During this period, worm egg counts were significantly lower than pretreatment WEC level (P<0.05). From the second month post-treatment (day 34), ewes markedly released worm eggs in feces in regard to a suitable level of 0-egg (P<0.05). This result could express the proportional remove of ivermectin efficacy that could be related to the pharmacokinetic of this ML. It showed that ivermectin could persist in animal body even days, until 35 days in matrices of animal [18]. In other hand, the presence of eggs after treatment from the first week post-treatment could induce some resistance of gastrointestinal nematodes against ivermectin [21], and justify the importance of monitoring of proportional efficacy of this drug according to their pharmacodynamics [21,22] such as performed hereinafter.
In the Table 2, ivermectin was greatly effective against strongyles as showed by an average reduction of about 96% of egg shedding on day 3 post-treatment at the time in which the drug has theoretically reached their maximal plasmatic concentration [11]. This ivermectin efficacy was similar to that (>95%) recorded in Algerian semiarid area [1]. Nevertheless, 14 % of ewes monitored for this parameter showed a persistence of egg shedding although for a very low level (3 eggs). It was suggested that a fully efficacy of an anthelmintic implies that no resistant worms persist treatment following usually by 48 h, the time taken to empty the intestines, and that no eggs should be found in the feces after 14 to 17 days [21].
At the day 22 after treatment, a diminution of average ivermectin efficacy was noted showing a possible resistance of strongyles against ivermectin. Any viable eggs and a small percentage of survivor worms may indicate a resistance problem that could develop with subsequent treatments [21]. However, this seems unjustifiable due to the presence of one ewe with a very low efficacy (36%) and to the late time of the current estimation (day 22) in comparison to the optimal time of resistance evaluation (14 to 17 days posttreatment) as proposed by WAAVP [16].
The average efficacy, on days 22 and 34 post-treatment, was maintained a lower level compared to WAAVP threshold percentage of reduction (95%) which could be related to a progressive remove of ivermectin anthelminthic effect and a diminution of its concentration from adipose tissue that is the main drug stock [17]. However, a perfect efficacy (100%) was obtained until 34 days in some ewes (Table 2) leading to suggest that ivermectin's effective concentrations had a long circulation time that expressing the time of remanence. For statistical reason (small sample size) and if we exclude the only case of one ewe showing a high resistance on day 22, a significant diminution of efficacy was noted on day 34 corresponding to the end of time of ivermectin remanence (2 to 4 week) reported in literature [19,20].

Relationship between BCS, BW and ivermectin efficacy
The Relationship between fat reserve and the dynamic of ivermectin effect on strongyles egg shedding was resumed in Table 3. The means of body condition score (1.79 ± 0.14) and body weight (39.83 ± 1.96 kg) recorded before treatment could indicate the energy deficit of postparturient ewes in comparison to an optimal score of 2.5. This state could be related to the inevitable peripartum negative energy balance [23] and the undernutrition condition characterizing the studied farm in which ration was based on pasture and irregular distribution of concentrate in rainy days. Results of Pearson correlation test (Table 3) showed the high significant and closely negative relationship (r=-0.91, P=0.01) between the resumption of a significant shedding of strongyles eggs (> 0 eggs) on day 34 and the pre-treatment BCS. This result could indicate the effect of fat reserve level on the time of drug distribution which caused extension of ivermectin remanence above one month after its subcutaneous injection. This is in agree with research indicating that the distribution of a greater proportion of ivermectin into lipid reservoirs from subcutaneous injection could increases the residence time in sheep [17]. Moreover, it was revealed in pigs that body condition has influenced the kinetic disposition of ivermectin after subcutaneous injection and the drug being less persistent in thin compared with fat animals [24]. This body condition effect was also reported in cattle [9].
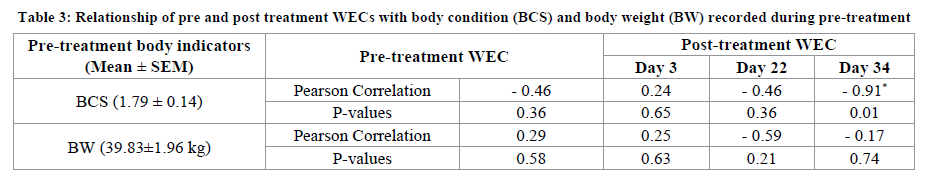
The present effect of body condition on egg shedding could explain the case of short effect of ivermectin in the case of one ewe on day 22 showing an early and important resumption of egg excretion (14 eggs) corresponding to a 36% of WEC reduction. This ewe showed a bad fattening state as expressed by a poor BCS (1.5) and it was the thinnest (34 kg) among those weighed before treatment. Therefore, ivermectin could be poorly stored and its elimination was probably faster.
However, means of worm egg counts recorded on day 3 and 22 post-treatment were not related to BCS. This could be related to the dependence of ivermectin more to its high plasmatic concentration on day 3 as reported previously [11,17] and to the possibility of presence of effective serum concentration on day 22 corresponding to time of remanence reported by some authors [19,20]. Similarly, it was reported that ivermectin partitioning into serum components was a determinative factor for ivermectin disposition and efficacy [22]. Furthermore, the non-significant relationship (P>0.05) between pre-treatment body weight and fecal egg counts through post-treatment days could lead to confirm that ivermectin early efficacy was depended to its serum concentration and its remanence time was more related to the fat reserve before the treatment in comparison to other body components.
Conclusion
This study has showed that dynamic of ivermectin effect could be affected by the status of postpartum energy deficit in underfeed ewes. This could lead to increase the proportion of gastrointestinal strongyles ivermectin resistant following next rounds of arbitrary deworming, mostly in the context of low infection in which the reappearance of eggs from resistant worm could be underestimated.
References
[1] A. Boulkaboul, A. Boucif, K. Senouci, 2010, 63 (3-4), 71-75.
[2] B. Bentounsi, H. Zouiouech, C. Benchikh-El Fegoun, K. Kohil, J. Cabaret, Rev. Méd. Vét., 2003, 154 (10), 649-652.
[3] J.A. Van Wyk, P.C. Van Schalkwyk, Vet. Parasitol., 1990, 35, 61-69.
[4] H. Zouiten, PhD thesis, Mohammed V-AGDAL University (Rabat, Morocco, 2006).
[5] B. Bentounsi, R. Trad, N. Gaous, K. Kohil, J. Cabaret. Veterinary Record, 2006, 158, 634-635.
[6] B. Bentounsi, B. Attir, S. Meradi, J. Cabaret, Vet. Parasitol., 2007, 144, 104-110.
[7] D.H. Bliss, W.G. Kvasnicka, Compendium (Beef Production Management), 1997, 104-109.
[8] D.H. Bliss, R.D. Moore, W.G. Kvasnicka, In: American Association of Bovine Practitioners (Ed.), 41st Annual Conference Proceedings of the American Association of Bovine Practitioners, Sep. 25-27, 2008, Charlotte, NC., USA (The AABP, 2008) 109-114.
[9] A.G. Canga, A.M.S. Prieto, M.J.D. Liébana, N.F. Martínez, M.S. Vega, J.J.G. Vieitez, The Vet. J., 2009, 179(1), 25-37.
[10] A.H. Atta, M.N. Abo-Shihada, J. Vet. Pharmacol. Therap., 2000, 23(1), 49-52.
[11] T. Bengone Ndong, Y. Kane, E.H.M. Diouf, M. Alvinerie, Vet. Res. Commun., 2007, 31, 739-747.
[12] R. Pérez, C. Palma, M.J. Núñez, J. Cox, M. Arboix, J. Vet. Pharmacol. Therap., 2008, 31, 71-76.
[13] D.M. Leathwick, C.M. Miller, A. Vlassoff, I.A. Sutherland, Int. J. Parasitol, 1997, 27(4), 411-416.
[14] R.L. Coop, I. Kyriazakis, Vet. Parasitol., 1999, 84(3), 187-204.
[15] J. Suiter, Farmnote, 1994, 69.
[16] G.C. Coles, C. Bauer, F.H.M. Borgsteede, S. Geerts, T.R. Klei, M.A. Taylor, P.J. Waller, Vet. Parasitol., 1992, 44, 35-44.
[17] S.E. Marriner, I. McKinnon, J.A. Bogan, J. Vet. Pharmacol. Therap., 1987, 10, 175-179.
[18] V. Cerkvenik, I. Grabnar, V. Skubic, D.Z. Doganoc, W.M.J. Beek, H.J. Keukens, M. Drobnič Košorok, M. Pogačnik, Vet. Parasitol., 2002; 104, 175-185.
[19] C. Mage, France Agricole Editions, 2008, 113.
[20] C. Richard, N. Kirschvink, Filière Ovine et Caprine, 2012, 42(4), 20-25.
[21] G. Cringoli, V. Veneziano, L. Mezzino, M. Morgoglione, S. Pennacchio, L. Rinaldi, V. Salamina, BMC Vet. Res., 2009, 5(1), 41.
[22] H.A. El-Banna, A. Goudah, H. El-Zorba, S. Abd-El-Rahman, Parasitol. Res., 2008, 102, 1337-1342.
[23] A. Cannas, In: G. Pulina, R. Bencini (Ed.), Dairy Sheep Nutrition (CABI Publishing, Wallingford, UK., 2004) 79-108.
[24] J. Craven, H. Bjørn, D.R. Hennessy, C. Friis, J. Vet. Pharmacol. Therap., 2002, 25(3), 227-232.



