Research Article - Der Pharma Chemica ( 2020) Volume 12, Issue 6
Fast and Accurate Quantitative Determination of Prednisolone and Chlorpheniramine in Active Pharmaceutical Ingredients and Parenteral Dosage Forms
Zeeshan Ali1, Javeria Tayyab1, Muhammad Zeeshan1 and Muhammad Sajid1*Muhammad Sajid, Department of chemistry, Institute of Chemical Sciences, Bahauddin Zakariya University Multan 60800, Pakistan, Email: dr.msajid@bzu.edu.pk
Received: 01-Jul-2020 Accepted Date: Oct 27, 2020 ; Published: 28-Oct-2020
Abstract
The research work entails the development of an efficient and facile method for the simultaneous quantitative analysis of Chlorpheniramine (CPM) and Prednisolone (Pred) with reasonable accuracy and precision both in active pharmaceutical ingredient (API) and its parenteral dosage form. C18 (5μm x 150 mm 4.6 mm) HPLC column was used using 50:50 v/v acetonitrile: buffer 0.008M sodium dihydrogen phosphate (NaH2PO4) as mobile phase. The run time for the analysis was 10 min. The retention times of CPM and Pred were 1 and 4 minutes. The developed method showed linearity, intermediate precision and accuracy within the defined limits set according to international standards. Verification of this method was done on parenteral dosage form of both the drugs. Linearity with R2 values of 0.991607 for CPM and 0.990061 for PRED was obtained. Limit of detection and limit of quantification for PRED were calculated 0.033 mg. mL-1 and 0.11 mg. mL-1 while for CPM, 0.08 mg. mL-1 and 0.27 mg. mL-1 were obtained. Accuracy parameters outcomes for PRED, were within range of 98.16576% to 98.46571% and the values for CPM were 98.10% to 99.57% Validation results of the developed method are accurate and within system suitability parameters provided by ICH and pharmacopeias.
Keywords
Pharmaceutical chemistry, Chlorpheniramine, Prednisolone, HPLC
Introduction
Pharmaceutical medicine is considered a chemical substance used to diagnose, prevent or cure a medical ailment. Most of the active components of the medicine are derived from natural sources like plants, herbs or animals. Some of the active components of medicines are made synthetically from different sources. A class of drugs can be further classified into sub-classes on the basis of their chemical composition, duration of action or the receptors in the body [1]. Antihistamines are the chemical agents that block action of histamine by blocking histamine receptors [2]. Antihistamines are actually antagonists to histamine receptors. Histamine receptors are G-protein coupled receptors in which primary binding ligand is histamine. At least four types of histamine receptors have been found to be present in the human body. They are named as H1, H2, H3 and H4 receptors.
H1 are one of the most important receptors for modulating internal clock, and are a main target for many clinical drugs. Excess activation of these receptors triggers the symptoms of hay fever and other seasonal allergies [3]. H2 are found on parietal cells located in the stomach lining, and are mainly responsible for regulating the levels of gastric acid. Histamine can also inhibit antibody and cytokine production by reacting with these receptors [4]. H3 are present throughout the nervous system, though most notably in the central nervous system [5,6]. The more of these receptors that are triggered by histamine, the less histamine is produced in the body [7,8]. H4 receptors regulate the levels of white blood cell release from bone marrow [9]. They are located in the thymus, small intestine and express similar expression configuration in the human oral epithelium spleen [10], the colon, bone marrow and basophils [11-13]. Antihistamines are great at soothing side effects of a hypersensitive response, for example, sneezing, running nose, edema, swelling, rashes, itching of eyes and irritation etc. [14]. They are more potent but cause more sedation. These drugs are very effective in the treatment of severe allergies [15,16].
Chlorpheniramine or chlorphenamine (shortly CP or CPM) is a drug that belong to primitive class of antihistamine [17]. It is widely used in the treatment of skin allergies [18]. It is combined with other drugs for treatment of medical ailments like cough and cold [19]. The drug is also commonly described as possessing weak anticholinergic activity by acting as an antagonist of the muscarinic acetylcholine receptors. In addition to acting as an inverse agonist at the H1 receptor [20], chlorphenamine has been found to act as a serotonin reuptake inhibitor (SRI) (Kd=15.2 nM for the transportation of serotonin [21]. It has only weak affinity for the norepinephrine and dopamine transporters [22]. Absorption is through the gut after first pass metabolism by the liver. The drug is bound to plasma protein up to 72%. Chlorpheniramine is mainly metabolized and excreted in urine as mono and di desmethyl chlorpheniramine and small amount of drug is excreted unchanged.
Prednisolone is a glaucocorticoid, both naturally occurring and synthetic that is absorbed from gastrointestinal tract and is widely used for the treatment of various medical conditions like inflammation, autoimmune disorders [23], adrenocortical insufficiency, uveitis, skin conditions and dermatitis. Prednisolone is a biologically active steroid that acts mainly as glucocorticoid [24,25]. It alters the biochemical reactions of lipids, carbohydrates and proteins. It alters and ceases formations of DNA. The mechanism of action in control of inflammation is by blocking the production of prostaglandins and leukotrienes by inhibition of arachidonic acid. The activity of prednisolone in immune suppression is by binding to lymphocytes altering their expression of immune response at cellular level [26]. In adrenal incompetency, prednisolone acts at steroid ligands. The drug is present freely too much extent and some of the drug is bound to proteins in the plasma.
Though the individual analysis of CPM and Pred, including both the spectrophotometric as well as chromatographic methods have been reported yet their simultaneous determination has not been sorted out. The combination of these two drugs are generally advised as a remedy to the patients with chronic cough and cold. One major objective for the development of new method for quantitative analysis of CPM and Pred is to design a pathway for their simultaneous analysis in single go, which is more efficient, cost effective in terms of chemical use and time saving with reasonable accuracy and precision for CPM and Pred in active pharmaceutical ingredient (API) and its parenteral dosage form.
Experimental
Chemicals
Chemicals used during the course of method development and validation of Chlorpheniramine maleate (99%) and Prednisolone (99%) in bulk as well as parenteral dosage form are Acetonitrile (HPLC grade), Sodium dihydrogen phosphate and water (MS grade). All these chemicals are purchased from Merck and Sigma Aldrich and were of high purity.
Method Development
Different trials were made to develop method for purpose of quantitative determination of Chlorpheniramine maleate and Prednisolone. Guidelines for the method were used as described in British Pharmacopeia. Optimizations in the flow rate and composition of mobile phase were done to get optimum results of Chlorpheniramine maleate and Prednisolone in active pharmaceutical ingredient and pharmaceutical dosage form. Different composition of mobile phase and flow rate were tested while keeping other parameters constant such as injection volume, temperature and detection wavelength, oven temperature and injection volume.
Mobile Phase and Solution Preparation
0.008M sodium dihydrogen phosphate (NaH2PO4) was prepared as a buffer. For this about 100 mL of HPLC grade distilled water was taken in 500 mL volumetric flask. 0.5 g of sodium dihydrogen phosphate was weighed and dissolved in water by mechanical shaking and pH was adjusted to 3.0 using phosphoric acid. Mobile phase for isocratic mode was buffer and acetonitrile (50:50 v/v). Mobile phase was filtered using nylon membrane filter of 0.2 μm pore size and 0.47 mm diameter and sonicated for 15 minutes at room temperature to degas.
HPLC Analysis
CPM and Pred standard solutions were analyzed using WATERS HPLC C18 5μm (150 mm x 4.6 mm, 5 μm) analytical column with mobile phase comprising ACN and buffer (50:50 v/v). Detection wavelength was set at 254 nm using Photodiode Array (PDA) detector. Total run time was in the range of 1 to 10 minutes.
Method Validation
Proposed method was validated according to guidelines by International Conference of Harmonization (ICH), British Pharmacopeia (BP) and US Pharmacopeia (USP). Specificity, Linearity, Range, Limit of Detection (LOD), Limit of Quantification (LOQ), Precision and Bias/Accuracy were studied during this validation process.
Result and Discussion
Method Development
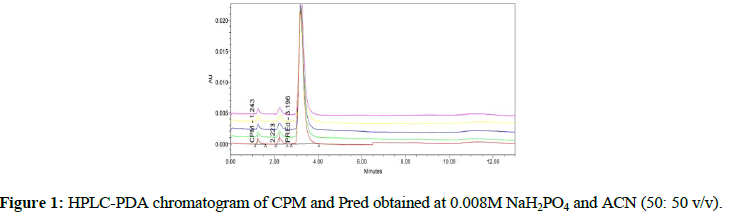
For the development of method different chromatographic conditions were tested. The best results were observed at buffer of 0.008M NaH2PO4 and ACN in concentration range of 50: 50 v/v. Chromatogram of the simultaneous analysis of CPM (0.40 mg/mL) and Pred (1.0 mg/mL) acquired from the method developed is shown Figure 1. Retention times (RT) of CPM and Pred are between 1 to 4 minutes. Samples analyzed in five replicates show excellent repeatability with RSD in the range of 1.1 % and 0.7% for CPM and Pred, respectively.
Method Validation
Analytical method is the path or way used for the detection or quantification of analyte in the sample. It gives detailed description of each step involved in the analysis. Here the developed method is validated for the parameters, linearity, range, specificity, limit of detection and quantification, precision and accuracy. Method validation is done according to the guidelines of International Conference of Harmonization (ICH), British pharmacopeia (BP) and U.S. pharmacopeia (USP). Verification is done by estimation of CPM and Pred in parenteral dosage form.
Linearity
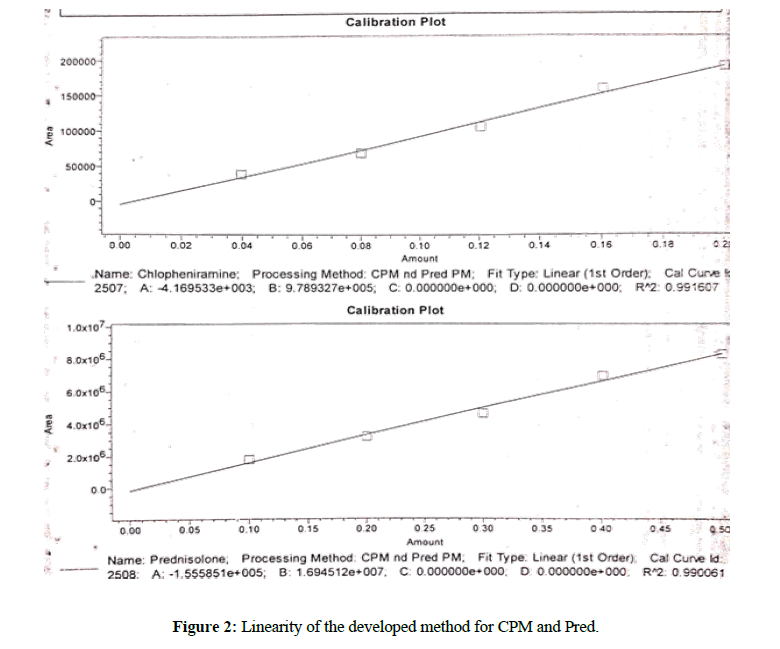
Linearity is usually calculated to check the response of method in different concentration ranges. The linearity of the method was calculated by analyzing the five concentrations of standard solutions. Calibration curve was drawn between the peak areas of the analyte peaks against the concentration of analyte in sample. For the entire range of concentrations for both analytes, linear correlation is observed with correlation coefficient of 0.9999 (Figure 2).
Limit of Detection and Limit of Quantification
The detection limit (LOD) of analytical procedure is a minimum amount of the analyte which can be detected by using that method. Limit of detection of analytical method can be calculated by comparing the signal of analyte and noise of the base line by analyzing the lowest amount of analyte [27]. In statistical terms, limit of detection may be calculated by calculating the slope of calibration curve and standard deviation of the replicates of lowest amount of analyte. The obtained LODs for CPM and Pred are 85 μg.mL-1 and 32 μg.mL-1 respectively. Similarly, the quantification limit (LOQ) of analytical procedure is also calculated. According to the international guideline (ICH Gridlines for method validation), in case of limit of quantification, the signal to noise ratio should be 10:1. Calculated LOQs for CPM and Pred are 275 μg.mL-1 and 108 μg.mL-1.
Repeatability
Repeatability is the degree of closeness of results obtained by analyzing the no of replicates of homogeneous sample under same conditions [28]. Repeatability of the developed method is calculated by preparing concentrations of standard solutions of Pred and CPM. Six replicates of CPM standard of 0.4 mg. mL-1 and 0.1 mg. mL-1 of Pred are analyzed. Obtained RSD values are 1.1% and 0.7%, respectively. According to international guidelines i.e. international conference of harmonization guidelines for method validation (Q2) and International pharmacopeias i.e. united states [pharmacopeia and British the value of relative standardization should be less than 2 for affective method. These results indicate that developed method is repeatable under optimized conditions.
Intermediate Precision
According to the ICH guidelines for method validation, intermediate precision of analytical method is an ability of method to produce precise results either by analyzing the sample on different equipment, by using different analyst or by analyzing the sample on different days. Here intermediate precision of developed method is calculated by using 0.4 mg. mL-1 concentration solution of CPM and 0.1 mg. mL-1 of Pred. Six replicates of prepared analyte standard are analyzed on two different days and results obtained are described in the Table 1. According to the international guidelines and pharmacopeias, the value of square root of Σ (%RSD)2 on different days should be less than 2. Obtained Ʃ (%RSD)2 of the developed HPLC based method is 1.4 and 1.2 for CPM and Pred, respectively.
| Replicates | Day One | Day Two | Day One | Day Two |
|---|---|---|---|---|
| Concentration of CPM (0.4 mg/mL) | Concentration of Pred (1 mg/mL) | |||
| Replicate 1 | 15474 | 166584 | 738796 | 738279 |
| Replicate 2 | 15784 | 164589 | 737761 | 720545 |
| Replicate 3 | 15727 | 166872 | 733817 | 732393 |
| Replicate 4 | 15686 | 164825 | 730924 | 737935 |
| Replicate 5 | 15363 | 167892 | 725986 | 736258 |
| Mean | 15606.8 | 166152 | 733456.8 | 733082 |
| SD | 179.7184 | 1405.592 | 5229.504 | 7387.464 |
| %RSD | 1.15 | 0.8 | 0.51 | 1.007 |
| (%RSD)2 | 1.32 | 0.71 | 0.508 | 1.01 |
| ⅀(%RSD)2 | 2.044 | 1.52 | ||
| SQRT of Sum | 1.4299 | 1.2 | ||
*SQRT = Square root
SQRT of Sum = Intermediate Precision
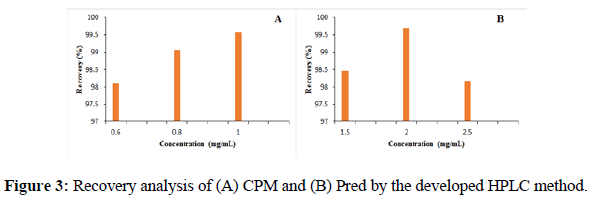
Accuracy The accuracy was calculated by spiking 50%, 100% and 150% amount of the targeted concentration of analyte. According to ICH guidelines for method validation the accuracy of analytical procedure should be calculated for at least nine concentrations i.e. three replicates of three different concentrations which cover entire range of the analyte. In this study targeted concentration of the analyte was calculated from three replicates of 0. 1mg.mL-1 standard solution of Pred and 0.4 mg. mL-1 of CPM. Three different concentration solutions are prepared by spiking 50%, 100% and 150% to the amount of targeted concentration in the sample. Three replicates of each concentration of analyte are analyzed and results obtained indicate that both samples show recovery from 98% to 100 % (Figure 3).
Uncertainty of the Method
Uncertainty of the method should be in the reference limits otherwise method validation is considered as null and void. Lower value of the measured uncertainty give surety about the authenticity of the results. Combined uncertainty of the results is calculated by taking the square root of the sum of the squares of the %RSD of all the validation parameters. Expanded uncertainty is also calculated which is double of the combined uncertainty (Table 2).
| Parameter | %RSD of CPM | Square of %RSD of CPM | % RSD of Pred | Square of % RSD of Pred |
|---|---|---|---|---|
| Linearity | 1.05633 | 1.11584 | 1.07219 | 1.1495 |
| Repeatability | 1.1 | 1.21 | 0.7 | 0.49 |
| Intermediate Precision | 0.8 | 0.64 | 1.2 | 1.44 |
| Practical Bias | 0.038029527 | 0.001446 | 0.0470914 | 0.0022175 |
| Sum of Square | 1.7 | - | - | 3.08 |
| Combined uncertainty | 1.3 | - | - | 1.73 |
| Expanded Uncertainty | 2.607 | - | - | 3.46 |
Uncertainty= √ (%RSD of Linearity)2+ (%RSD of Bais)2+ (%RSD of Precision)2
Method Verification
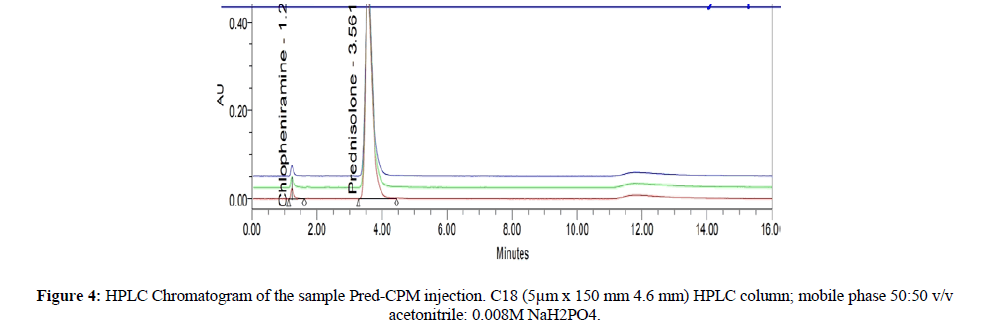
Method after validation studies is applied to analyze parenteral dosage form for qualitative and quantitative studies of PRED and CPM using a brand named as Pred CPM injection. Method was applied successfully for Pred CPM injection manufactured by Nova Med Pharma. The method responded well and there is no interference by excipients. Results of dosage forms are given in Figure 4. Method is verified by using Pred CPM injection 1 mg. mL-1 + 0.4 mg. mL-1. HPLC chromatogram indicates that method can successfully detect both analytes from commercially available products with being affected by excipients.
Conclusion
In this research, HPLC based method is developed for the analysis of Chlorpheniramine or (shortly CPM) and Prednisolone (Pred) For the purpose of separation, identification and determination WATERS HPLC was utilized for simultaneous analysis of Pred and CPM. Column employed is WATERS C18 5μm (150 mm x 4.6 mm). Mobile phase composition was 50: 50 ACN and Buffer. This method is found to be in limits defined by validation parameters Linearity, Intermediate precision and Accuracy. Verification of this method is done on parenteral dosage form named PRED-CPM, which is in accordance with defined guiding principle. Linearity was in range with r2 values of 0.991607 for CPM and 0.990061 for PRED. Precision results corresponds to the guideline as percent RSD is less than 2 (repeatability %RSD for PRED is 0.6 for CPM is 0.7 and intermediate precision %RSD for PRED is 0.164964 and for CPM is 0.203958). Outcomes of LOD for PRED are 0.03279 mg. mL-1 and for CPM are 0.0885 mg. mL-1. Outcomes for LOQ for CPM are 0.274 mg. mL-1 and for PRED are 0.108 mg. mL-1. Accuracy parameters outcomes for CPM are within range of 98.09446% to 99.5733% and for PRED are within range of 98.16576% to 98.46571%. Results obtained during verification process are accurate and within system suitability parameters provided by ICH and pharmacopeias. Results are reproducible at different days and time. This developed method is applicable for simultaneous determination of PRED and CPM in bulk and parenteral dosage form.
Acknowledgements
Authors acknowledge Higher Education Commission (HEC) of Pakistan for their financial support. Furthermore, author declares no conflict of interest.
References
[1] DF Guo, Z Lin, Y Wu. Diabetes. 2019, 68: p. 1591-1603.
[2] K Yamauchi and M Ogasawara. Int. J mol. sci. 2019, 20: p. 1733.
[3] A. Mandola, A Nozawa and T Eiwegger. J. Lym Sign. 2019, 6: p. 35-51.
[4] M. Durante, S. Sgambellone, C. Lanzi et al., Frontiers in pharmacology. 2019, 10: p. 525.
[5] JP Bolam and TJ Ellender. Neuropharmacology. 2016, 106: p. 74-84.
[6] B Sadek, A Saad and A Sadeq et al., Behavioural brain research. 2016, 312: p. 415-430.
[7] JM Arrang, M. Garbarg and JC Schwartz. histamine receptor. Nature. 1983, 302: p. 832.
[8] JH Cox, S Seri, AE Cavanna. Pediatric health, medicine and therapeutics. 2016, 7: p. 57.
[9] K Takeshita, KB Bacon and F Gantner. J. Pharm. Exp Ther. 2004, 310: p. 272-280.
[10] A. Salem, S Rozov, A. Al‐Samadi et al., British J. Derm. 2017, 176: p. 1213-1223.
[11] PJ Dunford, KN Williams, PJ Desai et al., J. all. Clin. imm. 2007, 119: p. 176-183.
[12] E Van Ganse, L Kaufman, MP Derde et al., J. Eur. Resp. 1997, 10: p. 2216-2224.
[13] WB Mendelson, T. Roth, J. Cassella et al., Sleep Medicine Reviews. 2004, 8: p. 7-17.
[14] R Csiernik. Canadian Scholars’ Press. 2019.
[15] R Gami-Yilinkou, A. Nasal and R. Kaliszan. J. Chrom. 1993, 633: p. 57-63.
[16] DS Pearlman. pharmacology and clinical use. Drugs. 1976, 12: p. 258-273.
[17] A. Mescheder and M Karrasch. Method of administering a cationic liposomal preparation. In: Google Patents. 2016.
[18] SL Gray, ML Anderson, S Dublin et al., JAMA internal medicine. 2015, 175: p. 401-407.
[19] SI Stephens, LG Hamel and GE Barclay. Wong PS. Chlorpheniramine therapy. In: Google Patents; 1989.
[20] AD Nebreda, CD Zappia, AR González et al., Eur. J. pharm. 2019, 847: p. 42-52.
[21] DG Adegoke. University of Saskatchewan. 2019.
[22] FER Simons and KJ Simons. J. All. Clin. Immun. 2011, 128: p. 1139-1150.
[23] CM Schempp, F. Schauer, CK Huhn et al., J. der Deut. Derma. 2019, 17:167-181.
[24] FER Simons. New England Journal of Medicine. 2004, 351: p. 2203-2217.
[25] C Stahn, M Löwenberg, DW Hommes et al., Mol. Cell. endo. 2007, 275: p. 71-78.
[26] XQ Fu, JY Cai and MJ Li. Molecular genetics & genomic medicine. 2019, 7: p. 00800.
[27] S. Reichgott, WJ Shakespeare, G. Kechter. In: Google Patents; 2019.
[28] B. Veeraswami and B Naveen. Plasma development. 2019, p.12.