Research Article - Der Pharma Chemica ( 2022) Volume 14, Issue 6
Formulation, Characterization and In-Vitro Evaluation of Herbal Sunscreen Cream Containing Anthocyanins
Praveenkumar V Vijapur1, Laxman S Vijapur1*, Anita R Desai1, Madhushree1, Sushmita V Mudalagi1, Apoorva P Daddenavar1, Laxmi M Hatti1 and Venkatesh Kustagi22Department of Pharmachemistry, BVVS Hanagal Shri Kumareshwar College of Pharmacy, Bagalkot, Karnataka, India
Laxman S Vijapur, Department of Pharmaceutics, BVVS Hanagal Shri Kumareshwar College of Pharmacy, Bagalkot, Karnataka, India, Email: laxman906@yahoo.co.in
Received: 28-May-2022, Manuscript No. dpc-22-65219; Editor assigned: 30-May-2022, Pre QC No. dpc-22-65219; Reviewed: 13-Jun-2022, QC No. dpc-22-65219; Revised: 15-Jun-2022, Manuscript No. dpc-22-65219; Published: 22-Jun-2022, DOI: 10.4172/0975-413X.14.6.1-6
Abstract
Herbal sunscreen cream containing anthocyanins extract of Punica granatum L and Brassica oleracea were successfully formulated and characterized. Anthocyanins were extracted from the Punica granatum L & Brassica oleracea L by maceration technique using 1% HCl (v/v) in methanol as a solvent. Anthocyanins cream were formulated using stearic acid, liquid paraffin, glycerin, cetyl alcohol, propyl paraben, propylene glycol, tri-ethanolamine, and distilled water. All the Anthocyanins cream formulations were evaluated for visual appearance and homogencity, pH, viscosity, spreadability, extrudability, centrifuge and in-vitro evaluation of sun protection factor was determined & formulations were evaluated for pH of which were about 6.6-6.9 within the range of skin pH. All the formulations were found to be homogeneous and there were no aggregate formation, there were no observable sediment in centrifuge tests. All the cream formulations showed good viscosity and they were capable to remain in the site of application for prolonged time. The values of spreadability indicated that the cream was easily spreadable by small amount of shear. All the cream formulations showed good extrudability. Among these formulations F2 showed excellent extrudability. In-vitro sun protection factor of F3 formulation showed enhanced protection against UV radiation as compared to F1 & F2.
Keywords
Anthocyanin; Punica granatum L; Brassica oleracea L; Topical Cream; SPF
INTRODUCTION
Anthocyanins are blue, red, or purple pigments found in plants, especially flowers, fruits and tubers. In acidic condition, anthocyanin appears as red pigment while blue pigment anthocyanin exists in alkaline conditions. Anthocyanin is considered as one of the flavonoids although it has a positive charge at the oxygen atom of the C-ring of basic flavonoid structure. It is also called the flavylium (2- phenylchromenylium) ion. The stability of anthocyanin is dependent on pH, light, temperature and its structure.
Anthocyanins are commonly found in flowers and the fruits of many plants. Most of the red, purple and blue-colored flowers contain anthocyanins. Red flowers are red hibiscus, red rose, red pineapple sage, red clover and pink blossom. These red flowers are edible. Blue (cornflower, blue chicory and blue rosemary) and purple (purple mint, purple passion flower purple sage, common violet and lavender) flowers are the common edible flowers.
Among the anthocyanin pigments, cyanidin-3-glucoside is the major anthocyanin found in most of the plants. The colored anthocyanin pigments have been traditionally used as a natural food colorant. The color and stability of these pigments are influenced by pH, light, temperature and structure [1].
Sun is the main source of Energy and life. Some research person says that sun has harmful effects including acute effects (Ex: Sunburn) and chronic risks of sunrays exposure like crack, melanoma, cancer and immune suppression [2]. Sun emits a constant flow of energy in the form of electromagnetic radiation spanning an enormous range of wavelength from 105 meters down to 10-6nm [3]. Sunlight is composed of about 40% visible light, 50% infrared light and 10% ultraviolet light [4].
All radiation is a form of energy most of which is visible to the human eyes. UV Radiation is measured on scientific scale called the electromagnetic spectrum [5]. UV Radiation is defined as Electromagnetic radiation lies between X-rays and visible light whose wavelength is from 200 to 400nm [6]. UV Radiation is essential to human health and it is necessary for the production of vitamin D3 in the skin. Vitamin D3 is essential for the intestinal absorption of calcium and phosphorus. Deficiency of vitamin D3 causes Osteoporosis in adults and growth retardation and skeletal deformities in children [4,7]. UV rays particularly with wavelength below 320 are most responsible for the therapeutic as well as noxious effects that we attribute to sunlight [8]. Exposure to solar UV radiation for a long time causes a variety of skin damage. Sunburn, skin pigmentation, premature aging and photo-carcinogenesis are some e.g. of skin damage due to UV radiation.
The main mechanism of Skin damage by UV radiation is the formation of Reactive Oxygen Species (ROS) that interact with proteins and lipids and subsequently alter them [9,10,11]. Solar radiation has the therapeutic effects on skin diseases such as psoriasis and eczema [4,7].
The demand for the herbal sunscreen increased over chemical based cream because of the advantage of herbal cream is having less or no side effects. The allergic reaction, negative side effects and comedogenic effects are not seen in sunscreen creams which are prepared with herbs [12,13]. The nature has gifted us with everlasting treasure of herbal ingredients which are easily available and found in large variety of plants thus they are more effective and reach with its stability, safety & purity. The cost effective of the herbal sunscreen also shows the properties other than UV absorbing property thus acts as antioxidants and Anti-inflammatory [14].
MATERIAL AND METHODS
Materials
Stearic acid obtained from Thermo fisher sci ind.pvt. Ltd. Mumbai, Cetyl alcohol and Propyl paraben obtained from Genuine chemical co. Mumbai, Propylene glycol obtained from LOBA Chemie. Mumbai, Glycerin, Triethanolamine and Liquid paraffin obtained from SDFCL. Mumbai.
Methods
Anthocyanin Extraction from Punica granatum
Anthocyanin was extracted from fresh arils of pomegranate by placing in methanol which was contained 1% v/v of HCl in a beaker. The lid of the beaker was closed with aluminium foil and placed in a dark room for 24 hours at room temperature. After 24 hours filtration was carried out using filter paper. Then filtrate was evaporated on steam bath at 40ºC to get viscous anthocyanin, later dry anthocyanin was obtained by freeze drying at -135ºC temperature and 2.58 mbar pressure [15].
Anthocyanin Extraction from Brassica oleracea L
Anthocyanin was extracted from fresh leaves of red cabbage by placing in one and half times volumes of methanol which was containing 1% v/v of HCl in a beaker. The lid of the beaker was covered with aliminium foil and placed in a dark place for 24 hours at room temperature. The filtrate was evaporated under 40ºC. Then the anthocyanin extract obtained was collected and stored in 4ºC [16].
Confirmatory Test for Anthocyanin [17]
• Presence of Anthocyanin in extract was confirmed by UV-Visible Spectrophotometer using methanol as blank. A spectrum was taken in UV-Visible range i.e. 200nm-800nm.
• To the Punica granatum L. extract, aluminium chloride added that will give a shift of 12 nm in spectrophotometer which confirms the Anthocyanin.
• One ml of the Punica granatum L. extract was mixed with 2M HCl and heated for 5min at 100ºC. When the extract remains the stable purple (magenta) color confirms the presence of Anthocyanin.
• One ml of the Punica granatum L. extract mixed with 2M NaOH and the formation of green color indicates the presence of Anthocyanin.
Phytochemical Tests for Extract [18]
• The Phytochemical screening of Punica Granatum L. extracts was carried out according to the methods described by Khandelwal and Firdouse et al. Sunscreen preparation method [2]
Step I: Aqueous Phase Preparation: anthocyanin extract, Propylene glycol, Propyl Paraben, Glycerin dissolved in de-ionized Water were heated to 80°C.
Step II: Oil Phase Preparation: Stearic acid, liquid paraffin, Cetyl alcohol was mixed and heated at 80ºC.
Step III: Mixing Phase: Oil phase was added to aqueous phase at 80ºC with continuous stirring for 10-15 min, Triethanolamine was added to adjust the pH and then it was homogenized till uniform emulsion formed. It was then poured into the wide mouth container and stored at temperature not exceeding 37°C. All the formulations composition is shown in the Table 1.
| SI. NO | Ingredients in grams | F1 | F2 | F3 |
|---|---|---|---|---|
| >1 | Anthocyanin of Punica granatum | 6 | - | 3 |
| 2 | Anthocyanin of Brassica oleracea | - | 6 | 3 |
| 4 | Stearic acid | 20 | 20 | 20 |
| 5 | Liquid paraffin | 7 | 7 | 7 |
| 6 | Glycerin | 20 | 20 | 20 |
| 7 | Cetyl alcohol | 4 | 4 | 4 |
| 8 | Propyl Paraben | 0.04 | 0.04 | 0.04 |
| 9 | Propylene glycol | 10 | 10 | 10 |
| 10 | Triethanolamine | qs | qs | qs |
| 11 | Water | qs | qs | qs |
| 12 | Fragrance | qs | qs | qs |
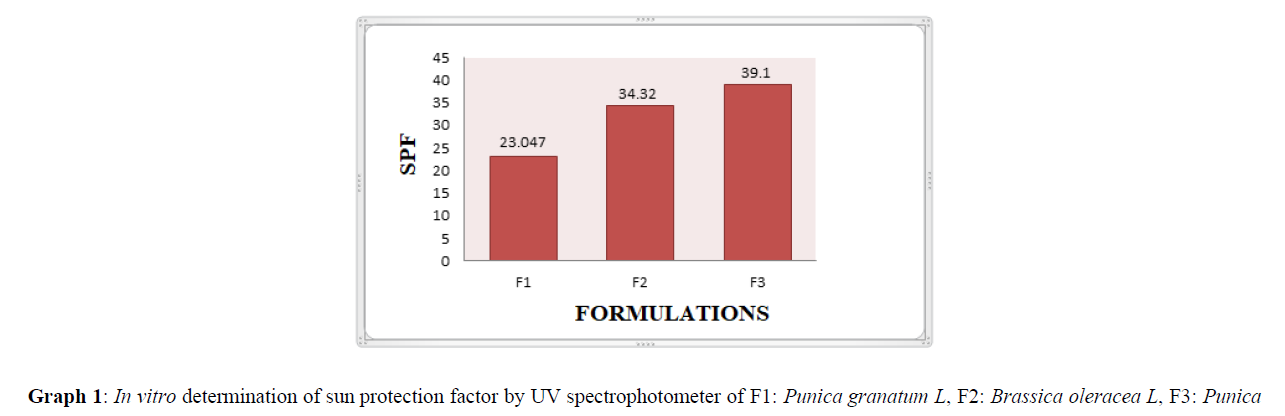
In-vitro Determination Sun Protection Factor (SPF) by UV- spectrophotometer [2]
Cream containing 6% w/w of anthocyanin was weighed, transferred to 100 ml volumetric flask and diluted to volume with methanol. Further, it was kept for ultra- Sonication for 5 mins. Afterwards 1 ml aliquot was transferred to 10 ml volumetric flask and the volume was adjusted with methanol. The absorption data obtained in the range of 290-320 nm every 5 nm interval and 3 determinations were made at each point. The absorbance values of anthocyanins are obtained. SPF was calculated using the formula,
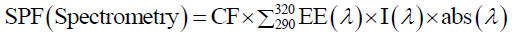
CF=10 (Correction Factor),
EE (λ) = Erythemogenic effect of radiation at wavelength λ,
I (λ) = Intensity of solar light at wavelength λ,
abs (λ) = Absorbance of wavelength λ
Evaluation of the formulation
Visual appearance and Homogeneity [19]
The prepared gels were visually analyzed for clarity, color and transparency. The prepared creams were also evaluated for the presence of any
aggregation. Creams were prepared and transferred into transparent container then observed under the microscope for the presence of any particle or grittiness.
pH determination [20]
The pH of various formulations was determined by using a digital pH meter. About 1 gram of cream was weighed and dissolved in 100 ml of distilled water and kept for 2 hours. The measurement of pH of each formulation was done in triplicate and average values were recorded.
Extrudability test [21]
About 5 gram of the cream formulation was filled in a clean, lacquered aluminum collapsible tube on crimped end of the tube then clamp was applied to avoid any roll back. And the cap was removed and cream was extruded. The extrudability was then determined by measuring the amount of cream extruded through the tip. The extruded cream was collected and weighed and the percentage of cream extruded was calculated and grades were allotted.
(>90% Extrudability: Excellent)
(>80% Extrudability: Good)
(>70% Extrudability: Fair)
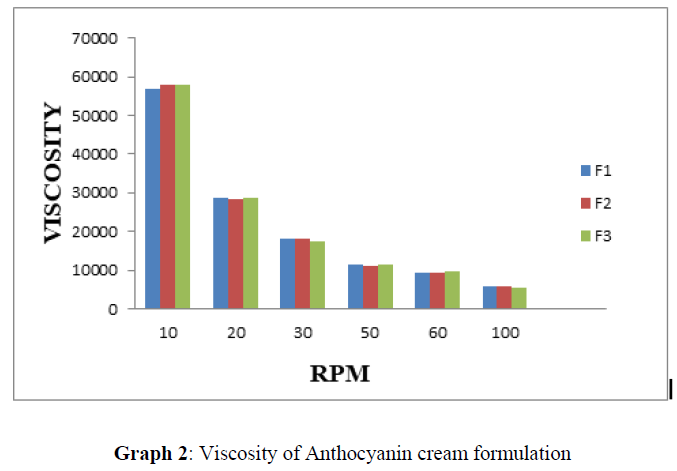
Viscosity [22]
Viscosity of formulated cream was determined by using Brookfield viscometer. The creams were rotated at 50 rpm using spindle no.64. At each speed, the reading was recorded. The viscosity determination of samples was repeated three times.
Spreadability [23]
Spreadability was determined by the apparatus which consists of a glass plate block, which was provided by a pulley at one end. By this method spreadability was measured on the basis of slip and drag characteristics of creams. An excess of cream (about 2 gm) under study was placed on this ground slide. The cream was then sandwiched between this slide and another glass slide having the dimension of fixed ground slide and provided with hook. About 20 grams weight was placed on the top of the two slides for 5 minutes to expel air and to provide a uniform film of the cream between the slides. Excess of the cream was scrapped off from the edges. The top plate was then subjected to pull of 20 gm. With the help of string attached to the hook and the time (in seconds) required by the top slide to cover a distance of 6.5 cm be noted. A shorter interval indicates better spreadability. Spreadability was calculated using the following formula:
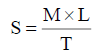
Where,
S = Spreadability,
M = Weight in the pan (tied to the upper slide),
L = Length moved by the glass slide (6.5 cm) and
T = Time (in sec.) taken to separate the slide completely each other.
Centrifuge test [20]
Prepared formulations were separately centrifuged in a test tube of 10 cm long and 1 cm width for 5, 15, 30 and 60 minutes with 2000 rpm and then studied for sedimentation and cream stability.
RESULTS AND DISCUSSION
The present investigation attempted to formulate suncreen using anthocyanin extract of Punica granatum L and Brassica oleracea by maceration method. Further cream were formulated by dispersion method and subjected for various evaluation parameters and optimized formulation was subjected for in-vivo determination of Sun Protection Factor (SPF) by UV- spectrophotometer method.
Confirmation test for anthocyanin
The Anthocyanin extract was analysed in UV-Visible spectrophotometer and found that maximum absorbance was at 532nm. Addition of aluminium chloride to the Punica granatum L. extract gave a shift of 12 nm in spectrophotometer due to the substitution or solvent effect it showed bathochromic shift on spectrophotometer, and may contain cyanidin, delphinidin, or petunidin. The shift of absorption towards a shorter wavelength basically called as blue shift. Punica granatum L. extract mixed with 2M HCl and heated for 5min at 100ºC. Extract remain in purple color due to the dominant form of anthocyanin. Punica granatum L. extract mixed with 2M NaOH, green color appeared due to the formation of quinoidal base.
Phytochemical investigation of Extract
Phytochemical screening is very important parameter to determine the phytoconstituents in the extract. Phytochemical study of Punica granatum methanolic extract contained Anthocyanin, flavonoids, flavanoidal glycosides, phenolic compound and glycosides as active phytoconstituents.
SPF
Sun protection factor is a metric for determining how effective a sunscreen product is. In this work, the sunscreen activity of a cream formulation containing Brassica oleracea L and Punica Granatum L anthocyanin extract was assessed using an in-vitro SPF technique [24]. Graph 1 displays the SPF value. Table 2 shows the stated SPF values of broad-spectrum sunscreens SPF of F1 was 23.047, F2 was 34.32 and F3 was 39.1, by comparing the SPF values of Table 2 it was evident that F3 was having the greater UV screening property than other two formulations.
| SI.NO | SPF | Percent of UV blocked |
|---|---|---|
| >1 | 2 | 50 |
| 2 | 4 | 75 |
| 3 | 8 | 87.5 |
| 4 | 16 | 93.75 |
| 5 | 32 | 96.88 |
| 6 | 64 | 98.44 |
Granatum + Brassica oleracea L.
Visual appearance and Homogeneity
Almost all the prepared cream formulations were visually analyzed they were found to be clear, transparent; there was no aggregation found and free from presence of particles.
pH determination: The pH of Anthocyanin cream was evaluated and it is constant throughout 1month.The results of cream pH are demonstrated in Table 3.
| Formulation code | pH (Mean Í?+SD) | Extrudability in percentage(MeanÍ?Í? Í?+ SD) | Spreadability in gm.cm/sec(Mean Í?+ SD) |
|---|---|---|---|
| >F1 | 6.9Í?+ 0.05 | 74.86Í?+0.003 | 14.44Í?+0.15 |
| F2 | 6.6Í?+ 0.1 | 90.69Í?+0.007 | 6.5Í?+0.05 |
| F3 | 6.8Í?+ 0.05 | 81.58Í?+0.010 | 32.5Í?+0.10 |
Extrudability test
The various Anthocyanins cream formulations were subjected to extrudability. The values of extrudability indicate that the creams were showed good Extrudability (Table-3). Among these F2 formulation showed excellent extrudability.
Spreadability
To determine the spreading ability of cream the test was performed (Table-3). The cream having low viscosity showed better spreadability. The various Anthocyanins cream formulations were subjected to spreadability studies. The values of spreadability show that the cream was easily spreadable by small amount of shear (Graph 2).
Viscosity
Centrifuge test
Centrifugation test for creams is performed to check the phase separation of oil and water in formulation. The prepared formulation was stable and there was not any separation of phases on centrifugation at different speeds which indicates the creams were stable.
CONCLUSION
Herbal sunscreen cream containing anthocyanin can be prepared using different anthocyanins of Punica granatum L and Brassica oleracea L. SPF of F3 formulation showed enhanced protection against UV radiation as compared to F1 and F2. The pH of the cream formulations was in the range of 6.6 to 6.9 which lies in the normal pH range of the skin. All the formulations were found to be homogeneous and there were no aggregate formation, there were no observable sediment in centrifuge tests. All the cream formulations showed good viscosity and they were capable to remain in the site of application for prolonged time. The values of spreadability indicate that the cream was easily spreadable by small amount of shear. All the cream formulations showed good extrudability.
ACKNOWLEDGEMENT
I wish to express my sincere gratitude to our beloved Principal, Guide, faculty members and non-teaching staff of department of pharmaceutics, Hanagal Shri Kumareshwar College of Pharmacy, Bagalkot. For providing necessary facilities to carry out this project work.
References
- Khoo HE, Azlan A, Tang ST, et al., J Food Nutri Res. 2017, 61(1): p. 1-21.
- Balogh TS, Velasco MV, Pedriali CA, et al., An Bras Dermatol. 2011, 86(4): p. 732-742.
- Butler H. Textbook of Pouchers, Perfumes, Cosmetics and soaps. Edition 10. 2000, p. 468
- Radice M, Manfredini S, Ziosi P, et al., Fitoterapia. 2016, 114: p. 144-62.
- https://www.fda.gov/radiation-emitting-products/tanning/ultraviolet-uv-radiation
- A textbook of cosmetic science by Dr.Aijaz A. Sheikh et al., p.191
- Gonzàlez S, Fernández-Lorente M, Gilaberte-Calzada Y. Clin Dermatol. 2008, 26: p. 614-626.
- A Handbook of cosmetics by B.M.Mithal and R.N.Saha edition 2000 page no.90
- Lakhdar H, Zouhair K, Khadir K, et al., J Eur Acad Dermatol Venereol. 2007, 21(6): p. 738-742.
- Wolf R. T zn B and T zn Y. Dermatol Ther. 2001, 14(3): p. 208-214.
- Imam S, Azhar I, Mahmood ZA. Asian J Pharm Clin Res. 2015, 8(3): p. 234-237.
- Kulkarni SS, Bhalke RD, Pande VV, et al., Indo Am j Pharm. 2014, 4(2): p. 1104-1108
- Prasanth B, Soman A, Jobin J, et al., W J Current Med Pharml R. 2020, 2(1): p. 14-20.
- Saewan N, Jimtaisong A. J cos derm. 2015, 14(1): p. 47-63.
- Vankar PS, Srivastava J. Int J Food Eng. 2010, 6(4): p. 01-11.
- Iosub I, Kajzar F, Makowska-Janusik MA, et al., Display Imaging. 2014, 1: p. 175-193.
- Harborne AJ. Spring Sci Bus Med. 1998.
- Firdouse S, Alam P. Int J Phyto med. 2011, 3(1): p. 32.
- Dhawan S, Medhi B, Chopra S. Sci Pharm. 2009, 77(2): p. 465-482.
- Aslani A, Zolfaghari B, Davoodvandi F. Adv Pharm Bull. 2016, 6(3): p. 391-398.
- Avinash S, Gowda DV, Suresh J, et al., Sch Res Libr. 2016, 8(8): p. 255-266.
- Jadhav VD, Talele Swati G, Bakliwal Akshada A, et al., J Pharm Biosci. 2015, 3: p. 65-72.
- Sumeet D, Shailesh G. Acta Chim Pharm Indica. 2012, 2(1): p. 54-59.
- Mark R. Eur J Dermatol. 1999, 9(5): p. 406-412.
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref