Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 7
Identification of Hepatoprotective Peptide Based Leads against Multiple Receptor Targets using Docking Strategy.
Nagarajan K*, Shikha, Grover P and Ghai R
Department of Pharmaceutical Chemistry, KIET School of Pharmacy, 13 Km. Stone, Ghaziabad Meerut Road, Ghaziabad-201206, Delhi-NCR, India
- *Corresponding Author:
- Nagarajan K
Department of Pharmaceutical Chemistry
KIET School of Pharmacy
13 Km. Stone, Ghaziabad Meerut Road
Ghaziabad-201206, Delhi-NCR, India
Abstract
Peptides are well known for bio-friendly properties are being designed as the hepatoprotective agents in this research study. After the analysis of all the results using Swiss Dock against different targets, it was observed that particular peptides (Arg-Lys-Pro, Lys-Glu-Leu) were identified as a best lead against the asialoglycoprotein receptor 1, TLR-2 and nulear receptor subfamily 5 group a member 2. Later, when these compounds were subjected to docking against the P-glycoprotein, it was found that Arg-Lys-Pro and Lys-Glu-Leu were having fullfitness energy as -175561.75 kJ/mol (lesser binding energy) and -155255.80 kJ/mol (lesser binding energy). In similarity, the same tripeptides (Arg-Lys-Pro, Lys-Glu-Leu) which are effective against and receptor targets are having very less binding energy with P-gp, indicating P-gp efflux mechanism for these drugs is low and hence they surely may have enhanced bioavailability even if tested in various phases of clinical trials. Hence, these peptides are discovered as one of the leads against their respective target sites, which can be utilised in the liver ailments and further needs investigation in pre-clinical & clinical studies as suitable hepatoprotective agents in near future.
Keywords
Asialoglycoprotein receptor 1, TLR-2, Nuclear receptor subfamily 5 group a member 2, P-glycoprotein.
Introduction
The liver performs a vital role in metabolism, secretion, storage, and detoxification of endogenous and exogenous substances. Hepatic damage occur by various mechanisms such as oxidative stress and free radicals enhance the severity of hepatic damage, hepatic infection by multiple viruses, hepatocellular carcinoma, hepatic fibrosis, cirrhosis, alcoholic fatty liver disease, non-alcoholic fatty liver disease, steatosis and xenobiotic induced.
Hepatic damage can be prevented by blocking these mechanisms with the help of various ligands which will bind at the specific receptor target site and stop the further process. Some of the receptor targets for hepatic damage includes Asialoglycoprotein receptor 1, TLR 2, Nuclear receptor subfamily 5 group a member 2 and P-glycoprotein.
The asialoglycoprotein receptor 1 (ASGR1) spans the plasma membrane which contributes to the serum glycoprotein homeostasis as it mediates endocytosis of glycoproteins followed by lysosomal degradation of glycoproteins with exposed terminal galactose or N-acetylgalactosamine residues [1]. Asialoglycoprotein receptor 1 autoantibodies have been considered specific markers of autoimmune hepatitis (AIH). The exact mechanisms responsible for the development of these autoantibodies and leading to autoimmunity to this peculiar liver receptor remain elusive [2].
The asialoglycoprotein (ASGPR), being a hepatic receptor is recycled through a process called receptor mediated endocytosis (RME). In a healthy cell, RME process aids in the proper routing of receptors and their ligands, hence any type of missorting or altered transport of proteins may become a probable reason for diseases connected with hepatocyte and liver dysfunction. Conformational studies have been conducted on knockout mice (who lack a functional ASGP receptor) being intoxicated using alcohol, anti-Fas (CD95) antibody, lipopolysaacharide/galactosamine and chemical carbon tetrachloride (CCl4). The results clearly exhibited that the knockout mice sustained augmented hepatic injury as seen by their enhanced indices of hepatic damage, like raised serum enzyme levels. Histopathological studies of liver of these knockout mice gives an evidence of association between hepatic receptors and liver injury [3].
Toll-like receptors (TLRs) recognize pathogen associated molecular patterns (PAMPs) to identify pathogens. TLRs besides important for innate immunity, plays a critical part in inflammation, regulation in injury and wound healing. TLR-mediated signals have been implicated in a number of chronic liver diseases such as, liver fibrosis and cirrhosis, viral hepatitis, alcoholic liver disease, non-alcoholic fatty liver disease and hepatocellular carcinoma.
Due to its anatomical connections with the gut, the liver is continously exposed to git-derived bacterial products and also functions as a major filter organ and a first line of defense and also functions as a major filter organ and a first line of defense. Moreover, the liver is an important site for bacterial phagocytosis and clearance as it hosts more than 80% of the body's macrophages. Specialized liver macrophages named Kupffer cells play a crucial role in host defense which is linked to their ability to phagocytose, process and present antigen, and secrete various proinflammatory mediators including cytokines, prostanoids, nitric oxide, and reactive oxygen intermediates. So they are able to efficiently take up endotoxin and phagocytose bacteria carried through the portal vein and helps in the clearance of systemic bacterial infection. Kupffer cells, also express TLR4 and are responsive to lipopolysaccharide. Kupffer cells mediated the majority of cytokine and chemokine expression in liver after lipopolysacchride injection (25 μg i. p.) as demonstrated by depletion experiments. Kupffer cells also functionally express TLR2 [4].
The healthy liver is found to contain low mRNA levels of TLRs such as TLR1, TLR2, TLR4, TLR6, TLR7, TLR8, TLR9, TLR10 and signalling molecules such as MD-2 and MyD88 in comparison to other tissues/organs. Low TLR expression may be responsible for high liver tolerance towards TLR ligands from the intestinal microflora to which the liver is all the time exposed.
Nuclear receptor subfamily 5 group a member 2 is a ligand modulated transcription factors that play diverse roles in cell-differentiation, development, proliferation and metabolism and are associated with numerous liver pathologies such as cancer, steatosis, inflammation, fibrosis, cholestasis and xenobiotics/drug induced liver injury. The network of target proteins microRNAs, and long noncoding RNAs. The importance of the nuclear receptors as targets of liver disease is exemplified by the number of nuclear receptors that are currently used in the clinics or in clinical trials with promising results. Understanding the regulation of nuclear receptor during pathophysiological conditions and identifying ligands for orphan nuclear receptor, points to a potential therapeutic approach for patients with liver diseases [5].
P-glycoprotein is encoded by the multi-drug resistance (MDR1) gene that influences drug pharmacokinetics and pharmacodynamics. P-gp is a 170 kDa membrane-bound protein, an ATP dependent efflux transporter responsible for multidrug resistance of many drugs. The presence of P-gp in the liver, kidney, blood brain barrier, spinal cord and placenta has been identified. P-gp is present in the biliary canalicular membranes of hepatocytes in the liver. Intestinal drug efflux by P-gp is widely recognized as a major determinant for low or variable oral absorption and bioavailabilty of several drugs. Physiologically, P-gp limits the noxious exposure of drugs, toxin& xenobiotics to the body by pumping out of the cells. Due to its modulating role in the pharmacokinetic properties of clinically significant therapeutic agents its screening becomes important in the drug discovery process.
Drug efflux Pgp transporters can be inhibited or induced by various drugs or herbs leading various drug–drug or drug–herb interactions which may affect ADME of many drugs. Moreover, reports on the studies of genetic polymorphism of P-gp also exist ultimately affecting drug disposition. As Drug interactions and genetic polymorphism are important factors requiring attention, P-gp binding studies are emphasized.
An appreciable number of docking web servers have recently emerged as observed from the currently available web services like:
DockingServer (http://www.dockingserver.com/web)
Blaster [6]
DockingAtUTMB (http://docking.utmb.edu/)
Pardock (http://www.scfbio-iitd.res.in/dock/pardock.jsp)
PatchDock (http://bioinfo3d.cs.tau.ac.il/PatchDock/)
MetaDock (http://dock.bioinfo.pl/)
PPDock (http://140.112.135.49/ppdock/index.html)
MEDock (http://medock.ee.ncku.edu.tw/)
The aim of the present study is to come up with the potent hepatoprotective peptide based leads against multiple receptor targets by docking strategy.
Materials and Methods
The system used in this research work is an Acer Laptop with Intel ® Core™ i3-2348M, having CPU of 2.30GHz, 32 bit operating system and windows 7. The various sites used in the research work to estimate different parameters are Lipinski rules of five, Molinspiration, Swiss dock.
Initially, 250 shorter chain peptides (dipeptide, tripeptide, tetrapeptide, pentapeptide and hexapeptide 50 each) were randomly selected, out of them only 38 shorter chain peptides were selected via Lipinski rules of five. These 38 molecules were further subjected to Molinspiration after which only 16 molecules were selected which were further subjected to Swiss docking against the selected hepatic receptors such as Asialoglycoprotein receptor 1, Toll - like receptor 2, Nuclear receptor subfamily 5 group a member 2, P-glycotein.
Target protein for docking and Swiss dock software [7]
With the Swiss Dock software, we can extend the use of small protein molecule docking software by providing suitable answers to many of the problems. Firstly, protein structures that are manually curated can be downloaded from the website, and their original protein database files (PDB) can be prepared through ad hoc scripts [8,9]. Next the docking software is easily accessible through either a programmatic interface or a web browser. Then with a simple embedded applet, the predicted binding modes (BMs) can be viewed online or analyzed in more details to a flawless integration with the UCSF Chimera molecular viewer [10], with the help of the online documentation and the user community. In our study, 16 leads were selected namely Glu-Pro, Gly-Trp, Ala-Trp, Tyr-Pro, Ile-Ala, Ala-Glu-Pro, Ile-Pro-Pro, Val-Trp, Phe-Pro, Val-Tyr, Leu-Ser-Pro, Ile-Asp-Pro, Ile-Gly-Val-Pro, Lys-Phe-Pro, Lys-Glu-Leu, Arg-Lys-Pro and were subjected to swiss dock strategy against asialoglycoprotein receptor 1, TLR 2, nuclear receptor subfamily 5 group a member 2 and P-glycoprotein.
Swiss dock
Swiss Dock is a docking web server. The structure of ligand as well as the target protein can be automatically prepared for docking. All computations are performed on the server site itself, so that there is no requirement for any computational power from the user for docking runs. The docking results interpretation and their integration into existing research pipelines is greatly facilitated by the flawless revelation of docking predictions in the UCSF Chimera molecular viewer, which can be initiated directly from the web browser.
Web interface
Inputs: To start a docking assay through the web interface of Swiss Dock, only three steps are required: users must define one or several putative ligands and docking parameters and a protein structure. Users are guided throughout this simple and short submission process by a complete contextual help.
Target selection: A target protein structure can be determined either by uploading structure files or by specifying its identifier from the Protein Data Bank [11]. The first option let the uploading of user-defined or edited target structures. The second option lets the users who are not known with 3D structure files to start a docking assay with only a PDB code. If several PDB records are available for the same target, those with a high resolution and a ligand similar to the one that will be docked should be considered first.
Since the result interpretations are performed in the CHARMM force field, Swiss Dock supports the uploading of CHARMM formatted files in addition to the commonly used PDB format. Protein structures can be uploaded as a set of protein structure file (PSF), extra topology (RTF), coordinate file (CRD) and parameter files (PAR), if needed.
Ligand selection: A ligand can be selected either by uploading structure files or by specifying its identifier from the ZINC database [12]. The latter possibility allows users who are not known to 3D structure files to start a docking assay with only a ZINC accession code (AC). The former allows uploading several ligands at once or uploading ligands that are not present in the ZINC database. As for the target protein, Swiss Dock supports the widely used Mol2 format and also the direct upload of CHARMM input files describing the ligand: a coordinate file (PDB), parameter (PAR) and extra topology (RTF) files. The ligand is instantaneously set up after its definition and its prepared structure can be downloaded and reviewed prior to the docking assay, if needed.
Docking type: Three docking parameter presets can be selected: fast, very fast and accurate [9]. Briefly, several docking parameters are adjusted in order to reach the desired docking time and exhaustiveness of the search: the number of sampled binding modes, the number of minimization steps that are performed to relax the ligand and the number of predicted binding modes. The very fast and fast modes are most probably sufficient, if the ligand has less than 15 rotatable bonds and/or is likely to fit exactly into a binding pocket of the target protein.
Outputs: After a docking assay has been processed, it can be trailed by a dedicated URL provided on the submission confirmation page. If an email address that is optional has been specified in the submission form, this URL is also sent to the user by email, as well as a link to the docking result web page once the docking is completed. This docking result web page features a Jmol applet [13] for the revelation of the predicted binding modes within the web browser. For further visual investigations, UCSF Chimera can be started by a single click, and the predicted binding modes are automatically loaded in its View Dock plugin.
Predicted binding modes are also available to be download as a ZIP file. It contains PDB files for the predicted binding modes as well as complex in the DOCK format. For experienced users, CHARMM PSF/CRD/PAR/RTF files are also provided for subsequent calculations.
Community: An online help is available using the form of a wiki. A searchable forum is proposed to promote communication among users, such as tips, tricks, feedbacks, questions and answers, so that the experience gained by some may be shared with others more broadly than what a mailing list would allow for [14].
Results
The results of docking of peptide based drugs against multiple receptor targets are expressed in Tables 1-4.
| S. No. | Molecule | Receptor | Full-fitness (kcal/mol) | Estimated ΔG (kcal/mol) | Image |
|---|---|---|---|---|---|
| 1. | Glu-Pro | ASGR1 | -200498.61 | -1490.58 |  |
| 2. | Gly-Trp | ASGR1 | -193769.64 | -1547.26 |  |
| 3. | Ala-Trp | ASGR1 | -194497.23 | -1567.35 |  |
| 4. | Tyr-Pro | ASGR1 | -193561.44 | -1541.5 |  |
| 5. | Ile-Ala | ASGR1 | -196829.15 | -1521.55 |  |
| 6. | Ala-Glu-Pro | ASGR1 | -196433.92 | -1609.62 |  |
| 7. | Ile-Pro-Pro | ASGR1 | -188368.68 | -1577.97 |  |
| 8. | Val-Trp | ASGR1 | -193781.86 | -1532.48 |  |
| 9. | Phe-Pro | ASGR1 | -191219.94 | -1588.94 |  |
| 10. | Val-Tyr | ASGR1 | -195378.6 | -1559.75 |  |
| 11. | Leu-Ser-Pro | ASGR1 | -187567.07 | -1562.18 |  |
| 12. | Ile-Asp-Pro | ASGR1 | -198832.59 | -1582.11 |  |
| 13. | Ile-Gly-Val-Pro | ASGR1 | -186163.39 | -1679.63 | 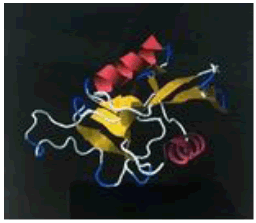 |
| 14. | Lys-Phe-Pro | ASGR1 | -189804.29 | -1669.15 | 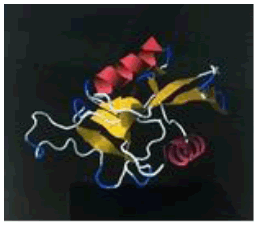 |
| 15. | Lys-Glu-Leu | ASGR1 | -200497.16 | -1725.55 | 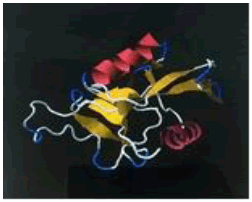 |
| 16. | Arg-Lys-Pro | ASGR1 | -223900.79 | -1688.72 | 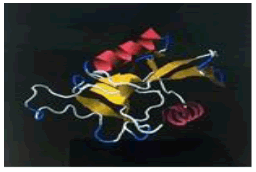 |
Table 1: The free energy estimated when the peptide hits are subjected for docking with the asialoglycoprotein receptor 1 (PDB code - 1DV8)
| S. No. | Molecule | Receptor | Full-fitness (kcal/mol) | Estimated ΔG (kcal/mol) | Image |
|---|---|---|---|---|---|
| 1. | Glu-Pro | TLR-2 | -300334.31 | -1592.16 |  |
| 2. | Gly-Trp | TLR-2 | -285951.15 | -1577.89 |  |
| 3. | Ala-Trp | TLR-2 | -293351.72 | -1623.8 |  |
| 4. | Tyr-Pro | TLR-2 | -292974.51 | -1640.82 |  |
| 5. | Ile-Ala | TLR-2 | -288587.55 | -1530.31 |  |
| 6. | Ala-Glu-Pro | TLR-2 | -295557.32 | -1653.45 |  |
| 7. | Ile-Pro-Pro | TLR-2 | -287510.1 | -1617.73 |  |
| 8. | Val-Trp | TLR-2 | -286042.87 | -1601.1 |  |
| 9. | Phe-Pro | TLR-2 | -290525.7 | -1653.83 |  |
| 10. | Val-Tyr | TLR-2 | -294745.95 | -1660.65 |  |
| 11. | Leu-Ser-Pro | TLR-2 | -286713.2 | -1651.28 |  |
| 12. | Ile-Asp-Pro | TLR-2 | -297774.8 | -1676.13 |  |
| 13. | Ile-Gly-Val-Pro | TLR-2 | -285211.82 | -1750.2 |  |
| 14. | Lys-Phe-Pro | TLR-2 | -287716.35 | -1757.47 |  |
| 15. | Lys-Glu-Leu | TLR-2 | -299233.58 | -1726.03 |  |
| 16. | Arg-Lys-Pro | TLR-2 | -322073.6 | -1768.96 |  |
Table 2: The free energy estimated when the peptide hits are subjected for docking with the toll-like receptor 2. (PDB code - 1FYW)
| S. No. | Molecule | Receptor | Full-fitness (kcal/mol) | Estimated ΔG (kcal/mol) | Image |
|---|---|---|---|---|---|
| 1. | Ile-Ala | Nuclear receptor subfamily 5 group a member 2 | -307966.89 | -1590.44 |  |
| 2. | Ala-Trp | Nuclear receptor subfamily 5 group a member 2 | -303667.43 | -1681.69 |  |
| 3. | Tyr-Pro | Nuclear receptor subfamily 5 group a member 2 | -300437.85 | -1670.03 |  |
| 4. | Gly-Trp | Nuclear receptor subfamily 5 group a member 2 | -299851.44 | -1658.77 |  |
| 5. | Glu-Pro | Nuclear receptor subfamily 5 group a member 2 | -305454.28 | -1627.52 |  |
| 6. | Val-Trp | Nuclear receptor subfamily 5 group a member 2 | -304941.71 | -1699.41 |  |
| 7. | Phe-Pro | Nuclear receptor subfamily 5 group a member 2 | -302968.07 | -1719.94 |  |
| 8. | Leu-Ser-Pro | Nuclear receptor subfamily 5 group a member 2 | -298808.02 | -1745.55 |  |
| 9. | Ile-Pro-Pro | Nuclear receptor subfamily 5 group a member 2 | -299161.5 | -1634.89 |  |
| 10. | Val-Tyr | Nuclear receptor subfamily 5 group a member 2 | -307184.34 | -1714.28 |  |
| 11. | Ala-Glu-Pro | Nuclear receptor subfamily 5 group a member 2 | -307014.88 | -1740.63 |  |
| 12. | Ile-Asp-Pro | Nuclear receptor subfamily 5 group a member 2 | -309290.43 | -1668.73 |  |
| 13. | Lys-Glu-Leu | Nuclear receptor subfamily 5 group a member 2 | -311483.63 | -1774.5 |  |
| 14. | Lys-Phe-Pro | Nuclear receptor subfamily 5 group a member 2 | -299626.66 | -1769.47 |  |
| 15. | Ile-Gly-Val-Pro | Nuclear receptor subfamily 5 group a member 2 | -293900.18 | -1731.67 |  |
| 16. | Arg-Lys-Pro | Nuclear receptor subfamily 5 group a member 2 | -334661.21 | -1762.74 |  |
Table 3: The free energy estimated when the peptide hits are subjected for docking with the nuclear receptor subfamily 5 group a member 2. (PDB code - 4IS8)
| S. No | Molecule | Receptor | Full-fitness (kcal/mol) | Estimated ΔG (kcal/mol) | Image |
|---|---|---|---|---|---|
| 1. | Ile-Ala | P-gp | -152230.33 | -1496.54 |  |
| 2. | Ala-Trp | P-gp | -150332.17 | -1578.66 |  |
| 3. | Tyr-Pro | P-gp | -148579.41 | -1502.33 |  |
| 4. | Gly-Trp | P-gp | -149671.8 | -1574.94 |  |
| 5. | Glu-Pro | P-gp | -152005.4 | -1398.73 |  |
| 6. | Val-Trp | P-gp | -149782.32 | -1583.2 |  |
| 7. | Phe-Pro | P-gp | -147076.37 | -1570.7 |  |
| 8. | Leu-Ser-Pro | P-gp | -142592.22 | -1590.15 |  |
| 9. | Ile-Pro-Pro | P-gp | -144824.52 | -1609.04 |  |
| 10. | Val-Tyr | P-gp | -150234.8 | -1575.39 |  |
| 11. | Ala-Glu-Pro | P-gp | -148515 | -1533.72 |  |
| 12. | Ile-Asp-Pro | P-gp | -151024.38 | -1557.01 |  |
| 13. | Lys-Glu-Leu | P-gp | -155255.8 | -1686.28 |  |
| 14. | Lys-Phe-Pro | P-gp | -145828.14 | -1772.17 |  |
| 15. | Ile-Gly-Val-Pro | P-gp | -139291.12 | -1636.72 |  |
| 16. | Arg-Lys-Pro | P-gp | -175561.75 | -1712.44 |  |
Table 4: The free energy estimated when the peptide hits are subjected for docking with the P-glycoprotein. (PDB code - 4KSD B)
Discussion
From the Tables 1-4 it is clearly identified that out of 16 candidates selected against asialoglycoprotein receptor 1, toll-like receptor 2, nuclear receptor subfamily 5 group a member 2 and p-glycoprotein. It was observed in the Table 1 that (Arg-Lys-Pro) having the fullfitness energy as -223900.79 kJ/mol, (Glu-Pro) having the full fitness energy as -200498.61 kJ/mol and (Lys-Glu-Leu) having the fullfitness energy as -200497.16 kJ/mol with respect to its docking with the receptor asialoglycoprotein receptor 1. From the Table 2 it was observed that (Arg-Lys-Pro) having the fullfitness energy as -322073.60 kJ/mol, (Glu-Pro) having the fullfitness energy as -300334.31 kJ/mol and (Lys-Glu-Leu) having the fullfitness energy as -299233.58 kJ/mol with respect to its docking with TLR-2 three candidates were selected from the results of docking against the receptor. From Table 3, it was observed that (Arg-Lys-pro) having the fullfitness energy as -334661.21 kJ/mol, (Lys-Glu-Leu) having the fullfitness energy as -311483.63 kJ/mol and (Ile-Asp-Pro) having the fullfitness energy as -309290.43 kJ/mol were observed to have the maximum fullfitness energy against nuclear receptor subfamily 5 group a member 2 as compared to the other molecules when compared with the rest of the molecules selected through molinspiration. At last, docking was performed for all the 16 molecules and it is found that the tetrapeptide Ile-Gly-Val-Pro was found to have the minimum fullfitness energy with regard to P-glycoprotein as -139291.12 kJ/mol, Leu-Ser-Pro was found to have the fullfitness energy as -142592.22 kJ/mol and Ile-Pro-Pro was found to have the fullfitness energy as -144824.52 kJ/mol respectively.
Conclusion
Peptides being the bio-friendly molecules are being desinged as the hepatoprotective agent. In analyzing all the results of Swiss Dock against various targets, we found that the particular peptides (Arg-Lys-Pro, Lys-Glu-Leu) were identified as a best lead against the asialoglycoprotein receptor 1, TLR-2 and nulear receptor subfamily 5 group a member 2. Later, when these compounds were subjected to docking against the P-glycoprotein, it was found that Arg-Lys-Pro and Lys-Glu-Leu were having fullfitness energy as -175561.75 kJ/mol (lesser binding energy) and -155255.80 kJ/mol (lesser binding energy). In similarity, the same tripeptides (Arg-Lys-Pro, Lys-Glu-Leu) which are effective against and receptor targets are having very less binding energy with P-gp, indicating P-gp efflux mechanism for these drugs is low and hence they surely may have enhanced bioavailability even if tested in various phases of clinical trials. Hence, these peptides are identified as the best lead against their respective target sites, which can be utilised in the liver ailments and have to be further explored for its pre-clinical and clinical studies as suitable hepatoprotective agents in near future.
Acknowledgement
The authors will always remain thankful to Dr. Jagannath Sahoo, Principal, KIET School of Pharmacy, Ghaziabad, Col. (Dr.) Amik K Garg, Director, KIET Group of Institutions, Ghaziabad and Dr. Manoj Goel, Director (Admin), KIET Group of Institutions for providing all the facilities.
References
- H. Li, J. Kim, L. Groy, J. Am. Chem. Soc., 2001, 123, 4867.
- ASGR1 asialoglycoprotein receptor 1 (Homo sapiens (human) Gene ID: 432, 2017.
- R. Dirk, M. Maria, G. Lapin, V. Sergey, C. Karsten, Springer Milan, Journal: Auto-Immunity Highlights., 2012, 23, 58.
- M.L. Serene, C. Lee, A Casey, B.L. McVicker, World J. Gastroenterol., 2009, 1194-1200.
- A. Mencin, J. Kluwe, R.F. Schwabe, Toll-like receptors as targets in chronic liver diseases, 2009, 58(5), 704-720.
- R. Swetha, Z. Xi, W.L Annu, Rev Pharmacol Toxicol., 2016, 56, 605-626.
- A. Grosdidier, V. Zoete, O. Michielin, J. Comput. Chem., 2009, 13, 2021–2030.
- J.J. Irwin, B.K. Shoichet, M.M Mysingery, N. Huang, F. Colizzi, P. Wassam, Y. Cao, J. Med. Chem., 2009, 52, 5712-5720.
- V.F. Zoete, M. Cuende, A. Grosdidier, O. Michielin, P. Swiss, J. Comput. Chem., 2011, 34, 698-701.
- A. Grosdidier, V. Zoete, O. Michielin, J. Comput. Chem., 2011, 35, 1013-1016.
- E.F. Pettersen, T.D. Goddard, C.C. Huang, G.S. Couch, D.M. Greenblatt, E.C Meng, T.E. Ferrin, J. Comput. Chem., 2004, 3, 1605-1612.
- H.M Berman, J. Westbrook, Z. Feng, G. Gilliland, T.N. Bhat,, H. Weissig, I.N. Shindyalov, P.E. Bourne,Nucleic Acids Res., 2000, 28, 235-242.
- J.J Irwin, B.K. Shoichet, J. Chem. Inf. Model., 2005, 45, 177-182.
- Jmol: an Open-Source Java Viewer for Chemical Structures in 3D, http://wwwjmol.org/ 2010.



