Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 3
In Vitro Antioxidant and Thrombolytic Activity of Artocarpus chaplasha Leaves
Bashudeb Talukder1, MD Forhad Hossain1, Rukhor Uddin Mubarok2, Nurul Absar2, Md Sahdat Hossain3, Tanzina Sharmin Nipun4 and SM Moazzem Hossain42Post Graduate Research Fellow, International Islamic University Chittagong, Bangladesh
3Post Graduate Research Fellow, BGC Trust University, Bangladesh
4Department of Pharmacy, University of Chittagong, Bangladesh
Abstract
Background: Herbal preparations were always used in our treatment from ancient times due to their convenience in case of little or no toxicity. This study was aimed to assess the antioxidant and thrombolytic properties of a plant Artocarpus chaplasha. Therefore, methanolic, ethanolic, ethyl acetate and chloroform extract from the leaves of Artocarpus chaplasha were studied. Methods: The extract were subjected to various in vitro methods like DPPH free radical scavenging activity, reducing power efficacy, total flavonoid content, total phenolic content to qualify and quantify the antioxidant potential. In addition, percent clot lysis activity of the extract were also studied. Result: From the result, it could be disclose that all the extract show antioxidant capacity and ethyl acetate extract show strongest antiradical activity with an IC50 value of 60 μg/ml in comparison to 6.12 μg/ml for standard ascorbic acid. The extract also possess flavonoid and phenolic content and among the extracts ethyl acetate extract also possess highest flavonoid and phenolic content. Percent clot lysis activity were in the range of 16.145% to 55.98% in comparison to 65% and 4.5% produced by positive control streptokinase and negative control water respectively. Percent clot lysis value accounted highest (55.98%) in case of ethyl acetate extract. Trial registration: For experimental clinical study on animal trial registration and permission was issued from departmental clinical ethical review committee, department of pharmacy, university of Chittagong. The trail registration reference number is ERC/DP/CU/2015/0012. Conclusion: Ethyl acetate extract exhibit maximum antiradical activity and thrombolytic activity, however lower than the positive control ascorbic acid and streptokinase.
Keywords
Herbal preparations, Antioxidant, Thrombolytic, Radical scavenging activity
Introduction
In order to discover and invent newer drug entity the contribution of nature is always acknowledged and natural resources like plants, herbs, microorganisms, marine products are of greater attention for medicinal chemists [1]. Herbal drugs due to their excellent therapeutic effects, cost effectiveness, minimal side effects have maintained their reputation as green medicine in case of health care system [2]. Herbal drugs were a inseparable part of human treatment due to their wide range of pharmacological activity like thrombolytic effect, antimicrobial activity, cytotoxic effect etc. [3].
The role of free radicals in cell inactivation through membrane lipid peroxidation has been documented in many diseases. That’s why; antioxidants are used to prevent cellular degeneration [4]. Antioxidants which obstruct the oxidizing chain reactions and ultimately block the oxidation of other molecules are of two types namely, synthetic and natural [5].
Synthetic antioxidants possess carcinogenic property and hence preference over natural antioxidants has been increased. Thus, with an aim to protect cell from oxidation plants sources which contain natural antioxidants are of greater concern [4]. In addition to the antioxidant activity simultaneous presence of certain other activities like antiinflammatory, antiatherosclerotic, antitumor, antimutagenic, anticarcinogenic, antibacterial, or antiviral activities in natural plant sources are also documented [5].
As like as synthetic antioxidants, the harmful effects of synthetic anti thrombolytic agents has been disclosed. Though, synthetic anti-thrombolytic agents like urokinase (UK), streptokinase (SK) or tissue plasminogen activators (t-PA) has gained popularity in different thrombotic diseases in developing countries due to their lower cost but their advantage does not outweigh their disadvantages. These agents are associated with nonspecific pharmacological action, intracranial hemorrhage, and severe anaphylactic reaction. Moreover, these agents are avoided in certain pathological conditions like a patient with a history of nervous lesions, gastrointestinal bleeding or hypertension. In this situation, natural resources have been utilized to evaluate safer and effective antiplatelet, anticoagulant and antithrombotic effect by many researchers. In addition, it is also evident foods that possess antithrombotic property could lower the thrombosis. Some examples of plants which contain antithrombolytic effect are Ageratum conyzoides L. (Goat weed or white weed or Chuva), Clausena suffruticosa (Kalomoricha), Leea indica (Burm.f.) Merr. (Achilagach or Arengi), Leucas aspera Willd. (Darkolos or Dandokolos), Senna sophera L. Roxb. (Kanduak or Khuksu), and Solanum torvum Swartz. (Titbegun) etc. [2].
As a part of our ongoing research with natural plants [6,7] we have evaluated the in vitro antioxidant and thrombolytic property of different organic extracts of Artocarpus chaplasha leaves in this study. Artocarpus chaplasha (Synonyms: A. chama) which is indigenous to northeastern India, lower Myanmar and the Andaman and Nicobar Islands belongs to the family Moraceae and the Bengali name of is Chapalish [1].
Methods
Collection of plant leaves
Artocarpus chaplasha leaves were collected from Jahangirnagar University, Savar and recognized with the help of Dr. Shaikh Bokhtear Uddin, Taxonomist and Associate Professor, Department of Botany, University of Chittagong. A voucher specimen (Accession number: DP/CU/2015/PS-00813) has also been maintained in the herbarium, department of pharmacy, university of Chittagong.
Chemicals
The solvents namely methanol, ethanol, ethyl acetate, chloroform which were used to prepare extract are of Merck, India origin. The reagents used in the antioxidant activity like Ascorbic acid, DPPH (1,1-diphenyl-2-picryl hydrazyl), Potassium ferricyanide [K3Fe(CN)6] were purchased from SD Fine Chem. Ltd., India, Sigma Chemical Co., USA, Loba Chemie Pvt. Ltd., Mumbai, India respectively and Quercetin, Folin-ciocalteu reagent were from Merck, Germany. To conduct the thrombolytic study lyophilized Streptokinase (SK) vial (Durakinase, Dongkook Phama. Co. Ltd., SouthKorea) were purchased from local market. In the Streptokinase vial (1500000 IU), 5 ml sterile distilled water was added and mixed in an attempt to prepare a suspension of stock solution. Moreover, other chemicals used in this study were of analytical and highest commercial grade.
Extract preparation
Though, in this study extracts of Artocarpus chaplasha leaves were prepared with different solvents, but the procedure is same for all. At first after collection, the leaves were dried in oven at a temperature of 25-30ºC for 24 h to make it suitable for grinding purpose. After grinding (Moulinex Blender AK-241, Moulinex, France), the leaves powder (approximately 250 g) were kept in four different clean flat bottomed glass containers filled with 1 L of solvents namely methanol, alcohol, ethyl acetate and chloroform. The leaves were kept in the solvent for seven days at room temperature with occasional shaking and stirring. Strict measures were taken to avoid the admixture among solvents. The openings of the glass containers were sealed with aluminum foil. After seven days, the mixture was filtered through cotton, cheesecloth and Whatman filter paper No. 1 respectively. Concentration of the filtrate then done with the aid of rotary evaporator (RE 200, Sterling, UK) at temperature below 50ºC and completely removed the solvent. The yielded extracts (yield 4-5.5% W/W) were then placed in glass Petri dishes (90 × 15 mm, Pyrex, Germany) and then 100 mg of the extract was mixed with 10 ml distilled water to prepare a suspension with the aid of a vortex mixer. The preparation were left for overnight and followed by decanted to separate the soluble supernatant which was filtered through a 0.22 μm syringe filter. Then 100 μl of this aqueous preparation were applied over the clots in the microcentrifuge tubes to check the thrombolytic activity. The antioxidant properties were also evaluated with the similar concentration (10 mg/ml) [2].
Study of antioxidant activity
Scavenging activity of DPPH free radical
This study was conducted according to a previously described method [8] by utilizing the scavenging activity of stable DPPH (1,1-diphenyl-2-picrylhydrazyl) free radical. At first, 0.1 ml extract of different concentrations (10, 50, 100 and 500 μg/ml) was mixed with 3 ml 0.004% methanol solution of DPPH. Then, the absorbance of the resultant mixture was assessed at 517 nm against a blank after 30 min and finally percent scavenging activities (% SCV) were measured by comparing the results of test samples and control with the following formula.

Where,
SCV=Radical scavenging activity
A0=Absorbance of the control
A1=Absorbance of the test (extracts/standard).
The value of IC50 (50% inhibition) was measured from the% SCV versus concentration curve by using Microsoft Excel 2007. In this study, ascorbic acid was used as a control and the test were performed in duplicate. The above described process was applied in case of methanol, alcohol, ethyl acetate and chloroform extracts.
Reducing power efficacy study
Reducing power of methanolic, alcoholic, ethyl acetate and chloroform extracts were analyzed with the help of an earlier method [9]. First of all, different concentration (125, 250, 500 and 1000 μg/ml) of extracts were taken in separate test tube and 1 ml distilled water was added in the test tube. Then 2.5 ml 0.2 M phosphate buffer and 2.5 ml 1% Potassium ferricyanide [K3Fe(CN)6] was poured in the test tube. To complete the reaction within the chemicals inside the test tube the mixture was incubated at 50ºC for 20 min. Later, in the mixture 2.5 ml 10% trichloro acetic acid was added prior to centrifugation of the resultant mixture at 3000 rpm for 10 min. From the upper liquid layer 2.5 ml of solution was taken and mixed with 2.5 ml distilled water, 0.5 ml 0.1% of ferric chloride (FeCl3) and finally the absorbance of the mixture was taken at 700 nm against a blank by using a UV spectrophotometer. The blank solution contains all the reagents described in the above described process without the plant extracts. Increased absorbance means higher reducing power and ascorbic acid was used as a reference.
Total flavonoid content study
Flavonoid contents in different organic extracts of A. chaplasha leaves was estimated by aluminum chloride colorimetric method [10] where quercetin was used as standard and the flavonoid content of the extractives was expressed as mg of quercetin equivalent/gm of dried extract. At first, 1.0 ml of plant extract (200 μg/ml) were taken in a test tube and followed by mixed with 3 ml methanol, 200 μl 10% aluminum chloride solution, 200 μl 1 M potassium acetate solution and 5.6 ml distilled water. Then the mixture was incubated to complete the reaction at room temperature for 30 min. After that, the absorbance was measured at 420 nm against blank in a spectrophotometer. The total flavonoid content in plant extracts in Quercetin equivalents was calculated by the following formula:
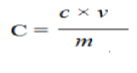
Where,
C=Total content of flavonoid compounds (mg/g) plant extract
c=The concentration of Quercetin established from the calibration curve (mg/ml)
v=The volume of extract (ml)
m=The weight of pure plant extracts (g)
Total phenolic content study
The total phenolic content of different extracts was estimated by using Folin–Ciocalteu reagent. In this case, gallic acid was used as standard. At first in a test tube, 0.5 ml of plant extract, 2.5 ml of Folin-Ciocalteu reagent, 2.5 ml of Sodium carbonate (7.5%) solution were taken and final volume were made up to 10 ml with distilled water. Then the mixture was shaken vigorously and kept for 2 hours. Then the absorbance of the resultant mixture and blank solution (contain standard) were measured at 760 nm by spectrophotometer. From the data of standard solution a standard curve was prepared and finally by comparing with the leave extracts data the total phenolic content was calculated [11]. The test was performed in triplicate.
Study of thrombolytic activity
Blood sample collection
This task was performed by the protocol set up by the Institutional Ethics Committee of University of Chittagong, Faculty of medicine Whole blood sample (6 ml) was collected from healthy human individuals (n=9) who were not under the prescription of contraceptive or anticoagulant. A consent form was collected from the faculty of medicine and supplied to the volunteers. Blood sample collection and followed by preservation was performed by Dr. Bijoy Talukder. Then the sample was transferred to 12 pre weighed micro centrifuge tubes (0.5 ml to each centrifuge tube) with the aim of preparing clots.
Clot lysis examination
This study was performed according to a previously described method [12]. Firstly, from the collected blood sample, 6 ml of blood were transferred to 12 pre-weighed centrifuge tubes (0.5 ml to each centrifuge tube) and centrifuged at 2,000 rpm for 5 min. The centrifuge-tubes were then incubated in simulated body temperature i.e., at 37ºC for 45 min in heat controlled incubator.
After 45 min, blood clot was formed at the bottom of each centrifuge tube. In the later stage, the serum was removed from the micro centrifuge tube without breaking the continuity of the clot. Then, the weights of the micro centrifuge tube which contain the clot (but no serum) were taken again to measure the weight of the clot. After that, in the micro centrifuge tubes 100 μl of plant extracts were added. The above process was conducted for each of the organic extracts of Artocarpus chaplasha leaves. As a positive and negative control, in the separate micro centrifuge tubes containing blood clot 100 μl of streptokinase and distilled water were added.
Then all the micro centrifuge tubes were incubated at 37ºC for 90 min to analyze the clot dissolving capacity. After 90 min, the released fluid from the micro centrifuge tubes along with the applied agents (extracts solution, streptokinase preparation, and distilled water) were removed carefully and completely and followed by weighted the micro centrifuge tubes. The weight differences before and after the clot lysis were estimated and expressed as percentage. The experiment was repeated for three times.
Statistical analysis
The data were plotted and converted to graph with the help of Microsoft Excel 2007.
Results
Scavenging activity of DPPH free radical
Percent scavenging activity and IC50 value of standard ascorbic acid and the extract are presented in Figure 1 and Table 1 respectively. From the figure, it is clear that all types of organic extract exhibit DPPH radical scavenging activity and this action is concentration dependent. DPPH radical scavenging activity accounted highest at each concentration points in case of ascorbic acid which is a well-known antioxidant [5] and then in the order of ethyl acetate extract, ethanolic extract, methanolic extract, chloroform extract respectively.
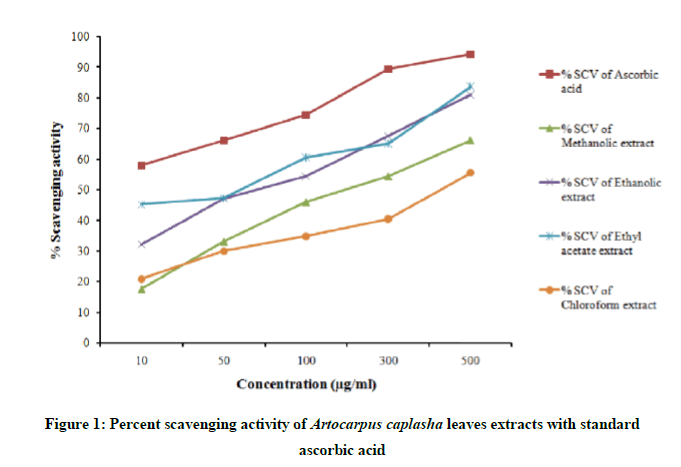

Ascorbic acid could scavenge 50% DPPH free radical at lowest inhibitory concentration (IC50) and IC50 value is reported as 6.12 μg/ml for ascorbic acid. The IC50 value is calculated as 60 μg/ml, 74 μg/ml, 202 μg/ml, 415 μg/ml for ethyl acetate extract, ethanolic extract, methanolic extract, chloroform extract respectively.
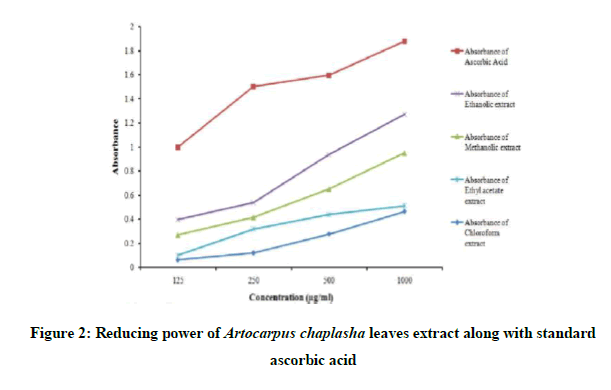
In the Figure 2, the reducing power of different organic extracts of Artocarpus caplasha leaves and standard ascorbic acid were displayed. The reducing power was increased with the increase of concentration. At a concentration of 1000 μg/mL level, the absorbance of standard ascorbic acid, methanolic extract, ethanolic extract, ethyl acetate extract, chloroform extract was 1.878, 0.950, 1.271, 0.511 and 0.464 respectively. Among the organic extracts, ethnolic extract exhibit the maximum reducing power and then in the order of methanolic extract, ethyl acetate and chloroform respectively. Though, the reducing power of standard ascorbic acid is higher than that of organic extracts but the difference in minimum in case of ethanolic extract, higher in case of others.
Total flavonoid content study
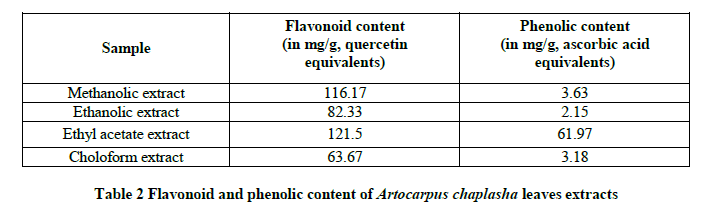
Total flavonoid content in different organic extracts of Artocarpus caplasha leaves is presented in the Table 2 and expressed as Quercetin equivalent of flavonoids per gram of extracts of the sample. The total flavonoid content obtained were 116.17 mg/quercetin equivalent, 82.33 mg/quercetin equivalent, 121.5 mg/quercetin equivalent, 63.67 mg/quercetin equivalent for methanolic extract, ethanolic extract, ethyl acetate extract and chloroform extract respectively. The value is higher in case of ethyl acetate extract and followed by methanolic extract.
Total phenolic content
In the Table 2, phenolic content value was presented as mg/g ascorbic acid equivalent for different organic extracts of Artocarpus caplasha leaves. The value is significantly higher only in case of ethyl acetate extract and accounted 61.97 mg/g ascorbic acid equivalents. While in case of methanolic extract, ethanolic extract and chloroform extract the phenolic content value is very poor and reported as 3.63 mg/g ascorbic acid equivalents, 2.15 mg/g ascorbic acid equivalents and 3.18 mg/g ascorbic acid equivalents respectively.
Clot lysis
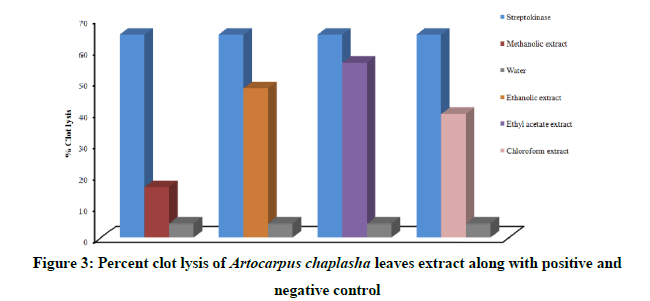
The resulted percent of clot lysis upon treating with different organic extracts and controls is presented in Figure 3. Addition of positive and negative control namely streptokinase and distilled water to the clots showed 65% and 4.5% of clot lysis activity respectively. On the other hand, percent clot lysis for Artocarpus caplasha leaves extracts accounted 16.145%, 47.783%, 55.98%, and 39.59% for methanolic, ethanolic, ethyl acetate and chloroform extracts respectively. The percentage of clot lysis for ethanolic, ethyl acetate and chloroform extracts were significant but insignificant for methanolic extract in comparison to positive and negative control.
Discussions
From a very ancient time, for the prevention and treatment of diseases human races are dependent on medicinal plant and today most of the Food and Drug Administration (FDA) approved pharmaceuticals are of plant origin [2]. In this study, extracts of Artocarpus chaplasha leaves were tested by various in vitro means not only for their antioxidant property but also for their thrombolytic activity.
Among the various techniques which are employed to measure the antioxidant property, the most quickest, simple and cost effective method is the application of DPPH (2,2-diphenyl- 1- picrylhydrazyl) free radical. DPPH is not only used to estimate the free radical scavenging activity or hydrogen donating potential of a compound but also quantify antioxidants in biological systems [13]. The IC50 value of Artocarpus chaplasha leaves extracts are listed in Table 1. Ethanolic solution of DPPH upon presence of hydrogen donation antioxidants change the color from purple to yellow and converted into a nonradical form diphenylpicrylhydrazine after reduction [5]. The extract of Artocarpus chaplasha was able to decolorize purple colored DPPH radical to yellow color. In a previous study [5], the antioxidant activity of cysteine, glutathione, ascorbic acid, tocopherol, gallic acid has been linked to phenolic contents. In this study, we have tested the phenolic content also and all extract exhibit positive findings (Table 2). Close similarity in the value of% scavenging activity and IC50 among ascorbic acid and ethyl acetate extract could be linked to high value of phenolic content of this extract (Table 2) in comparison to others.
Another parameter which helps in determining the antioxidant properties is the reducing power and was correlated with the phenolic content of plants due to redox properties [13]. In reducing power analysis, the amount of oxidized form of iron (Fe3+) could be lowered after conversion to its reduced form (Fe2+) by donating an electron if there is a presence of antioxidants in the extract of Artocarpus caplasha leaves. From the result (Figure 2) it is accepted that the antioxidants present in the extract aided in the reduction of ferricyanide complex (Fe3+) to ferrous form. Therefore, by measuring the formation of Perl’s Prussian blue at 700 nm the ferrous complex can be monitored and higher value indicates greater reducing capability [5].
Flavonoids which possess basic diphenylpyrans structure are the main classes of polyphenols found in plants and known as free radical scavengers. Flavonoids have multifarious antioxidant activities. Firstly, they exhibit free radical scavenging activity towards reactive oxygen species (e.g. .OH, O2.-, .O2) or towards lipid peroxidizing radicals (e.g. R., RO., and ROO.) and secondly, prevent the production of reactive species. Moreover, they also interact with other antioxidants (such as cooperative actions) and facilitate localization, and mobility of the antioxidant at the microenvironment. From the result (Table 2) it could be propose that the flavonoid present in Artocarpus caplasha leaves extracts is responsible for the antioxidant activity [5].
In earlier study [5], a close relationship between phenolic content of fruits and vegetables and antioxidant activities has been documented. The antioxidant activity of the phenolic compound is mainly due to its redox properties helps in the adsorption and followed by neutralization of free radicals, peroxides decaying also aided in the singlet and triplet oxygen suppression. Moreover, therapeutic activities of the phenolic compounds in diseases like cardiovascular disease, aging and cancer have also been reported.
In the analysis of thrombolytic activity, the clot lysis percentage value of positive and negative control clearly reveals that water could not influence the dissolution of clots. Moreover, a substantial differences in clot lysis value among the controls (positive and negative) and different organic extracts of Artocarpus caplasha leaves was exposed.
In a previous study, relationship between antibacterial activity and thrombolytic activity has been reported [2] and antibacterial properties of Artocarpus caplasha have studied earlier [1]. Many plants could possess bacterial contaminants having plasminogen receptors that bind plasminogen and such cell surface bound plasminogen could result in fibrinolysis upon activation to plasmin [14]. Some molecules namely staphylokinase, streptokinase known as bacterial plasminogen activator, activates plasminogen to dissolve clots [15,16]. Thus, it could be assumed that antibacterial effect of Artocarpus caplasha is related to the evaluated thrombolytic effect. In a previous study [13], antibacterial properties of Artocarpus heterophyllus linked with the presence of phenolic content of the plants. As in this study we have evaluated the presence of phenolic content in the Artocarpus caplasha leaves extracts, antimicrobial properties of this plant could also be assumed. But it’s worth of doing further research to discover the antibacterial effect of Artocarpus caplasha.
Conclusion
From the results it is clear that, the extracts possess both antioxidant and thrombolytic activity and the intensity of these activity varies with the change of extract. Natural resources of this extract are accounted for safety to the people. This is a preliminary study and it is necessary to assure the nontoxic nature and dose-activity relationships of Artocarpus caplasha. As the active constituents of the extracts are crucial in governing the antioxidant and thrombolytic activity efforts should be continued to understand and collect the active principles of Artocarpus caplasha leaves and in vivo studies are needed to interpret the mechanism of action.
Competing Interests
The authors declare that they have no competing interests.
Authors Contributions
BT and SMMH contributed in the contributed to conception and design of study; assist in drafting and revising the manuscript. MFH participated in the design of study; acquisition, analysis and interpretation of data. RUM performed in data collection and manuscript preparation. NA and MSH participated in experiments, literature search and manuscript preparation.
Acknowledgments
The authors would like to thank Dr. Bijoy Talukder for assisting in the blood collection.
References
[1] F. Chowdhury, S. Pal, T. Sharmin, R.B. Rashid, M.A.A. Sikder, S. Kabir, Bangladesh Pharmaceut. J., 2013, 16(1), 63-68.
[2] M.A. Rahman, R. Sultana, T.B. Emran, M.S. Islam, M.A. Rahman, Chakma JS, BMC. Compl. Alter. Med., 2013, 13:25.
[3] Rishikesh, D.R. Ghosh, M.M. Rahman, Int. J. Pharm., 2013, 3(2), 308-311.
[4] L. Nahar, F.A. Ripa, Rokonuzzaman, M.A.A. Al-Bari, J. Appl. Sci. Res., 2009, 5(12), 2285- 2288.
[5] T. Ahmed, M.N. Uddin, M.K. Hossain, M.I. Islam, N. Uddin, M.E.H. Mazumder, J. Appl. Pharmaceut. Sci., 2012, 2(12), 89-95.
[6] B. Talukder, J. Ara, A. Walliullah, D.M.S. Ali, The Experiment., 2013, 13(1), 829-883.
[7] M.A.M. Bhuiya, B. Talukder, M. Ajrin, S. Akter, R. Sen, The Experiment., 2013, 14(2), 888-893.
[8] A. Braca, N.D. Tommasi, L.D. Bari, C. Pizza, M. Politi, I. Morelli, J. Nat. Prod., 2001, 64(7), 892-895.
[9] M. Oyaizu, The Japanese J. Nutrit. Dietetics., 1986, 44(6), 307-315.
[10] C. Chang, M. Yang, H. Wen, J. Chern, J. Food Drug Anal., 2002, 10, 178-182.
[11] L. Yu, S. Haley, J. Perret, M. Harris, J. Wilson, M. Qian, J. Agr. Food. Chem., 2002, 50(6), 1619-1624.
[12] S. Prasad, R.S. Kashyap, J.Y. Deopujari, H.J. Purohit, G.M. Taori, H.F. Daginawala, Thrombosis. J., 2006, 4, 14.
[13] M.R. Loizzo, R. Tundis, U.G. Chandrika, A.M. Abeysekera, F. Menichini, N.G. Frega, 2010, 75(5), M291- M295.
[14] M. Pantzar, A. Ljungh, T. Wadström, Infection and Immunity., 1998, 66(10), 4976-4980.
[15] M.A.A. Parry, X.C. Zhang, Trends. Biochem. Sci., 2000, 25(2), 53-59.
[16] D. Collen, Annals of Internal Medicine., 1990, 112(7), 529-538.



