Research Article - Der Pharma Chemica ( 2020) Volume 12, Issue 7
In Vitro Interaction of 5-aminoorotic Acid and Its Lanthanum (III) Complex With Superoxide and Hypochlorite Radicals
Lozan T. Todorov1*, Dr. Maria L. Traykova2 and Prof. Irena P. Kostova12Department of Physics and Biophysics, Faculty of Medicine, Medical University - Sofia, Sofia, Bulgaria
Lozan T. Todorov, Department of Chemistry, Faculty of Pharmacy, Medical University - Sofia, Sofia, Bulgaria, Email: ltodorov@pharmfac.mu-sofia.bg
Received: 19-Aug-2020 Accepted Date: Nov 21, 2020 ; Published: 28-Nov-2020
Abstract
Superoxide and hypochlorite radicals are involved in both normal and pathological processes in the human body. Excessive production of any of them may result in malignization of healthy cells. The ideal chemotherapeutic must exhibit prooxidant action in the tumor and antioxidant behavior in healthy tissues. Lanthanum (III) salts have been investigated for their antibacterial and antitumor activity. 5-aminoorotic acid (HAOA) has antioxidant properties. In this work the interaction of HAOA and its Lanthanum (III) complex (LaAOA) with with superoxide and hypochlorite radicals was evaluated in vitro by way of two superoxide-generating and one hypochlorite-generating systems at normal physiological pH. Luminol-dependent chemiluminescence (LDCL) and UV spectroscopy were used to estimate the radicals scavenging properties of the investigated compounds. The ability of both compounds to exchange electrons and to donate hydrogen was estimated by the utilization of two models of stable free radicals. Both HAOA and LaAOA were capable of participating in electron transfer and hydrogen donation, therefore they behave like promising antioxidants. The latter was proven by monitoring their interactions with superoxide (potassium superoxide and xanthine/xanthine oxidase generated) and hypochlorite (NaOCl generated) radicals in vitro. The complex LaAOA behaved as a better antioxidant than 5-aminoorotic acid itself. This suggested that these compounds would protect healthy tissues from oxidative damage. The antioxidant behavior of LaAOA and HAOA was in agreement with the 3D structure and UV-spectra of the compounds. It was suggested that LaAOA might be a promising anticancer agent with antioxidant properties.
Keywords
Ethnomedicine, Acaciaspecies, Flavonols, Spectroscopic analysis, In Vitro antimicrobial activity.
Introduction
Although the malignization of tissues requires a high level of oxidative stress, the survival, proliferation and spread of malignant cells is propagated by low contents of free radicals in their environment [1,2]. Malignant tumors produce high level of extracellular antioxidant enzymes to maintain a tumor-friendly environment [3-9] which often leads to chemotherapeutic resistance [10].
The formation of the superoxide radical (O2•-), inevitable in the living body, is involved in many normal and pathological reactions [11], including cellular malignization, tumor proliferation and death of malignant cells [1,12-16]. On the other hand, it is a component of the host’s immune defense [17]. Superoxide can be generated by two major sources in a biological system: the mitochondrial respiratory chain and phagocytic nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase). The human body generates approximately 5 grams of reactive oxygen species (ROS) daily, mainly superoxide and hydrogen peroxide, via leakages of the electron transport chain [18, 19]. Along with a physiological role, beneficial for the host, superoxide is involved in several harmful reactions with normal bioactive molecules resulting in protein denaturation, lipid peroxidation and DNA damage [20,21]. It has been known for a long time that the generation of O2•--derived reactive species in a living body is strictly controlled and dynamically balanced [22-27]. Control over the accumulation of extracellular superoxide is among the requirements for a successful chemotherapeutic [28]. A perfect anticancer drug must maintain high level of O2•- in the tumor, but low concentrations in healthy tissues. In order to achieve that, it has to be capable to eliminate free radicals in healthy tissues while being toxic in tumors. To do so the anticancer substance must be capable in participating in reactions of electron transfer or/and hydrogen donation. As superoxide leads to the formation of large variety of toxic and aggressive reactive species this way resulting in oxidative tissue damage, the interaction of a substance with O2•- is important for the biological outcome to a chemotherapeutic.
The production of hypochlorite radical (ClO-) is an important component of the self-defense of a living body [29-35]. However, the excessive generation of hypochlorous acid (HOCl) and ClO- contributes to tissue injury - non-associated [36-40] and associated [31,41-43] with inflammation, renal disease [44-47] and lung cancer [31,43]. Hypochlorite is generated from the reaction of hydrogen peroxide (H2O2) and Cl- catalyzed by the myeloperoxidase (MPO) enzyme in neutrophils [31,48-51]. HOCl damages DNA, proteins, glutathione and protein thiols by reacting with amino-acid chains, backbone cleavage and attacking C=C bonds, this leading to formation of free radicals various in size, geometry and structure (including nitrogen-centered), their yield increasing together with high HOCl/target ratios and enzymatic digestion. High concentrations of HOCl production are associated with cellular death (apoptosis or necrosis) and DNA damage resulting in an increased cellular mutation rate. Modest production of hypochlorite is associated with cellular growth arrest. The prevalence of the positive or negative effect of hypochlorous acid on the host depends on the health status and on the activity of neutrophils, leading to rising or decreasing levels of hypochlorite [31], as well as on the content of antioxidants capable of eliminating the hypochlorite anion (such as glutathione, lycopene etc.) [52,53]. The expression of hypochlorite by neutrophils is part of the self-defense of a living body against xenobiotics. Along with many other factors, the efficacy of an anticancer therapy might be affected by the interaction between biologically active substance and hypochlorite radical released by the neutrophils.
The unique properties of Lanthanides (Ln), their salts and organic complexes make them promising components of anticancer drugs [54,55]. Depending on environmental conditions Ln complexes may behave either as antioxidants or as pro-oxidants [55]. The oxidative behavior of Ln (III) complexes may be controlled by the selection of appropriate organic ligands. It has been observed that the La (III) complex with 5-aminoorotic acid inhibited the Fenton reaction [56].
The aim of this investigation was to monitor in vitro the interaction of the La (III) complex (LaAOA) of 5-aminoorotic acid (HAOA) with superoxide and hypochlorite free radicals at normal homeostatic pH of 7.45. The information about interactions with O2•- and hypochlorite radicals would indicate if the compounds investigated would act as antioxidants in close vicinity to healthy cells, this way protecting healthy tissues from oxidative damage. In the present work, the participation of HAOA and LaAOA in electron transfer and hydrogen donation was estimated by monitoring their interactions with model stable free radicals. The elimination of superoxide and hypochlorite radicals was estimated in in vitro model systems. The ability of 5-aminoorotic acid and its complex with La (III) to eliminate the superoxide radical was monitored in two superoxide generating in vitro model systems. In one model system the O2•- was generated by decomposition of KO2, while the other it was a by-product of the enzymatic transformation of xanthine (X) by xanthine oxidase (XO) (the X/XO model system). The elimination of the hypochlorite radical (ClO-) by the two compounds investigated was estimated in a model system generating hypochlorite from NaOCl. The in vitro interaction of HAOA and LaAOA with superoxide and NaOCl was monitored using Luminol-dependent chemiluminescence (LDCL) and UV-spectroscopy. The ability of HAOA and LaAOA to participate in electron transfer and hydrogen donation was estimated by measuring their reaction with two stable free radicals – 2,2'- azino-bis (3-ethylbenzothiazoline-6-sulfonic) acid (ABTS) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) through the use of UV spectroscopy.
Materials and Methods
All materials and compounds were of finest grade (p.a.) purchased from SIGMA- ALDRICH. Bi-distilled water was used for the preparation of the solutions. The impact of 5-aminoorotic acid and its complex with La (III) on the specific activity of xanthine oxidase in the model system containing xanthine and xanthine oxidase (X/XO) was investigated in aqueous solutions of HAOA and LaAOA within a concentration range of between 10-4 M and 10-6 M. The required concentrations of the compounds were achieved by diluting standard aqueous solutions with concentrations 10-3 M (HAOA and LaAOA). 25.4 mU/mL xanthine oxidase was dissolved in 50 mM K, Na- phosphate buffer of pH 7.45 (PBS) and used in the LDCL and UV- spectroscopic measurements. 5-Amino-2,3-dihydro-1,4-phthalazinedione (luminol) was dissolved in a small amount of 0.01 M NaOH, further diluted to 1*10-3 M in 50 mM K, Na-phosphate buffer (PBS) of pH 7.45, and pH was adjusted again to 7.45. 1 mM KO2 solution in dehydrated dimethyl sulfoxide (DMSO) was prepared directly prior use. The 3 mM solution of xanthine was prepared by dissolving the compound in 0.1 N NaOH, and diluting with bi-distilled water. 4*10-4 M aqueous solution of HOCl was prepared prior measurements.
The antioxidant capacity by way of hydrogen donation is often estimated by measuring the radicals scavenging activity (RSA) toward the stable 2,2-diphenyl-1-picrylhydrazy radical (DPPH•) [57-59], while participation in electron-exchange reactions may be tested by way of the ABTS•+ (2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic) free radical) assay [60-63].
The complex was synthesized by the reaction of La (III) nitrate and the ligand in amounts equal to metal:ligand molar ratio of 1:3 using an earlier described procedure [64-66]. The reaction between La (III) and 5-aminoorotic acid resulted in the formation of a complex which turned out to be quite stable both in solid state and in solution. The preparation of La (III) complex could be summarized in the next equations:
HAOA ↔ H+ + AOA-
[La (H2O) n]3+ + 3AOA-→La (AOA-)3.H2O,
HAOA = C5N3O4H5 and AOA- = C5N3O4H4-
The new La (III) complex was characterized by elemental analysis, FTIR, Raman and UV-VIS spectroscopies. The used spectral analyses confirmed the nature of the complex.
Luminol-dependent chemiluminescence was utilized to estimate the radicals scavenging activity in presence of the in vitro model systems containing KO2 and X/XO and NaOCl. Apparatus LUMAT LB9507 was used for the LDCL investigations involving KO2 and X/XO derived superoxide. Kinetics were measured with delay time 2 s, measurement time 3 s for a total measurement time of 600 s. The integral intensities for the first 10 seconds (s) were used in data management. The interaction between the tested substances and NaOCl was measured using LKB 1251 luminometer (Bioorbit, Turku, Finland) set at 37°C and connected with IT-type computer via serial interface. The flash assay option of the apparatus of the Multiuse 1.08 program was applied every 50 milliseconds (ms).
The specific activity of the XO in the model system X/XO was estimated by UV spectrophotometric measurement of the relative change of the characteristic signal of the uric acid (UA) at 293 nm. This experiment was performed using UV 1650PC Shimadzu spectrophotometer. The delay time was 10 s, and the activity of the UA formation was computed by the program subroutine for the period 10 - 90 s. Data for 600 s was collected as well. One unit of XO activity was defined as the amount of XO which transformed 1 μmol of xanthine for 1 minute in 1 mL reaction mixture at 298 K.
The UV-VIS spectra of HAOA and LaAOA were recorded using Shimadzu 1601 spectrophotometer equipped with PC, using very low speed. The background was eliminated using the background elimination ability of the apparatus. Instrumental errors in the intensity and position of the spectroscopic bands were found by scanning the spectrum of the solvent vs itself. It was found that the error in position was ±2 nm, while the error in intensity was ±0.0004 a.u.
The UV spectra of HAOA and LaAOA alone at each desired concentration were recorded 5 times, and average spectra were used in discussion. Initially spectra within 800 and 200 nm were recorded, but as no characteristic bands were found above 400 nm, the interval 400 to 200 nm was used in further discussions. The spectra were resolved in accordance with literature [64,65-75].
In order to compare the hydrogen-donating and electron-transfer properties of both investigated substances to an established standard, their C50 trolox equivalents in presence of DPPH• and ABTS•+ were calculated. Standard solutions of Trolox (6-hydroxy-2,5,7,8- tetramethylchroman-2-carboxylic acid) were prepared in ethanol (DPPH•) and water (ABTS•+) at the following micromolar concentrations: 1, 5, 10, 20, 30, 40, 60, 100, 200, 300, 400, 600, 800,1000, 1200, 1400, 1600, 1800, 2000.
RSA assay with ABTS•+
Two solutions named R1 and R2 were prepared as described in [61]. The solution R1 was Na-acetate buffer with pH 5.8. The solution R2 contained ABTS•+ dissolved in acetate buffer with pH 3.8. The interaction of the HAOA and LaAOA with ABTS•+ was evaluated at 660 nm as that wavelength did not coincide with the characteristic bands of the tested compounds. Sample compositions are described in Table 1.
| Sample content | ‘Blank’ sample | ‘Control’ sample | ‘Sample’ sample |
|---|---|---|---|
| Investigated substance |
0.04 mL | none | 0.04 mL |
| R1 | 0.96 mL | 0.94 mL | 0.94 mL |
| R2 | none | 0.02 mL | 0.02 mL |
| Bi-distilled water | none | 0.04 mL | none |
Table 1: Sample composition for the ABTS·+ assay
The radicals scavenging activity RSA was calculated using the formula:
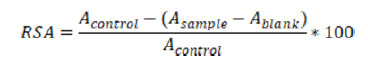
Acontrol, Asample and Ablank represented the corresponding results for the control, sample or blank measurement. The “control” measurement helped evaluate the absorbance of the characteristic signal of the stable free radical itself in the medium. The “blank” measurement accounted for the effect of the medium itself on the absorption at the radical’s characteristic wavelength, the radical being absent. Therefore (Asample - Ablank) demonstrated the diminishing of the characteristic absorption due to radical's scavenging only. The higher RSA, the higher the antioxidant activity was.
RSA assay with DPPH•
A standard solution of DPPH• was prepared as has been previously described [66]. The absorption of the characteristic band for DPPH• at 517 nm was measured. The relative decrease in the absorption was monitored for 5 min after a 10 s lag time using the kinetic software of the apparatus. The absorption at 517 nm was recorded every 60 s. Sample compositions are described in Table 2.
| Sample content | ‘Blank’ sample | ‘Control’ sample | ‘Sample’ sample |
|---|---|---|---|
| Investigated substance | 0.02 mL | none | 0.02 mL |
| Ethanol | 1.98 mL | none | none |
| DPPH· solution | none | 1.98 mL | 1.98 mL |
| Bi-distilled water | none | 0.02 mL | none |
Table 2: Sample composition for the DPPH• assay.
Radicals scavenging activity, RSA (%) was calculated in the following way:

Data from the five parallel measurements were used in order to perform a statistical evaluation of relative differences between the RSAs of HAOA and LaAOA at different concentrations.
Assay for CL in the presence of KO2
One mL of the control sample contained 0.05 mL KO2 solution, 0.05 mL luminol and PBS. One mL of the sample volume contained 0.05 mL KO2, 0.05 mL luminol, the compound investigated in the desirable concentration and PBS. The results were presented as Chemiluminometric Scavenging index (CL-SI) calculated as follows:
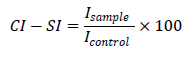
Icontrol and Isample being the integral intensities measured for the KO2 alone and in the presence of the compound in a desirable concentration. The background measurement showed integral intensity of 10 and was subtracted from both control and sample measurements. For each compound at each desirable concentration 5 parallel measurements were performed. Average values and standard deviations were used for further comparisons.
Assay for detection of interactions with NaOCl
One mL of the sample contained PBS (pH 7.45), 10-4 M luminol, and the tested compound at a desirable concentration. The tested compound was omitted in the control sample. The background was measured in the cuvette with NaOCl omitted, the background data being subtracted from both control and sample data. The CL-SI was calculated using the same formula as in the measurements for the KO2 effects. For each desired concentration of a compound investigated 5 parallel measurements were performed and 5 CL-SI data were calculated. The average value and standard deviation were calculated and used in further comparisons.
LDCL in the presence of X/XO model system
One mL of the cuvette for the control measurement contained 0.02 mL XO solution, 0.1 mL xanthine, 0.1 mL luminol and PBS. One mL solution of the sample measurement contained 0.02 mL XO, 0.1 mL X, 0.1 mL luminol the compound investigated in the desirable concentration and PBS. The results were presented as Chemiluminometric Scavenging index (CL-SI) calculated as follows:
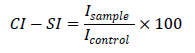
Icontrol and Isample being the integral intensities measured for the X/XO alone and in the presence of the compound at a desirable concentration. For each desirable concentration of the compounds investigated, 5 parallel measurements were performed. The average value and standard deviation were calculated and used in further comparisons.
Assay for UV determination of XO activity in the presence of X/XO model system
One mL of the reaction mixture for the control measurement contained 0.02 mL XO, 0.1 mL xanthine and PBS. One mL of the cuvette for the sample measurement contained 0.02 mL XO, 0.1 mL X, the compound investigated in a desirable concentration and PBS. As no additional components in this model system were present only UA and O2•- were produced (Scheme 1):
As the final product of xanthine transformation was uric acid (UA), the absorption at 293 nm (characteristic wavelength for UA) was measured for 10 minutes, using the molar extinction coefficient of 1.22*104 M-1.cm-1 [76]. The activity of XO was defined as the amount of enzyme needed to convert 1 μmol of xanthine for 1 minute in 1 mL reaction mixture at 298 K. Data for the activity of XO were presented as percentage of XO activity seen in the control measurement.
Trolox equivalent calculations
The interaction between the various trolox standard solutions and the stable radicals were investigated using the aforementioned DPPH• and ABTS•+ assay methods (sections 2.1 and 2.2 respectively). The compositions of the trolox samples is described in the Tables 3 & 4 below.
| Sample content | ‘Blank’ sample | ‘Control’ sample | ‘Sample’ sample |
|---|---|---|---|
| Trolox | 0.02 mL | none | 0.02 mL |
| Ethanol | 1.98 mL | none | none |
| DPPH· solution | none | 1.98 mL | 1.98 mL |
| Bi-distilled water | none | 0.02 mL | none |
Table 3: Sample composition for the trolox DPPH• assay.
| Sample content | ‘Blank’ sample | ‘Control’ sample | ‘Sample’ sample |
|---|---|---|---|
| Trolox | 0.04 mL | none | 0.04 mL |
| R1 | 0.96 mL | 0.94 mL | 0.94 mL |
| R2 | none | 0.02 mL | 0.02 mL |
| Bi-distilled water | none | 0.04 mL | none |
Table 4: Sample composition for the trolox ABTS•+ assay.
For each trolox concentration, five parallel measurements were performed for the purpose of statistical evaluation. The RSAs were calculated as described previously (section 2.1). The two RSAs closest to the calculated C50 RSA values of HAOA and LaAOA were estimated.
Data Management
For each concentration of HAOA and LaAOA 5 parallel measurements of LDCL were performed, each CL-SI being calculated. Then the average and standard deviation for this concentration were calculated and used in the further discussion. A comparison between data for HAOA and LaAOA of same concentration was performed using a non-parametric t-test with Welch correction. The effect of the concentration of a compound on CL-SI was statistically evaluated using One-Way ANOVA with Bonferroni post-test. The Bartlett test showed that all standard deviations were from the same population. The C50 for both HAOA and LaAOA were calculated too. Similar approach was applied to the measurements of the activity of the system
X/XO in the presence of the compounds investigated. In this case, for each concentration of the compounds, 5 parallel data points of XO activity were established. The statistical evaluation of relative differences was done in a similar way as this in the case of LDCL and UV measurements. Chem Office program package v. 3.01 was used to build molecule models of the compounds investigated, as well as to illustrate their interactions with solvent molecules. The solvent effect on the molecular geometry was illustrated by presenting the interaction of one solvent molecule with each of one HAOA or AOA- ligand. The milieu was presented by one dissociated KNaHPO4 molecule:
KNaHPO4 → K+ + Na+ + HPO 2-
Results
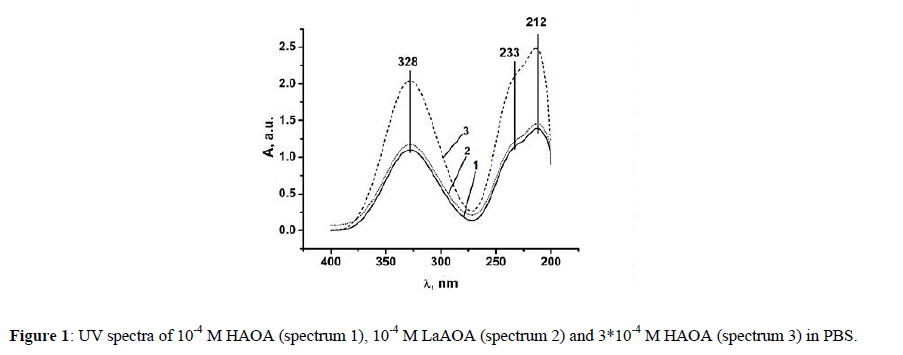
The UV spectra of HAOA and LaAOA aqueous solutions diluted to 10-4 M in PBS are presented in Figure 1.
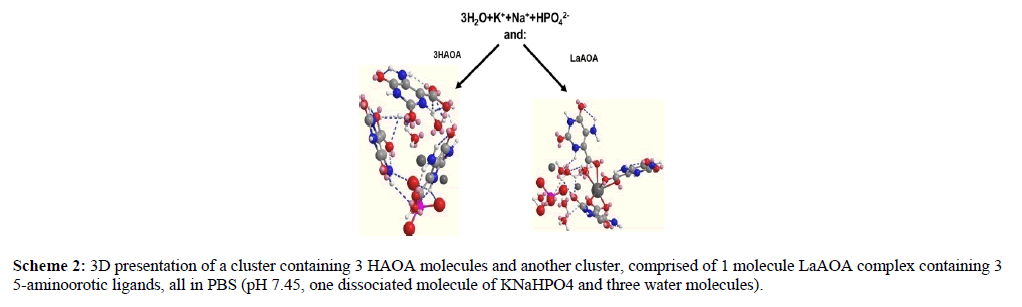
The band at 328 nm was associated with the aromatic ring in the HAOA and LaAOA. The band at 233 nm was related with π→π* transition in the conjugated double-bond systems and this at about 212 nm was associated with NH2 group. As LaAOA contained three AOA- ligands, the spectrum of 10-4 M LaAOA (spectrum 1 in Figure 1) was expected to be very similar by bands positions and intensities to this of the 3*10-4 M HAOA (Spectrum 3 in Figure 1). Instead the spectrum of the 10-4 M LaAOA was very similar by intensity to this of 10-4 M HAOA (spectrum 2 in Figure 1). This indicated that the geometry of LaAOA resulted in a dipole moment in the complex. This was proven by the 3D models of this ligand surrounded by the ions of PBS being compared to a 3D model of three HAOA in the same environment (Scheme 2).
The environment was modeled by three water molecules and one dissociated KNaHPO4 molecule. The complicated geometry of the LaAOA complex in PBS suggested varying activity of the three ligands with regard to the interaction between the complex and different free radicals. Despite containing three 5-aminoorotic ligands, the reactivity of LaAOA might not be three times that of an equimolar solution of HAOA.
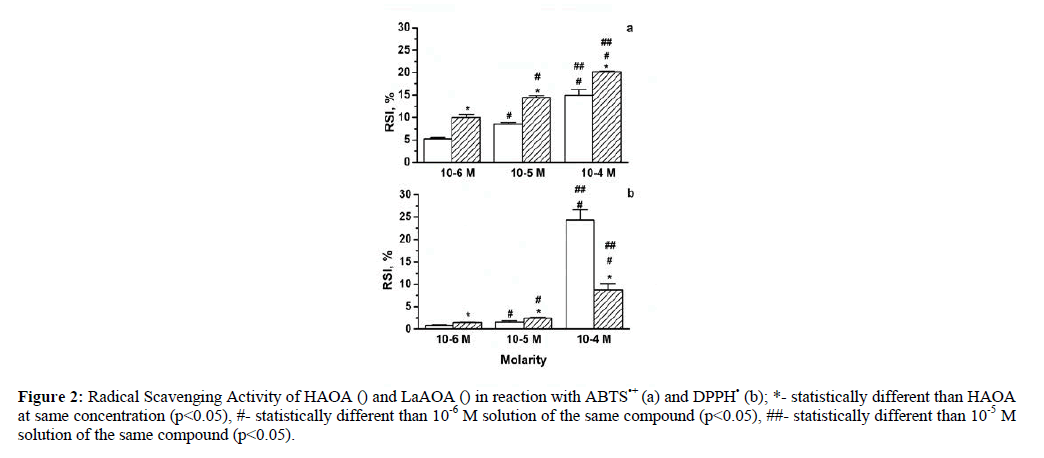
The Radicals scavenging activities of HAOA and LaAOA toward the two large stable free radicals were presented in Figure 2.
Figure 2: Radical Scavenging Activity of HAOA () and LaAOA () in reaction with ABTS•+ (a) and DPPH• (b); *- statistically different than HAOA at same concentration (p<0.05), #- statistically different than 10-6 M solution of the same compound (p<0.05), ##- statistically different than 10-5M solution of the same compound (p<0.05).
Data, represented by figure 2 suggested that both compounds investigated can participate in electron transfer (Figure 2-a) and hydrogen donation (Figure 2-b). Figure 2-a indicated that LaAOA was the more active participant in electron transfer reactions compared to HAOA at equimolar concentrations. Figure 2-b showed that at concentrations below 10-4 M LaAOA was more active in hydrogen donation than HAOA. Comparing the data, presented in Figure 2-a and Figure 2-b it was observed that below a concentration of 10-4 M both HAOA and LaAOA tended to participate better in electron transfer rather than in hydrogen donation reactions. The RSA of HAOA at a concentration of 10-4 M (Figure 2-b) toward DPPH• was higher than that of LaAOA. That might be related to a better accessibility of the active center of the DPPHxx to HAOA compared to LaAOA, probably due to the size and geometry of the complex.
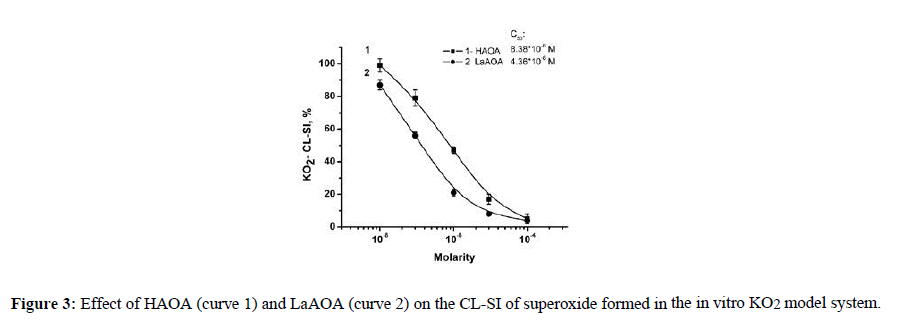
The in vitro interaction between superoxide and the compounds investigated in presence of KO2 model system was represented by Figure 3. The C50 were found to be 8.38*10-6 M and 4.36*10-6 M for HAOA and LaAOA, respectively.
Figure 3 demonstrated that at equimolar concentrations LaAOA was a better (but not three times better) scavenger of the superoxide radical than HAOA. The C50 were found to be 8.38*10-6 M and 4.36*10-6 M for LaAOA and HAOA, respectively.
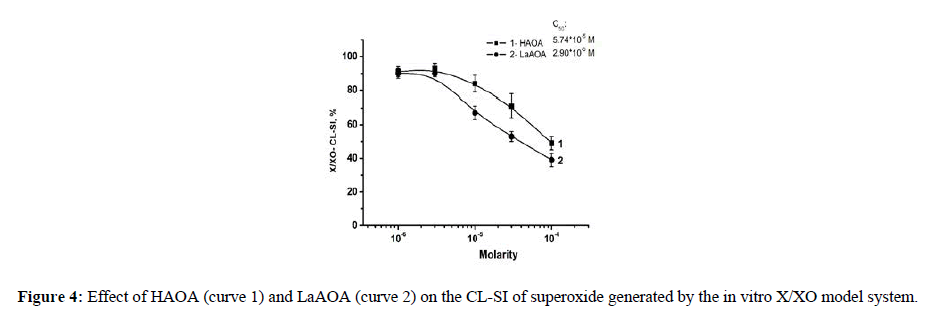
The in vitro interaction of the compounds investigated with superoxide generated by the X/XO model system was displayed in Figure 4. Within concentrations of 10-6 and 10-4 M both compounds were scavengers of the X/XO generated superoxide. At equimolar concentrations HAOA (curve 1) was a better antioxidant than LaAOA (curve 2). The C50 values calculated from data in Figure 4 were 5.74*10-5 M and 2.90*10-5 M for HAOA and LaAOA, respectively.
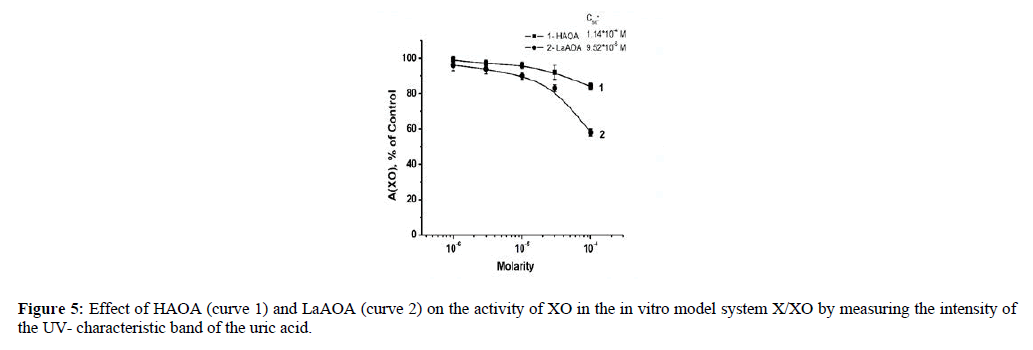
The concentration-dependent radical scavenging properties presented in Figure 4 (based on LDCL measurements) align with the data in Figure 5 (based on the UV measurements). That indicated interaction of the compounds investigated with the superoxide formed as a by-product of the transformation of xanthine to uric acid and/or possible interaction with the X/XO system (either with xanthine, or with xanthine oxidase, or with both) as the production of uric acid changes with the content of a compound in a concentration-dependent manner.
The results presented in Figures 4 and 5 showed that at equimolar concentrations LaAOA was better antioxidant toward X/XO- generated superoxide. The relative differences between CL- SI of the compound, seen in Figures 2 and 4 might be a result the disparate natures of the model systems, generating superoxide.
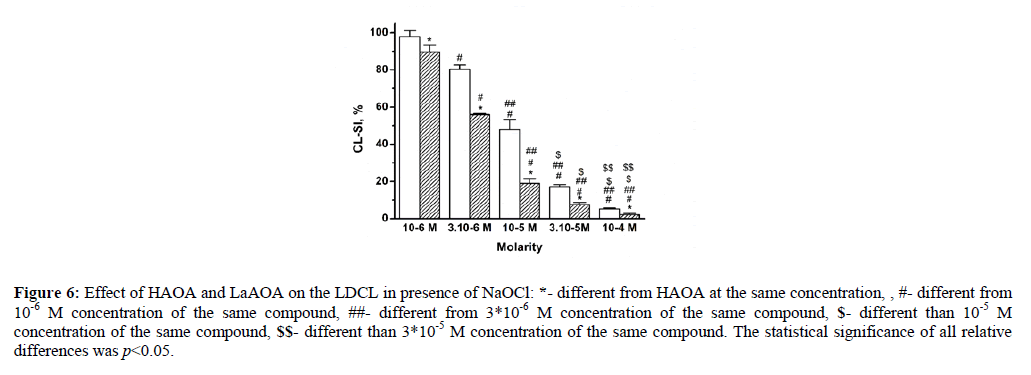
The effect of HAOA and LaAOA on the luminol-dependent chemiluminescence in presence of NaOCl is illustrated in Figure 6.
Figure 6: Effect of HAOA and LaAOA on the LDCL in presence of NaOCl: *- different from HAOA at the same concentration, , #- different from 10-6 M concentration of the same compound, ##- different from 3*10-6 M concentration of the same compound, $- different than 10-5 M concentration of the same compound, $$- different than 3*10-5 M concentration of the same compound. The statistical significance of all relative differences was p<0.05.
In presence of both HAOA and LaAOA, CL-SI decreased in a concentration-dependent manner. This indicated that both compounds exhibited antioxidant effect in presence hypochlorite. At equimolar concentrations, the CL-SI of LaAOA was lower than that of HAOA, suggesting a better antioxidant activity of LaAOA than this of HAOA. The calculated C50 values were 1.012*10-5 and 4.58*10-6 for HAOA and LaAOA, respectively.
The C50trolox equivalents of both investigated substances were displayed on Table 5.
| Sample | DPPH· | ABTS+· |
|---|---|---|
| HAOA | 474 ± 44 | 365 ± 37 |
| LaAOA | 207 ± 12 | 386 ± 23 |
Table 5: C50trolox equivalents for HAOA and GaAOA.
Data in Table 5 suggested that the elimination of free radicals in the presence of HAOA would be possible via both hydrogen donation and electron transfer, the former more evident than the latter. In the presence of LaAOA the electron transfer pathway was more evident than that via hydrogen donation.
Discussion
According to relevant literature, one result of hyper-production of superoxide and hypochlorite radicals is profound damage of biomolecules, causing oxidative stress, increased cellular mutation rate and malignization [77, 78, 79, 80, 81]. Therefore, eliminating the excessive amounts of these radicals in the extracellular fluid would be beneficial to healthy tissues. 5- aminoorotic acid, both as an individual compound and as a ligand in La (III) complex, acted as an antioxidant toward superoxide (Figures 2 – 5) and hypochlorite radicals (Figure 6). Figures 2 to 5 illustrated that scavenging of superoxide radical occurs in any selected model O2•- generating system. Comparisons between equimolar solutions of HAOA and LaAOA indicated the complex to be a better antioxidant than the 5-aminoorotic molecule alone. The greater antioxidant activity of LaAOA was in agreement with its better ability to participate in electron transfer and hydrogen donation (Figure 2) compared to 5-aminoorotic acid itself. The relative difference in antioxidant activities of both compounds suggested that not all ligands in the complex play the same role in the scavenging of radicals. This was in agreement with the geometry of LaAOA in PBS as illustrated by the 3D model (Scheme 2), as well as with the spectra presented in Figure 1. The La (III) ion as a component in salts and complexes is a well-known toxic agent with anticancer activity [54,55]. Taking into account the radicals scavenging properties of LaAOA observed in our investigation it could be proposed that this complex might be a promising antioxidant carrier of the toxic La (III) to the targeted tumor. At normal physiological environment the complex would eliminate the excessive superoxide and hypochlorite radicals, thus preserving healthy cells from oxidative stress, mutation and malignization. In the vicinity of the tumor, La (III) would act as a killer of the malignant cells.
The C50 trolox-equivalent calculations performed (Table 5) showed several tendencies:
-HAOA was a scavenger of both DPPH• and ABTS+• stable free radicals. Hydrogen donation seemed to be slightly more pronounced as an antioxidant mechanism of action compared to electron transfer. Both mechanisms could be possible, dependent on conditions in the medium.
-LaAOA demonstrated a slightly more pronounced tendency for participation in electron transfer reactions, compared to hydrogen donation.
The aforementioned observations were made with the consideration that both radicals are very large in size and complex in structure. For that reason, we propose that conclusions could be drawn only regarding large, complex free radicals in an actual biological system.
Our investigation proved that both HAOA and LaAOA were scavengers of the superoxide radical at normal physiological pH (7.45). In healthy tissues these compounds could probably manifest as antioxidants, eliminating superoxide, this way preventing oxidative cellular damage. The complexity of the content of the environment imposes a risk of various effects on the radicals scavenging activity of HAOA and LaAOA. Both 5-aminoorotic acid and its complex with La(III) may exhibit antioxidant action via hydrogen donation and electron transfer, the former more probable in presence of HAOA, the latter more evident in presence of LaAOA.
Conclusions
5-aminoorotic acid and its complex with La(III) were significant scavengers of superoxide in two in vitro superoxide-generating model systems: KO2 and X/XO at normal homeostatic pH. The complex of La(III) with 5-aminoorotic acid was a better scavenger of superoxide and hypochlorite radicals than 5-aminoorotic acid itself. The antioxidant effect of 5-aminoorotic acid and its complex with La(III) in different superoxide-generating model systems might be impacted by interactions between the compounds investigated and the components of the model system.
Acknowledgements
The authors gratefully acknowledge the financial support from the Medical University- Sofia Grant Commission (Grant No. 130/2019).
References
[1] RM Day, YJ Suzuki. Dose-Response, 2005, 3(3): p. 425- 442.
[2] LA Del Rio. Free Rad. Biol. Med. 1992,13: p.557-580.
[3] B. Grigolo, G. Linsigoli, S. Toneguzzi et al., Anticancer Res. 1998, 18(2A): p. 1175-1180.
[4] AM. Jansen, CB Bosman, L.Kuidenier et al., J. Cancer Res. Clin. Oncol. 1999, 125(6): p. 327-335.
[5] B. Palazotti, G. Pane, R.Colavitti et al., J. Cancer. 1999, 82(1): p. 145-150.
[6] M. Malafa, J. Margenthaker, B. Webb et al., J. Surg. Res. 2000, 88(2): p. 130-134.
[7] JQ Yang, S. Li, Y. Huang et al., Antiox. Redox. Signal. 2001, 3(4): p. 697-709.
[8]J Chung-ma Ho, S Zheng, SAComhaer et al., Cancer Res. 2001, 61(23): p. 8578-8585.
[9] GC Hur, SJ Cho, CH. Kim et al., Clin. Cancer Res. 2003, 9(15): p. 5768-5775.
[10] A Suresh, L. Guedez, J Moreb et al., Br. J. Haematol. 2003, 120(3): p. 457-463.
[11] M Hayyan, MA Hashim, IM Al Nashed.Chem.Rev., 2016, 116(5): p. 3029-3085.
[12] RS Weening, R Wever, D Doos, J. Lab. Clin. Med. 1975, 85(2): p. 245-252.
[13] T Matsubara, MZiff.J. Immunol. 1986,137(10): p. 3295-3298.
[14] GA Murrell, MJ Francus, L Btuneley. Biochem. J.2016, 265(3): p. 659-665.
[15] PF Li, R Dietz, R von Harsdorf. Cummulation. 1997, 96(10): p. 3602-3609.
[16] RH Burdon. Free Rad. Biol.Med. 1995, 18(4): p. 775-794.
[17] J Su. JT Groves. Inorg. Chemistry. 2010, 49: p. 6317- 6329.
[18] TP Steen. Nutrition. 2002, 18(10): p. 867-871 (2002).
[19]B Halliwell. Nutr. Rev. 1997, 55: p. S44-S49.
[20] A Tetsuya, KHiroakiK. Chem. Pharm. Bull. 2001, 49(5): p. 541-545.
[21] HC Birnboim. Carcinogenesis. 1986, 7: p. 1511-1517.
[22] I Fridovich. The biology of oxygen. Radicals Science. 1987, 201:875-880.
[23] MN Kondrashova.Biokhimiya. 1999, 64:361-363.
[24] I Fridovich.Biokhimiya.1999, 64: p. 361-363.
[25] MC Symons. Nature. 1987, 325: p. 659-660.
[26] SL Marklund. J. Biol. Chem.1992, 267(10): p. 6696-6701.
[27] B Bohm, Heinzelmann, MMotz et la., Biol. Chem.2015, 396 (12): p. 1339-1356.
[28] E Rybalka, CA Timpani, BD CheregiMitochondrion.2018, 42: p. 33-49.\
[29] CC Zhang, YJ Cong, Y Yanwt al., Anal. Methods. 2014, 6: p. 609-614.
[30] XH Li, GH Zhang, HM Ma et al., J. Am. Chem. Soc. 2004,126(37): p. 11543-11548.
[31] JM Pullar, MC Vissers, CL Winterbourn et al., IUBMB Life. 2000, 50(5): p. 259-266.
[32] C Chiang, JA Ledermann, E Aitkens et al., Clin. Cancer Res. 2008, 14(15).
[33] ZM Prokopowicz, F Arce, RBiendon. The J. Immunol. 2009, 184(2): p. 824-835.
[34] CL Chiang, JA Ledermann, AN Rad et al., Cancer Immunol, Immunother., 2006, 55: p. 1384-1395.
[35] MS Sampson, S. Panicheva, CSchokenmoehj et al., E. Pat. No.2017.
[36] S Hammerschmidt, NBuchler, H Wahn. Chest. 2002, 121: p. 573-581.
[37] CJ Venglarik, J. Giron-Calle, AF Wigley et al., J. Appl. Physiol. 2003, 95: p. 2444-2452.
[38] S Hammerschmidt, T Vogel, S Jokel et al, Respir. Med. 2007, 101: p. 1205-1211.
[39] T Hasegawa, E Malle, A Farhood et al., Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 289: p. G760-G767.
[40] M Whiteman, JPE Spencer, HH Szeto. Antioxid Redox Signal. 2008, 10(3): p. 641-650.
[41] YW Yap, M Whiteman, NC Cheung et al., Cell Signal. 2007, 19(2): p. 219-28.
[42] X Jiao, C Liu, Q Wang. Anal. Chim. Acta. 2017, 969: p. 49-56.
[43] PC Ikwegbue, PMasamba, LS Mbatha et al., Am. J. Cancer Res. 2019, 9(2): p. 242-249.
[44] E Malle, T Buch, JGrone. Kidney Int. 2003, 64(6): p. 1956-1967.
[45] SV Shah, RBaliga, MRajapurkar, JASN. 2007, 18(1): p. 16-28.
[46] H Honda, M Ueda, S Kojima, CJASN.2009, 4(1): p. 142-151.
[47] GM Virzi, A Clementi, A Brocca et al., Oxid. Med. Cell. 2015.
[48] P Panizzi, M Nahrendorf, MWieldgruber et al., J. Am. Chem. Soc. 2009,131: p. 15939-15744.
[49] K Cui, DQ Zhang, GH Zhang et al., Tetrahedron Lett. 2010,51(46): p. 6052-6055.
[50] SG Weiss, A Klein, MSlivka, J. Clin. Invest. 1982,70(3): p. 598-605.
[51] BS Rayner, Y Zhang, BE Brown et al., Free Rad. Biol. Med. 2018,129: p.25-34.
[52] JM Pullar,MCMVissers, CCWinterbourn et al., J. Biol. Chemistry. 2001, 276: p. 22120-22125.
[53] S Pennathur, D Maitra, J Byun et al., Free Rad. Biol. Med. 2010, 49(2): p. 205-213.
[54] M Valcheva-Traykova, L Saso, IKostova. Med. Chem. 2014, 14: p. 2508-2519.
[55] L Todorov,I Kostova, M Traykova. Curr. Med.Chem. 2019, 26: p. 1-16.
[56] J Martin, P Mladenka, L Saso. Report. 2016, 21(2): p. 84-89.
[57] SB Kedare, RP Singh. J. Food Sci.Technol. 2011, 48(4): p. 412-422.
[58] P Molyneux. J. Sci. Technol. 2004, 26: p. 211-219.
[59] J Chrzczanowicz, AGawron, AZwolinska et al., Clin. Chem. Lab. Med., 2008, 46(3): p. 342-349 (2008).
[60] O Erel. Clin. Biochem. 2004, 37(2): p. 112-119.
[61] O Erel. Et al., Clin. Biochem.2004, 37: p. 277-285.
[62] G Vural, SGumusyayla, H Bektas et al., World J. Neurosci. 2016, 6(3): p. 214-219.
[63] S Celic, B Baysal, S Sen. Adv. Biosci. Biotechnol. 2019, 10(11): p. 389-404.
[64] I Kostova, M. Valcheva-Traykova. Appl. Organomet. Chem. 2015, 29(12): p. 815–824.
[65] I Kostova, M Valcheva-Traykova, J. Coord. Chem. 2015, 68: 4082-4101.
[66] I Kostova, MValcheva-Traykova, S Balkansky. et al., J. Iran. Chem. Soc. 2016, 13: p. 891–902.
[67]NPL Kale&Laby, UV spectroscopy.2015.
[68] F Smidt. Nature Publishing Group.2001.
[69] LT Todorov, BBChifchiev, MLValcheva-Traykova. 2018, p. 207-212.
[70] K Xiong, C Yin, J Chao et al., Molecular and Biomolecular Spectroscopy. 2016, 166: p. 79-83.
[71] LT Molina, MJ Molina. J. Phys. Chem. 1978, 82(22): p. 2410-2414.
[72] M Aldrich, CA McCarthy, JK Hurst. PNAS. 1981,78(1): p. 210-214.
[73] SJ Weiss, R Klein, ASlivka et al., J. Clin. Inv. 1982, 70: p. 598- 600.
[74] CI Hawkins, MJ Davies.et al.Biochem. J.1998, 332(3): p. 617-625.
[75] CL Hawkins,MJDavies. Biomed. J. 1999, 340: p. 539-548.
[76] S Shintani.2013, p.S7:004.
[77] L Benov. Protoplasma. 2001, 217: p. 33-36.
[78] YA Suh,RSArnold, B Lassegue. Nature. 1999, 401: p.79-82.
[79] RH Butdon. Free Rad. Biol.Med., 1995, 18(4): p. 775-794.
[80] L Le Marchand, LA Deifried, A Lum et al., Cancer Epidemiol Biomarkers Prev.2000, 9(2): p.181-184.
[81] N Gungor, Ad Knaapen, A Munnia. Mutagenesis. 2010, 25(2): p. 149-154.