Research Article - Der Pharma Chemica ( 2022) Volume 14, Issue 10
Intermolecular Free Length And Molar Volume Of Binary Liquid Mixtures Of Ethyl Acetate With 1-alkanol At 303.15K
Dhirendra Kumar Sharma*, Seema Agarwal and Akil KhanDhirendra Kumar Sharma, Department of Chemistry Institute of Basic Science, Bundelkhand University, Jhansi (U.P), India, Email: dhirendra.dr@rediffmail.com
Received: 23-Sep-2022, Manuscript No. dpc-22-75723; Editor assigned: 26-Sep-2022, Pre QC No. dpc-22-75723; Reviewed: 10-Oct-2022, QC No. dpc-22-75723; Revised: 12-Oct-2022, Manuscript No. dpc-22-75723; Published: 19-Oct-2022, DOI: 10.4172/0975-413X.14.10.1-9
Abstract
The ultrasonic velocity (u) and density (ρ) have been measured for ethyl acetate with 1-alkanols at 303.15K, over the entire range of composition. The thermodynamic parameter such as intermolecular free length (Lf), molar volume (Vm), isentropic compressibility (βs) and internal pressure (Pi) have been obtained for all mixtures from experimental data with a view to investigating the exact nature of the molecular interaction. The excess values of the said parameter have also been calculated. The deviation from ideality of these parameters is explained on the basis of molecular interactions between the constituent components of the mixture.
Keywords
Internal pressure; Isentropic compressibility; Ultrasonic velocity; Density; Mole fraction; Binary mixtures; Molecular interaction
INTRODUCTION
During last three decades [1,2], studying thermo acoustical parameters have gained much importance. These parameters include intermolecular free length (Lf), molar volume (Vm), isentropic compressibility (βs) and internal pressure (Pi) [3,4], has been made to study the intermolecular interaction in mixtures. Fundamental thermodynamic and thermo physical properties are essential sources of information necessary for a better understanding of the non-ideal behaviour of complex systems because physical and chemical effects which are caused by molecular interactions, intermolecular forces etc. of unlike molecules. In recent years the ultrasonic study of properties of liquid mixtures and solvation find direct application in chemical and biochemical industry. Thermodynamic and transport properties of liquid of liquid mixtures have been extensively used to study the departure of a real liquid mixture behavior from ideality. The measurements of ultrasonic velocity, viscosity and density have been adequately employed in understanding the molecular interactions in liquid mixtures.
Investigations on binary liquid mixtures of ethylacetate with 1- alkanols at 303.15K by calculation excess thermodynamic parameters are found to be highly useful in understanding the solute-solvent interactions in these mixtures. The measured ultrasonic parameters are being extensively used to study intermolecular process in liquid system [5,6].The sign and magnitude of non-linear deviations from ideal values of velocities and inter molecular free length are attributed to the difference in molecular size and strength of interaction between unlike molecules[7-10].Ultrasonic velocity and density of the six binary mixtures are measured at 303.15K for different composition of the components. Intermolecular free length (Lf), molar volume (Vm), isentropic compressibility (βs) and internal pressure (Pi) and excess values are calculating using the standard equation.
The present note deals with the study of ultrasonic velocity (u) , density (ρ), Intermolecular free length (Lf), molar volume (Vm), isentropic compressibility (βs) and internal pressure (Pi) and excess values of excess intermolecular free length (LEF), excess molar volume (VEM), excess isentropic compressibility (βES) and excess internal pressure (Pi E) for six binary liquid mixtures. The results were analyzed in terms of changes in intermolecular free spaces between the pairs of molecules due to addition of second component.
EXPERIMENTAL
Material
The chemicals used in the present work were high purity laboratory reagent grade samples of ethyl acetate, 1-methanol, 1-ethanol, 1-propanol, 1-butanol, 1-hexanol, 1-octanol purchased from Merck Chem. Ltd India. The purity of the chemicals is shown in table-1. All the chemicals were kept in tightly sealed bottles to minimize the absorption of atmospheric moisture
| Component | CAS number | Source | Initial mass fraction purity |
|---|---|---|---|
| >Ethyl acetate | 141-78-6 | Merck Chem. Ltd India | 0.998 |
| 1-Methanol | 67-56-1 | Merck Chem. Ltd India | 0.995 |
| 1-Ethanol | 64-17-5 | Merck Chem. Ltd India | 0.995 |
| 1-Propanol | 71-23-8 | Merck Chem. Ltd India | 0.995 |
| 1-Butanol | 71-36-3 | Merck Chem. Ltd India | 0.995 |
| 1-Hexanol | 111-27-3 | Merck Chem. Ltd India | 0.995 |
| 1-Octanol | 111-87-5 | Merck Chem. Ltd India | 0.995 |
The purity of the solvent was ascertained by comparing the measured density, and sound velocity of the pure component at 303.15K with the available literature [11-18], as shown in Table 2. The reported experimental values of density (ρ) and sound velocity (u) conform closely to their corresponding literature values.
| Component | Density (ρ) | Ultrasonic Velocities (u) | ||
|---|---|---|---|---|
| kg-m-3 | m.s-1 | |||
| Observed | Literature | Observed | Literature | |
| >Ethyl acetate | 0.882 | 0.8885[13] | 1125 | 1115.0[11] |
| 1-Methanol | 0.784 | 0.7817 [13] | 1084 | 1084.0 [17] |
| 1-Ethanol | 0.772 | 0.7807 [12] | 1141 | 1144.3 [14] |
| 1-Propanol | 0.807 | 0.8003 [16] | 1182 | 1182.6 [14] |
| 1-Butanol | 0.804 | 0.8020 [14] | 1196 | 1196.6 [14] |
| 1-Hexanol | 0.8128 | 0.8118 [14] | 1298 | 1282.0 [18] |
| 1-Octanol | 0.8242 | 0.8187 [15] | 1327 | 1330.8 [15] |
Measurements
Six binary system viz. ethyl acetate + 1-methanol, ethyl acetate + 1-ethanol, ethyl acetate +1- propanol, ethyl acetate + 1-butanol, ethyl acetate + 1-hexanol and ethyl acetate + 1-octanol were studied. Each sample mixture was prepared, on mass basis, by mixing the calculated volume of liquid components in specially designed glass stoppered bottles. All binary mixtures were prepared by weight covering the entire mole fraction range. The components of binary mixtures were injected by means of syringe in to the glass vials of sealed with rubber stopper in order to check evaporation losses during sample preparation. The mass measurements were carried out using an single pan analytical balance ( Model K-15 Deluxe, K Roy Instruments Pvt. Ltd. Varanasi, India.) with an accuracy of ± 0.00001×10-3 kg as described elsewhere [19]. The possible error in the mole fraction was estimated to be less than 1×10 -4. Five samples were prepared for one system, and their density and sound velocity were measured on the same day.
Density
Densities of pure liquids and their binary mixtures were determined by using a double-arm pycnometer made of Borocil glass [12], with a bulb of 25 cm3 and a capillary of an internal diameter of about 1 mm is used to measure the densities (ρ) of pure liquids and binary mixtures. The pycnometer was calibrated by using conductivity water (Conductance less than 1×10-6ω-1) with 0.99705 g cm-3 as its density at 303.15K. The pycnometer was kept for about 30 min. in an electronically controlled thermostated water bath (303.15 + 0.02K) and the position of the liquid level on the capillary was noted. The volume of the pycnometer at each mark was calculated by using the literature value of the density of pure water at 303.15K. The pycnometer filled with air bubbles free liquids is kept in a thermostate water bath (MSI Goyal Scientific, Meerut, India) controlled with a thermal equilibrium. The precision of the density measurements was estimated to be ±0.0002 g cm-3. The observed values of densities of pure ethyl acetate, 1-methanol, 1-ethanol, 1-propanol, 1-butanol, 1-hexanol and 1-octanol at 303.15K were 0.8820, 0.7840, 0.7720, 0.8070, 0.8040, 0.8128 and 0.8242 kg.m-3 which compare well with corresponding literature values of respectively.
Sound velocity
The ultrasonic velocities were measured using a multi frequency ultrasonic interferometer (Model F-80D, Mittal Enterprise, New Delhi, India) working at 3 MHz. that has reproducibility of + 0.5 m s-1 at 300C. The meter was calibrated with water and benzene at 303.15K. For all the measurements, temperature was controlled by circulating the water through a thermo state water bath (MSI Goyal Scientific, Meerut, India) controlled with a thermal equilibrium keeping temperature accuracy within + 0.02K. The measured values of ultrasonic velocities of pure ethyl acetate,1- methanol, 1-ethanol, 1-propanol, 1-butanol, 1-hexanol and 1-octanol at 303.15K were 1125, 1084, 1141, 1182, 1196, 1298 and 1327 m.s-1 respectively, which compare well with the corresponding literature values.
Theoretical
In the year 1952, Jacobson [15,16], suggested an empirical relation for calculating the free length (LF) of liquids. Intermolecular free length (LF), can be calculated from the isentropic compressibility (βs) by the relation given below
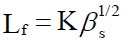
Where K is temperature dependent constant, called the Jacobson constant. Jacobson determined the value of K empirically between 0 to 50ºC and βs is the isentropic compressibility. The isentropic compressibility (βs) has been calculated from the sound velocity (u) and the density (ρ) of the medium using the equation as
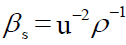
The molar volume Vm calculated from the measured values of density (ρ), molar volume (Vm) was calculated using the relation
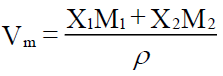
Where X1, X2 and M1, M2 are the mole fraction and molecular weight of the component 1 and 2 respectively.
Suryanarayana and Kuppuswami [20,21], suggested a method for evaluation of internal pressure from the knowledge of ultrasonic velocity, u, density, ρ, and viscosity, η, the relation proposed is expressed as
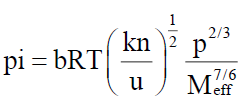
Where b is packing factor, which is assumed to be 2 for all liquid and solution. k is a constant, independent of temperature and its value is 4.28 × 109 for all liquids, R is universal gas constant and T is absolute temperature.
The excess value of ultrasonic related parameters have been calculated by using the following relation
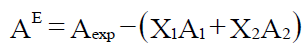
Where A represents the parameter such as intermolecular free length, molar volume, isentropic compressibility and internal pressure and X1 and X2 are the mole fractions of components whose parameters.
RESULT AND DISCUSSION
The experimental values of density and ultrasonic velocity and calculated values of intermolecular free length, isentropic compressibility, molar volume and internal pressure at 303.15K over the entire composition range are presented in table 3. The excess function, excess intermolecular free length (LEF ), excess molar volume (VEM), excess isentropic compressibility (βES ), and excess internal pressure (Pi E) for six binary liquid mixtures evaluated using equation (5) are summarized in table 4. The excess parameters, excess intermolecular free length (LEF ), excess molar volume (VEM), excess isentropic compressibility (βES) and excess internal pressure (Pi E) have been plotted against mole fraction of ethylacetae in figures 1,2,3,4. It is evident from table 3 density, molar volume and sound velocity increases while intermolecular free length, isentropic compressibility and internal pressure decrease with increase in concentration of ethylacetae in all the mixtures. The pronounced increase or decrease in these parameters with composition of the mixtures indicates the presence of interaction between the component molecules in the binary liquid mixture. The resultant interaction an a liquid mixture is not solely dependent on the molecular structure of component liquids but is also influenced by other factors like dispersion forces, dipole-dipole interaction, dipole-induced dipole interaction, hydrogen bonding, charge transfer interaction and a complex formation. Normally dispersion forces make a positive contribution to excess values, while other makes a negative contribution.
| Mole fraction x1 |
Density(ρ) k.gm-3 |
Sound Velocity(u) m / s |
isentropic compressibility βs×10-8 m2 N-1 |
intermolecular free length Lf×10-9 m |
Molar volume vm×106 m3 mol-1 |
Internal pressure (pi×104) atm |
|---|---|---|---|---|---|---|
| Ethyl acetate+ 1-Mehanol | ||||||
| >0.0000 | 784.0 | 1084 | 108.54 | 2.22022 | 40.8673 | 1.85827 |
| 0.1039 | 796.8 | 1099 | 100.39 | 2.13515 | 47.5245 | 1.39542 |
| 0.2248 | 819.2 | 1103 | 100.33 | 2.13451 | 54.4992 | 1.07580 |
| 0.3129 | 839.5 | 1105 | 96.54 | 2.09380 | 59.0643 | 0.91572 |
| 0.4370 | 848.3 | 1110 | 95.67 | 2.08435 | 66.6542 | 0.73366 |
| 0.5474 | 867.5 | 1114 | 92.88 | 2.05373 | 72.3090 | 0.62282 |
| 0.6409 | 870.9 | 1117 | 92.02 | 2.04420 | 78.0508 | 0.53580 |
| 0.7128 | 879.0 | 1118 | 91.01 | 2.03295 | 81.9179 | 0.49096 |
| 0.8164 | 879.2 | 1122 | 90.34 | 2.02545 | 88.5081 | 0.43096 |
| 0.9104 | 880.5 | 1123 | 90.05 | 2.02220 | 94.3665 | 0.38473 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.8979 | 0.34494 |
| Ethyl acetate+ 1-Ethanol | ||||||
| 0.0000 | 772.0 | 1141 | 99.49 | 2.12563 | 59.6761 | 1.48567 |
| 0.1049 | 802.5 | 1137 | 96.38 | 2.09216 | 62.9054 | 0.99503 |
| 0.2090 | 815.7 | 1135 | 95.16 | 2.07882 | 67.2489 | 0.84487 |
| 0.3105 | 827.8 | 1134 | 93.93 | 2.06531 | 71.4184 | 0.71041 |
| 0.4166 | 839.2 | 1133 | 92.81 | 2.05305 | 75.7609 | 0.62822 |
| 0.5094 | 849.6 | 1132 | 91.84 | 2.04230 | 79.4294 | 0.56071 |
| 0.6076 | 860.4 | 1131 | 90.85 | 2.03118 | 83.2250 | 0.49618 |
| 0.7150 | 863.9 | 1130 | 90.65 | 2.02893 | 88.1223 | 0.44587 |
| 0.8069 | 877.6 | 1128 | 89.54 | 2.01653 | 91.1441 | 0.40652 |
| 0.9030 | 882.7 | 1126 | 89.35 | 2.01434 | 95.1972 | 0.37581 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.8979 | 0.34494 |
| Ethyl acetate + 1-Propanol | ||||||
| 0.0000 | 807.0 | 1182 | 88.68 | 2.00681 | 74.4563 | 1.12536 |
| 0.1074 | 813.3 | 1173 | 89.35 | 2.01439 | 77.5792 | 0.88659 |
| 0.2086 | 826.2 | 1169 | 88.56 | 2.00541 | 79.7993 | 0.72795 |
| 0.3145 | 832.1 | 1161 | 89.15 | 2.01209 | 82.8013 | 0.62680 |
| 0.4099 | 842.8 | 1159 | 88.33 | 2.00280 | 84.9257 | 0.54900 |
| 0.4758 | 850.9 | 1154 | 88.24 | 2.00185 | 86.2868 | 0.51357 |
| 0.54307 | 860.9 | 1150 | 87.82 | 1.99707 | 87.4748 | 0.46810 |
| 0.6127 | 864.7 | 1142 | 88.67 | 2.00668 | 89.3453 | 0.44151 |
| 0.7564 | 868.5 | 1138 | 88.90 | 2.00933 | 93.5897 | 0.39664 |
| 0.9126 | 878.8 | 1134 | 88.48 | 2.00458 | 97.4750 | 0.36363 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.8979 | 0.34494 |
| Ethyl acetate + 1-Butanol | ||||||
| 0.0000 | 804.0 | 1196 | 86.95 | 1.98712 | 92.1902 | 0.93886 |
| 0.1063 | 805.6 | 1194 | 87.06 | 1.98837 | 93.8435 | 0.72809 |
| 0.2151 | 816.8 | 1184 | 87.33 | 1.99147 | 94.4294 | 0.61710 |
| 0.3213 | 826.9 | 1180 | 86.85 | 1.98595 | 95.0707 | 0.53084 |
| 0.4327 | 832.2 | 1176 | 86.88 | 1.98633 | 96.3355 | 0.51618 |
| 0.5192 | 842.0 | 1170 | 86.75 | 1.98486 | 96.6507 | 0.43625 |
| 0.6266 | 842.4 | 1167 | 87.16 | 1.98949 | 98.3882 | 0.39871 |
| 0.7124 | 858.1 | 1154 | 87.50 | 1.99344 | 97.9889 | 0.38101 |
| 0.8127 | 866.6 | 1142 | 88.47 | 2.00441 | 98.6404 | 0.36421 |
| 0.9044 | 874.2 | 1134 | 88.94 | 2.00981 | 99.2555 | 0.35557 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.8979 | 0.34494 |
| Ethyl acetate +1- Hexanol | ||||||
| 0.0000 | 812.8 | 1298 | 73.02 | 1.82102 | 125.6914 | 0.76533 |
| 0.0996 | 821.4 | 1292 | 72.92 | 1.81983 | 122.6634 | 0.62247 |
| 0.2225 | 833.8 | 1287 | 72.40 | 1.81331 | 118.7742 | 0.54978 |
| 0.3149 | 835.5 | 1275 | 73.62 | 1.82849 | 116.9760 | 0.49429 |
| 0.4151 | 840.6 | 1257 | 75.29 | 1.84907 | 114.5933 | 0.45038 |
| 0.5186 | 846.6 | 1247 | 75.95 | 1.85723 | 112.0864 | 0.41469 |
| 0.6083 | 854.4 | 1240 | 76.11 | 1.85917 | 109.5595 | 0.39077 |
| 0.7096 | 861.7 | 1222 | 77.70 | 1.87853 | 106.9770 | 0.37353 |
| 0.8066 | 867.2 | 1210 | 78.75 | 1.89111 | 104.7255 | 0.36080 |
| 0.9041 | 878.0 | 1192 | 80.15 | 1.90786 | 101.8771 | 0.34774 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.8979 | 0.34494 |
| Ethyl acetate + 1-Octanol | ||||||
| 0.0000 | 824.2 | 1327 | 68.89 | 176878 | 158.0052 | 0.66872 |
| 0.1056 | 825.9 | 1312 | 70.33 | 1.78718 | 152.2800 | 0.55668 |
| 0.2095 | 830.0 | 1294 | 71.94 | 1.80754 | 146.2509 | 0.49024 |
| 0.3174 | 831.8 | 1275 | 73.94 | 1.83249 | 140.4730 | 0.43728 |
| 0.4286 | 838.7 | 1239 | 77.66 | 1.87799 | 133.7374 | 0.37509 |
| 0.5083 | 840.0 | 1225 | 79.32 | 1.89800 | 129.5386 | 0.38467 |
| 0.6196 | 844.4 | 1214 | 80.35 | 1.91025 | 123.3172 | 0.35719 |
| 0.7090 | 858.6 | 1192 | 81.96 | 1.92927 | 145.9131 | 0.37519 |
| 0.8064 | 865.1 | 1164 | 85.31 | 1.96830 | 111.2675 | 0.34396 |
| 0.9044 | 871.6 | 1148 | 87.05 | 1.98825 | 105.7013 | 0.35364 |
| 1.0000 | 882.0 | 1125 | 89.58 | 2.01695 | 99.89795 | 0.34494 |
Alkanols are liquids which are associated through the hydrogen bonding and in the pure state; they exhibit an equilibrium between the monomer and multimer species. Also, they can be associated with any other group having some degree of polar attraction [22]. Due to polar nature of ethyl acetate and alkanols, the dipole-dipole interaction prevail in these mixtures. When the compounds are mixed the changes the occur in association equilibria are evidently repture of the hydrogen bonds [17] in pure ethyl acetate and alkanols, dipole-dipole interactions and the formation of O ÃÂ?¶ H----O hydrogen bonds between the components.
The 1-alkanols are associated through hydrogen bonding.

and ethyl acetate – 1-alkanols interactions are due to hydrogen bonding between the oxygen atom of the ethyl acetate and the proton of hydrogen group of alkanols.
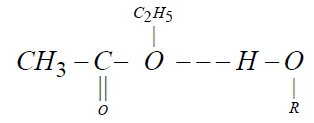
The result present in Table 3, show non-linear behaviour of isentropic compressibility, intermolecular free length, molar volume and internal pressure which is further substantial by their excess values (Table 4). All the seven organic compounds namely ethyl acetate, methanol, ethanol, propanol, butanol, hexanol and octanol are a polar organic compounds having dipole moment 1.78 D, 1.70 D, 1.69 D, 1.68 D, 1.66 D, 1.60 D and 1.68 D respectively. Normally more the dipole moment, stronger is the intermolecular attraction, which results in decreasing of free space between molecules and increase in the ultrasonic velocity (Table 4).
| Mole Fraction x1 |
βES×10-7 m2 N-1 |
LEF×10-10 m |
VEM m3 mol-1 |
PEi×104 atm. |
|---|---|---|---|---|
| Ethyl acetate + 1-Methanol | ||||
| >0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.1039 | -61.8 | -0.6391 | -0.52201 | -0.3055 |
| 0.2248 | -39.4 | -0.6993 | -0.66072 | -0.4421 |
| 0.3129 | -60.6 | -0.7077 | -0.77383 | -0.4689 |
| 0.4370 | -45.8 | -0.7300 | -0.80962 | -0.4732 |
| 0.5474 | -52.8 | -0.7517 | -0.87129 | -0.4070 |
| 0.6409 | -43.6 | -0.4569 | -0.95002 | -0.2967 |
| 0.7128 | -40.1 | -0.4232 | -0.62853 | -0.2885 |
| 0.8164 | -27.1 | -0.2876 | -0.55257 | -0.1923 |
| 0.9104 | -12.2 | -0.1289 | -0.54600 | -0.0956 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| Ethyl acetate+ 1-Ethanol | ||||
| 0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.1049 | -20.6 | -0.2204 | -0.99258 | -0.3708 |
| 0.2090 | -22.6 | -0.2408 | -1.83336 | -0.4023 |
| 0.3105 | -24.8 | -0.2654 | -1.94880 | -0.4209 |
| 0.4166 | -25.4 | -0.2722 | -2.67661 | -0.4438 |
| 0.5094 | -25.9 | -0.2794 | -2.73675 | -0.4821 |
| 0.6076 | -26.2 | -0.2839 | -2.88955 | -0.3438 |
| 0.7150 | -17.5 | -0.1896 | -1.31421 | -0.2963 |
| 0.8069 | -19.4 | -0.1137 | -0.99000 | -0.2240 |
| 0.9030 | -11.9 | -0.1013 | -0.79942 | -0.1585 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| Ethyl acetate + 1-Propanol | ||||
| 0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.1074 | -5.75 | -0.0650 | -0.39267 | -0.1542 |
| 0.2086 | -3.10 | -0.0849 | -0.43678 | -0.2345 |
| 0.3145 | -4.85 | -0.1210 | -0.64416 | -0.2530 |
| 0.4099 | -7.22 | -0.1873 | -0.84248 | -0.2564 |
| 0.4758 | -8.65 | -0.1978 | -0.97433 | -0.2640 |
| 0.5430 | -13.4 | -0.1524 | -1.80306 | -0.2334 |
| 0.6127 | -5.62 | -0.0634 | -0.69878 | -0.2056 |
| 0.7564 | -4.57 | -0.0515 | -0.41016 | -0.1384 |
| 0.9126 | -3.16 | -0.0148 | -0.19909 | -0.0495 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| Ethyl acetate + 1-Butanol | ||||
| 0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.1063 | -1.69 | -0.0190 | -0.83451 | -0.1476 |
| 0.2151 | -1.84 | -0.0204 | -0.98179 | -0.1939 |
| 0.3213 | -9.46 | -0.1073 | -1.38436 | -0.2171 |
| 0.4327 | -12.0 | -0.1368 | -1.81050 | -0.2656 |
| 0.5192 | -15.6 | -0.1772 | -2.45928 | -0.2942 |
| 0.6266 | -14.3 | -0.1630 | -1.36900 | -0.1679 |
| 0.7124 | -13.1 | -0.1491 | -1.30851 | -0.1347 |
| 0.8127 | -6.17 | -0.0693 | -0.88668 | -0.0919 |
| 0.9044 | -3.81 | -0.0286 | -0.79466 | -0.0460 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| Ethyl acetate + 1-Hexanol | ||||
| 0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.0996 | -17.4 | -0.2069 | -0.45637 | -0.1009 |
| 0.2225 | -43.0 | -0.5129 | -1.17608 | -0.1219 |
| 0.3149 | -46.1 | -0.5421 | -1.59146 | -0.1386 |
| 0.4151 | -46.0 | -0.5828 | -1.88840 | -0.1404 |
| 0.5186 | -56.5 | -0.6538 | -2.25452 | -0.1623 |
| 0.6083 | -69.8 | -0.8103 | -1.43844 | -0.1326 |
| 0.7096 | -60.6 | -0.7152 | -1.40800 | -0.1187 |
| 0.8066 | -56.2 | -0.4784 | -0.75911 | -0.0934 |
| 0.9041 | -38.4 | -0.3028 | -0.49263 | -0.0654 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| Ethyl acetate + 1-Octanol | ||||
| 0.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
| 0.1056 | -7.43 | -0.0781 | -0.42861 | -0.0778 |
| 0.2095 | -12.8 | -0.1323 | -0.43692 | -0.1106 |
| 0.3174 | -15.1 | -0.1505 | -0.92541 | -0.1286 |
| 0.4286 | -16.97 | -0.1684 | -1.04906 | -0.1548 |
| 0.5083 | -10.81 | -0.1308 | -1.07905 | -0.1694 |
| 0.6196 | -9.50 | -0.1229 | -1.32444 | -0.1194 |
| 0.7090 | -8.90 | -0.1146 | -0.85666 | -0.1109 |
| 0.8064 | -7.65 | -0.0160 | -0.72863 | -0.0632 |
| 0.9044 | -5.53 | -0.0096 | -0.25366 | -0.0222 |
| 1.0000 | 0.00 | 0.0000 | 0.00000 | 0.0000 |
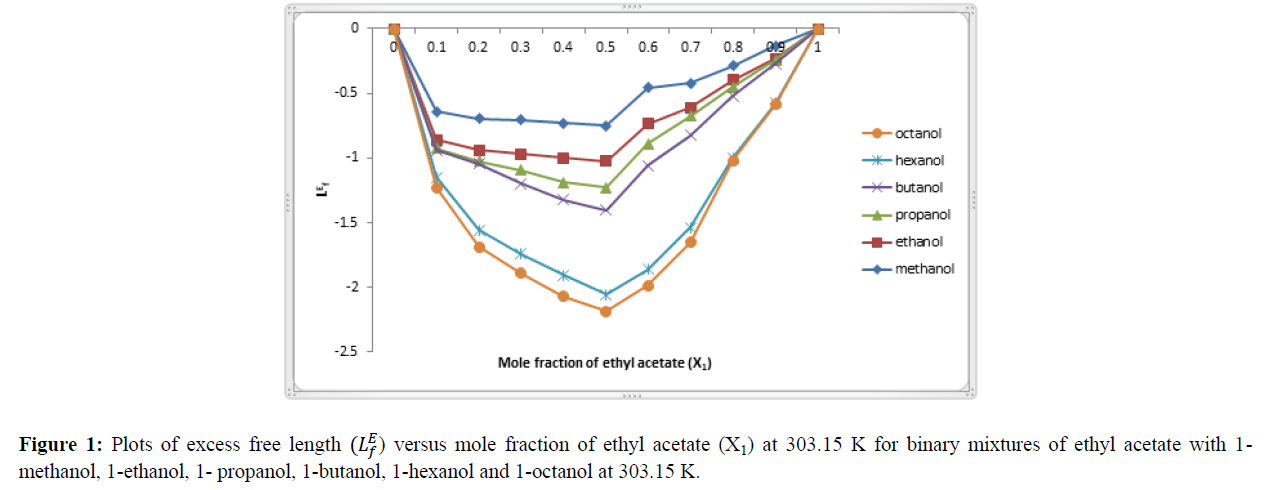
A perusal of table 4 shows that the values of excess intermolecular free length for all the six binary system are negative. These negative values of excess intermolecular free length are shown in figure 1. From figure 1 shows that the excess intermolecular free length (LEF ) values are negative for all binary systems but the magnitude of the negative values increase with increasing chain length of alcohols, the order are given below- 1-Methanol < 1-Ethanol < 1-Propanol < 1-Butanol < 1-Hexanol < 1-Octanol
The above order indicates the strength of interactions between component molecules decreases due to decrease in polarizability of alkanol molecules.
The negative values of excess intermolecular free length (LEF ) play a very important role in description of molecular interaction in liquid mixtures through dipole-dipole interaction and hydrogen bonding. Due to polar nature of ethyl acetate and alkanols, the dipole-dipole interactions prevail in these mixtures. When the compounds are mixed the changes the occur in association equilibria are evidently rapture of the hydrogen bonds in pure ethyl acetate and alkanols, dipole-dipole interactions and the formation of O ÃÂ?¶ H----O hydrogen bonds between the components. This suggests the existence of strong interaction between the components in all the binary systems. The values of LEF suggest that strong specific interaction like the formation of H- bond association through weaker physical forcers of attraction [23].
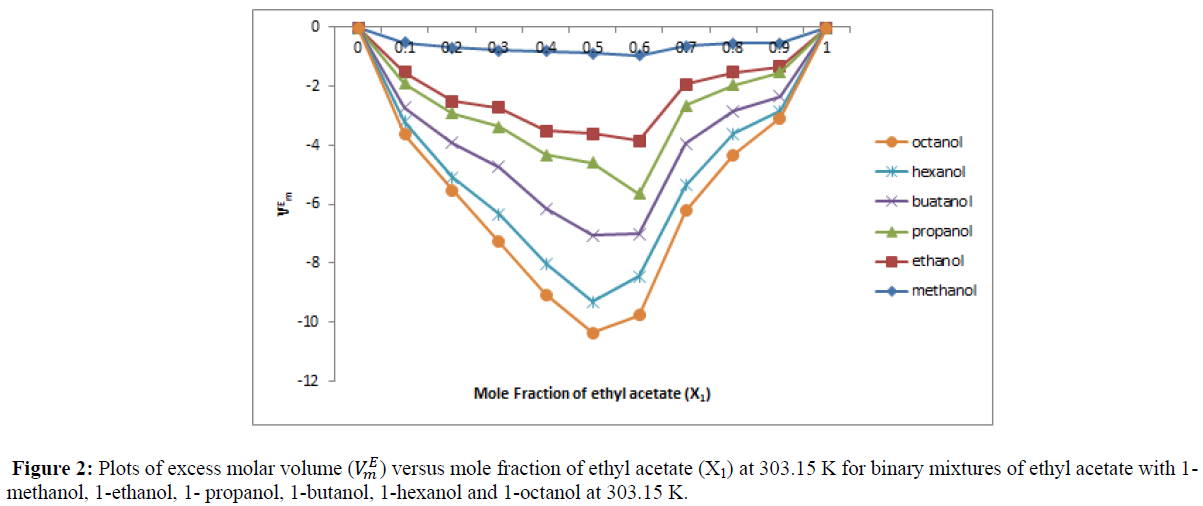
A perusal of Table 4 reveal that the value of excess molar volume (VEM) are negative in all binary liquid system over the entire range of composition at 303.15 K. The value of VEM are plotted against the mole fraction of ethyl acetate (x1) and are shown in Figure 2. In the present investigation the negative VEM values for binary mixtures of ethyl acetate with 1- alkanols may be attributed to hydrogen bond formation through dipole-dipole interaction between 1- alkanol and ethyl acetate molecule or to structure contribution arising from geometrical fitting of one component (1-alkanol) in to the other (ethyl acetate) due to differences in the molar volumes between components. Excess molar volume value of all binary liquid systems increases with an increases of concentration of ethyl acetate (x1). This is attributed to decreased ester-ester and alkanol-alkanol contacts with an increase the concentration of ethyl acetate (x1).
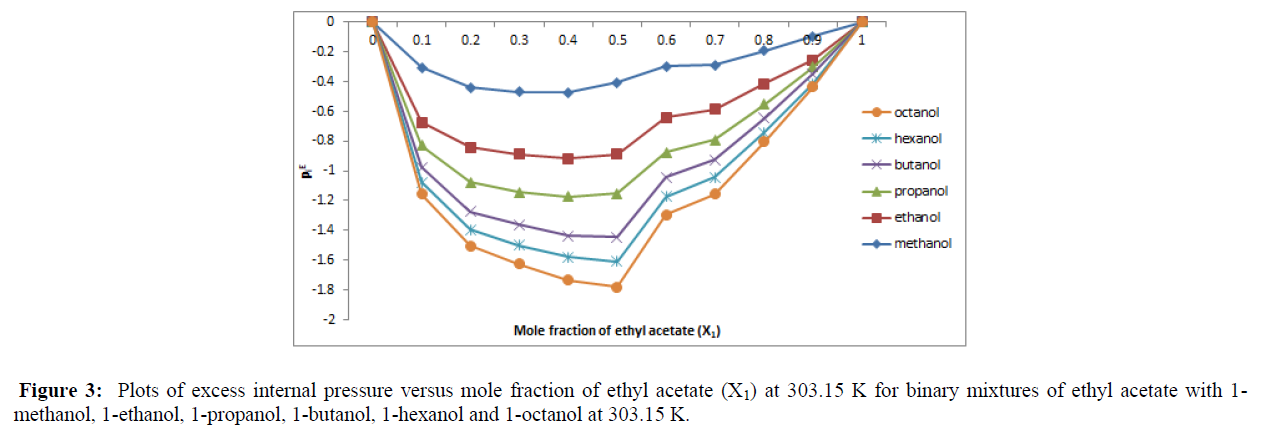
The excess internal pressure (Pi E ) is another important parameter through which molecular interactions can be explained. In the present investigation for the six binary systems it is observed that, as the mole fraction of ethyl acetate increase, the Pi E values decreases. The values of Pi E are almost negative and gradually decrease and move towards the positive values by the increase of mole fraction of ethyl acetate. More over the Pi E decrease with increase in x1. This situation is observed for all six binary system under study and can be viewed from plots Figure 3.
The negative values of excess internal pressure (Pi E) indicate the presence of strong molecular interaction. We may conclude that alkanols, which is a self – associating polar organic liquid has a tendency to form complexes with ethyl acetate and the increase in its dilution causes disruption of aromatic C – H bond stretching as the self – association of alkanes is disrupted. It is also concluded that suryanarayana approach for estimating internal pressure of binary liquid mixtures, based on dimensional analysis using thermodynamic consideration is very well applicable in the present case.
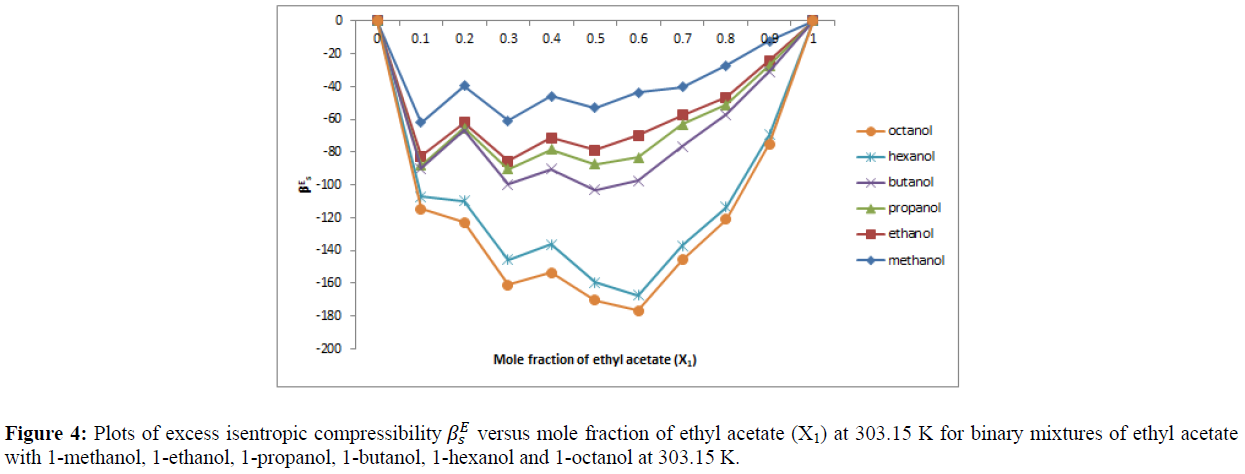
A perusal of Table 4 also reveal that the values of excess isentropic compressibility (βES) are negative over the entire range of concentration in all the six binary system at 303.15 K. The value of excess isentropic compressibility (βES) are plotted against the mole fraction of ethyl acetate and are shown in Figure 4.
The sign of excess isentropic compressibility play a vital role in assessing the compactness due to molecular interaction in liquid mixtures through charge transfer, dipole-dipole interaction and dipole induced dipole interactions interstitial accommodation and orientational ordering leading to more compact structure making, which enhances excess isentropic compressibility to have negative values. Fort and Moore [18] suggested that the liquid having different molecular size and shape mix well there by reducing the volume which causes the values of βES to be negative. It is also suggested that liquids are less compressible when compared to their ideal mixtures signifying the chemical effects including charge transfer forces, formation of hydrogen bond and other complex forming interactions. It can also be said that the molecular interaction are strong in these binary liquid mixtures and that the medium is highly packed. Similar results were obtained by earlier worker [24]. The negative value of βES in these mixtures can be associated with a structure forming tendency.
The charge in intermolecular free length, molar volume and internal pressure with change in composition in these systems, indicate the variations in cohesive forces of these system. A perusal of the sign and magnitude of the excess values of the parameters (LEF), (VEM), (βES) and (PEi ) show that the strength of the molecular interaction between the component molecules. The present study point at clearly that in all six binary systems ethyl acetate is involved in the charge transfer interaction with alcohols. The oxygen atom of ethyl acetate being strongly electronegative would have tendency to attract electropositive hydrogen of alcohols resulting in the formation of new species, showing the negative value of excess thermodynamic properties.
The negative deviations of high magnitude lead to the unstable complex formation between the hetero molecules of the mixtures. In the present study the negative excess volume indicates the breaking of hydrogen bond of alcohols polymers as alcohol is diluted with ethyl acetate and association between unlike molecules causing contraction with increasing concentration of ethyl acetate. Therefore it can be concluded that AB type strong specific interaction take place leading to a bonding like ( ÃÂ?¶ H---O ÃÂ?¶ ) Which results in the formation of a complex of the type
The results reveal that the intermolecular interaction decreases with the increasing size of the alcohol molecules. Hydrogen bonding strength also decreases with decreasing dipole moment of the alcohol molecules.
CONCLUSIONS
The ultrasonic method is powerful tool for characterizing physic-chemical properties and existence of molecular interaction in the mixture. In this paper, an attempt is made to measure densities (ρ) and ultrasonic velocity (u) at 303.15 K over the entire range of mixture composition of ethyl acetate with 1-methanol, 1-ethanol, 1- propanol, 1-butanol, 1-hexanol and 1-octanol out of these measured data, excess intermolecular free length (LEF), excess molar volume (VEM), excess isentropic compressibility (βES) and excess internal pressure (PEi) have been calculated.
The negative deviations are observed in the case of excess intermolecular free length (LEF), excess molar volume (LEF), excess isentropic compressibility (βES) and excess internal pressure (PEi) are observed for all binary mixtures of ethyl acetate with 1-methanol, 1-ethanol, 1-propanol, 1-butanol, 1-hexanol, 1-octanol.The results are analyzed in the sight of molecular interactions between the components. It may be concluded that the interaction resulting in the interstitial accommodation of ethyl acetate in to 1-alkanols are the predominant factor over dipole-dipole interaction. All the experimental determinations of excess intermolecular free length (LEF), excess molar volume (VEM) excess isentropic compressibility (βES) and excess internal pressure (PEi) are strongly correlated with each other.
Acknowledgement
The authors are very much thankful to the Head of the Department of Chemistry Bundelkhand University, Jhansi (U.P.) India. For proving the facilities for Research work.
Declarations conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Funding
The author(s) reported there is no funding associated with the work featured in this article.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Author Contribution Statement
Dhirendra Kumar Sharma, Research design, Investigation, Writing-Original draft preparation and Manuscript correction.
Seema Agarwal, Data Analysis and Mathematical Calculation
Akil Khan, Tables and Graph preparation.
References
- Pandey JD, Dey R, Bhatt BD. J Phys chem Comm. 2002, 5: p. 37.
- Pandey JD, Dey R. J Phys chem Comm. 2003, 6: p. 55.
- Pal A, Das G, Kumar H. J Pure Appl Ultrason. 1987, 10: p. 23.
- Ali A, Tiwari K, Nain AK et al., Indian J Phys. 2000, 74: p. 351.
- Ali A, Hyder S, Nain AK. J Solut Chem. 2003, 32: p. 865-877.
- Banipal TS, Toor A, Rattan PVK. J Solut Chem. 2002, 31: p. 905-915.
- Fort R, Moore WR. Trans Faraday Soc.1965, 61: p. 2102.
- Kannappan AN, Palani R. Indian J Phys. 1996, 70B: p. 531-536.
- Rodriguez S, Lafuente C, Artigas H, et al., J Solut Chem. 1997, 26: p. 207–215.
- Gascon I, Pardo I, Santafe JM. Fluid Phase Equilibria. 2001, 180: p. 211-220.
- Yasmin M, Singh KP, Parveen S, et al., Acta Physica Polonica. 2009, 115: p. 890-900.
- Sathyanarayan B, Ranjith Kumar B, Savitha Jyostna T, et al., J Chem Thermodyn. 2007, 39: p. 16-21.
- Suindells JR, Godfray TB. J Res Natd Bur Stand. 1952, 48: p1.
- Nikam PS, Hasan M. J Chem Eng Data. 1998, 43: p. 732-737.
- Jacobson B. J Chem Phys. 1952, 20: p. 927.
- Jacobson B. Acta Chem Scand. 1952, 5: p. 1214.
- Vekatesu P, Chandra Sekhar G, Prabhakar Rao MV. Phys Chem Liqs. 2006, 44: p. 287-291.
- Fort RJ, Moore WR. Trans Faraday Soc. 1965, 61: p. 2102-2110.
- Yadava RR, Singh VN, Yadava SS. J Chem Eng Data. 1994, 39: p. 705-707.
- Suryanarayana CV and Kuppusami J. Indian J Acoust Soc.India. 1977, 5: p. 102.
- Suryanarayana CV and Kuppusami J. Indian J Acoust Soc.India. 1976, 4: p. 75.
- Thirumaran S and Earnest Jaya Kumar J. Ind J Pure & Appl Phys. 2009, 47: p. 265-272.
- Garcio B, Aparicio S, Navarro AM, et al., J Phys Chem B. 2004, 108: p. 15841-15850.
- Iloukhani H, Zoorasna N, Sloeimani R. Phys Chem Liq. 2005, 43: p. 391-401.
Indexed at, Google Scholar, Crossref