Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 3
In-Vitro Alpha-Amylase Inhibitor Activity and Antioxidant Activity of Actinobacteria Isolated from Marine Sediment Samples
Elizabeth Mathew, Haimanti Mondal, Priyanka Kharat, Purbasha Saha and KV Bhaskara Rao*KV Bhaskara Rao, Department of Biomedical Sciences, School of Bioscience and Technology, VIT University, Vellore, Tamil Nadu-632014, India, Email: dean.acad@vit.ac.in
Abstract
Marine environment is a diverse and untapped source of a wide variety of biologically active compounds. Among several organisms, marine actinomycete has been known to be a potential source for novel enzymes. Diabetes mellitus is a long term metabolic disorder characterized by hyperglycaemia. In the present study, marine actinobacteria isolated from the marine sediments of the coastal areas of Kothapattanam, Ongole, Andhra Pradesh, were used to screen for alpha-amylase inhibitor and antioxidant compounds, which are known to manage type 2 diabetes. A total of 20 actinobacteria were isolated from the marine sediments. Among the 20 isolates, an actinobacterial isolate designated VITMA1 showed 58.16% inhibition against alpha amylase enzyme. Further, VITMA1 also exhibited antioxidant activity. It showed 61.82% of DPPH radical scavenging activity, reducing power at 1.933 and a metal chelating activity of 20.33% respectively. The combined effect of antioxidant activity and alpha-amylase enzyme inhibition makes the secondary metabolite obtained from marine actinobacterial isolate, a potent anti-diabetic drug. Based on phenotypic characteristics, VITMA1 was identified upto the genus level as Streptomyces sp.
Keywords
Marine actinobacteria, Diabetes mellitus, Antioxidant, α-Amylase inhibitor
Introduction
Diabetes mellitus is considered as one of the major endocrine disorder which is characterized by chronic hyperglycaemia. Hyperglycaemia promotes auto-oxidation of glucose to form free radicals. Type 2 diabetes is the most predominant form of diabetes and affects more than 90% of the total diabetic patients [1]. This form of diabetes causes our body to not use the insulin properly and hence the blood glucose reaches to abnormal levels. Pancreatic alpha-amylase and alpha-glucosidase are the key enzymes present in the small intestine that plays a major role in digestion of starch yielding glucose and maltose and therefore, leading to the increase of postprandial glucose levels and decrease postprandial hyperglycaemia [2-4]. Alpha amylase converts starch into simple sugars which are utilized by the body. It catalyses the hydrolysis of (α-1,4) glycosidic linkages of various carbohydrate compounds such as starch, amylose, amylopectin, glycogen, and several others like malto-dextrins [5]. In diabetic patients low insulin level prevents the extracellular glucose to get clear quickly through the blood. Therefore diabetics’ treatment requires low α-amylase levels so that our blood glucose levels remains under control. Similarly, alpha glucosidase enzyme found in the brush border of enterocytes in the intestinal villi; cleave diasachharides and oligosachharides, increasing the blood glucose level [6-8]. Hence, the best way to manage type 2 diabetes is by reducing the starch digestion rate through inhibition of the enzymes alpha-amylase and alpha-glucosidase. Enzyme inhibitors are important biochemical tools which can be used for the study of several enzymatic structures and their mechanisms. They can be applied in fields such as medicine, agriculture as well as biotechnology i.e., they have the potential utility in the treatment of diseases. Alpha amylase inhibitors help in reduction of the glucose peaks which occur after consumption of a meal. Amylase inhibitors are considered as starch blockers as they results in reducing the conversion rate of the starch by inhibiting the activity of amylase.
The microbial origin for the enzyme inhibitors have very low molecular weight in comparison to that of both animal as well as plant inhibitors due to its origin from the hydrolysis of the macromolecular compounds [9]. Actinobacteria, which are known to be one of the important producers of antibiotics and suppliers to the pharmaceutical industry, produces a broad variety of secondary metabolites. These microbes are now gaining much importance for their taxonomic and ecological perspectives, and also for their ability to produce certain novel bioactive compounds. Actinobacteria are gram positive, aerobic, non-motile, with high Guanosine-Cytosine (GC) content in their DNA (70-80%) and are phylogenetically related to the bacteria based on the evidence of their 16S rRNA cataloguing studies. Bioactive compounds from several marine actinomycetes have become a remarkable source of drugs for potential targets [10]. These bioactive compounds are considered as the secondary metabolite which enhances the survival of fitness under extreme conditions in the ocean and have the potentiality to serve chemical weapons against specified targets [11]. The conditions of marine environments are totally discrete from terrestrial environment. Thus, most marine actinobacteria are diverse genetically and metabolically with several distinct features resulting in the production of a variety bioactive compound. These secondary metabolites from marine actinomycetes have become the main subject of commercial interest. They have been the prime target of screening programs in several researches due to their potential to produce a diverse range of novel secondary metabolites and other molecules [12]. It is anticipated that isolation and characterization of the marine actinomycetes could be useful in the discovery and develpoment of novel bioactive compounds. Imada and Simidu reported the α-amylase enzyme inhibitor producing marine Streptanyces corchorushii subsp. rhodomarinus subsp.nov [13]. Several other reports have been made on the production of alpha amylase inhibitor from the genera Streptomyces [14-17].
Oxidative stress induced by free radicals is also considered as one of the causative factors for type 2 diabetes. Therefore antioxidant compounds play a major role for the regulation of the oxidative stress related diabetes by scavenging free radicals. Antioxidants are compounds that remove free radicals, and delays or inhibit the oxidation of nucleotides, lipids and proteins. Thus, they can prevent damage to the body cells done by reactive oxygen speices [7,8].
Taking this into account, the present study is aimed at isolating and screening of amylase inhibitory activity for marine actinobacteria and also performing several antioxidant assays to develope a novel drug for the treatment of type 2 diabetes.
Materials and Methods
Collection of samples
For the purpose of sample collection, marine sediment samples were obtained aseptically from the coastal areas of Kothapattanam, Ongole, Andhra Pradesh (15.5057ºN, 80.05ºE). The samples were kept in sterile bags and transported to the laboratory under low temperature conditions for further use.
Isolation of marine actinobacteria
Marine sediment samples were used for the isolation of actinobacteria. Sample was serially diluted followed by plating on Starch Casein Agar plates (composed of soluble starch: 10 g, K2HPO4: 2 g, KNO3: 2 g, casein: 0.3 g, MgSO4.7H2O: 0.05 g, CaCO3: 0.02 g, FeSO4.7H2O: 0.01 g, sea water: 500 ml, distilled water 500 ml at pH: 7.0 ± 0.1). The petriplates were then incubated at 28ºC for 7- 10 days.
Isolates were repeatedly sub cultured on Starch Casein Agar slants for maintaining pure cultures of the isolates.
Preparation of crude extract
The potential isolates were inoculated in Soluble Starch broth (SS broth) (composed of Starch: 25 g, Glucose: 10 g, Yeast: 2 g, trace salts: 1 ml, CaCO3: 3 g, Sea Water: 500 ml, Distilled Water: 500 ml) followed by incubating in rotary shaker (110 rpm) for 7 days at 28ºC. After 7 days, fermented broth was centrifuged at 10,000 rpm for 10 min. After centrifugation, supernatant was collected and extracted with equal amount of ethyl acetate. The crude extract thus obtained was then dried to obtain the powder form of the extract.
A stock concentration of the extract was prepared at 100 mg/ml to carry out further assays.
Alpha-amylase inhibition assay
Alpha amylase enzyme solution (0.5 mg ml−1) was prepared in sodium phosphate buffer (0.02 M). The enzyme solution (500 μl) was then mixed with the actinobacterial crude extract (500 μl) and incubated for 10 min at 25ºC. Then, 1% starch solution was prepared in sodium phosphate buffer (0.02 M). Starch solution was added to each of the test tubes and incubated again at 25ºC for 10 min. The ongoing reactions in the test tubes were then stopped by addition of 1 ml dinitrosalicylic acid. Incubation was done by keeping in boiling water bath for 5 min and then cooled to room temperature. The reaction mixture was subsequently diluted with distilled water (10 ml). The absorbance of the solution in each test tube was measured at 540 nm. The presence of starch in the reaction mixture is indicated by a dark blue colour. The colour changes to yellow in the absence of starch.
The percentage of alpha-amylase inhibition was calculated by the following formula:
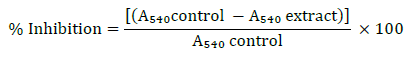
Antioxidant assays
DPPH radical scavenging assay
Broth culture of the isolates was first centrifuged at 5000 rpm for 10 min. The resulting supernatant was collected for DPPH radical scavenging assay. In each of the test tubes, 1 ml of supernatant and 2 ml of DPPH reagent (1,1-Diphenyl-2-picrylhydrazyl) were added under dark conditions. The test tubes were then incubated for 30 min at room temperature in dark. DPPH reagent is a stable free radical and when scavenged turns from red to yellow colour. When antioxidant activity is present it reduces DPPH to DPPH-H and as a result the absorbance decreases. After incubation, absorbances of the samples were measured at 517 nm. The absorbance of the control was also noted [18].
The percentage of radical scavenging activity of the extract was calculated by the following equation:
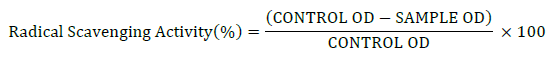
Reducing power assay
Assay was performed by dissolving 0.1 ml of the samples in 1.0 ml of phosphate buffer in separate test tubes and was mixed well with 1.0 ml of 1% potassium ferricyanide. The tubes were then incubated at 50ºC for 20 min in water bath. The tubes were then cooled rapidly and mixed with 1.0 ml of trichloroacetic acid, and 1.0 ml of milliQ water. Finally, 0.1 ml of 0.1% ferric chloride was mixed to each tube. After 10 min of incubation period, absorbance of the reaction mixtures were measured at 700 nm. The change in colour to green indicates a positive test. Substances having reduction potential react with potassium ferricyanide to form potassium ferrocyanide. Potassium ferrocyanide reacts with ferric chloride to form ferric-ferrous complex. The resulting reaction mixture has an absorption maximum at 700 nm [19,20].
Metal chelating assay
For this assay, the chelating effect on ferrous ion was estimated. Actinobacterial extract (50 μl) was dissolved in 1.6 ml of milliQ water. To each test tube 0.05 ml of FeCl₂ was added. To this mixture 0.1 ml of 5 mM ferrozine was added to initiate the reaction. The test tubes were then shaken vigorously and left standing at 40ºC for 10 min. The absorbance of the reaction mixture was then measured at 562 nm using a spectrophotometer. Ferrozine quantitatively chelates Fe2+ and form a red coloured complex. Compounds with metal chelating ability compete with ferrozine for the ferrous ions. They then form a coordinate complex with the metal ions and inhibit transfer of the electrons. Thereby, oxidation is prevented and no free radicals are formed [19,21].
The ferrous ion chelating ability was calculated by the following equation
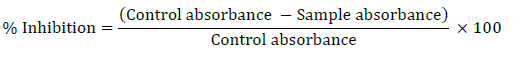
Phenotypic characterization of VITMA1
Morphological characterization is an important characteristic in the classification of actinomycetes. For this purpose some basic tests such as, gram character, aerial mass colour, reverse side pigments, spore chain morphology under light microscope, spore surface morphology under scanning electron microscope were performed.
Results
Isolation of marine actinomycete
A sum of 20 actinobacterial isolates was obtained from the marine sediments of the coastal areas of Kothapattanam, Ongole, Andhra Pradesh (15.5057ºN, 80.05ºE). The isolates were selected on the basis of distinct morphological characteristics.
Alpha-amylase inhibition assay
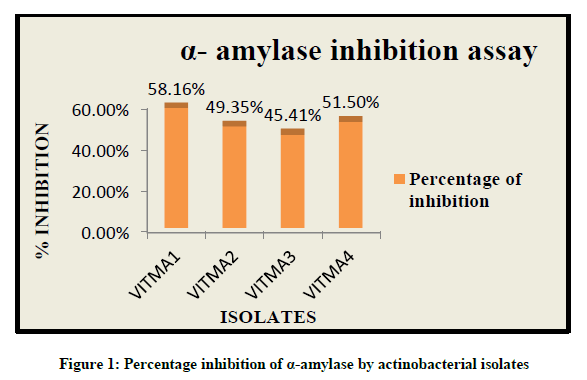
Alpha amylase inhibitory activity can be measured by the amount of starch in presence of alpha amylase enzyme. The reduced intensity of blue colour in the reaction mixture indicates the hydrolysis of starch into mono-saccharides by alpha amylase enzyme. While increased intensity of blue colour indicates the presence of starch due to the presence of alpha amylase inhibitor activity. Thus it was observed that out of all the isolates, VITMA1 showed higher percentage of alpha amylase inhibitory activity of 58.16%, while VITMA2, VITMA3 and VITMA4 showed 45.41%, 51.50% and 49.35% respectively (Figure 1).
Antioxidant assays
DPPH radical scavenging assay
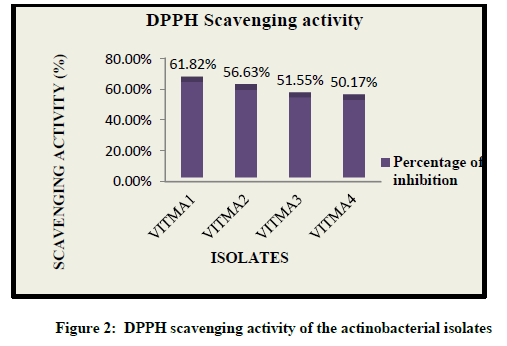
The radical scavenging activity of different extract was determined for particular isolates. The degree of discoloration of the reaction mixture indicates the scavenging potential of the extracts and its ability to donate hydrogen. Out of all the isolates, VITMA1 showed the highest scavenging activity of 61.82%, while VITMA2, VITMA3, and VITMA4 showed 56.63%, 51.55% and 50.17% scavenging activity respectively (Figure 2).
Reducing power assay
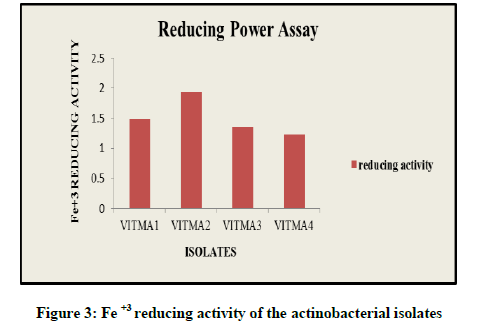
This assay is an indicator for the antioxidant activity. Increase in the intensity of green colour indicates efficient reducing power of the sample. Thus in reducing power assay, VITMA2 showed the highest reducing power of 1.933, while VITMA4, VITMA3, and VITMA1 showed reducing power results of 1.231, 1.362, and 1.487 respectively (Figure 3).
Metal chelating assay
The metal chelating activity of the ethyl acetate extracts is reflected in the decreasing intensity of red colour of the reaction mixture. The highest chelating activity was showed by VITMA3 at 46.23%, while VITMA1, VITMA2 and VITMA4 showed 20.33%, 21.62% and 22.23% respectively (Figure 4).

Phenotypic characterization of VITMA1
The potent isolate VITMA1 was a gram positive, non-motile, aerobic actinobacteria. The colonies were white in colour and raised when grown on starch caesin agar. It showed spiral spore chain morphology when observed under light microscope. SEM analysis revealed the spore surface morphology of VITMA1 which was found to be smooth and individual mature spores were 2 μm in diameter (Figure 5). There was absence of any diffusible pigments. Based on these phenotypic characteristics, VITMA1 was tentatively identified as belonging to the genus of Streptomyces sp.
Discussion
The drugs which are responsible for lowering the glucose levels are inhibitors of α-amylase and α-glucosidase enzymes. These enzymes catalyze the breakdown of complex carbohydrates into simple absorbable monosaccharides [22]. Enzyme inhibitors delay the absorption of ingested carbohydrates and thus are involved in the reduction of the increased blood glucose level. Hence, investigation of such kinds of agents from new and unexplored sources has become beneficent. Previously, numerous researches have focused on alpha amylase inhibitors from microbial origin, but relatively less has been studied about marine actinobacteria as a source of antidiabetic enzyme inhibitors. Therefore in this present study we investigated the inhibitory effect of the marine actinobacterial extracts on α-amylase. The crude extracts obtained with ethyl acetate solvent were screened for the enzyme inhibitory activity, which demonstrated that VITMA1 gave the higher inhibitory activity against alpha amylase enzyme. This helps us to include that it may have certain potential to manage disorders related to type 2 diabetes and the purified inhibitor could be of some use as an indicator for the combinational therapy. Marine actinomycetes producing enzyme inhibitor have been reported earlier in other assays [13,23-28].
Revathy et al. reported the alpha-amylase inhibitory potential activity of Streptomyces sp. for VITMSS05 strain, being isolated from Marakkanam, southern coast of India which reported that the extract gave 64.1% inhibition on α-amylase at 100 μg/ml [18]. Another research reported of an actinomycete Micromonospora sp. showing 74.32% inhibition against alpha amylase. In the present study, ethyl acetate extract of VITMA1 showed 58.16% inhibitory activity against α-amylase indicating that it has the potential to be used as antidiabetic drug [29].
Further, the actinobacterial extracts also possessed antioxidant activity. Antioxidant activity was confirmed by in vitro assays including, DPPH radical scavenging assay, reducing power assay and metal chelating assay. Out of the four potent isolates which showed alpha amylase inhibitory activity, VITMA1 scavenged 61.82% of DPPH radical. Earlier studies have reported DPPH radical scavenging activity from marine actinomycets. Sourav and Kannabiran reported 44.13% radical scavenging activity by Streptomyces sp. [30]. Similarly, antioxidants herbimycin A and dihydroherbimycin A produced by Streptomyces sp. exhibited 61% and 72% radical scavenging activity [31]. Highest reducing power activity was shown by the isolate VITMA2 at 1.933. While Vanamthi et al. reported a reducing power activity by Streptomyces at 0.16 [32]. In the case of metal chelating activity, VITMA3 showed 46.23% activity which was comparable to a previous paper which reported [33] that the marine actinomycetes Streptomyces sp. isolated from the marine sediments showed 51% metal chelating activity.
VITMA1 showed the maximum alpha amylase activity and thus was chosen for further identification. Based on the observed phenotypic characteristics, it was identified to be belonging to the genus of Streptomyces sp.
Conclusion
The present study finding suggests that the tested isolates produce antidiabetic compound as well as possesses antioxidant activity. From the results, it has been confirmed that the actinobacterial isolate VITMA1 produces potent α-amylase inhibitor and possess highest DPPH scavenging activity. Thus, VITMA1 may be used as a source to obtain novel antidiabetic compounds which can be used to treat Type 2 diabetes. Further, VITMA1 was tentatitvely identified to be belonging to the genus of Streptomyces sp. Future prospects of the study involve identification of the antidiabetic compound. Finally, by structural and in vivo studies, these compounds can be successfully used for new drug development.
Acknowledgment
The authors are very much grateful to the management and staff of VIT University Vellore, Tamil Nadu, India for supporting this study.
References
[1] S.M. Lakshmi, K.S.S. Rani, T.U.K. Reddy, Asian. J. Pharmaceut. Clin. Res., 2012, 5, 15.
[2] E.S. El Ashry, N. Rashed, A.H. Shobier, Pharmazie., 2000, 55, 251.
[3] O.L. Franco, D.J. Rigden, F.R. Melo, M.F. Grossi-de-sa, Eur. J. Biochem., 2002, 269, 397.
[4] M.A. Jayasri, A. Radha, T.L. Mathew, J. Herb. Med. Toxicol., 2009, 3, 91.
[5] R. Heidari, S. Zareae, M. Heidarizadeh, Pak. J. Nutr., 2005, 4, 101.
[6] A.A. Moneim, H. Fayez, Int. J. Bioassays., 2015, 4, 4002.
[7] S. Tachakittirungrod, S. Okonogi, S Chowwanapoonpohn. Food. Chem., 2007, 103, 381.
[8] H. Nasri, H. Shirzad, M. Rafieian-kopaei, J. Res. Med. Sci., 2015, 20, 1112.
[9] K. Alagesan, P.K. Raghupathi, S. Sankarnarayanan, Int. J. Pharm. Life. Sci., 2012, 3, 1407.
[10] H.P. Fiedler, C. Bruntner, A.T. Bull, A.C. Ward, M. Goodfellow, O. Potterat, C. Puder, G. Mihm, Antonie van Leeuwenhoek., 2005, 87, 37.
[11] P. Proksch, R.A. Edrada, R. Ebel, Appl. Microbiol. Biotechnol., 2002, 59, 125.
[12] K.S. Lam, Curr. Opin. Microbiol., 2006, 9, 245.
[13] C. Imada, U. Simidu, Nippon Suisan Gakkaishi., 1988, 54, 1839.
[14] A.A. Kulkarni-Almeida, M.K. Brahma, P. Padmanabhan, P.D. Mishra, R.R. Parab, N.V. Gaikwad, C.S. Thakkar, P. Tokdar, P.V. Ranadive, A.S. Nair, A.A. Damre, U.A. Bahirat, N.J. Deshmukh, L.S. Doshi, A.V. Dixit, S.D. George, R.A. Vishwakarma, K.V.S. Nemmani, G.B. Mahajan, AMB Express., 2011, 1, 42.
[15] J.V. Akshatha, M.S. Nalini, C.D. Souza, H.S. Prakash, Lett. Appl. Microbiol., 2013, 58, 433.
[16] P. Sanjebam, M. Thenmozhi, K. Kannabiran, Int. J. Pharm. Res. Rev., 2013, 2, 5.
[17] Y. Yang, L. Yu, H. Komaki, N. Oku, Y. Igarashi, The J. Antibiot., 2016, 69, 69.
[18] T. Revathy, M.A. Jayasri, K. Suthindhiran, Am. J. Biochem. Biotechnol., 2013, 9, 282.
[19] L. Karthik, G. Kumar, K.V.B. Rao, Asian Pacific J. Tropical Med., 2013, 6, 325.
[20] L.W. Chang, W.J. Yen, S.C. Huang, P.D. Duh, Food. Chem., 2002, 78, 347.
[21] G.R. Zhao, Z.J. Xing, T.X. Ye, Y.J. Yuan, Z.X. Guo, Food. Chem., 2006, 99, 767.
[22] A.R. Stuart, E.A. Gulve, M. Wang, Chem. Rev., 2004, 104, 1255.
[23] H Sugino; A Kakinuma. J. Biol Chem., 1978, 253, 1546.
[24] S. Sakuda, A. Isogai, S. Matsumoto, A. Suzuki, K. Koseki, Tetrahedron Lett., 1986, 27, 2475.
[25] C. Imada, U. Simidu, Nippon Suisan Gakkaishi., 1992, 58, 2169.
[26] T. Aoyama, F. Kojima, C. Imada, Y. Muraoka, H Naganawa, Y Okami, T Takeuchi, T Aoyagi, J. Enzy. Inhibit., 1995, 8, 223.
[27] C. Imada, Y. Okami, J. Mar. Biotechnol., 1995, 2, 109.
[28] C. Imada, Antonie van Leeuwenhoek, 2005, 87, 59.
[29] K.R. Suthindhiran, M.A. Jayasri, K. Kannabiran, Int. J. Integrative. Biol., 2009, 6, 115.
[30] K. Sourav, K. Kannabiran, Saudi J. Biol. Sci., 2012, 19, 81.
[31] H.B. Chang, J.H. Kim, Biotechnol. Lett., 2007, 29, 599.
[32] K. Vanmathi, K. Saraswathi, J. Jayabharath, P. Arumugam, Int. J. Appl. Sci. Eng. Res., 2016, 5, 107.
[33] M. Thenmozhi, K. Kannabiran, Oxidants and Antioxidants Medical Science., 2012, 1, 51



