Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 3
In-Vitro Cytotoxic Effect of Linderina madayiparense Extracts against Human Lung Carcinoma Cell Lines (A-549)
Umakrithika Chiranjeevi1*, Anandarajagopal Kalusalingam2, Ajaykumar Thankakan Vimala1 and Kannan Kamarajan12Department of Pharmaceutical Chemistry, Arulmigu Kalasalingam College of Pharmacy, Krishnankoil-626190, Tamil Nadu, India
Umakrithika Chiranjeevi, Department of Pharmacy, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India, Email: scgrcdde@gmail.com
Abstract
Lung cancer is one of the most lethal worldwide and has a dismal prognosis. This disease has become important as a public health concern. Conventional radio-chemotherapies cause adverse side effects ranging from gastro-intestinal complications to bone marrow failure. The inadequate availability of efficient and safe treatment for the cancer intends the scientific researchers to focus the phytochemicals derived from medicinal plants. Lindernia madayiparense (Family: Linderniaceae) is widely distributed in the laterite hills, Kerala, India. Petroleum ether, ethyl acetate and ethanol extracts of L. madayiparense were prepared by continuous hot extraction method using Soxhlet apparatus. All the extracts were assessed for their in-vitro cytotoxic effect against Human lung carcinoma cell lines (A-549 cell line) by MTT assay. The maximum cytotoxic effect (90.96%) was observed by ethyl acetate extract of L. madayiparense at a concentration of 1000 μg/ml against the A-549 cell lines followed by pet ether extract and ethanol extract. The CTC50 value for ethyl acetate extract of L. madayiparense was found to be 594.81 μg/ml.
Keywords
Linderina madayiparense, Cytotoxic, Human lung carcinoma, MTT assay
Introduction
Cancer is the leading cause of death, and the prevalence of cancer continues to go up even though advances in earlier diagnosis and clinical intervention. Chemotherapy is one of the commonly-used strategies for the treatment of cancer. Other standard cancer therapies involved is surgery to remove the major tumor masses and subsequent radiotherapy to kill potential cancer cells present remaining and to prevent cancer recurrence [1]. But the lack of specificity is the main drawback of these conventional radio-chemotherapies which cannot discriminate cancer cells from normal cells, resulting in the substantial death of normal cells, and causes adverse side effects ranging from gastro-intestinal complications such as nausea, vomiting, appetite loss and ulceration, mucositis, necrosis to bone marrow failure [2]. Moreover, they complicate the therapies and thereby affect the prognosis and survival of cancer patient. It may affect the survival and quality of the life of the cancer patients. Therefore, the ideal anticancer therapeutics should exclusively target cancer cells while sparing normal cells.
Among all types of cancers, lung cancer is one of the most lethal worldwide and has a dismal prognosis. It is the most common source of brain metastases in adult patients, which occurs in 30-50% of total lung cancer cases. Lung cancer can be divided into two types, small-cell lung cancer and non–small-cell lung cancer. Non-small-cell lung cancer is the most common type of lung cancer which accounting for 80% of total lung cancer cases [3]. The inadequate availability of efficient and safe treatment for the cancer invites the scientific researchers to intend the development of novel therapeutic agents from the plants for the treatment of lung cancer.
Since, human lives on earth, plants have been the primary source of medicines for the treatment of various ailments [4]. For many years herbal medicines have been used and are still used in developing countries such as India as the primary source of medical treatment.
Plant-derived drugs are preferred for health care, prevention and the treatment of various diseases and ailments as they are natural and readily available. They can be easily administered orally as part of patient’s dietary intake. Phytochemicals and their derivatives have a key role for the development of anticancer drugs. Also, being naturally derived compounds from plants are generally more tolerated and non-toxic to normal human cells [5].
India is one of the countries with rich biodiversity and massive treasure of herbal plants. Many indigenous systems such as Ayurveda, Yoga and Siddha are more prominent and prevailing in India from decades [6]. Consequently, lot of researches are conducting by experimental studies to demonstrate the epidemiological evidence of some plants as a whole, their parts or identified ingredients with specific biological properties which have substantial protective effects on human carcinogenesis. The use of medicinal herbs is still a tradition adopt by ethnic communities who are living in undulating plains and at foothills of dense forest and some of the traditional medical practitioner [7].
Lindernia madayiparense (Family: Linderniaceae) is a plant discovered first from the laterite hills, Madayipara, Kannur District, Northern Kerala, India. The Plant is used by the tribal inhabitants and local villagers for therapeutic purposes since long time [8]. But there is no scientific evidence for the pharmacological effects of this plant. Therefore, a detailed phytopharmacological study is warranted to explore the major active compounds and related bioactivities of this plant. Hence, the present study was undertaken for first time to explore the in-vitro cytotoxic effect of the extracts of the plant, L. madayiparense against human lung cancer cell lines (A-549 cell line).
Materials and Methods
Collection and identification of plant materials
Wild crafted plant, Linderina madayiparense was collected during its flowering season in the month of October to December, 2013 in Kannur District, Kerala, India. The plant material was identified and authenticated by botanist Mr. P. Biju, Assistant Professor, Government College, Kasaragod, Kerala, India.
Preparation of plant extracts
Various extracts of Linderina madayiparense were prepared by continuous hot extraction method using Soxhlet apparatus [9]. The whole plant, Linderina madayiparense was washed thoroughly with distilled water and dried under air-shade and pulverized mechanically into coarse powder. The coarse powder (1000 g) has been successively extracted with three different solvents by changing the solvent polarity from non-polar to polar solvents such as petroleum ether, ethyl acetate and ethanol respectively. At the end of each successive extraction, the mixture was collected and concentrated using rotary vacuum evaporator under reduced pressure to remove the excessive solvents. All the extracts obtained were freeze dried to get an absolute extracts and stored properly and preserved in 8ºC until further use. The % yield of all the extracts was calculated.
Preliminary phytochemical studies
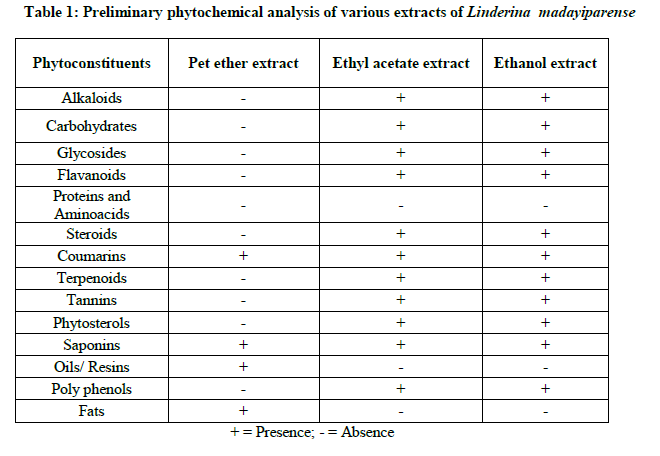
The various extracts of Linderina madayiparense were subjected for preliminary qualitative analysis to identify the presence of phytochemical constituents such as alkaloids, carbohydrates, glycosides, flavonoids, steriods, triterpenoids, phenols, proteins, tannins etc. as per the standard methods [10]. The presence and absence of the secondary metabolites in the extracts was noted in Table 1.
Evaluation for cytotoxic activity
Chemicals
All the chemicals used were analytical grade. 3-(4,5–dimethyl thiazol–2–yl)–5–diphenyl tetrazolium bromide (MTT), Fetal Bovine serum (FBS), Phosphate Buffered Saline (PBS), Dulbecco's Modified Eagle Medium (DMEM) and Trypsin (Sigma Aldrich); EDTA, Glucose and Antibiotics (Hi-Media); Dimethyl sulfoxide (DMSO) and Propanol (E. Merck) were used.
Cell lines
Human lung carcinoma cell lines (A-549 cell line) were used in this study and procured from National Centre for Cell Sciences (NCCS), Pune, India.
Culture medium
Stock cells of A-549 cell line were cultured in DMEM and supplemented with 10% inactivated FBS, penicillin (100 IU/ml), streptomycin (100 μg/ml) and amphotericin B (5 μg/ml) in a humidified atmosphere of 5% CO2 at 37°C until confluent. The cells were dissociated with TPVG solution (0.2% trypsin, 0.02% EDTA, 0.05% glucose in PBS). 25 cm2 culture flasks were used to grow the stock cultures and all experiments were carried out in 96 microtitre plates (Tarsons India Pvt. Ltd., Kolkata, India).
Preparation of test solutions
Each weighed pet ether, ethyl acetate and ethanol extracts were separately dissolved in distilled DMSO and the volume was made up with DMEM supplemented with 2% inactivated FBS to obtain a stock solution of 1000 μg/ml concentration and sterilized by filtration. From this stock solution, four different lower dilutions (500, 250, 125 and 62.5 μg/ml) were prepared [11,12].
Determination of cell viability by MTT Assay
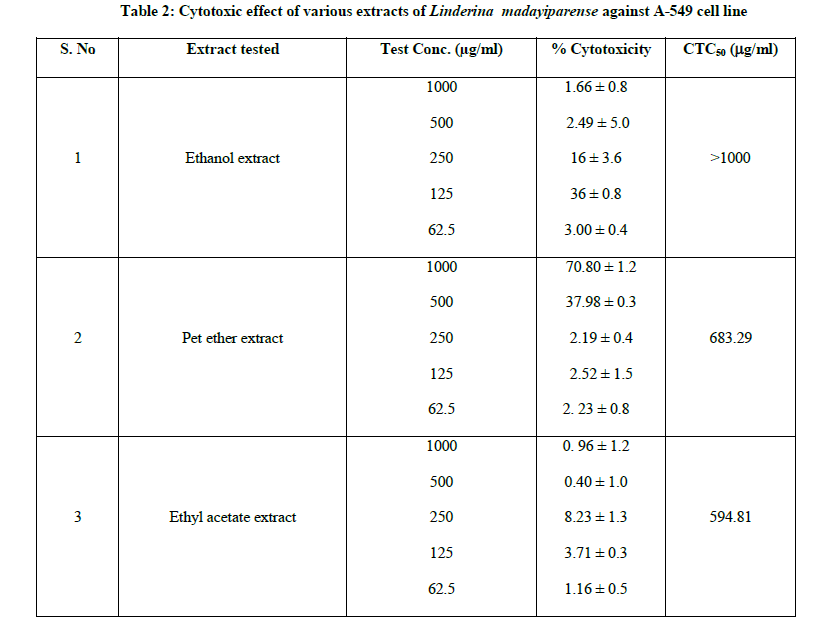
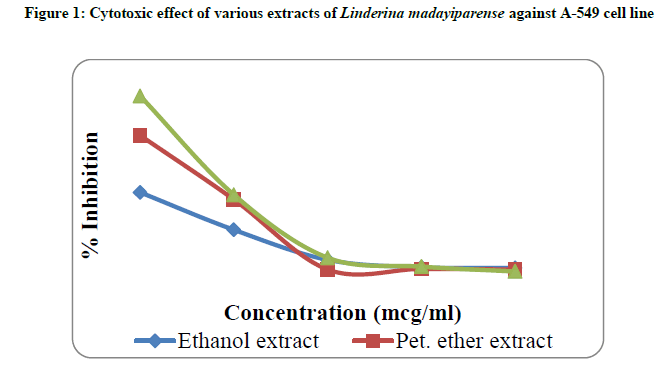
The monolayer cell culture was trypsinized and the cell count was adjusted to 1.0 × 105 cells/ml using DMEM containing 10% FBS. 0.1 ml of the diluted cell suspension (approximately 10,000 cells) was added to each well of the 96 well microtitre plates. After 24 h, when a partial monolayer was formed, the supernatant was flicked off and washed the monolayer once with medium. To this, 100 μl of different test concentrations of pet ether, ethyl acetate and ethanol extracts of L. madayiparense were added respectively on to the partial monolayer in microtitre plates. The plates were then incubated at 37ºC for 3 days in 5% CO2 atmosphere, and microscopic examination was carried out and observations were noted every 24 h interval. After 72 h, the test substance in the wells was discarded and 50 μl of MTT in PBS was added to each well. The plates were gently shaken and incubated for 3 h at 37ºC in 5% CO2 atmosphere. The supernatant was removed and 100 μl of propanol was added. All the plates were gently shaken to solubilize the formed formazan. Then the absorbance was measured using a microplate reader at a wavelength of 540 nm using UV-visible spectrophotometer. The percentage growth inhibition was calculated using the following formula and recorded (Table 2). The concentration of the extract needed to inhibit the cell growth by 50% (CTC50 value) was determined from the dose-response curve (Figure 1) for the cell line [11-14].
% Growth Inhibition = 100 – Mean OD of individual test group/ Mean OD of control group × 100
Results
The percentage yield of pet ether, ethyl acetate and ethanol extracts of Linderina madayiparense were ranged from 4.78%-26.40%. Among these three extracts obtained ethanol extract had the highest percentage yield (26.40%) followed by ethyl acetate extract (15.46%) whereas pet ether extract had the lowest percentage yield (4.78%).
Phytochemical tests revealed that the presence of coumarins and saponins in all the three extracts while the absence of proteins and amino acids were found in these all extracts. Oils and fats were oresent only in pet ether extracts. Alkaloids, carbohydrates and glycosides, flavonoids, steroids, terpenoids, tannins, and phytosterols are present in both ethyl acetate and ethanol extracts of L. madayiparense (Table 1).
The in-vitro cytotoxicity effect of all the extracts of Linderina madayiparense was studied on Human lung carcinoma cell lines (A-549) by MTT assay method. The cytotoxic effect of pet ether, ethyl acetate and ethanol extracts of L. madayiparense was shown in Table 2 and Figure 1. The maximum cytotoxic effect (90.96%) was observed by ethyl acetate extract at a concentration of 1000 μg/ml against A-549 (Human lung carcinoma cell line) cell line followed by pet ether extract and ethanol extract (Table 2). CTC50 value for ethyl acetate extract of L. madayiparense was found to be 594.81 μg/ml (Figure 1).
Discussions
Pet ether, ethyl acetate and ethanol extracts of Linderina madayiparense were obtained by continuous hot extraction method using Soxhlet extractor. The type of solvents used for the extraction procedure plays a key role to obtain a maximum quantity of extract because every phytochemical constituent has its own solubility competency in different solvent [15]. Therefore, the order of increasing polarity of solvents used in the present study is pet ether, ethyl acetate and ethanol.
The in-vitro cytotoxic effect of the various extracts of Linderina madayiparense was determined by 3-(4,5-dimethyl thiazol-2-yl)- 2-4-diphenyl tetrazolium bromide (MTT) assay. This colorimetric assay is based on the capacity of the enzyme, mitochondria succinate dehydrogenase in living cells to reduce the yellow water soluble substrate, 3-(4, 5-dimethyl thiazol- 2-yl)-2,5-diphenyl tetrazolium bromide (MTT) into an insoluble, colored formazan product which is measured spectrophotometrically. The ability of the cells to survive a toxic insult is the basis of most cytotoxic assays. This assay is based on the assumption that dead cells or their products do not reduce tetrazolium. The assay depends on both the mitochondrial activity per cell and number of cells present. The cleavage of 3-(4, 5 dimethyl thiazole-2yl)-2, 5-diphenyl tetrazolium bromide (MTT) to a blue formazan derivative by living cells is clearly a very effective principle on which the assay is based. The reduction of MTT can only take place in metabolically active cells and the level of activity is a measure of the viability of the cells. The amount of cells was found to be proportional to the extent of formazan production by the cells used. MTT reduction as a cell viability measurement is now extensively chosen as the most advantageous end point to assess the cytotoxicity [16,17].
The result reveals that the ethyl acetate extract of the plant, Linderina madayiparense effectively inhibits the A-549 cell line. It might be due to the cytotoxic action of the phytoconstituent(s) present in the ethyl acetate extract of Linderina madayiparense. The cytotoxic selective activity of the phytoconstituent might depend on the type of chemical composition and their concentration as well as the type and developmental stage of the cancer [18]. The findings suggest that A-549 cell line is susceptible due to the presence of cytotoxic phytoconstituent(s) in the plant, Linderina madayiparense. The present study demonstrates first time for the cytotoxic potential of the plant, Linderina madayiparense extract against human lung carcinoma cell lines (A-549 cell line).
Conclusion
From the present findings, it can be concluded that the plant, Linderina madayiparense showed cytotoxic effect against Human lung carcinoma cell lines (A-549 cell line). Further investigations are necessary to assess the cytotoxic effect on animal models and also to identify the secondary metabolites which might be responsible for the cytotoxic activity of this plant. The isolation and characterization of the specific bioactive principle is in process to exploit the potential therapeutic uses of the plant, Linderina madayiparense.
Acknowledgment
The authors are grateful to the Management and Department of Pharmacy, Annamalai University for their continuous encouragement and providing necessary facilities. Authors are also thankful to Mr. P. Biju, Assistant Professor, Government College, Kasaragod, Kerala for his support during the collection and identification of the selected plant.
References
[1] J.S. Park, C.J. Lim, O.S. Bang, N.S. Kim, BMC Compl. Altern. Med., 2016, 16, 115.
[2] P. Graidist, M. Martla, Y. Sukpondma, Nutrients., 2015, 7, 2707.
[3] Y.C. Chen, Y.H. Kuo, N.C. Yang, C.W. Liu, W.T. Chang, C.L. Hsu, J. Chin. Med. Assoc., 2014, 77, 535.
[4] N.A. Jaradat, R. Ramahi, A.N. Zaid, O.I. Ayesh, A.M. Eid, BMC. Compl. Altern. Med., 2016, 16, 93.
[5] S. Sarojini, V. Ramesh, P. Senthilkumaar, Int. J. Adv. Sci. Res., 2016; 2(7), 139.
[6] A.D.B. Vaidya, T.P.A. Devasagayam, J. Clin. Biochem. Nutr., 2007, 41, 1-11.
[7] D. Savita, A. Huma, J. Cancer Ther., 2010, 1, 87.
[8] M.K.R. Narayanan, C.N. Sunil, M.K. Nandakumar, K.A. Sujana, J.P. Joseph, N. Anilkumar, Int. J. Pantl. An. Env. Sci., 2012, 2(3), 59.
[9] P.A. Egwaikhide, C.E. Gimba, Middle-East. J. Sci. Res., 2007, 2 (3-4), 135.
[10] M. Rajan, V. Kishorkumar, P. Satheeshkumar, T. Venkatachalam, V. Anbarasan, Int. J. Pharmacog. Phytochem. Res., 2011, 3(3), 47.
[11] J. Pan, J. Liu, G. He, Pract. J. Cancer., 2009, 24(5), 441.
[12] N. Manglani, S. Vaishnava, P. Dhamodaran, S. Hemant, Int. J. Phar. Pharm. Sci., 2014, 6(3), 70.
[13] R. Archana, B. Shabana, Eur. J. Pharm. Med. Res., 2016, 3(10), 337.
[14] L.L. Ramesh, S.A. Basavarajeshwari, Int. J. Curr Microbiol. Appl. Sci., 2016, 5(8), 414.
[15] R.S.M. Abdel, S.A.A. Ellatif, S.F. Deraz, A.A. Khalil, Afr. J. Biotechnol., 2011, 10(52): 10733-10743.
[16] K. Khairunnisa, D. Karthik, J. App. Pharm. Sci., 2014, 4(8), 11.
[17] S.I.A. Wahab, A.B. Abdul, S.M. Mohan, A.S.A. Zubairi, M.M. Elhassan, Res. J. Biol. Sci., 2009, 4, 209.
[18] M. Suganya, M. Megala, Rubalakshmi, Int. J. Rec. Sci. Res., 2016, 7(5), 10954.



