Research - Der Pharma Chemica ( 2022) Volume 14, Issue 1
Isolation of Antispasmodic Active Constituent from the N-Butanol Portion of the Leaf Extract of Stachytapheta Angustifolia (Verbenaceae)
Musa Mohammed*Musa Mohammed, Department of Chemistry, Faculty of Science, Ibrahim Badamasi Babangida University, Lapai Niger State, Nigeria, Email: contactdrmusa@gmail.com
Received: 23-Dec-2021, Manuscript No. dpc-21-50521; Accepted Date: Dec 25, 2021 ; Editor assigned: 25-Dec-2021, Pre QC No. dpc-21-50521; Reviewed: 11-Jan-2022, QC No. dpc-21-50521; Revised: 17-Jan-2022, Manuscript No. dpc-21-50521; Published: 22-Jan-2022, DOI: 10.4172/0975-413X.14.1.22-31
Abstract
Extraction of the leaf extract of S. angustifolia followed by an extensive column chromatography of n-butanol portion of the extract on silica gel, purification over sephadex LH – 20 and subsequently (PTLC) was conducted using standard chromatographic and spectroscopic protocols. A bioactive constituent (compound S) from the n-butanol portion of the leaf extract of S. angustifolia was isolated. The structure was elucidated mainly by using IR, FTIR and combination of 400MHZ and 100MHZ 1D and 2D NMR techniques. Thus compound S was determined as 2'-O-p-hydroxybenzoyl-6'-O-trans- Caffeoyl-8-epiloganic acid. The Antispasmodic studies of the various partition portion of the leaf extract were conducted using rabbit jejunum. The n-butanol leaf portion of the extract produced a concentration dependent contraction of the rabbit jejunum (0.04mg/ml-0.32mg/ml). The contraction of the leaf extract at (0.032mg/ml) was blocked or antagonized by atropine at (0.1mg/ml). The preliminary phytochemical screening of the partition portion of the leaf extract conducted reveals the presence of Tannins, Glycosides, Flavonoids, Saponin, Steroids, Alkaloids, and Terpenoids.
Keywords
S. angustifolia; Leaf; Antispasmodic; Phytochemistry; Verbenaceae; Spectral data (1D and 2D)
Introduction
Medicinal plants are believed to be important sources of new chemical substances with potential therapeutic effect [1]. The research into plants with alleged various folkloric uses in the cure of various ailments should therefore be viewed as a fruitful and logical for the treatment of various ailments [2]. The World Health Organization (2005) recently recognized many herbal remedies and hence encouraged the developing countries to incorporate the use of folkloric plants for obvious reasons of their availability and affordability apart from being effective alternative to allopathic practice [3,4]. In Nigeria today, ethno medical treatment using herbal remedies has succeeded in the treatment of diseases like diabetics, mental disorder, breast cancer, sickle cell anemia and other forms of viral infections [5].
Stachytarpheta angustifolia has been used in the management of some diseases related to bacterial infections by the folkloric healers [6]. The decoction of the whole shrub mixed with natron is taken as a remedy for dysentery and also for similar condition in horses. The cold infusion of the plant when mixed with natron is taken as a remedy for, gonorrhea and other forms of venereal diseases. It is also taken as a vermifuge or a purging vehicle for other vermifuge. The boiled leaf portion of the plant is taken as a remedy for diabetes [7]. In Asia and America, the aerial parts of Stachytarpheta angustifolia is boiled and taken traditionally as a remedy for diarrhea, intestinal parasites, and skin ulcer and as an abortificient [8]. The whole plant extract of Stachytarpheta angustifolia was reported to have abortificient property [9]. The aqueous leaf portion of S. angustifolia has been reported also to have Laxative and anti-diabetic property [10,6], anti-pyretic and hepatoprotective, anti-helminthes and anti- diarrhea properties [11]. In Brazil, the triturated fresh leaf is applied locally for the treatment of ulcer and also as a good remedy for rheumatism. The leaf has also been used for the relief of sprain. The plant has been reported to contain a glucosidal substance called stachytarphine which has been reported to have abortificient property (7). In Ghana, the leaf juice is used as a remedy for eye trouble such as cataracts and also applied to sores in children’s ear. The aqueous leaf extract of Stachytarpheta angustifolia is also used to cure heart problems. In papua – New – Guinea, oral infusion of the whole plant is used as an anti-fertility agent [12].
The decoction of the aerial parts of Stachytarpheta jamaicensis (L) Vahl is used for the treatment of many ailments e.g. diarrhea, intestinal parasite, skin ulcer and as an Abortificient [8]. The Iridoid glycosides isolated from Stachytarpheta indica have been found to possess antimicrobial, antitumor, laxative, hemodynamic, chlorotic, hepatoprotective and anti-inflammatory activity [12]. Stachytarpheta cayennensis has been used by the Brazilian folk medicine as an anti-inflammatory, antipyretic, hepatoprotective, laxative and to treat gastric disturbances [13]. The infusions of the root and stem of L. Javanica spreny are used for the treatment of cough, colds and other related bronchial problem while the leaves are used as tea substitute [9]. A similar member of the genus (Stachytarpheta cayennensis) has been reported to have anti-inflammatory property [8], while the root portion of Stachyterpheta jamaisensis has been found to contain anti-diarrhea properties [9,13].
Literature search shows that no much work has been reported on this plant. Consequent upon this, the present research work was aim at carrying out a pharmacological study on the leaf extract of the plant and also isolating active principles from the n-butanol portion of the leaf extract of Stachytarpheta angustifolia (Verbenaceae). This is with a view to validate scientifically the use of the drug plant by the traditional healers and its safety or order wise, using standard chromatographic, spectroscopic and pharmacological protocols.
Material and Methods
Experimental
General experimental procedure: All melting points (mp) were determined on Gallenkemp melting point apparatus and results are uncorrected. All solvents of analytical grade were purchase from BDH chemical Ltd. Poole England. TLC analysis was carried out using cellulose (TLC) and Silica gel 60 F254 (TLC) plates (Merck, Darmstadt, Germany). Polyamide (Roth, England) and Sephadex LH20 (Fluka, Switzerland) were used for open column chromatography (CC). Chromatographic system:TLC: Visualization of the plate were performed using visible light UV fluorescence and or spraying with the following reagent i=2% AlCl3 and ii= 10% H2SO4 by heating at 110ºC for (5 – 10min). Column chromatography (CC): Chloroform, Chloroform/Ethyl acetate mixture, Ethylacetate, Ethylacetate/ methanol and methanol 100% was used base on increasing gradient polarity system. UV spectra were recorded on specord 40 UV –VIS spectrophotometer (Jena Analytik AG Germany) and HREI-MS was measured on Finnigan MAT GCQ and JOEL JMS-700. Optical rotations were measured with JASCO DIP polarimeter while IR spectra were measured on a Nicolet Avalar 320 FT-IR spectrometer.1H NMR and 13C NMR experiments were performed on Bruker spectrometer 400MHZ for 1H and 100MHZ for 13C NMR. Spectra were referenced to the CD3OD solvent, signals at δ3.30 (`H) and 49.00 (13C) with TMS as an internal solvent standard. Chemical shift – values (δ) were reported in parts per million (ppm) in relation to the appropriate internal solvent standard (TMS). The coupling constants (J – values) were given in Hertz – TOF spectrometer.
Collection of Plant Materials
Leaf portion of Stachytarpheta angustifolia (Verbenaceae) was collected from a farm land in Bomo, a village outskirt of Zaria in Kaduna State of Nigeria in the month of August, 2020. Botanical identification of the plant was performed at the herbarium section of the Department of biological science, Ahmadu Bello University, Zaria, Nigeria. A voucher specimen No. (DC 90188) was deposited.
Extraction and Isolation
Leaf part of Stachytarpheta angustifolia plant was obtained, cut and sliced into pieces. This was then air dried at room temperature for 7 days after which the material was crushed into powder to afford 1040g. The powdered material was then subjected to cold maceration techniques at room temperature with EtOH (4.0L) at 45ºC for 72hrs with intermittent agitation. The extract was concentrated to dryness under reduced pressure to afford a semi solid material. This was further re-suspended in water (700ml) and partitioned successively with n-hexane, (3×600ml), Ethyl acetate (4×500ml) and N-butanol (5×400ml). The various extracts were concentrated using rotary evaporator to afford n-hexane, (3.94g), ethyl acetate (4.34g), n-butanol (6.81g) and aqueous residue (9.20g) respectively. The various fractions of the extract were subjected to phytochemical screening using standard protocols [14-16].
The n-butanol soluble portion (3.4g) was subjected to column chromatography on a silica gel (70-230 mesh) and the column was exhaustively eluted with gradient solvent system of n–hexane (100%,400ml), Chloroform/EtOAC (10:1-1:5), EtOAC/ MeOH (10:1-1:5) and MeOH(100%) . Similar fractions from each portion were pooled together on the basis of TLC analysis to afford 93 fractions of 20ml aliquot (F1 –F93). Fractions (F15-88= 179mg) was further re-subjected to repeated gel filtration techniques using sephadex LH-20 and RP – 18 column chromatography using (100% Methanol) as an eluting solvent to obtain compound S (32mg, Rf 0.6). The progress of elution was monitored by TLC analysis using precoated plate in different solvent system of Chloroform:Methanol:Water (3:3:1),Ethyl acetate : Methanol: Water( 100:16.5:13.5) and EtOAC: MeOH (70:30). The chromatogram was spread with 10% H2S04 and kept in an oven at temperature of 1050C for 5min after which it was removed to ascertain the compounds on the plate [17].
Chemical Test
Ferric Chloride Test
5.0% ferric chloride in 0.5N HCl was sprayed on the chromatogram, fluka-silica gel precoated glass plate of compound S. This was done to test for the presence of phenolic [18].
Vanillin/Sulphuric Acid Test
4.0g solution of vanillin was dissolved in 100ml of Tetraoxosulphate (VI) acid (H2SO4). This was spread on the chromatogram precoated glass plate of compound S in a fume chamber with the aid of a spray canister. The plate was taken to the oven and heated to 110oc, for about 5-10min after which it was removed to ascertain the color formed [17].
Liebermann Buchard’s Test
1ml of anhydrous Acetic acid was added to 1ml of chloroform, and cooled to 0oC in a test tube. Few drops of concentrated H2SO4 was added to the test tube containing solutions of compound S [17].
Determination of Sugar in Compound S
Compound S (4mg) was dissolved in H2O (2.5ml) and 2N aqueous solution of CH2F2-COOH (2.5ml) was added and then refluxed on a water bath for 3hrs each. After this period, the reaction mixture was diluted with H2O (10ml) and extracted with CH2Cl2 (2 x 5ml). The CH2Cl2 extract was washed with H2O and then evaporated to dryness in vacuo. The concentrated aqueous layer was passed through a short Amberlite column and evaporated to dryness to give a sugar fraction (1.2mg). This was further analyzed by HPLC using CH3CN/H2O (85:15). The sugar identified in compound S was D-glucose based on the HPLC retention time of tR of 11.65 (D- glucose tR-11.55min). Co-TLC of the sample with an authentic sample was carried out. The sugar was analyzed by silica gel TLC comparison with standard sugars using solvent system as above [19].
Studies on isolated rabbit jejunum
The rabbit was starved for 24 hours and sacrificed by a blow on the head and exsanguinated. The abdomen was dissected carefully to reveal its abdominal cavity. The jejunum was carefully removed and cleaned free of its content, and 2-3 cm were cut [20,21]. The tissue was mounted into a 25 ml organ bath containing Tyrode solution of the following composition (mm):D-glucose 10.0g, NaCl 90.0g, KCl (10% solution) 20 ml, NaH2PO4.2H2O (10%) 5ml, NaHCO3 10 g, MgCl2.6H2O 1.0 ml and CaCl2 (10% solution) 20ml (24). This was aerated with air maintained at 370C ± 10C. The initial concentration of acetylcholine (0.05-0.8 ug/ml) and contractile responses were recorded isometrically through an isometric micro dynamometer 7004 on an Ugo Basile uni recorder 7050 model. The tissue was equilibrated for 60 - 90 minutes during which the physiological solution was replaced every 15 minutes [22,23]. At the end of the equilibration period, the effect of the ethanol extracts of the leaves, stem bark and root bark at concentration of 0.04 – 0.32 mg/ml each were investigated. The chloroform, ethyl acetate, n-butanol and aqueous fractions of the leaf were also investigated on the isolated jejunum at concentration of 0.04 mg/ml – 0.32 mg/ml. The effect of the leaf ethanol extract of S. angustifolia was also investigated in the presence of atropine on the isolated rabbit jejunum. This was repeated four times [24,25].
Results
The results are given in the following Tables 1 and 2.
| CONSTITUENTS | TEST | OBSERVATION | PORTIONS OF EXTRACTS | |||||
|---|---|---|---|---|---|---|---|---|
| Carbohydrate | HE | Et | CL | Eta | n-But | Aq | ||
| General Test | Molisch | Red colouring | - | + | - | - | - | + |
| Sugar Test | Aniline | Red colour | - | - | - | - | - | + |
| Sugar (Monosaccharide) | Barfoed’s | Red ppt | - | + | - | - | - | + |
| Red. Sugar | Fehling’s | Red ppt | - | + | - | - | - | + |
| Tannins | Lead Ethanoate Iron (III) Chloride Ethanoic acid Methanol’s |
White ppt Blue – Black White ppt Red ppt |
- - - - |
+ + + + |
- - - - |
- - + - |
+ + - + |
+ + + + |
| Saponins Sterols |
Frothing Liberman B. |
Persist frothing Blue or green |
- - |
+ + |
- + - |
- | + + |
+ + |
| Saponin Glycoside | Fehlings Solution Tetraoxosulphate(iv) acid |
Red ppt Brick red |
- - |
+ + |
- - |
+ + |
+ + |
- - |
| Phlobatannins | Hydrochloric Acid | Red ppt | - | - | - | - | + | - |
| Carotenoids | Carr price’s | Blue to red colour | - | + | - | - | - | + |
| Emodol | Borntrager’s | Red colour | - | - | - | - | - | + |
| Flavones aglycones | Shibata’s | Red to Orange | - | - | + | + | + | - |
| Terpenoids | Liebermann B. | Pink to Red colour | + | + | + | + | + | - |
| Alkaloids | Mayer’s Wagner’s Dragendorff’s |
Buff ppt Dark brown ppt Redish brown |
- - - |
- + + |
+ + + |
+ + + |
+ + - |
- - - |
| Flavonoids | Shinoda Tetraoxosulphate (vi) acid |
Deep red Deep Yellow |
- - |
+ + |
- - |
- + |
+ - |
- - |
| Cardiac glycoside | Legal’s Kedd’s Keller – kilanis Baljet Lieberman |
Deep red colour Violet colour Reddish brown Orange to Deep red Bluish green |
- - - - - |
+ + + + + |
_ - _ _ + |
+ + + + + |
+ + + _ + |
+ - + - + |
| Aglycone | δH(ppm) | δC(ppm) |
|---|---|---|
| 1 | 5.30 d (4.0) | 96.02 |
| 2 | - | - |
| 3 | 7.08 br.s | 151 |
| 4 | - | 113.8 |
| 5 | 2.84 m | 32 |
| 6 | 1.91 m | 41.1 |
| 7 | 1.72 m | - |
| 8 | 3.70 m | 79.5 |
| 9 | 2. 03 dd(14.3,7.02) | 45.4 |
| 10 | 2.39 m | 43.2 |
| 11 | 0.98 d (7.2) | 15.1 |
| 1' | 4.98 d (7.9) | 98.4 |
| 2' | 4.93 d d (9.5, 8.0) | 74.3 |
| 3' | 3.70 m | 75.7 |
| 4' | 3.53 dd(9.6, 9.2) | 72.3 |
| 5' | 3.64 m | 76.4 |
| 6' | 4.52 d d (12.0,2.0) | 64.2 |
| 4.40 (12.0,6.0) | ||
| 1" | - | 122.3 |
| 2" | 7.81d (8.4) | 132.8 |
| 3" | 6.80 d (8.2) | 116.4 |
| 4" | - | 163.5 |
| 5" | 6.81 d (8.2) | 116.4 |
| 6" | 7.82d (8.4) | 132.8 |
| 7" | - | 168.3 |
| 1'" | - | 128.2 |
| 2'" | 7.06 br s | 115.2 |
| 3'" | - | 147.3 |
| 4'" | - | 149.5 |
| 5'" | 6.73 d (8.2) | 116.7 |
| 6'" | 6.91dd (8.6,1.7) | 123.4 |
| 7'" | 7.61d (15.7) | 147.3 |
| 8'" | 6.40d (15.01) | 144.9 |
| 9'" | - | 169 |
Key: - = Absent, + = Present.
HE=n-Hex, Et=Methanol, CL=Chloroform, Eta=Ethylacetate, n-But=n-Butanol, Aq=Aqueous
TLC profile of n-butanol leaf fraction showing various spots (Figure 1).
Acute toxicity study
The intreperitonial LD50 of the Leaf, Stem bark and Root bark extract were found to be 118.32 mg/kg -1. 150 mg/kg -1 and 145 mg/kg -1 respectively. The acute toxicity studies showed that the mice were observed to show general CNS depression, restlessness and subsequently death. The whole plant extract, leaf and n-butanol soluble fraction of the leaf extract produced a concentration dependent increase in spontaneous perpendicular movement of the rabbit jejunum.
Gastrointestinal activity study
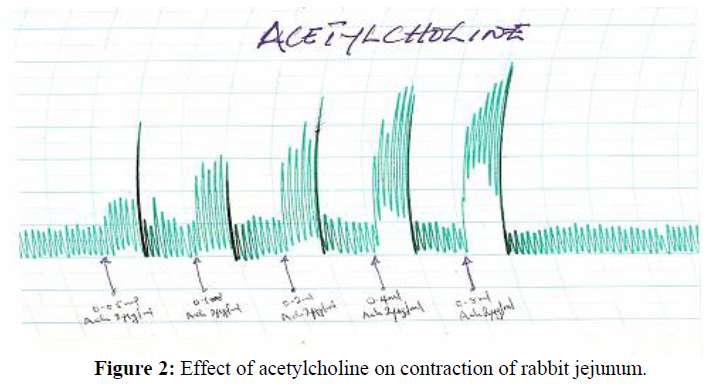
Effect of Acetylcholine on rabbit jejunum: Acetylcholine produces concentration dependent increase in spontaneous contraction of the rabbit jejunum at the concentration of (4.0x10-3-6.4x10-3ug/ml) (Figure 2).
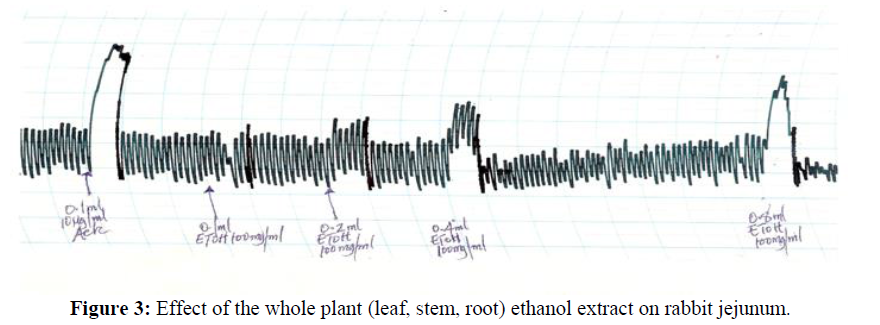
The whole plant (leaf, stem, root) methanol plant extract produces a concentration dependent increase in spontaneous contraction of the rabbit jejunum (0.004 mg/ml-0.32 mg/ml) (Figure 3).
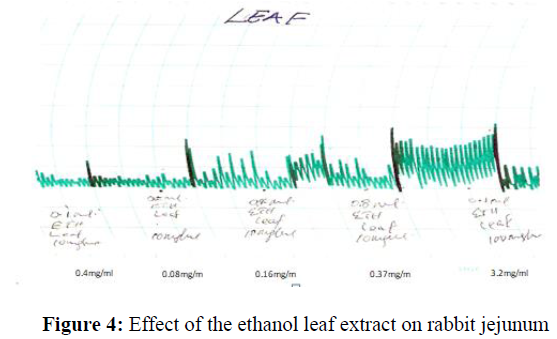
The leaf ethanol plant extract produces a concentration dependent increase in spontaneous contraction of the rabbit jejunum (0.04 mg/ml-0.32 mg/ml) (Figure 4).
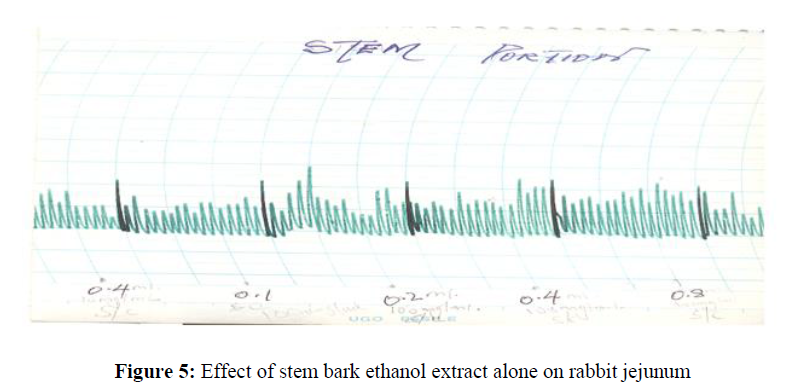
The ethanol stem bark extract of S. angustifolia do not have a marked effect on rabbit jejunum at concentration tested (0.04 mg/ml-0.32 mg/ml) (Figure 5).
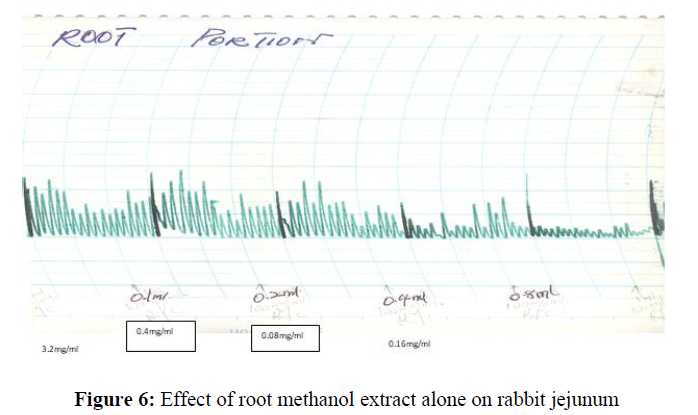
The ethanol root extract produced a concentration dose dependent (0.04 mg/ml-3.2 mg/ml) relaxation of the rabbit jejunum (Figure 6).
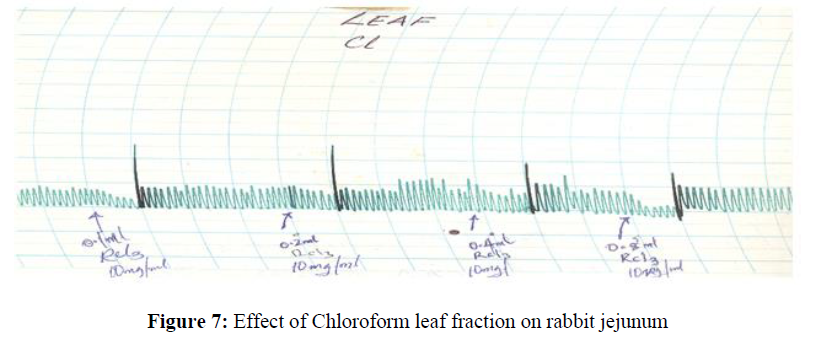
The chloroform fraction of the ethanol leaf extract produced a dose concentration dependent (0.004 mg/ml-0.32 mg/ml) relaxation of the rabbit jejunum (Figure 7).
The ethyl acetate fraction of the leaf extract of S. angustifolia also produces dose concentration dependent relaxation of rabbit jejunum at (0.16 mg/ml-0.64 mg/ml) (Figure 8).
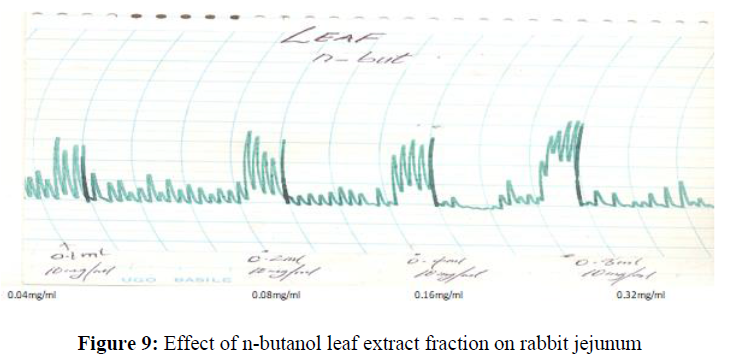
The n-butanol soluble leaf fraction of S. angustifolia produced a concentration dependent increase in spontaneous contraction of rabbit jejunum at (0.04 mg/ml-0.32 mg/ml)b (Figure 9).
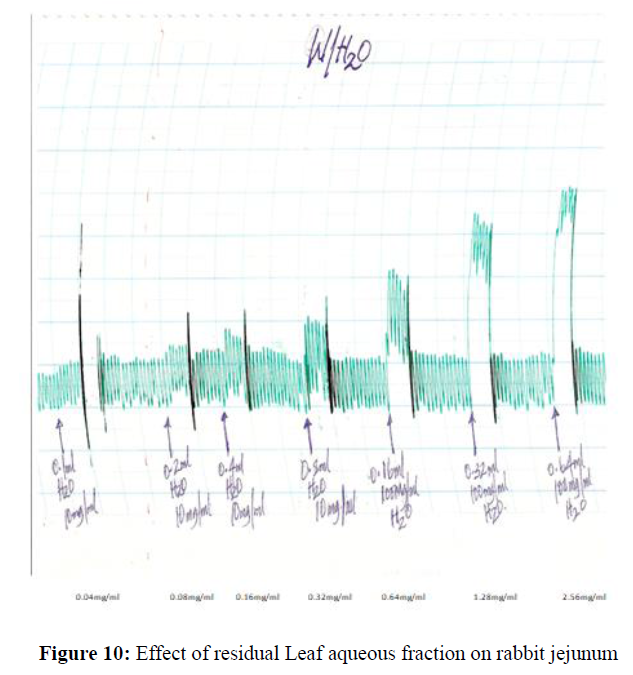
The residual aqueous fraction of the ethanol extract S. angustifolia produced a concentration dependent increase in spontaneous contraction of rabbit jejunum at 0.04 mg/ml-2.56 mg/ml (Figure 10).
The effect of concurrent administration of Atropine with acetylcholine at (0.4 mg/ml) was found to block the spontaneous contraction amplitude of the rabbit jejunum (Figure 11).
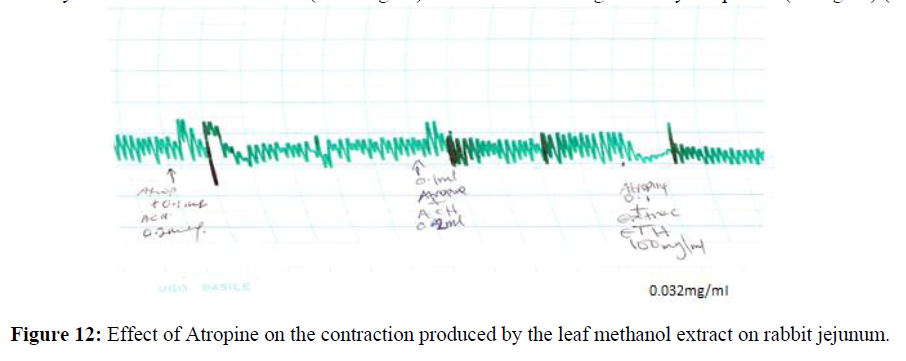
The contraction produced by the leaf methanol extract at (0.032mg/ml) was blocked or antagonized by atropine at (0.1mg/ml) (Figure 12).
Discussion
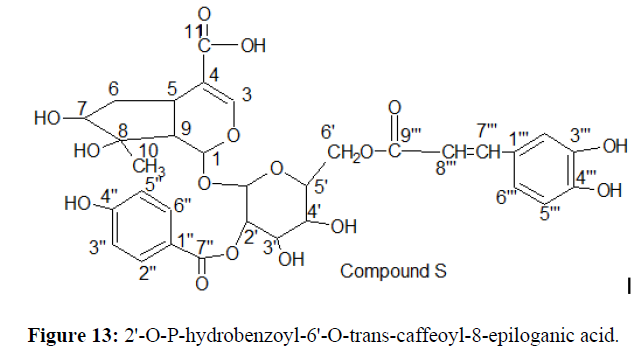
Extraction of the leaf extract of S. angustifolia followed by an extensive column chromatography of n-butanol portion of the extract on silica gel, purification over sephadex LH - 20 and subsequently (PTLC) resulted in the isolation of compound S. The isolate gave a positive colour test with ferric chloride test suggesting the compound to be phenolic; it also gave a red colour at the interphase indicating the presence of glycoside [18]. Compound S was obtained as an amorphous pale yellow powder. The molecular formula was established as C32H34O15 by FABMS with [m/z 657(M-H)]. (FABMS) displayed the following intensive fragmentation M/z 83,8936, 96.3143, 133.3942, 184.9125, 234.0915, 325.4634, 428.5422, 485.2282, 551.3470, 577.1345, 368.3165, 677.4815, 635.4830, 734.5628, 817.2214, 834.9410 and 967.5227 with M/z (100%) at 635.4830 [26].
The IR spectrum of compound S showed the presence of hydroxyl group (3353 cm-1), an aromatic ring at (1617, 1608, 1498 cm-1) and methylenedioxy (1035, 936 cm-1) groups [27]. The UV spectrum shows absorption maximum at λmax 322 and 347 nm which was in conformity with 2 –phenylbenzo furan skeleton [28]. The I.R Spectrum displayed absorption due to hydroxyl at 2447cm indicating an hydroxyl functionality. The absorption at 2954cm indicate a (C – H / stretch) for a methyl group while peak at 14626cm (C=C) indicate several bands on Aromatic rings with many peaks [29]. Bands on 1462cm-1 corresponds to (C=O) an ester group without an enol ether system [30]. The 1H NMR spectrum of compound S shows four aromatic singlet signals at δH 7.81ppm, (IH,s), δH 7.82ppm, - δH 6.82ppm and δH 6.80ppm [31]. The multiplet signal on δH 7.61ppm and δH 6.40ppm are characteristics of an α and β Methylene proton assign to H-7' and H-8' of a benzene ring signifying the presence of a vinylic proton [32]. Signal at δH4.98ppm on H-1' is a characteristic of sugar proton which is in conformity with a β– configuration of an anomeric proton. The glucosyl anomeric proton on δH4.98ppm is linked to the caffeoyl moiety by a glycosidic bond [33]. The spectra data (table 2 ) of compound S have similar signals with those of cyclopentane ring of the iridiod nucleus. The 1H NMR spectrum showed the presence of a secondary methyl signal at 0.98 (3H,d,J=7.2 Hz, H3-10), a hydroxymethine proton (1H,δH3.70, m, H-7) and a methane proton signal at δH2.03ppm (1H,dd,j=14.0,7.0Hz,H-8(35). In 13C NMR, peaks were assigned on the basis of Chemical shift consideration and comparison with data for glucose. 13C NMR spectrum was found to exhibit 32 Carbon signals, 9 corresponding to caffeoyl moiety, 17 to aglycone and 6 to glucose moiety. Signals observed at δC 147.3ppm, δC 149.5ppm, δC 116.7ppm on C,-3", C - 1" and C – 5" are characteristic carbon of aromatic nucleus. Those at δC147.3ppm and δC114.9ppm indicate the presence of an – α and β vinyl carbon on δC169.0ppm which exhibits the presence of a carbonillic carbon on C – 9 [34]. The hydroxyl substitute on C-3 and C – 4 are similar to the corresponding – β – (3'", 4'" dihydroxy phenyl on the aglycone unit. The coupling constant of the anomeric carbon of glucose is completely in consistence with the – β – configuration for the former and α for the latter [35,36]. The 13C NMR data supported the presence of secondary methyl (δc15.1ppm, C-10) at C-8 (δc 45.4ppm) and a hydroxymethine carbon (δc 79.5ppm,C-7). In the HMBC spectrum, the methyl group (δ��0.98, d,j=7.5 Hz,��3-10) showed a correlation with a hydroxy methine carbon (δc 79.5ppm,C-7), hence supporting the presence of an 8-epiloganic acid nucleus [37]. The chemical shift of the glucose and the aromatic structure of compound is in conformity those of the 8-epiloganic acid. Thus, the structure of compound S is determined as 2'-O-p-hydroxybenzoyl-6'-O-trans-caffeoyl-8-epiloganic acid (Figure 13).
The phytochemical screening of the partition portion of the leaf extract reveals the presence of Tannins, Glycosides, Flavonoids, Saponin, Steroids, Alkaloids, and Terpenoids. Tannins are known to be antimicrobial agent that tends to inhibit the growth of microorganism by precipitating out the microbial protein and thus depriving them of development [38]. In the present study, tannins were detected in the ethanol and n-butanol extract fraction of the extract. This can also explain the comparatively better antimicrobial activity of the extract on tested pathogens. This seems further to highlight the limitation of ethylacetate, n-butanol and residual aqueous portion of the extract in traditional management of that ailment which include diarrhea, inflammation of the mouth and throat that were reported to be treated with tannins [39].
Steroids on the other hand increase nitrogen level in the body there by producing protein that helps in the production of muscles. Steroids were found to enhance metabolism and thus inhibit the accumulation of fat, adjust disorders like aneamia by increasing the production of red blood cells in the treatment of arthritis, asthma, brain injury and other type of cancer [40]. However, steroids play an important role in the body as it enhances the onset and progression of cardiovascular and liver diseases as well as acne (by stimulating the serum to produce oil). In the present study, steroid was found to be present in n -hexane, methanol and ethylacetate extract which could be preferred in the management of cardiovascular and liver ailment [41,42].
The presence of carbohydrate and reducing sugars in the leaves extract indicate high energy content of the leaves that could be exploited as a source of edible food or raw materials for industries that utilized carbohydrates reducing sugars to produced food, drugs or biodiesel [42].
Flavonoids are ubiquitous in plants they are known to be common part of human diet and could significantly inhibit microbes which are known to be resistance to the conventional antibiotics [43]. Some of the recently isolated flavonoids were reported to exhibit antimicrobial activities [44]. In addition flavonoids through their free – radical scavenging activity have evoked multiple biological functions which include anti-inflammatory, anti-bactericidal, anti-carcinogenic, Vasodialotory, immune stimulatory, anti-allergic and anti-viral functions [45]. Consequent upon this, the presence of flavonoids in the ethylacetate and n-butanol extract in our study has exhibited the therapeutic efficacy of the extract and has accounted for the folkloric use of the plant in the treatment of related ailment especially the inhibitory effect of pathogens e.g S. aureus, S. typhi, E.coli and P. aeruginosa. However, the antifungal activity of saponin has been reported by [46-48], though saponins has been detected in the ethanol extract of S. angustifolia in a previous study but was found not to elicited any antifungal activity against the fungi and as such were not lethal enough to elicit a measurable fungi static effect.
The observed Antispasmodic effect on the rabbit jejenum could also be attributed to the presence of flavonoids, glycosides, tannins, saponins. Terpenoids, carbohydrate and Alkaloid which have been shown to possess pharmacological activities [49]. The contractions observed by the whole plant extract, leaf extract and n-butanol soluble fraction were similar to those produced by Acetylcholine [50]. Acetylcholine induces contraction of the smooth muscles via activation of the muscarinic receptors and differences in muscarinic receptors are known to exist [51]. The contraction induced by the plant extract and other fractions of the leaf extract might be mediated via the muscarinic receptors [52]. The contractile responses exhibited by the leaf extract and n-butanol soluble fraction were blocked by atropine suggesting that the observed pharmacological effect, were mediated through the muscarinic receptors [53]. The gastrointestinal activities of the leaf extract, chloroform and Ethylacetate fraction of the leaf were assessed for their pharmacological activities on the rabbit jejunum. The chloroform and ethylacetate fractions were found to relax the jejunum concentration dose dependently. The elevated tone of the leaf extract was competitively blocked by atropine (Figure 12). The attenuated rhymic contraction produced by atropine above, signifies that the action might be mediated through the cholinergic receptors [54]. The medium inhibitory contraction of the extract on each of the spasmogen was observed as result of the extract antagonizing the muscarinic receptors [55]. The extract was found to act through the musculotropic route on the rabbit jejunum. The extract was found to block the response of the spasmogen contraction as in (Figure 12). This further confirms its activities via the muscolotropic route. The phytochemical screening of the ethanol and patition portion of the plant extract was conducted using standard protocols. This reveals the presence of steroids, terpenoids, saponins, tannins and glycosides. The active principles present in the extract are apparently acting on the tissue through the cholinergic receptors and hence are responsible for the actions on the tissue [56,57].
Conclusion
This study indicates that, the leaf extracts contain some active components which can induce concentration - dependent contraction of rabbit jejunum. The contraction observed suggest that, they are inactivated in the absence of other portions of the plant. The active principles contain in the plant (leaf) extract of S. angustifolia are apparently mediated through muscarinic receptors other than MI receptors. Therefore, the study has now justified the use of the leaf portion of this plant by the folkloric healers as a purgative agent.
References
- Farnsworth NR. NAPRALET. 2006.
- Farnsworth NR and Soejarto DD. Cambridge Uni Press Publishers New. 1991, p. 25-41.
- Elisabetsky E, Amador TA, Albuqueir que RR et al., J Ethnopharmacol. 2015, 48(2): p. 77-83.
- WHO. WHO technical report services. 2016, 823, p.44-75.
- WHO. Reports of WHO global survey. 2005.
- Ganapaty S, Babu GJ and Naidu KC. J. Medicinal Aromat. Plants. 1998, 203: p. 697- 699.
- Sofowora A. Spectrum Books Limited, Ibadan, Nigeria. 3rd Edition. 2008, p. 200-202.
- Dalziel JM. Crown Agents London. 1999, p. 432 - 434.
- Alberto R, Janet P, Angel V et al., J Phytotherapy Res. 2010, 15: p. 360 - 365.
- Watt JM and Breyer - Brandwijk MG. ES Living Stone Edinburg publishers. 1963, p. 1046 - 1088.
- Sophon R, Kasan S, Nongmy J et al., Anal Sci. 2002, 5: p. 1063-1068.
- Mesia - Vela S, Souccar C, Lima L et al., Phytomedicine. 2004, 2: p. 616 - 624.
- Chanch H, Koffi Y and Chanch APH. Planta medica. 1988, 54: 244 - 296.
- Veena Sharma and Prucheta janmeda. Arab J Chem. 2014, 25(8): p. 25-34.
- Samarya Krishna and Sarin Renal. J Drug Delivery and Therapeutics. 2013, 3(2): p. 109-113.
- Trease GC and Evans WC. Trease and Evans pharmacognosy. 2002, p. 61-66.
- Richard JPC. Humana press Totowa New Jersey, USA. 1988, p. 209 - 230.
- Manguro LO and Lemmen P. Nat Prod Res. 2007, 21(1): p. 56-68.
- Schlemper VA, Rihas M, Nicolas P et al., Phytomedicine. 1996, 3: p. 211 - 216.
- Akah PA, Oni AN, Enwerem NM et al., J Phytotherapy Res. 1997, 43: p. 27-331.
- Kitchen I. Blackwell Scientific Publishers, London, United Kingdom 1st Edn. 1994, p. 40 - 60.
- Samuelson G. Academic Press. New York. USA. 2011, 6: p. 261-280.
- Vongtau HO, Amos S, Binda L et al., J Ethnopharmacol. 2006, 72: p. 207-214.
- Mitchelson FJ. Trends in Pharmacology Science. 2004, 5: p. 12-16.
- Oliveira FC, Ferreira MJP, Nunez CV et al., ELSEVIER. 2000, 37: p. 1-45.
- Wei Dong xie, Cheng Wu weng, Yu fang wiu et al., Chem Biodivers. 2014, 73: p. 4328-4338.
- Zheng-Xiang Xiat, Dan-Dan Zhang, Shuang Liang Yuan - Zhi Laol et al., J Nat Prod. 2012, 75(8): p. 1459-1464.
- Hostettmann K and Wolfende JL. Taylor and Francis. 2004, p. 32-68.
- McLafferty FW and Turecek F. University Science Books, California, USA 1993, p. 226-236.
- Eberhard Breimaier and Wolfgang Voelter. Methods and Applications in Organic Chemistry and Biochemistry. Third completely reversed edition New york. 1990.
- Brown GD. J Phytochem Res. 2003, 5: p. 45-59.
- Francis A Carey. Organic Chemistry N York America. 2003.
- Kemp W. Organic Spectroscopy. Macmillan Education Ltd. 1991, p. 186-197.
- Silverstein RM, Webster FX and Kiemele DJ. John Wiley and sons Inc., New Jersey, USA. 2005, p. 1-71.
- Mohammed M and Idris Bugaje and Garba M. J Chemistry and Biochemistry. 2015, 3: p. 145-157.
- Biswanath D, Debashis RC and Biskas CM. Chemical pharm Bull. 2009, 8: p. 765-796.
- Masaki K, Hide K and Daisuke U. J Nat Prod. 2018, 64: p. 1090-1092.
- Schippman U, Leaman DJ and Cunningham AB. FAO Rome, Italy. 2002, p. 1-21.
- Kayser O and Arndt SR. Pharmaceutical and pharmacological Letter. 2010, 10: p. 38-40.
- Parekh J, Jadeja D and Chanda S. Turk J Biol. 2005, 29: p. 203- 210.
- Prashanth KV, Chauhan NS, Padh H et al., J Ethnopharmacol. 2006, 107: p. 182–188.
- Mohammed M, Bugaje I and Garba MA. J Chemistry and Biochemistry. 2015, 3(1): p. 47-62.
- Cushinie TPT and Lamb AJ. Int J Antimicro Agent. 2005, 26: p. 343-356.
- Kim HP, Son KH, Chang HW et al., J Pharmaco Sci. 2004, 96: p. 229-245.
- Oyvind M Anderson and Kenneth R Markhan. Tyylorandfrancis group, Boca Raton London, New York. 2006.
- Mohammed M. International journal of service and Technology. 2013, 3(1): p. 68-74.
- Michal Araboki, Aneta Wegierek Cluk, Grzegorz Czerwonka et al., J Biomed Biotechnol. 2012, 12: p. 6.
- Edeoga HO and Eriata DO. Medicinal and Aromatic Plants Sciences. 2005, 23: p. 244-249.
- Binda L, Kunle OF, Okafor I et al., J Phytotheraphy Res. 2003, 17: p. 792-796.
- Amos S, Gammaniel K, Adamu M et al., J Herbs, spices and medicinal Plants. 2008, 7(3): p. 45-53.
- Augustine A, Haruna AK, Garba M et al., NJNPM. 2003, 7: p. 13-15.
- Bolton TB. British Medical Bulletin. 1979, 35: p. 275-283.
- Bolton TB. Physiol Rev. 1979, 59: p. 606-718.
- Bonner TI. J Neurosci Res. 1989, 10: p. 197 – 236.
- Nathanson NM. J Neurosci Res. 1987, 6: p. 197-236.
- Nkeh BN, Kamanyi A and Bopelet M. Phytotherapy Research. 1993, 7: p. 120-123.
- Pohocha N and Grampurohit ND. Phytother Res. 2001, 15: p. 49-52.
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref
Indexed at Google Scholar Crossref