Review Article - Der Pharma Chemica ( 2024) Volume 16, Issue 1
Microsponge: An Innovative and Novel Strategy for Drug Delivery System
Shweta Saboo1* and Yogesh Bhise22Department of Pharmacy, Governmeent College of Pharmacy, Aurangabad, India
Shweta Saboo, Department of Pharmaceutics, Dr. Babasaheb Ambedkar Marathwada University Aurangabad, Maharashtra, India, Email: precisewritingsolutions@gmail.com
Received: 22-Dec-2023, Manuscript No. DPC-23-123824; Editor assigned: 27-Dec-2023, Pre QC No. DPC-23-123824 (PQ); Reviewed: 10-Jan-2024, QC No. DPC-23-123824; Revised: 20-Jan-2024, Manuscript No. DPC-23-123824 (R); Published: 26-Feb-2024, DOI: 10.4172/0975-413X.16.1.211-223
Abstract
The microsponge approach has become a highly competitive and rapidly evolving technology. They are porous, highly cross-linked, polymeric microspheres that can be efficiently incorporated into topical drug delivery systems for the purpose of release and retention of dosage form on the skin over a long period of time. Furthermore, they may modify drug release, enhance stability, improve patient compliance and reduce side effects. microsponge systems are non-toxic, non-irritating, non-mutagenic and non-allergenic. In general, there are multiple methods used to manufacture microsponges, such as the quasi-emulsion solvent diffusion method and liquid-liquid suspension polymerization. The present review focuses on the microsponge preparation methods along with its principle and characterization such as Particle size and distribution, porosity, density and surface morphology and release mechanis and drugs incorporated in the Microsponges Drug Delivery System (MDS).
Keywords
Microsponge delivery system; Controlled release; Microsponge preparation methods; Drugs used in MDS
Introduction
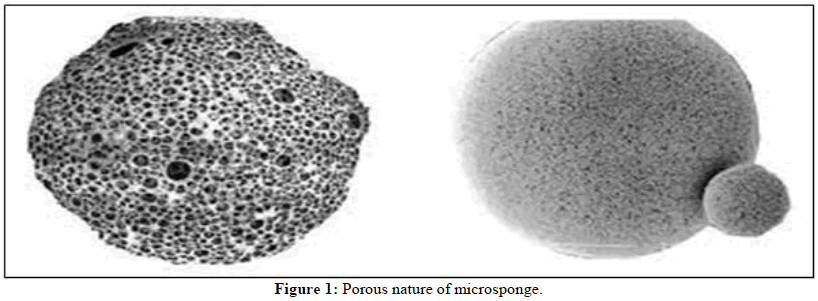
The area of drug delivery mechanisms is now quite challenging and evolving rapidly. Many advancements in medication delivery systems are integrated to maximize the cost-efficiency and effectiveness of therapy. Medicine delivery systems that offer precise control over the rate of release to a particular part of the body have a significant impact on the health care system. Conventional dosage forms such as tablets, capsules, gels, creams and lotions have an instantaneous release and exhibit numerous inadequacies such as low bioavailability, gastrointestinal and skin irritation, unpleasant reactions and toxicological outcomes of active ingredients. One of the significant fields of pharmaceutical sciences is represented by the modified medication release strategy, which benefits both human and animal health. The Microsponges Delivery System (MDS) is a "highly porous, cross-linked, polymeric system composed of porous microspheres that can entrap a wide range of active chemicals and then release them into the skin layers in effective ways over time and in response to a trigger event". Microsponge polymers are versatile enough to accommodate a variety of active ingredients, enhancing product efficacy, stability and extended wear for a variety of skin therapies. Won developed microsponge technology in 1987; it was later patented as a sophisticated polymer that may be used for over-the-counter products, prescription pharmaceuticals and cosmetics (Figure 1) [1-5].
Microsponges typically have a diameter of 5 μm to 300 μm and an average pore size of 0.25 μm, which is much smaller than the average size of many different microorganisms and prevents their penetration. Because of this, microsponges are referred to as self-sterilizing and do not require any form of excipients to maintain their stability. Each particle has a surface that is highly porous and is made up of interconnected channels that form a non-collapsible structure. Depending on the pore size, the pores form an uninterrupted arrangement open to the outer surface of the micro particles, allowing the controlled diffusion of the medication contained. In addition, environmental factors like pH and temperature might affect the release and friction during application, as well as autocatalytic degradation at the application site. The permeation and release can also be stimulus-responsive and release the active ingredients in response to changes in pH, temperature, rubbing and time. These delivery systems have a porous polymeric structure that resembles a sponge-like sphere particle with interconnecting spaces inside of its rigid form and a substantial porous surface. This structure controls the release of the medicine from the delivery system. Microsponges are porous microsphere-based polymeric delivery methods for active pharmacological ingredients that have the potential to be included in a variety of pharmaceutical dosage forms, such as gels, emulsions, tablets and capsules [6-10]. Microsponges are used in oral, topical and ocular administration systems. For topical distribution, microsponges can suspend or entrap a wide range of compounds that can be made into gel, cream, liquid or powder form. A Microsponge Delivery System (MDS) can hold a variety of active substances, including fragrance, sunscreen, emollients, anti-fungal, anti-infective and anti-inflammatory compounds. Drug delivery systems that may regulate therapeutic release rates or direct medications to a specific body site increase drug efficacy, drug therapy cost-effectiveness and patient compliance. This technique is frequently used to entrap medicines that are insoluble or very slightly soluble, such as antibacterial, non-steroidal anti-inflammatory, antihistaminic, antidepressant, antiemetic. Microsponges are suitable topical carriers because they release their encapsulated active ingredient gradually without penetrating the skin or mucous membranes. Accordingly, this MDS system has been commercially employed for chronotherapy of topical drug delivery and is considered for oral, parenteral and pulmonary drug delivery [11-13].
Literature Review
Advantages of microsponges’ technology
• An improvement in product performance.
• Microsponges prolong the duration of drug action and maintain drug release for up to 12 hours.
• They have a large surface area, which increases their capacity for trapping.
• Microsponges are non-toxic, hypoallergenic and free of irritants and mutagenic substances.
• Stability in terms of physical, chemical and thermal factors can be improved.
• Increased solubility and bioavailability of drugs.
• The ability to absorb oily skin secretions, results in a shining appearance for the skin.
• Immiscible products can be easily entrapped by microsponge.
• Microsponges decreased toxicity.
• It can prevent a lot of substances from accumulating on the skin's surface without compromising their effectiveness.
• Greater consumer acceptability results from decreased irritability and improved tolerance.
• Enhances the processing of materials, for example, by turning liquids into powders.
Characteristics of microsponges
• In the pH range of 1 to 11, formulations are stable.
• At temperatures as high as 130°C, microsponge compositions remain stable.
• The majority of vehicles and components are compatible with microsponge compositions.
• Microsponge compositions are self-sterilizing because of the inability of germs to pass through their typical pore size of 0.25 μm.
• Microsponge compositions offer a greater payload (50%-60%), are still free-flowing and may be economical.
• Oil can be absorbed by microsponges up to six times its weight.
• The entrapment efficiency of microsponge formulations is high, reaching 50 to 60 percent.
• They are more flexible in their formulation.
Benefits
The following benefits of the microsponge drug delivery system may exist:
• Without the use of preservatives, shelf life and product stability can be extended because bacteria cannot fit within the microsponge due to their
size.
• Microsponges have a huge internal surface area, a high pay loading capacity and are highly compartmentalized.
• Ingredients are suitable for topical application to the skin when their undesirable properties, such as oiliness and tackiness, as well as their
disagreeable feel and odor, are greatly decreased.
• The delayed release of medications produced for topical use is made possible by MDS, increasing their efficacy.
• Microsponges are non-collapsible structures comprised of interconnected gaps with a broad porous surface.
Limitations of microsponge delivery system
• Organic solvents are typically used as the porogen in the preparation process for microsponge, which poses risks to the environment and public
safety because they can catch fire easily.
• Very little residual monomer residue that could be poisonous and life-threatening has been observed in certain situations.
Properties of the actives for the entrapment into the microsponge
• It must either be completely miscible in the monomer or have the ability to become miscible by adding a small amount of a solvent that is not miscible with water.
• It must be either water-insoluble or poorly soluble.
• It shouldn't make the mixture more viscous during formulation and should be inert to monomers.
• It must maintain its stability when in contact with the polymerization catalyst and when polymerization is occurring.
• Microsponges shouldn't lose their spherical shape.
• For prolonged effect, the API's half-life should be less than 5 hours.
• For ease of penetration, a drug's molecular weight should be less than 600 g/mole.
• The microsponge can include both hydrophilic and hydrophobic materials (Tables 1-3).
| Sr.No. | Drug Name | Sr. No. | Drug Name |
|---|---|---|---|
| 1 | Benzoyl peroxide (Anti-acne) | 17 | Diclofenac (NSAID) |
| 2 | Retinol (Vitamin-A) | 18 | Ibuprofen (NSAID) |
| 3 | Lornoxicam (NSAID) | 19 | Paracetamol (NSAIDS) |
| 4 | Indomethacin (NSAID) | 20 | Trolamine (Analgesic) |
| 5 | Flurbiprofen (NSAIDS) | 21 | Etodolac (Anti-inflammatory) |
| 6 | Ketoprofen (NSAIDS) | 22 | Curcumin (Anti-inflammatory) |
| 7 | Tretinoin (NSAID) | 23 | Dicyclomine (Anti-inflammatory) |
| 8 | Mupirocin (Anti-bacterial) | 24 | Acyclovir sodium (Anti-viral) |
| 9 | Diacerein (Anti-bacterial) | 25 | Fluconazole (Anti-fungal) |
| 10 | Mometasone furoate (Corticosteroid) | 26 | Luliconazole (Anti-fungal) |
| 11 | Prednisolone (Corticosteroid) | 27 | Itraconazole (Anti-fungal) |
| 12 | Fluocinolone acetonide (Corticosteroid) | 28 | Tioconazole (Anti-fungal) |
| 13 | Erythromycin (Anti-biotic) | 29 | Oxiconazole (Anti-fungal) |
| 14 | Clindamycin (Anti-biotic) | 30 | Miconazole nitrate (Anti-fungal) |
| 15 | Paeonol (Anti-cancer) | 31 | Nebivolol (Anti-hypertensives) |
| 16 | Loratidine (Antihistaminics) | 32 | Meloxicam (NSAIDS) |
Table 1: List of drugs in the preparation of microsponges.
| Sr.No. | Polymer name | Sr.No. | Polymer name |
|---|---|---|---|
| 1 | Ethylcellulose | 8 | Sodium Alginate |
| 2 | Eudragit RSPO | 9 | Carbopol 940 |
| 3 | Eudragit EPO | 10 | Carbopol 934 |
| 4 | Eudragit RS 100 | 11 | HPMC E15 |
| 5 | Eudragit RL 100 | 12 | Acrylic polymer |
| 6 | Eudragit S100 | 13 | Propylene glycol |
| 7 | Eudragit L100 | 14 | Polystyrene |
Table 2: List of polymers used in the preparation of microsponges.
| Sr.no. | Specification | Optimum value |
|---|---|---|
| 1 | Drug and Polymer ratio | 1:1,1:2,1:3 and 2:1,3:1 |
| 2 | Amount of drug (mg) | 100-300 |
| 3 | Polyvinyl alcohol (mg) | 100 |
| 4 | Amount of inner phase solvent (ml) | 100 |
| 5 | The temperature of the inner phase | 25°C |
| 7 | Type of process | Magnetic stirrer and bath sonicator |
| 8 | Amount of water in the outer phase (ml) | 100 |
| 9 | Magnetic stirrer speed | 1000 rpm |
Table 3: Optimum values for microsponges formulation.
Discussion
Method of preparation of microsponges
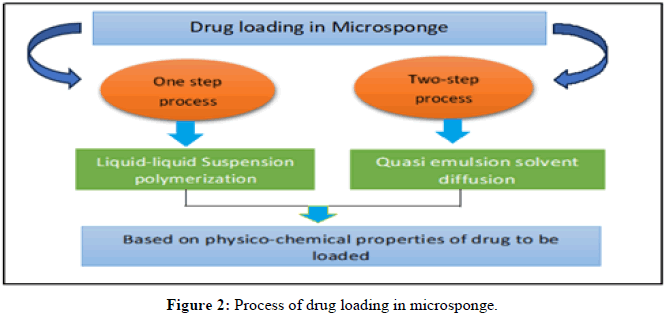
Preparation methods of microsponges: In accordance with the drug's physical-chemical properties, the procedure for loading the medication into microsponges is depicted in Figure 2.
• One-step process: Through this procedure, a non-polar, inactive medication is loaded. The porogen produced by this kind of medication has a porous structure. The porogen medication is neither affected nor activated by the polymerization process and it is resistant to free radicals.
• Two-step process: When the medicine is sensitive to a polymerization environment, this technique is used. In this procedure, a replacement porogen is employed during polymerization and is replaced with an active ingredient in a low-stress experimental setting.
Following are some preparation methods for microsponges:
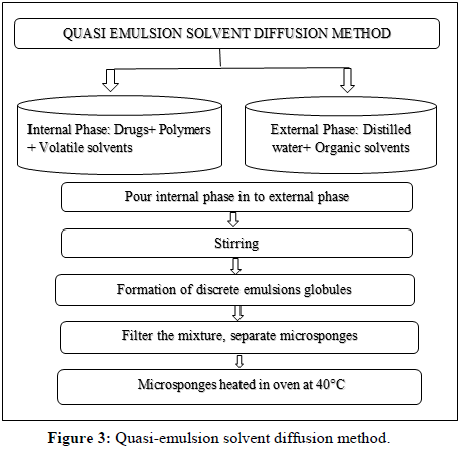
Quasi-emulsion solvent diffusion method: The previous two stages (external and internal phase) are constructed using this procedure. The internal phase contains the active therapeutic agent, polymer, solvent and plasticizer, while the external phase consists of organic solvents and distilled water containing surfactants. As depicted in Figure 3, the quasi-emulsion solvent diffusion is a two-step procedure [14-16].
The steps involved in preparation can be described as (Quasi-emulsion solvent diffusion method):
Preparation of the internal phase through the polymer's dissolution in a volatile solvent, such as acetone, dichloromethane or ethyl alcohol.
• The drug was slowly added and dissolved into the above solution.
• PVA is dissolved in water to prepare the outer phase.
• Pouring the inner phase into the outer phase while stirring vigorously for 60 minutes at a specific RPM.
• Quasi-emulsion globules are separate globules that form as a result of vigorous swirling.
• Creating hard, insoluble microsponges by extracting solvent from globules.
• By filtration, the created microsponges are separated.
• Microsponges should be cleaned using a suitable solvent.
• Microsponges can be dried in an oven for up to 12 hours at 40°C.
• Percentage yield determined by weighing.
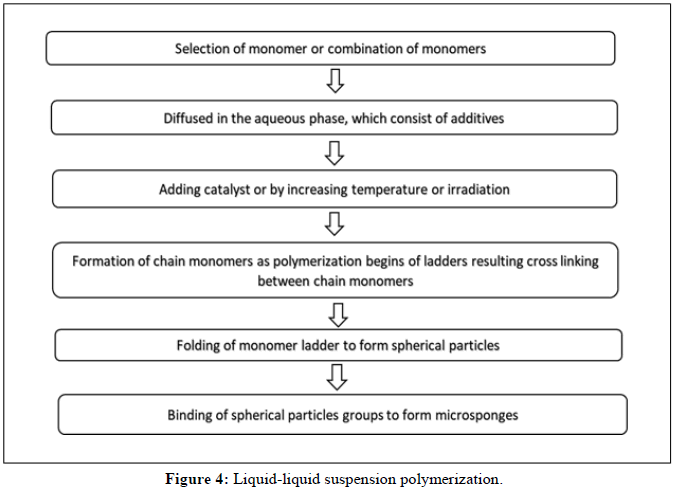
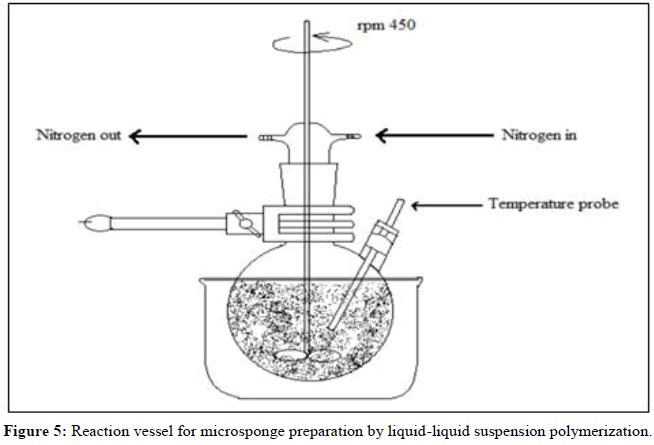
Liquid-liquid suspension polymerization method:
• The suspension polymerization method used to create microsponges is based on the free radical suspension polymerization technique depicted in Figure 2.
• This method uses three bare round-bottom flasks with a stirrer, coupled to a water condenser and a thermometer to measure the temperature
(Figures 4 and 5).
The steps involved in the suspension polymerization method can be described as follows:
Monomer choice (single or numerous).
• Chain monomers form once the polymerization process gets going.
• The development of a ladder due to the cross-linking of chain monomers.
• Development of sphere-shaped particles (microspheres).
• The clumping together of spherical particles to form bunches.
• Microsponges are created when microsphere clusters come together.
Multiple-emulsion solvent diffusion
To create biodegradable porous microspheres, a unique approach was devised. This approach involved dispersing an internal aqueous phase in an organic polymeric solution that contained an emulsifier such as span, polyethylenemine and stearyl amine. After that, a double emulsion was created by dispersing this non-emulsion once more in an external aqueous phase that included PVA. This approach offers the benefit of entrapping both medications that are water soluble and those that are not. Additionally, it can be utilized to trap thermolabile substances like proteins. According to some authors, xanthan gum acts as an emulsifier to stabilize the internal w/o emulsion.
Addition of porogen: In this method, a porogen like a hydrogen peroxide or sodium bicarbonate substituted internal numerous emulsions. For this, a single-phase system consisting of the porogen was created in the polymeric solution and then redispersed in an aqueous phase containing PVA. The multiple emulsions were then given an initiator and the organic solvent was allowed to evaporate, leaving the micro particles to make the microsponges [17-19].
Oil in oil emulsion solvent diffusion: Contrary to the w/o/w approach, an Oil-in-Oil (o/o) emulsion was created by utilizing a volatile organic liquid as the internal phase, which was then allowed to slowly evaporate at a regulated rate while being continuously stirred. As stated, the procedure used a mixture of fixed oil (corn or mineral) and dichloromethane containing span 85 as the external phase solvent, dichloromethane as the internal phase solvent and polylactide glycolic acid as the polymer. To create the microsponges, the internal phase was continuously stirred into the dispersion medium while being added dropwise. Using acetone as the dispersing solvent and liquid paraffin as the continuous medium, this method was used to create hydroxyzine HCl-loaded Eudragit RS-100 microsponges. The drug's physicochemical characteristics and the polymer used to make microsponges have an impact on the choice of organic solvent and external phase.
Lyophilization technique: The gelation process was utilized to create porous microspheres from the gelation procedure-prepared microspheres. In this procedure, the microspheres were lyophilized after being incubated in a chitosan hydrochloride solution. Rapid solvent removal caused the microspheres to develop pores. Due to the speedy removal of the solvent, this process is quick and rapid but has the drawback of producing broken or shrunken microparticles.
Vibrating orifice aerosol generator technique: For the first time, a Vibrating Orifice Aerosol Generator (VOAG) was used to create lipid-bilayered mesoporous silica particles. The procedure entailed creating porous particles by the use of VOAG-driven surfactant evaporation in microdroplets. Tetra-ethyl-orthosilicate core particle preparation used stock solution made by refluxing ethanol, water and diluted hydrochloric acid. To create monodisperse droplets using VOAG, the stock solution was diluted with the solvent containing surfactant and agitated. The liposomes contained the created microspheres. The tailored drug distribution of active ingredients is possible with these encapsulated particles.
Ultrasound-assisted production: By modifying the liquid-liquid suspension polymerization method, this technique was developed. The monomer Beta-Cyclodextrin (BCD) and the cross-linking agent diphenyl carbonate are used to create the microsponges. The reaction mixture was heated and sonicated to control the size of the microparticles. After the reaction mixture cooled, it was milled to produce rough particles, which were subsequently cleaned with ethanol and distilled water. Cross-linked cyclodextrin microparticles have pores that can act as carriers for medication loading.
Electrohydrodynamic atomization method: By using this technique, chitosan microspheres with pores were created. After being sonicated to create bubbles, the chitosan solution was drawn into a syringe, perfused via a steel capillary using a syringe pump and then electro hydrodynamic atomization was applied. The capillary's diameter was selected such that it would hold onto every bubble in the suspension as it passed through it. The sole factor affecting the voltage utilized in the studies is the amount of chitosan present in the solution. In each example, the flow rate and applied voltage produced a steady cone-jet mode, except in the situation when the maximum concentration was used, which was challenging to electrospray. The sodium hydroxide aqueous solution at 4% weight percent was used to cross-link the chitosan microspheres [20].
Effect of formulation variable on microsponges
Effect of composition of internal and external phases: The mean particle size of the microsponge will be higher if there is a greater difference between the apparent viscosities of the dispersed and continuous phases. Due to the higher viscosity of the internal phase, the globules of the produced emulsion can scarcely be split into smaller particles and bigger droplets are found, increasing mean particles when the dispersion phase is more viscous is poured into the continuous phase (external phase). Only 3 to 5 ml of internal phase can be used to create the best microsponges. The manufacturing yield and drug content of microsponges are shown to decrease when the internal phase is raised from 5 to 15 ml. This is because the internal phase has a larger concentration while the drug concentration is lower.
Effect of drug-to-polymer ratio: Particle size is the variable that was impacted by the change in the drug: Polymer ratio. The microsponge’s particle size increases along with the amount of medicine being used. The loading capacity is not much impacted but the production yield can change significantly from a minimum ratio to a maximum ratio when the amount of polymer concentration is constant but the drug-to-polymer ratio is altered.
Effect of stirring rate: With an increased stirring rate, smaller microsponges are produced. The production yield decreases but the drug content rises when the stirring rate is increased, indicating that there is less drug loss when the stirring rate is raised. This is brought on by the external phase turbulence, which makes the polymer adhere to the paddle and lowers production yield (Figure 6).
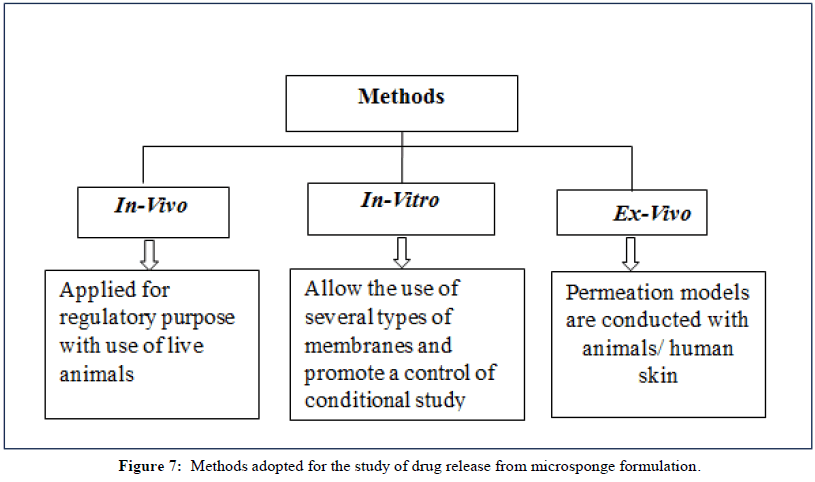
Methods of drug release
There are three categories for the techniques used to analyze drug release from microsponges and penetration through the skin depicted in Figure 7.
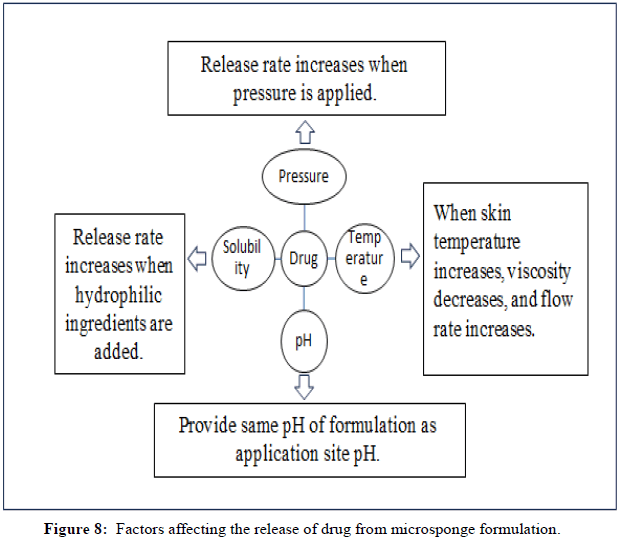
Factors affecting release mechanism
Pressure triggered systems: In this method, when the dosage form is rubbed against the skin, tiny sponges that are entrapped with the medication release it. Process parameters, robustness and the type of material used are only a few of the microsponge's characteristics that affect how much medication is delivered.
Temperature triggered systems: When the temperature changes, the active ingredient are released into the body. Some drugs are too viscous to flow at room temperature without interacting with the porous system. However, when the substance is applied to the skin, a rise in skin temperature leads to an increase in flow rate, which results in a continuous release of the medication.
pH triggered systems: In this procedure, drug release is triggered by a change in pH and is accomplished by altering the coating on microsponges for pH-based actives.
Solubility triggered system: When exposed to water, porous systems that contain a water-soluble excipient release the medication. Release can occasionally be brought on by diffusion mechanisms, which include the partition coefficient between the drug and the external system (Figure 8) (Tables 4-7).
| Drug used | Polymer used | Solvent used | Method of preparation | Result |
|---|---|---|---|---|
| Mometasone furoate | Eudragit RS 100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | Increasing the ratio of the drug to polymer will decrease the release rate of the drug from microsponges. |
| Etodolac | Ethyl cellulose, Eudragit RS 100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | Formulation of etodolac with ethyl-cellulose gives the maximum drug release of 99.3% within 8 h. |
| Diclofenac sodium | Ethylcellulose | Dichloromethane | Quasi-emulsion solvent diffusion | Increasing the drug and polymer ratio will increase their release rate followed by Higuchi diffusion kinetic. |
| Acyclovir sodium | Ethylcellulose | Dichloromethane | Quasi-emulsion solvent diffusion | Optimized F1 released 50.85% drug at 8 and Fick's law of diffusion was not followed. |
| Prednisolone | Eudragit RS 100 | Ethyl alcohol | Quasi-emulsion solvent diffusion | Cumulative release of microsponges 48.87% at 8 h. |
| Domperidone | Eudragit RS 100 | Dichloromethane | Quasi emulsion solvent diffusion | Drug; polymer ratio of 1:2 is more efficient and 76.38% drug release at 8 h. |
| Oxybenzone | Ethylcellulose | Dichloromethane | Quasi-emulsion solvent diffusion | The controlled release of drugs from microsponges promotes the retention of drugs with reduced permeation activity. |
| Fluconazole | Eudragit s 100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | Microsponges-loaded gel releases 85.38% drug at 8 h. |
| Sertaconazole nitrate | Eudragit RS-100 | Dichloromethane | Quasi-emulsion solvent diffusion | Batch F5 releases 69.38% drug at 8 h. |
| Famotidine | Eudragit RS 100 | Dichloromethane | Quasi-emulsion solvent diffusion | % Entrapment efficiency was 88.83% and % cumulative release 86.9% for F6 formulation. |
| Risperidone | Ethyl cellulose and Eudragit RS 100 | Ethyl alcohol | Quasi-emulsion solvent diffusion | Ethyl cellulose and Eudragit RS 100 gave better drug release and encapsulation efficiency as compared to their single-use. |
| Nateglinide | Eudragit RS-100 | Dichloromethane | Quasi-emulsion solvent diffusion | Microsponge with a drug-polymer ratio of 1:3 was more proficient in giving controlled release at the end of 12 h. |
| Betamethasone | Eudragit RS-100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | The pH of microsponges gel was 6.8 and 73% drug release |
| Piroxicam | Eudragit RS-100, RL, S-100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | Piroxicam microsponge carbopol 934 gel produced a significant (p<0.05) improvement of the in-vitro release than pure piroxicam gel. |
| 5-Fluorouracil | Eudragit RS 100 | Acetone | Quasi-emulsion solvent diffusion | MS-loaded 5-FU was more effective than 5-FU itself. |
| Curcumin | Ethylcellulose | Dichloromethane and methanol | Quasi-emulsion solvent diffusion | Following zero-order release kinetics, the curcumin microsponges placed inside the capsule shells demonstrated a 93.2 % curcumin release in an 8-hour examination. |
| Tazarotene | Eudragit RS-100 | Dichloromethane and methanol | Quasi-emulsion solvent diffusion | Preparation and development of 0.1% tazarotene-loaded microsponge gel for increased drug availability at the site of action. |
| Posaconazole | Eudragit S 100 | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | Microsponge formulations demonstrated linked diffusion, non-fickian (anomalous) release and polymer matrix relaxation. |
| carbamazepine | Ethyl cellulose | Dichloromethane and methanol | Quasi emulsion solvent diffusion | Increasing the concentration of ethyl cellulose increased entrapment efficiency and particle size. The release rate subsequently decreased by increasing ethyl cellulose concentration. |
| Tazarotene | Ethylcellulose | Dichloromethane and ethanol | Quasi-emulsion solvent diffusion | The use of TZR-loaded microsponge-based gel as a controlled-release drug delivery system has been demonstrated to be effective in reducing the negative effects associated with traditional TZR formulations. |
| Azithromycin | Ethylcellulose | Dichloromethane and methanol | Quasi-emulsion solvent diffusion | Azithromycin in a sustained release pattern for an extended period reduces the frequency of application and improves patient compliance. |
Table 4: Summary of recent research on microsponges drug delivery system.
| Microsponges | Microcapsules | Liposomes |
|---|---|---|
| Porous in structure | Shell-like structure | Bilayer vesicles |
| Size range 5 um-300 um | Size range 50 nm-2 nm | Size range 40 nm-180 nm |
| Both hydrophilic and hydrophobic drugs can be entrapped | Solid or droplets of liquids and dispersion can be entrapped | Can entrap both hydrophilic and hydrophobic drugs |
| Chemically inert across temperature and pH | Chemically and thermally stable | Controlled drug release |
| Preservative is not required in the formulation | Preservatives are required in the formulation | Preservatives are required in the liposome formulation |
| Drug entrapment is about 50%-60% | Drug entrapment is about 30% | Drug entrapment is about 50% |
| Controlled drug release | No controlled drug release | Controlled drug release |
Table 5: Comparison between microsponge, microcapsules and liposomes.
| Sr. No. | Micropsonge Delivery System | Name of product | Drug | Treatment | Advantages | Manufacturer |
|---|---|---|---|---|---|---|
1 |
Cream | Retin-A-Micro 1 | Tretinoin | Acne-vulgaris | 0.17 and 0.04% tretinoin entrapped into a microsponge containing methyl methacrylate/glycol and di methacrylate polymer. | Ortho-McNeil Pharmaceuticals, Inc. |
2 |
Cream | Line eliminator dual retinol | Retinol | Anti-wrinkle | Lightweight cream delivers both immediate and time-released wrinkle-fighting action. | Avon |
3 |
Cream | Carac cream 0.05% | Fluorouracil | Actinic keratoses | 0.5% Fluorouracil incorporated into a microsponge composed of glycolmethacrylate/methyl methacrylate and dimethicone. | Dermik laboratories Inc |
4 |
Cream | EpiQuin micro 8 | Hydroquinone and retinol | Hyperpigmentation | Microsponges release active ingredients into the skin gradually throughout the day which minimizes skin irritation. | Skin medica Inc |
5 |
Cream | Retinol 15 night cream 3 | Retinol | Anti-wrinkle | Retinol 15 Night cream result in the visible diminishment of fine lines and wrinkles and improve skin discoloration. | Bio-medic, Sothys |
6 |
Cream | Neobenz | Benzyl peroxide | Anti-acne treatment | Reduce the amount of acne-causing bacteria by causing the skin to dry. | Skin media, Inc |
7 |
Moisturizing cream | Lactrex 12% (moisturizing cream) 7 | Lactic acid and ammonium lactate | Moisturizer | Moisturize dry, flaky, cracked skin. | SDR pharmaceuticals, Inc |
8 |
Lotion impregnated Wipes | Ultra-guard 5 | Dimethicone | Protect babies’ skin | Dimethicone that helps to protect babies’ skin from a diaper rash. | Scott paper |
9 |
Lotion | Oil control lotion | Natural antibodies | Tightness to promote healing, acne-prone, oily skin condition | Absorb oil on the skin surface. | Fountain cosmetics |
10 |
Spray | Aramis fragrances | Antiperspirant spray gives sustained release of fragrance | The ultra-light powder absorbs fragrance oil easily. | Aramis Inc | |
11 |
Gel | Salicylic peel 20 and 30 | Salicylic acid | Excellent exfoliation | Excellent exfoliation and stimulation of the skin which improves fine lines, pigmentation and acne concerns. | Bio-medic |
Table 6: Various marketed formulations based on microsponges delivery technique.
| Sr.No. | Patent Number | Inventors | Publication date |
|---|---|---|---|
| 1 | US4997753 | Dean RC, et.al | 1991 |
| 2 | US5135740 | Katz, et al. | 1992 |
| 3 | US5100783 | Robert, et al. | 1992 |
| 4 | US5679374 | Fanchon, et al. | 1994 |
| 5 | US5316774 | Robert P, et al. | 1994 |
| 6 | US5725869 | Ray JR, et al. | 1996 |
| 7 | US5851538 | Forix M, et al. | 1998 |
| 8 | US6395300 | Straub, et al. | 1999 |
| 9 | US6211250 | Tomlinson, et al. | 2001 |
| 10 | US20030232091 | Shefer, et al. | 2002 |
| 11 | US20030008851 | Singh | 2003 |
| 12 | US20040247632 | Cattaneo and Maurizio | 2004 |
| 13 | US20050271702 | Steven G, et al. | 2005 |
| 14 | US7098315 | Schaufler A, et al. | 2006 |
| 15 | US20070141004 | Malek | 2007 |
| 16 | US20080160065 | Halliday | 2008 |
| 17 | US7426776 | Franklin SL, et al. | 2008 |
| 18 | US7604814 | Karykion Inc. | 2009 |
| 19 | US7740886 | Sara Vargas | 2010 |
| 20 | US7749489 | Celmatrix corporation | 2011 |
| 21 | US8323672 | Karykion corporation | 2012 |
| 22 | US8361273 | Ferring BV, et al. | 2013 |
| 23 | US8758728 | Stiefel research Australia Pvt ltd. | 2014 |
| 24 | US8936800 | Galderma research and development | 2015 |
| 25 | US9764316 | Eugenia P, et al. | 2017 |
Table 7: Patent numbers, inventors and publication dates are shown in table.
Evaluation parameters of microsponges
Particle size (microscopy): The particle size distribution is studied with optical or electron microscopes. The particle size of microsponges can also be determined using other imperative techniques, such as laser light diffractometry. By using any relevant technique, including laser light diffractometry, particle size analysis is carried out. For all formulations, the values (d50) can be represented as a mean size range. To explore the impact of particle size on drug release, the cumulative percentage of drug release from microsponges of various particle sizes will be plotted against time. Particles between 10 m and 25 m in size are preferred for use in the final topical formulation because particles bigger than 30 m can cause a gritty feeling.
Scanning Electron Microscope (SEM) study: Prepared microsponges can be coated with gold-palladium at room temperature in an argon environment to study their morphology and surface topography. SEM can then be used to examine the microsponges' surface morphology (SEM). It is also possible to use SEM to show the ultrastructure of a shattered microsponge particle.
Characterization of pore structure: Depending on the microsponges' pore size, the rate of medication release may change. Aspects of microsponges' porosity, such as apparent density, mass, total pore surface area, intrusion-extrusion isotherms, pore size distribution and average pore diameters, are also investigated.
Loading efficiency and production yield: The following equation can be used to compute the loading efficiency (percent) of the microsponges:
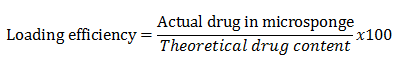
Production yield of the micro particles can be calculated accurately by taking the initial weight of the raw materials and the final weight of the produced microsponge.

Production yield is influenced by the drug-to-polymer ratio as well; an increase in this ratio results in an increase in production yield.
Determination of true density
Microsponges' real density can be determined using an ultra-pycnometer under helium gas and is estimated from the mean of several measurements.
Compatibility studies
Through the use of Thin Layer Chromatography (TLC) and Fourier Transform Infrared spectroscopy, the compatibility of medicine with reaction adjuncts can be investigated (FT-IR). Powder X-Ray Diffraction (XRD) and Differential Scanning Colorimetry (DSC) can be used to investigate the impact of polymerization on the drug's crystallinity. For DSC, 5 mg samples can be accurately weighed into aluminum pans, sealed and heated at a rate of 15°C per min over 25°C-430°C in a nitrogen atmosphere.
Polymer/monomer composition
The drug release from microspheres is governed by elements such as microsphere size, drug loading and polymer composition. The MDS's polymer composition can have an impact on the entrapped drug's partition coefficient between the vehicle and the microsponge system, directly affecting how quickly the drug is released. Plotting cumulative percent drug release versus different polymer compositions will allow you to study the release of drugs from microsponge systems.
Resiliency
According to the demands of the final formulation, microsponges' resilience (viscoelastic characteristics) can be changed to produce beadlets that are either softer or stiffer. The rate of release tends to be slowed down by increased cross-linking. Thus, by taking into account release as a function of cross-linking with time, the resiliency of Microsponges is evaluated and optimized as per the requirements.
Drug release study
Utilizing the dissolving apparatus USP XXIII and a modified basket made of 5μm stainless steel mesh, the dissolution profile of microsponges can be examined. The rotational speed is 150 rpm. To achieve sink conditions, the solubility of the actives is taken into consideration while choosing the dissolution media. At various periods, samples from the dissolution medium can be examined using an appropriate analytical technique.
In vitro diffusion studies
Through a cellophane membrane, the prepared microsponges gel was used to conduct in vitro diffusion tests in a Keshary-Chien diffusion cell. The receptor compartment was made up of 100 ml of phosphate buffer and the membrane was then evenly coated with 500 mg of a gel containing 10 mg of medication. The donor compartment was kept near the receptor compartment and the temperature was held constant at 37 ± 0.50 degrees. At regular intervals, Teflon-coated magnetic bars that were driven externally stirred the solution on the receptor side. Pipetting out 5 ml of the solution from the receptor compartment, the old 5 ml of phosphate buffer was then immediately replaced.
Stability study
The durability of the gel formulation is assessed under ICH recommendations. After being maintained in sterile, lacquered, collapsible aluminium tubes for 30, 60 and 90 days at a temperature of 40°C ± 2°C and relative humidity of 75% ± 5%, the in vitro release profile, pH and outer shell of the gel were all evaluated (Tables 8 and 9).
Safety considerations
Safety studies of microsponges can be confirmed by:
• Eye irritation studies in rabbits.
• Skin irritation studies in rabbits.
• Oral toxicity studies in rats.
• Allergen city in guinea pigs.
• Mutagenicity in bacteria.
Applications of microsponge
Topical drug delivery using microsponge technology:
| Sr.No. | Category | Drug | Applications |
|---|---|---|---|
| 1 | Anti-fungal | Fluconazole, miconazole, clotrimazole, econazole | Give sustained release of drug. |
| 2 | Anti-acne | Tretinoin, benzoyl peroxide | Reduce skin irritation and sensitization. |
| 3 | Anti-dandruff | Selenium disulfide, zinc pyrithione | Enhanced safety and efficacy of drugs with reduced irritation and odor. |
| 4 | Anti-wrinkle | Retinol | Time-released delivery into the skin. |
| 5 | Anti-actinic keratoses | 5-fluorouracil | Treat actinic keratoses with reduced dosage form. |
| 6 | Anti-inflammatory | Piroxicam, Hydro-cortisone | Extended drug release with reduced dermatoses and allergy. |
| 7 | Moisturizer | Lactic acid and ammonium lactate | Moisturize dry, cracked and flaky skin. |
| 8 | Skin pigmentation | Hydroquinone | Improve aesthetic appeal by reducing oxidation. |
Table 8: List of topical drug delivery using microsponge technology.
| Sr.No. | Category | Drugs | Applications |
|---|---|---|---|
| 1 | Anti-pyretic | Paracetamol | Time-release dosage form with a pH-dependent polymer coating. |
| 2 | Anti-inflammatory | Indomethacin | Reducing side effects like G.I. irritation with modified release. |
| 3 | Colon targeting | Paracetamol | Time-release dosage form with a pH-dependent polymer coating. |
| 4 | Musculoskeletal pain | Ketoprofen | Provided modified release while reducing the severity of side effects. |
| 5 | Anti-cholinergic | Dicyclomine | Effective local action with prolonged drug release. |
Table 9: Oral drug delivery using microsponge technology.
Long-lasting-colored cosmetics
A range of colored cosmetic products, including rouge and lipsticks, can have their colors trapped by microsponges so they remain on the skin for a longer amount of time. As already said microsponge contributes to more even spreading and enhanced covering power. As a result, colored cosmetics made with microsponges would be exceedingly luxurious.
In bone and tissue engineering:
A porous microsponge was created by mixing two acrylate derivative monomers with two aqueous dispersions of tricalcium phosphate grains and hydroxyapatite powder. Basic Fibroblast Growth Factor (bFGF) was applied to a collage sponge sheet, which has a limited angiogenic effect in a dose-dependent way and has a prolonged release within the mouse subcutis due to matrix biodegradation. It suggested that type I collagen could be exploited as a bFGF reservoir for therapeutic purposes because it showed improved blood flow in the ischemic hind leg of mice.
Therapeutic application of microsponges
Diabetes wound healing with microsponges: Microsponges containing nebivolol have been developed and are being utilized to treat diabetic wound healing. The medication primarily acts as a vasodilator in diabetic wounds and restores endothelial function. In particular, the drug facilitates wound closure by promoting a healing environment that is perfect for diabetic wounds.
Microsponges in a fungus infection: When compared to using plain gel loaded with the medication, using microsponges anti-fungal gel formulation has been shown to extend the duration of medication release. The formulation of oxiconazole nitrate microsponges, used to treat fungal infections, allowed for a controlled release of the medication for up to 12 hours.
Microsponges for arthritic pain: In situations of arthritis, diclofenac is applied topically using a microsponge. Diclofenac-containing microsponges can be applied topically to patients to improve compliance by reducing gastrointestinal discomfort and first-pass metabolism. Diclofenac dimethylamine microsponges gel was created using the quasi-emulsion diffusion of solvent approach and it demonstrated sustained and controlled release for the treatment of arthritis. Skin protection with microsponges: Sunscreen protects our skin from UV radiation, avoiding sunburns and some malignancies, such as malignant melanoma and basal carcinoma. Oxybenzone microsponge sunscreen was created to offer increased UV protection and SPF rating.
Microsponges for acne: A dangerous skin ailment that can irritate the skin is acne. An anti-acne cream developed by rizkalla et al., contains miconazole nitrate. They produced microsponges with eudragit RS 100 and added them to the cream to ensure prolonged release. Studies showed that although conventional formulations released 83.089 % of the medicine in four hours, microsponges offered sustained drug release (78.28 %) for up to eight hours.
Microsponges in psoriasis: Psoriasis is an inflammatory skin disorder. Microsponges are used to treat psoriasis as well. In research to treat psoriasis, clobetasol propionate was added to microsponges. They saw the drug lasting up to 12 hours, which is longer than the typical form, which delivers the medication over a period of 2.5 hours.
Microsponges for skin infections: In addition, cream forms of microsponges were created to treat a variety of skin conditions, including eczema and atopic dermatitis. Amrutiya et al. combined mupirocin with microsponges to generate a stable and non-irritating formulation.
Microsponges in melanoma: Hydroquinone microsponges containing 4% hydroquinone and 0.15 % retinol were used to treat melanoma and Pro-Inflammatory Hyperpigmentation (PIH). Only one patient experienced an adverse reaction to the formulation and the medicine was released gradually with little skin irritation.
Microsponges for colon cancer: A microsponges formulation of the chemotherapy medication 5-fluorouracil (5-FU) has been developed for the treatment of colon cancer. The formulation boosted the activity of the medicine by increasing the relative accumulation in cancer areas and decreasing the toxicity. According to drug release studies, pure 5-FU released the medication for up to 5 hours, as opposed to the microsponges.
Microsponges in the treatment of gastric ulcer: The famotidine-loaded gastro-retentive microsponges for the treatment of stomach ulcers. Eudragit RS100 as a polymer and polyvinyl alcohol as a stabilizer were used in various ratios to create the floating microsponges utilizing a quasi-emulsion solvent diffusion process. According to reports, the microsponges followed zero-order kinetics and released the medication in a sustained manner for a duration of 12 hours.
Microsponges in hyperpigmentation: Glabridin microsponge has been created by Deshmukh et al. with great success for the efficient control of hyperpigmentation conditions. By employing ethyl cellulose as the polymer and a quasi-emulsion solvent diffusion process, microsponges were created. The produced microsponges were mixed with the Carbopol gel to make topical application simpler. In guinea pigs, the glabridin microsponge-based gel's ability to whiten the skin was evaluated. In guinea pigs, UV B radiation was utilized to cause hyperpigmentation. The skin of the animal was examined histopathologically after the course of treatment. Animals treated with microsponge-based gel experienced an effective reduction in melanin density. Finally, writers demonstrated how well microsponges can treat hyperpigmentation problems.
Recent advances in microsponge drug delivery system
Various techniques were made by modifying the methods to form nanosponges, nanoferrosponges and porous microbeads.
Nanosponges: The nanoformulations employed in topical drug delivery, particularly the passive targeting of cosmetic chemicals, are known as nanosponges. These are useful for prolonged retention in the skin layer and absorption via the skin. The Solvent diffusion method was modified to create these nanosponges. Nanosponges can be easily generated by changing the agitation, amount of polymer or emulsifying agent. Researchers have also demonstrated that nanosponges are effective delivery systems for active ingredients that are available in gaseous form. Targeting malignant cells is another function of these nanosponges carriers.
Nanoferrosponge: Due to the external magnetic trigger that forces the carriers to penetrate deeper tissue and then results in the removal of magnetic material from the particle, leaving a porous system, nanoferrosponge a novel approach, constitutes the self-performing carriers with better penetration to the targeted site.
Porous microbeads: The microbeads with a high pore count are created by improving the properties of porous microspheres. The development of solid porous microbeads uses cross-linking and polymerization processes. For topical, buccal and oral medication delivery systems, these microbeads are employed.
Microsponge approach in future prospective
In addition to regulated oral peptide delivery, tissue engineering in cell culture media (cellular regeneration and stem cell culture) and transdermal delivery method, microsponge is a unique drug delivery system. Extended-release formulations without additives, increased stability and reduced irritability were developed. In toothpaste, cosmetics or mouthwash, lipsticks, long-lasting colored cosmetics and concealing powder, microsponge technology serves as a carrier system. We can create new drug delivery methods like the parenteral and pulmonary routes.
Conclusion
The detailed data summarized in this article suggest that many various pharmaceutical drugs are explored in microsponge delivery systems. MDS holds major potential in both pharmaceutical industries due to its novel release methodology and its ease of administration with fewer side effects. It is a peculiar and novel methodology for the controlled release of topical agents which can entrap the actives that are released onto the skin over time. Various literatures have reported that the microsponge systems are non-irritating or soothing, non-allergic, non-mutagenic and non-toxic. MDS technique is used currently in over-the-counter skin care products, sunscreens and prescription and cosmetic products. Therefore, microsponge has a lot of prospective and is a very rising field which is needed to be examined and it is probable to become a precious drug delivery matrix substance used for different therapeutic applications in the upcoming future.
Acknowledgment
The authors are very much thankful to management and principal, Dr. Bothara, Government college of pharmacy, Chh. Sambhaji Nagar for providing the necessary facility for doing the literature of this review manuscript.
References
- Jadhav N, Patel V, Mungekar S, et al. J Sci Innov Res. 2013; 2(6): p. 1097-1110.
- Junqueira MV, Bruschi ML. AAPS Pharm Sci Tech. 2018; 19: p. 1501-1511.
[Crossref] [Google Scholar] [PubMed]
- Kaundal A, Bhatia R, Sharma A, et al. Int J Adv Pharm. 2014; 4: p. 177-181.
- Kaity S, Maiti S, Ghosh AK, et al. J Adv Pharm Technol Res. 2010; 1(3): p. 283.
[Crossref] [Google Scholar] [PubMed]
- Aloorkar NH, Kulkarni AS, Ingale DJ, et al. Int J Pharm Sci Nanotechnol. 2012; 5(1): p. 1597-1606.
- Patil RS, Kemkar VU, Patil SS. Am J Pharm Tech Res. 2012; 2: p. 227-251.
- Gangadharappa HV, Vishal Gupta N, Chandra Prasad M S, et al. Curr Drug Deliv. 2013; 10(4): p. 453-465.
[Crossref] [Google Scholar] [Pubmed]
- Srivastava R, Pathak K. Expert Opin Drug Deliv. 2012; 9(7): p. 863-878.
[Crossref] [Google scholar] [Pubmed]
- Nokhodchi A, Jelvehgari M, Siahi MR, et al. Micron. 2007; 38(8): p. 834-840.
- Ravi R, Senthilkumar SK, Parthiban S. Int J Pharm Rev Res. 2013; 3(1): p. 6-11.
- Angamuthu M, Nanjappa SH, Raman V, et al. J Pharm Sci. 2014; 103(4): p. 1178-1183.
- Crcarevska MS, Dimitrovska A, Sibinovska N, et al. Int J Pharm. 2015; 489(1-2): p. 58-72.
[Crossref] [Google Scholar] [Pubmed]
- Lalitha SK, Shankar M, Likhitha D, et al. Eur J Mol Biol Biochem. 2016; 3(2): p. 88-95.
- Kumari A, Jain A, Hurkat P, et al. Crit Rev Ther Drug Carrier Syst. 2016; 33(1).
- Kumar PM, Ghosh A. Eur J Pharm Sci. 2017; 96 (56): p. 243-254.
[Crossref] [Google Scholar] [Pubmed]
- Amrutiya N, Bajaj A, Madan M. AAPS Pharm Sci Tech. 2009; 10(2): p. 402-409.
[Crossref] [Google Scholar] [Pubmed]
- Osmani RA, Aloorkar NH, Ingale DJ, et al. Saudi Pharm J. 2015; 23(5): p. 562-572.
[Crossref] [Google Scholar] [Pubmed]
- Osmani RA, Aloorkar NH, Thaware BU, et al. Asian J Pharm Sci. 2015; 10(5): p. 442-451.
- Orlu M, Cevher E, Araman A. Int J Pharm. 2006; 318(1-2): p. 103-117.
[Crossref] [Google Scholar] [Pubmed]
- Patravale VB, Mandawgade SD. Int J Cosmet Sci. 2008; 30(1): p. 19-33.
[Crossref] [Google Scholar] [Pubmed]