Research Article - Der Pharma Chemica ( 2022) Volume 14, Issue 11
Nano-Zinc Oxide particle: synthesis by Sol-gel and optical properties study
Shurouq Salem Alshehri, Sarah S. Alghamdi, Fedaa I. Alatiequ, Ghidaa Alshikh, Khulood Batayah and Maymona M. Abutalib*Maymona M. Abutalib, Department of physics, University of Jeddah, College of science, Jeddah, Saudi Arabia, Email: mmabotaleb@uj.edu.sa
Received: 01-Nov-2022, Manuscript No. dpc-22-78841; Editor assigned: 03-Nov-2022, Pre QC No. dpc-22-78841; Reviewed: 17-Nov-2022, QC No. dpc-22-78841; Revised: 19-Nov-2022, Manuscript No. dpc-22-78841; Published: 26-Nov-2022, DOI: 10.4172/0975-413X.14.11.5-7
Abstract
Zinc oxide nanoparticles were synthesized using Sol-gel method with zinc acetate and sodium hydroxide. The synthesized sample was calcined at different temperatures for half hour. The samples were characterized by UV-Vis. The strong absorption below 430 nm of all samples was confirmed by UV–Vis., due to the interband electronic transitions through the band gap edges.
Keywords
Zinc oxide nanoparticles; Sol-gel; Uv Vis; Optical energy gap
INTRODUCTION
Nanomaterials are materials with at least one external dimension that measures 100 nanometers (nm). Nanoscience’s and nanotechnology have become popular in recent years, and nanotechnology is an inherently interdisciplinary field that has generated significant scientific and engineering interest. There are many processes for creating different nanomaterials, categorized as "top-down" or "bottom-up" nanoobjects of interest, including quantum dots and nanocrystals of metals, semiconductors, oxide, and other materials one dimensional nanostructures such as nanotubes and nanowires. These nanomaterials' synthesis, characterization, and applications are being discovered widely [1]. The impact of nanotechnology in all fields of science and technology is clear, as it stimulated researchers to take advantage of its applications in the medical field, energy, pollution treatment, and food because of its importance in improving products and producing environmentally friendly waste and preserving energy production through renewable energy systems and improvement of medicinal properties [2]. Among the most widely used nanomaterials, zinc oxide has gained significant interest in the scientific and medical communities due to its important use in many biomedical and antibacterial applications. Moreover, ZnO- NPs is generally considered non-toxic and safe. ZnO-NPs are one of the most widely used semiconductors in various applications. ZnO is deemed more favorable for creating UV light-emitting diodes and laser diodes, Due to its unique optical properties. ZnO- NPs are used in many different medical industries, such as pharmaceuticals and cosmetics, UV blockers in sunscreens, and antimicrobials. There are many ways to manufacture ZnO nanoparticles. They are classified into chemical or physical methods, such as physical vapor deposition (PVD), chemical vapor deposition (CVD), nanolithography, hydrothermal synthesis, the sol-gel process, and precipitation in water solution method. In this research, the manufacturing method used is the Sol-gel process. Sol-gel processing is a wet chemical route for synthesizing colloidal dispersions of inorganic and organic- The reason for choosing the Sol-Gel process is that it has many advantages, Simple and economical, and efficient, including low processing temperature. It can produce a thin coating to ensure excellent adhesion between the substrate and the top layer [3]. Sol-gel processing is beneficial in making complex metal oxides, temperature- inorganic hybrid materials, mainly oxides and oxide-based hybrids. Sensitive organic-inorganic hybrid materials and thermodynamically unfavorable or metastable materials. ZnO-NPs were prepared to study the optical properties at different temperatures using a spectrophotometer. According to the results, ZnO-NPs can be used as a possible and promising application in sensors [4].
EXPERIMENTAL WORK
Material
Zinc acetate dihydrate (molecular mass = 297.47 g/mol) (Merck). Sodium hydroxide. Ethanol (Ethyl Alcohol).
Syntheses of ZnO nanoparticles
Sol gel method was used to prepare Zinc oxide (ZnO) nanoparticle. Briefly, ZnO NPs was prepared at 60 °C for 2 h, 7.22 and 3.73 mmol of NaOH and zinc acetate dihydrate, respectively, as initial starting material quantities. To study this reaction and to obtain the best product, the synthesis procedure was modified by changing precursor concentrations, reaction time and temperature as well as pH. Final products were white precipitates. The obtained white precipitate was dissolved in 20ml of ethanol until it was dissolved. A clean substrate was placed in the spin coater; a few drops zinc was placed with determine the spin duration and revolution per time. Then it was placed at different temperatures for 30 minutes.
Characterizations
A spectrophotometer was used to characterize the optical absorption properties of ZnO, and the spectra of samples in the wavelength range (400-600) were recorded using a V-770 spectrophotometer.
RESULT AND DISCUSSION
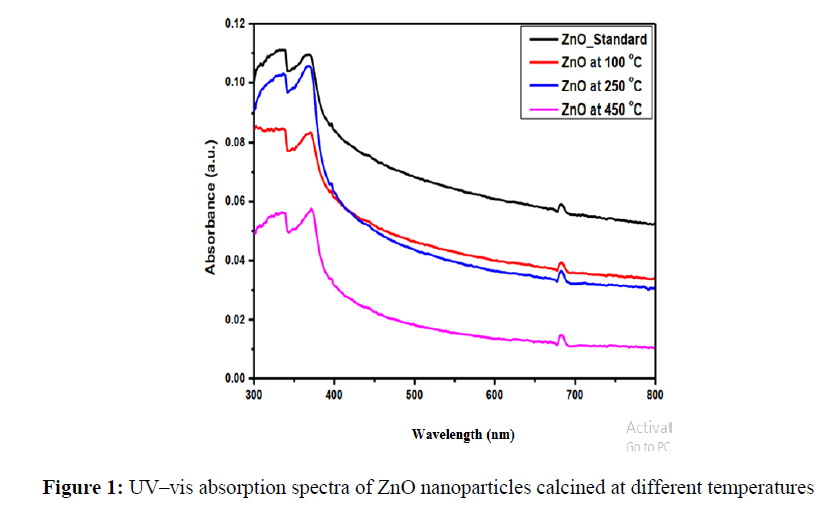
UV–vis diffuses reflectance spectra of ZnO NPs. The diffuse reflectance spectra of ZnO nanoparticles calcined at different temperatures are shown in Figure 1. All samples show a strong absorption below 430 nm due to the interband electronic transitions. All samples show a strong absorption below 430 nm due to the interband electronic transitions through the band gap edges [1]. With increasing the calcination temperature, the absorption band shifts to lower wavelength (blue-shift). Also, the intensity of the absorption band increased with increasing the calcination temperature.
The band gap energy can be estimated according to the relation [2,3]:
αhv =A (hv-Eg)n
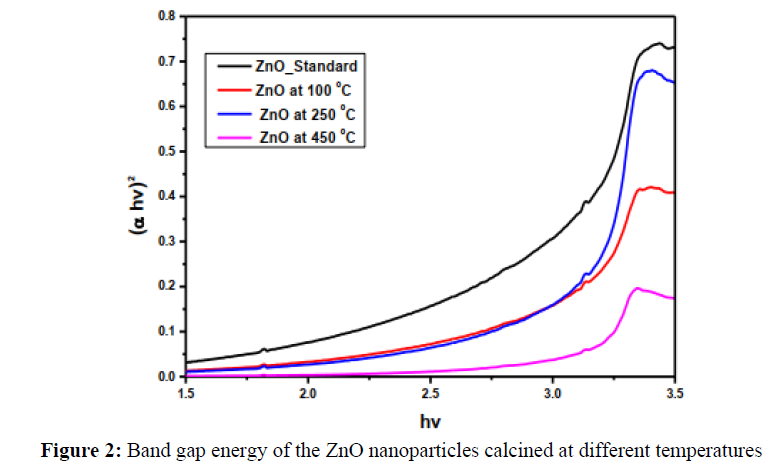
where α is absorption coefficient, h is Planck constant, v is the wavenumber, A is a constant and Eg is the energy band gap in which n = 1/2 for direct band gap materials and n = 2 for indirect band gap [4]. The band gap energies (Eg) values of ZnO unclaimed and that calcined at 100º, 250º and 450ºC were estimated from the plot of (αhν)1/2 versus photon energy in electron volts as shown in Figure 2. The obtained band gap energy values are listed in Table 1. Figure 2 and Table 1 illustrate that the band gap energy of the samples became wider with increasing the calcination temperature.
| Sample name | Zno standard | Zno 100ºC | Zno 250ºC | Zno 450ºC |
|---|---|---|---|---|
| Band gap (eV) | 3.08 | 3.01 | 3.15 | 3.17 |
CONCLUSION
In this paper, ZnO nanoparticles were successfully prepared by the Sol-gel method. The effects of temperature change on the absorption spectra for ZnO, which were deposited on the substrate by spin coating was studied. The strong absorption below 430 nm of all samples was confirmed by UV– Vis., due to the interband electronic transitions through the band gap edges. The intensity of the absorption band increased with increasing the calcination temperature. The absorption band shifts to a lower wavelength with increasing the calcination temperature. From the relation αhv =A (hv-Eg)n, we conclude that the ZnO nanoparticles values of the energy gaps increase as the calcination temperature of the samples increases.
References
- Rajeh A, Hassan SM, Ahmed AI. J Sci Adv Mater Devices. 2019, 4: p. 400–412.
- Hassan SM, Ahmed AI. Ceram Int. 2018, 44: p. 6201–6211.
- Hassan SM, Ahmed AI. Eng Asp. 2019, 577: p. 147-157.
- Alsulami QA, Rajeh A, Mannaa MA, et al., Int J Hydrogen Energy. 2021, 46: p. 27349-27363.