Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 2
Nematicidal Activity of Four Medicinal Plants Extracts against Meloidogyne Spp.
Senhaji B1*, Mziouid A1, Chebli B1, Mayad EH1,2,3 and Ferji Z3
1Biotechnology and Environmental Engineering Team, National School of Applied Sciences, PO Box: 1136/S, Agadir, Morocco
2Department of Biology, Faculty of Sciences, Ibn-Zohr University, PB. 8106-Cité Dakhla Agadir, Morocco
3Plant Protection Department/Nematology, Horticultural Complex of Agadir, Hassan II Institute of Agriculture & Veterinary Medicine-Agadir, Morocco
- *Corresponding Author:
- Senhaji B
Biotechnology and Environmental Engineering Team
National School of Applied Sciences
PO Box: 1136/S, Agadir, Morocco
Abstract
The aim of this study is to evaluate the effect of organics and aqueous extracts of some aromatic and medicinal plants harvested in Agadir region (Morocco), against second stage juveniles of Meloidogyne spp. The four plants used are: Asteriscus imbricatus, Lavendula dentata, Pulicaria mauritanica and Globularia alypum. The best results were obtained with A. imbricatus extracts: petroleum ether and chloroform extracts induced respectively 89, 31% and 92, 71% of mortality at 2000 ppm, while the aqueous induced 78, 76% of mortality at 20000 ppm. For this reason these extracts have been tested against Meloidogyne spp in vivo. The results of in vivo test showed that both nonpolar extracts petroleum ether and chloroform of A. imbricatus have an important nematicidal activity at 6000 ppm similar to the effect of chemical nematicid, while the aqueous extract was ineffective. Then the organic extracts of this plant were subjected to a phytochemical screening to get an overview on their metabolites secondary composition. The results showed that the effective extracts in vitro and in vivo are distinguished by the presence of terpenes. The activity demonstrated by A. imbricatus was good enough to propose this plant for further studies at greenhouse and field stages to determine its efficacy in soil.
Keywords
In vitro, In vivo, Meloidogyne spp, Asteriscus imbricatus, Pulicaria mauritanica, Lavendula dentate, Globularia alypum
Introduction
Currently the plant pests cause significant losses in crops. These parasites cause yield losses of up to 45% [1] or even a total loss of production. Meloidogynes is a phytophagous pest which cause the onset of deformation in the roots of infected plants called galls. Meloidogyne parasitize more than 5500 species of plants [2], they are widely spread over the globe and are a dangerous group for the damage it causes [3,4]. Yield losses can reach 65% depending on the level of infestation [5]. The control methods used against these pests are pesticides synthesis, especially methyl bromide remains the most effective remedy for both disinfect the soil nematode that not only to eliminate any kind of terrestrial pathogens and weeds.
Methyl bromide and other nematicidal products are actually withdrawn from the market because of their adverse effects on human health and the environment. Therefore, the search for alternative methods of control, cleaner and cheaper, proves mandatory [6]. The use of plant extracts is one of the solutions that can help reduce the problem of nematodes [6,7]. The Agadir region contains a variety of species having toxic effect against root-knot nematodes. However, although some have been the subject of several studies in the laboratory of Nematology in Hassan II Agriculture and veterinary Institute, Agadir Morocco, but a large number of species have never been evaluated for their toxic effect against root-knot nematodes Meloidogyne. It is in this context that fits this study which aims to evaluate the effectiveness of organic and aqueous extracts of four aromatic and medicinal plants against root-knot nematodes Meloidogyne spp in vitro and in vivo, and characterize phytochemically the most effective extracts.
Materials and Methods
Collection and preparation of plant
The aerial part of plants samples were collected randomly in April 2012 in three different areas near Agadir (Cape Ghir, Imozzer idaoutanane, Tmanar). The spices of all samples were identified and were deposited in the herbarium in the Mechanics and Process Laboratory, Energy and Environment in National School of Applied Sciences Agadir, Morocco.
The botanical name family, the collecting zone and GPS coordinates are summarized in Table 1. Plant samples were cleaned, and air dried in the shade then grounded to a fine powder using an electric laboratory grinding mill and stored in the dark at 4°C until use [8].
| Botanical name | Family | The collecting areas | GPS coordinates |
|---|---|---|---|
| Asteriscus imbricatus | Asteraceae | Cape Ghir, 40 km northwest of the city of Agadir | 30° 37' 48" N 9° 54' 00" W |
| Pulicaria mauritanica | Asteraceae | Imozzer idaoutanane,60 km northwest of the city of Agadir | 30° 40' 23'' N -9° 28' 48''W |
| Lavendula dentata | Lamiaceae | Tmanar,103 km northeast of the city of Agadir | 31°00'0 00"N -9°39'59 99" W |
| Globularia alypum | globulariaceae |
Table 1: The botanical name family, parts used and the collecting zone of plant samples
Preparation of plant extracts
Extraction using organic solvents
We conducted a hot extraction using Soxhlet apparatus, using a cellulose cartridge we filled with plant material powder (Aerial part). Solvents we used are in order of increasing polarity: petroleum ether, the Chloroform, ethyl acetate and methanol [9]. The extraction is complete when the extraction solvent becomes increasingly clear that is to say, without a significant proportion of solute [10]. The final extracts were obtained after concentration and removal of the solvent by rotary evaporation.
Aqueous extraction
The aqueous extraction was performed using 20 g of the powdered herb to 50 ml of distilled water after three hours of maceration using magnetic stirring plaque. The mixture was filtered through filter paper Whatman paper number 4. The remaining cake is recovered and the operation is repeated five times to make five exhaustion. Extracts obtained were stored at 4°C and protected from light until use [11].
In vitro test
Extracting eggs of Meloidogyne spp
The roots from a tomato field infested with Meloidogyne spp typical galls are rinsed with water, cut into small pieces. Then we proceeded to mix for 2 min in a solution of bleach to 1%. The mixture is filtered through a series of sieves (250 mesh size ranging from microns to 5 μm), and the filtrate containing the nematode eggs are collected on a coarse sieve 2 lined paper handkerchiefs cellulose deposited in a receptacle, the second stage juveniles (J2) of migrating through the cellulose is recovered after 48 h.
Nematicidal assay
Approximately 100 larvae (J2) of Meloidogyne spp were placed in an aqueous suspension of 9 ml of H2O per Petri dish and then added one ml of the extract tested.
The organics extracts were tested against Meloidogyne spp, at 20, 200 and 2000 ppm. While the aqueous extract was tested at 20, 200, 2000 and 20000 ppm. Each treatment is carried out in three replicates. The boxes are arranged in a completely randomized device at room temperature (25 ± 2) for 72 h. Distilled water was used as a negative control. During in vitro incubation, juvenile (J2) of Meloidogyne react differently to plant extracts studied depending on the concentration and the incubation period used (24 h, 48 h and 72 h). Immobile J2 larvae were identified under an optical microscope at a magnification of 40 x. Dead larvae (J2) are identified based on the appearance of needle still they present. A correction is made with respect to the control treatment that distilled water is according to the formula [12].
Tim (%)=(Tr-Tm)/(Tm-100) × 100
With, Tr=Percentage of J2 motionless in the extract, Tim=Percentage of J2 immobile in distilled water.
In vivo test
To confirm the results obtained in vitro, petroleum ether chloroform and aqueous extracts of A. imbricatus plant were selected for in vivo test. The experiment was conducted on the culture of tomato at the experimental greenhouse at of Horticultural Complex Agadir, Morocco. The trial lasted from 27/02/2015 (the date of the application of treatment) to 27/04/2015 (date grubbing plants). Plants aged two weeks were transplanted in black plastic pots filled with pretreated substrate by various treatments. The transplantation took place one week after the application of treatments. The transplantation substrate includes 1/3 of peat and 2/3 of a soil naturally infested with Meloidogyne spp. We applied eight treatments which are listed in Table 2.
| Treatment number | Type of treatment | Concentration(ppm) |
|---|---|---|
| T1 | Petroleum ether extrait of Astericus imbricatus | 6000 |
| T2 | Petroleum ether extrait of Astericus imbricatus | 4000 |
| T3 | Chloroform extrait of Astericus imbricatus | 6000 |
| T4 | Chloroform extrait of Astericus imbricatus | 4000 |
| T5 | Aqueous extract of Astericus imbricatus | 40000 |
| T6 | Aqueous extract of Astericus imbricatus | 30000 |
| T7 | A chemical nematicid: vydate. | 5 l/hectare |
| T8 | Positive control consisting of naturally soil infested with Meloidogyne spp but untreated | - |
Table 2: The treatments used in vivo
Each treatment was replicated eight times, and pots were arranged in a completely randomized design in a greenhouse.
Phytochemical screening
The extracts which gave the best results in vitro and in vivo, were selected for phytochemical screening. Each dry residue is dissolved in methanol to prepare a solution of 1 mg/ml. This solution is used to realize all the phytochemical tests.
Characterization of flavonoids
The extracts were subjected to Thin Layer Chromatography (TLC) examination using commercial silica plates for the determination of the secondary metabolites. We tried different solvent systems to achieve better migration and components separation of the studied extracts. For each sample and analysis the same volume 20 μl was used.
Concerning flavonoids, the best separation of the constituents of studied extracts was obtained using the solvent system toluene / acetic acid / water (125:72:3) [13]. The revelation of different fluorescences was carried out under UV light at 365 nm after spraying with the NEU reagent (2 amino ethyl diphenylboric). The frontal report (Rf) was calculated as: The distance line-compound deposition on the line distance of solvent.
Characterization of terpenes and alkaloids
For terpenes, the benzene was used as a migration solvent. After migration, the silica gel plate was sprayed with antimony chloride, and then placed in an oven at 110°C for 10 min. Any fluorescence detected, under UV at 365 nm, after this treatment proves that the tested material contains terpenes (Randerath 1971). For alkaloids we used the solvent system consisting of (AcEt/MeOH/NH4OH) (9/1/1). The detection was carried out by spraying the Dragendorff reagent. The presence of alkaloids was shown by a bright orange color on the chromatogram [14].
Test for tannins
A few drops of 0.1% ferric chloride was added to 5 each organic extract and observed for brownish green or a blue-black coloration [15].
Statistical analysis
The data were statistically analysed using SPSS 16.0 software. We performed an analysis of variance (ANOVA). Student-Newman-Keuls tests were used to segregate treatments which were significantly different at P < 0.05.
Results and Discussion
Results of in vitro tests
Nematicidal activity of organics extract against Meloidogyne spp
The results presented in Table 3 show the nematicidal activity of organic extracts of four plant species belonging to three botanical families, against Meloidogyne spp, after 72 h evaluated at 20, 200 and 2000 ppm. Among the four species tested, three had some nematicidal activity. cc, A. imbricatus and L. dentata. While the fourth plant G. alypum showed no effect against Meloidogyne spp. Organics extracts of A. imbricatus showed the highest nematicidal activity (mortality rate ranging from 2,41 to 92,71%) followed by P. mauritanica plant who showed moderate activity (mortality rate ranging from 2,21 to 37.5%) and finally L. dentata who showed low activity (mortality rate ranging from 0,08 to 2,32%).
| Asteriscus imbricatus | Pulicaria mauritanica | Lavendula dentata | Globularia alypum | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Concentration (ppm) | PE | CHL | ACT | MT | PE | CHL | ACT | MT | PE | CHL | ACT | MT | PE | CHL | ACT | MT |
| 20 | 2,41c | 2,95c | -3,78b | -3,73b | 4,19b | 2,21b | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 200 | 45,2b | 45,88b | 4,71b | 5,57ab | 6,71b | 11,43b | 3,64b | 0 | 0,08 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2000 | 89,31a | 92,71a | 36,24a | 17,42a | 37,05a | 36,35a | 19,47a | 13,8a | 1,31 | 2,32 | 0,66 | 0 | 0 | 0 | 0 | 0 |
(PE): Petroleum ether; (CHL): Chloroform; (ACT): Ethyl acetate; (MT):Methanol; C: Concentration of organic extracts.
Table 3: In vitro effects of organic plants extracts on mortality rate (%) of Meloidogyne spp, after 72 h evaluated at 20,200 and 2000 ppm
The statistical analysis for each plant extract according to the concentration (results obtained in each column) showed a significant difference between the concentrations tested. Indeed the mortality rate of Meloidogyne spp increases with increasing concentration. In addition the interesting results were observed at 2000 ppm.
Each value represents the mean of three replicates. Means followed by different letters in each column are significantly different at P < 0.05 according to Newmanand Keuls test. In a second statistical analysis for each plant we evaluated the influence of the type of extract on the mortality of Meloidogyne spp at the same concentration of 2000 ppm (The results are represented in each column of Table 4).
| Concentration extracts (ppm) | Asteriscus imbricatus | Pulicaria mauritanica | Lavendula dentata | Globularia alypum |
|---|---|---|---|---|
| Petroleum ether | 89, 31a | 37,05a | 1,31a | 0 |
| Chloroform | 92, 71a | 36,35a | 2,32a | 0 |
| Ethyl acetate | 36, 24b | 19,47b | 0 | 0 |
| Methanol | 1,742c | 13,8b | 0 | 0 |
Each value represents the mean of three replicates. Means followed by different letters in each column are significantly different at P<0.05 according to Newmanand Keuls test.
Table 4: A mortality rate (%) of Meloidogyne spp at 2000 ppm depending on the type of organic extracts
A significant difference was observed between the extracts tested. The Newmanand Keuls test has shown the presence of at least two homogeneous groups for both Asteraceae plants: A. imbricatus and P. mauritanica. The first one is constituted by no polar extracts petroleum ether and chloroform. While the others groups are constituted by ethyl acetate and methanol extracts, which means that the mortality rate of Meloidogyne spp increased with decreasing polarity.
Effect of aqueous extracts against Meloidogyne spp
Table 5 shows the results of the effect of aqueous extracts of four plants studied on the mortality percentage of Meloidogyne spp after 72 hours, evaluated at different concentrations (20000, 2000, 200 and 20 ppm). Exposure of J2 larvae of Meloidogyne spp during 72 h to the aqueous extracts of these four plants at 20000 ppm, showed a different nematicidal effect: the higher nematicidal activity was detected with the aqueous extracts of A. imbricatus 78.76% of mortality, followed by the aqueous extract of P. mauritanica 9.5% of mortality while the two plants Lavandula dentata and G. alypum showed no nematicidal effect (Table 5).
| Concentration (ppm) | Asteriscus imbricatus | Pulicaria mauritanica | Lavendula dentata | Globularia alypum |
|---|---|---|---|---|
| 20 | 0c | 0b | 0 | 0 |
| 200 | 2,25c | 1,11a | 0 | 0 |
| 2000 | 14,99b | 1,94a | 0 | 0 |
| 20000 | 78,76a | 9,5a | 0 | 0 |
Each value represents the mean of three replicates. Means followed by different letters in each column are significantly different at P < 0.05 according to Newmanand Keuls test.
Table 5: In vitro effects of aqueous extracts on mortality rate (%) of Meloidogyne spp
Based on the results of in vitro tests, petroleum ether, chloroform and aqueous extracts of A. imbricatus were selected to testing their effect on the damage of tomato plants which is manifested by the appearance of galls on roots.
Results of in vivo test
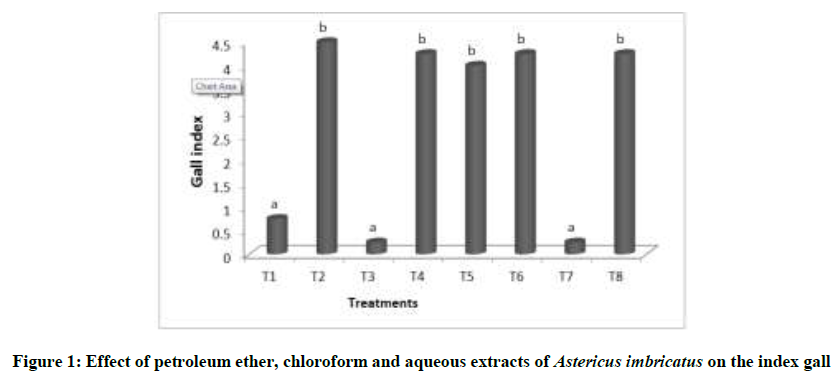
The effectiveness of various treatments against Meloidogyne spp in vivo was evaluated based on the index of gall. At the end of the test plants were uprooted and roots were carefully washed with tap water. The number of galls on roots of each treatment was counted. Then the gall index was determined based on a 0-5 scale [4]. A significant difference was observed between the treatments tested. The Newmanand Keuls test has shown the presence of two homogeneous group’s. the first one is constituted by petroleum ether and chloroform extracts at 6000 ppm in the addition of chemical nematicid. The second group contains the positive control and the other treatments (Figure 1).
Results of phytochemical screening
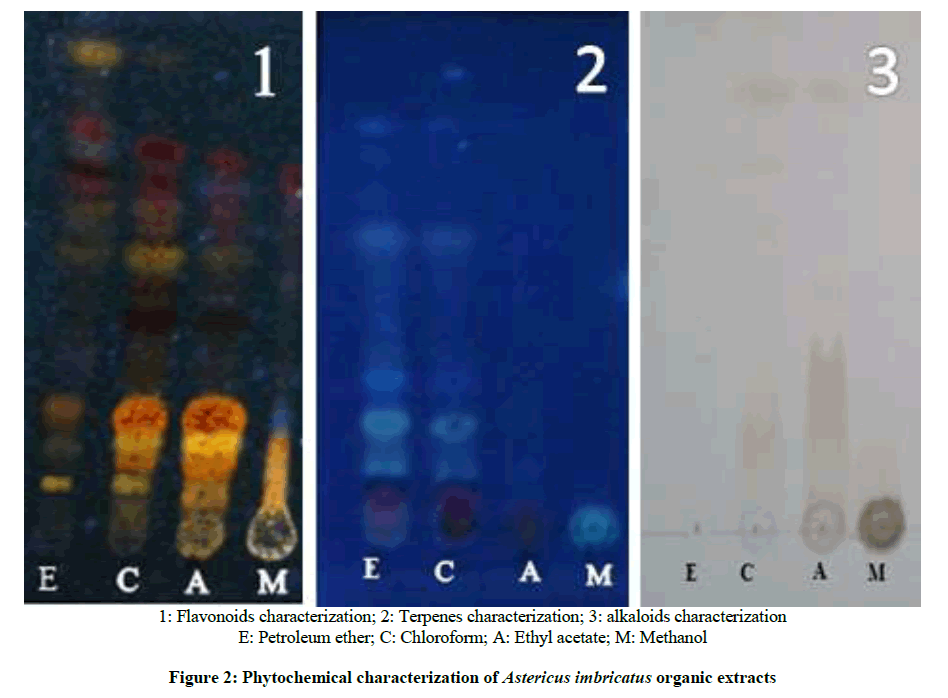
Flavonoids characterization using TLC
The results of flavonoids characterization of A. imbricatus organic extracts are represented in the Figure 2 (Picture 1). The analysis of TLC under UV light after revelation shows: Firstly the fluorescence appears in the four organic extracts, showing that they all contain flavonoids but in various proportions. The ethyl acetate extract has more bands with intense fluorescence compared to other extracts. Secondly this fluorescence is intensified when the polarity of the solvent increases, from petroleum ether chloroform and ethyl acetate extract, but it decrease for the methanol.
Terpenes and alkaloids characterization
For this plant, a blue fluorescence is observed in both petroleum ether extract and chloroform extract which means that these extracts contain terpenes while the two other extracts do not contain terpens Figure 2 (Picture 2). The presence of alkaloids is demonstrated by spraying the chromatogram with Dragendorff reagent. For A. imbricatus, we notice the appearance of orange spot in chloroform and ethyl acetate extracts, which indicate that these extracts contain the alkaloids. While the petroleum ether and methanol extracts showed no stain Figure 2 (Picture 3).
Tannins
The presence of tannins was observed at the ethyl acetate and methanol extracts which showed brownish green coloration. The results of the phytochemical screening are summarized in Table 6.
| Flavonoids | Terpenes | Alkaloids | Tannins | |
|---|---|---|---|---|
| Petroleum ether | + | + | - | - |
| Chloroform | + | + | + | - |
| Ethyl acetate | + | - | + | + |
| Methanol | + | - | - | + |
+: Present - : Absent
Table 6: Phytochemical constituents of Asteriscus imbricatus organic extracts
Discussion
The results of in vitro and in vivo experiments show that the A. imbricatus plant is more effective against Meloidogyne spp compared to the other species tested. These results are consistent with several previous researches that has shown the effectiveness of Asteraceae plants to controlling nematodes: Tagetes patula for example has long been known as nematicidal plant [16-18]. Nematicidal activity of Artemisia genus was reported by some studies [19]. The effectiveness of plants against nematodes is due to their richness in secondary metabolites. Indeed several previous researchers proved nematicidal activity of diverse classes of these compounds: Saponins [20], alkaloids [21,22], phenolics [23], polyacetylenes, fatty acids, terpenoids [24,25], and others. For Asteraceae plants some nematicidal compounds have been isolated. The most studied are Polythienyls, especially α-terthienyl isolated from Tagetes species [26-29]. For this reason, the A. imbricatus organic extracts of this plant have been phytochemically screened to see what are the compounds contained in these extracts and how they relate to the activity studied. The results of this study showed that flavonoids are present in the four organic extracts of A. imbricatus. The terpenes are present only in petroleum ether and chloroform extracts, the alkaloids are present in chloroform and ethyl acetate extract and finally tannins are present in ethyl acetate and methanol extracts. The analysis of all the results revealed a correlation between the phytochemical content of these extracts and the nematicidal activity. Indeed a remarkable correlation appears between the nematicidal power of the extracts and the presence of the terpenes: the two extracts of petroleum ether and chloroform which contain the terpenes gave a strong nematicidal activity, whereas this activity was very low for the two extracts d ethyl acetate and methanol which do not contain. Terpenes have been reported to possess nematicidal properties: β-sitosterol, stigmasterol, daucosterol, carvacrol, thymol, geraniol, camphor and linalool [29], Gossypol, hemigossypol, methoxygossypol, methoxyhemigossypol dimethoxygossypol [25]. Borneol, carvedel, citral, geraniol, and α-terpineol [30].
Our phytochemical study revealed also the presence of flavonoids in petroleum ether and chloroform which have given a better nematicidal effect. These results suggest that flavonoids may contribute to nematicidal effect. In this context several works in the literature have proved the effectiveness of these metabolites against nematodes: Flavonoids are synthesized by leguminous plants in response to infection by nematode [31]. glyceolline inhibits the oxidation, respiration and motility of Meloidogyne incognita [32]. González et al. have shown that the (E) -chalcone (trans-1,3-diphenylpropenone) inhibited the eggs hatching of Globodera pallida et Globodera rostochiensis. isoflavonoïds like quercétin are nématotoxic to Meloidogyne incognita [33,34].
Conclusion
Nematicidal In vitro and in vivo tests showed that both petroleum ether extracts and chloroform of A. imbricatus plant have an interesting nematicidal power. The phytochemical screening, realized by chromatographic characterization, showed the richness of these two extracts into different secondary metabolites essentially terpenes and flavonoids. Who are probably responsible for this activity. To confirm this hypothesis, further studies are needed to complete our results, especially the evaluation of the isolated and combined effect of these metabolites against Meloidogyne spp in vitro and in vivo. It would also be interesting to test these extracts at greenhouse and field stages to evaluate their effectiveness against nematodes in soil, and based on the results obtained, the plant could be recommended for use in agriculture in the future. Finally the results of this work, encourage the investigations of aromatic and medicinal plants from the Souss-Massa region.
References
- J. Sasser, J Nematology., 1977, 9, 1, 26.
- V. C. Blok, J. T. Jones, M. S. Phillips, D. L. Trudgill, Bio Essays., 2008, 30, 249.
- ]C. Netscher, Les nématodes parasites des cultures maraîchères au Sénégal, Cahiers ORSTOM Série Biologie., 1970, 11, 209.
- J. Sasser, M. Kirby, 1979.
- A. T. Ploeg, M. S. Phillips, Nematology., 2001, 3, 151.
- Y. Oka, Nematology., 2001, 3, 2, 159.
- M. Takasugi, Y. Yachida, M. Anetai, T. Masamune, K. Kegasawa, Chemistry Letters., 1975, 4, 1, 43.
- I. Talibi, L. Askarne, H. Boubaker, E. Boudyach, F. Msanda, B. Saadi, A. Aoumar, Crop Protection., 2012, 35, 41.
- N. Dohou, K. Yamni, K. Badoc, A. Douira, Bull. Soc. Pharm Bord., 2004, 143, 31.
- S. Harnett, V. Oosthuizen, M. Van de Venter, J Ethnopharmaco., 2005, 96, 1, 113.
- O. Senhaji, M. Faid, M. Elyachioui, M. Dehhaoui, J Medical Mycology., 2005, 15, 4, 220.
- W. Abbott, J. Econ. Entomol., 1925, 18, 2, 265.
- K. R. Markham, Techniques of flavonoid identification, London: Academic press, 1982, 31.
- K. Randerath, Chromatographie sur couches minces, Paris, 1971, Édition Gauthier-Villars, 337.
- G. Ayoola, H. Coker, S. Adesegun, A. Adepoju-Bello, K. Obaweya, E. Ezennia, T. Atangbayila, Tropical. J. Pharm. Res., 2008, 7, 1019.
- J. Good, N. Minton, C. Jaworski, Phytopathology., 1965, 55, 9, 1026.
- G. Poinar, P. Johnson, How to neutralize nematodes. Rodale's organic gardening, USA, 1986.
- L. Reynolds, J. Potter, B. Ball-Coelho, Agronomy J., 2000, 92, 5, 957.
- E.H. Mayad, Z. Ferji, L.M. Idrissi, B. Chebl, Reviews in Biol Biotechnol., 2006, 5, 2, 37.
- M. P. Argentieri, T. D’Addabbo, A. Tava, A. Agostinelli, M. Jurzysta, P. Avato, Europ. J. Plant. Pathology., 2008, 120, 2, 189.
- B. Zhao, J. Chem. Ecol., 1999, 25, 10, 2205.
- T. Thoden, M. Boppré, Nematology., 2010, 12, 1, 1.
- R. Mahajan, D. Kaur, K. Bajaj, Nematologia Mediterranea. 1992., 20, 2, 217.
- J. Veech, J Nematology., 1977, 11, 3, 240.
- N. Ntalli, F. Ferrari, I. Giannakou, U. Menkissoglu Spiroudi, Pest Management Science., 2011, 67, 3, 341.
- C. Wat, S. Prasad, E. Graham, S. Partington, T. Arnason, G. Towers, J. Lam, Biochemical Systematics and Ecology., 1981, 9, 1, 59.
- F. Gommers, J. Bakker, Diseases of Nematodes., 1988, 1, 3, 22.
- D.J. Chitwood, Annual review of phytopathology., 2002, 40, 1, 221.
- P. Ohri, S. K. Pannu, Environmental Res. Development., 2009, 4, 171.
- S. Echeverrigaray, J. Zacaria, R. Beltrão, Phytopathology., 2010, 100, 199.
- P. Hutangura, U. Mathesius, M. G. Jones, B. Rolfe, Funct. Plant Biol, 1999, 26, 221.
- D. Kaplan, N. Keen, I. Thomason, Physiological Plant Pathol., 1980, 16, 309.
- M. Begum, M. Sivakumar, Nematol., 2004, 15, 73.
- T. Tsay, S. Wu, Y. Lin, J. Nematol., 2004, 36, 1, 36.