Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 4
New pyrazolo[3,4-b]pyridines: Synthesis and antimicrobial Activity
Shawkat A Abdel-Mohsen* and Talaat I El-Emary
Chemistry Department, Faculty of Science, Assiut University, Assiut-71516, Egypt
- *Corresponding Author:
- Shawkat A Abdel-Mohsen
Chemistry Department
Faculty of Science
Assiut University
Assiut-71516, Egypt
Abstract
Basic hydrolysis of the starting 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile 1, afforded the corresponding carboxylic acid 2, which was in turn converted to its methyl ester 3 and subsequently reacted with hydrazine hydrate to afford the corresponding 5- carbohydrazide 4. Compound 4 was used as a key intermediate for the synthesis of some novel series of pyrazolo[3,4-b]pyridines and related heterocycles such as pyrazoles 6-8, oxadiazoles 9-12, thiadiazoles 16, triazoles 13, 15, 17a-c, triazolothiadiazoles 18a-c and triazolothiadiazines 19a,b. The structures of all the new compounds have been established on the basis of their analytical and spectral data. Twenty two of the synthesized compounds were also evaluated for their antibacterial and antifungal activity using Streptomycin and Clotrimazole as the reference drugs.
Keywords
Pyrazolo[3,4-b]pyridines, Oxadiazoles, Thiadiazoles, Triazoles, Antimicrobial activity
Introduction
Pyrazolopyridine and related fused heterocycles are of interest as potential bioactive molecules. They are known to exhibit pharmacological activities such as CNS depressant [1,2], neuroleptic [3] and turberculostatic [4]. Pyrazolo[3,4-b]pyridines were reported as antimicrobial agents [5], inhibitors of Glycogen Synthase Kinase-3 (GSK-3) [6] and potent antitumor agents [7]. Also, pyrazolo[3,4-d]pyrimidines were identified as a general class of adenosine receptors [8-10]. They show analgesic and antinociceptive activity [11]. Also, they act as selective serotonin reuptake inhibitors [12], corticotrpin-releasing factor (CRF) antagonists in treating cardiovascular diseases osteoporosis ulcer [13] and they are effective in the treatment alzheimers disease, gastrom-testinal disease, hemorrhagic stress, drug addiction and infertility [14]. pyrazolo[3,4- b]pyridine were believed to be effective as antileishmanial agents [15], inhibitors of erectile dysfunctions [16]. Taking all the above into consideration and in continuation of our previous work directed to synthesis of new heterocycles engaged with pyrazole nuclei [17-21], we describe herein the utilization of the 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide 4 as the key intermediate in the synthesis of novel pyrazolo[3,4-b]pyridine and their related heterocycles along with studies of the effect of some of them as antibacterial and as antifungal agents.
Experimental
Melting points are uncorrected and determined using a Gallenkamp melting point apparatus. IR spectra were recorded on a Pye-Unicam SP 3- 100 spectrophotometer using the KBr Wafer technique. 1H-NMR spectra were recorded on a Varian EM-390 90 MHz spectrometer and on GNM-LA (400 MHz) in DMSO-d6 as a solvent and TMS as internal standard. Chemical shifts are expressed in ppm. 13C-NMR spectra were measured on a Varian EM-200, 100 MHz spectrometer. Mass spectra were determined on a JEOL JMS-600 spectrometer. Elemental analyses were carried out in the Microanalytical Unit at Assiut University (Egypt). The results were found to be in good agreement (±0.4%) with the calculated value. Compound 1 was prepared according to reported procedure [23].
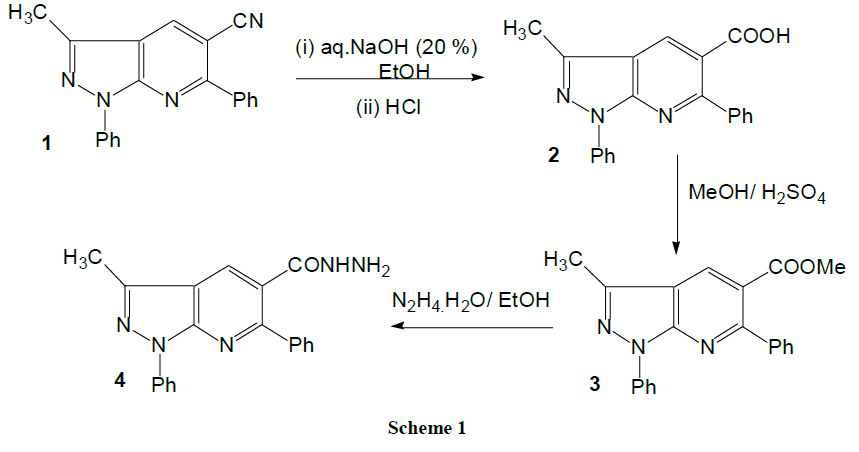
Synthesis of 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carboxylic acid (2): A mixture of compound 1 (3.1 g, 0.01 mol) and aqueous sodium hydroxide (40 ml 10%) and ethanol (40 ml) was heated under reflux for 3 h. The reaction mixture was filtered while hot and the filtrate was cooled and then neutralized with diluted 6 N HCl. The solid precipitate that formed was filtered off, washed with water and crystallized from dioxane-water (3:1) to give pale yellow needles. M.p. 185-187°C, yield (2.50 g, 81%). IR: νmax cm-1: 3450-3250 (br, OH), 3070 (CH-aromatic), 2995 (CH-aliphatic), 1715 (C=O). 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.77 (s, 3H, CH3), 5.45 (s, 1H, OH), 7.23-8.08 (m, 10H, Ar-H), 8.30 (s, 1H, H-pyridine); MS m/z: 284.11[M+-COOH, 67%]; Anal. Calcd. for C20H15N3O2 (329.35): C, 72.94; H, 4.59; N, 12.76. Found: C, 72.61; H, 4.70; N, 12.49.
Synthesis of ethyl 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carboxylate (3): A mixture of compound 2 (6.6 g, 0.02 mol), absolute methanol (50 ml) and concentrated sulfuric acid (1.5 ml) was refluxed for 3 h. The reaction mixture was concentrated under reduced pressure, cooled and then poured onto a cold solution of NaHCO3. The solid product was filtered, washed with water, dried and recrystallized from ethanol to give pale yellow flakes. M.p. 98-100°C, yield (4.8 g, 70%). IR: νmax cm-1: 3065 (CH-aromatic), 2987 (CH-aliphatic), 1710 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.80 (s, 3H, CH3), 3.85 (s, 1H, CH3), 7.23-7.90 (m, 10H, Ar-H), 8.15 (s, 1H, H-pyridine); Anal. Calcd for C21H17N3O2 (343.38): C, 73.45; H, 4.99; N, 12.24. Found: C, 73.30; H, 5.13; N, 12.01.
Synthesis of 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (4): Method A: A mixture of the ester 3 (6.9 g, 0.02 mol) and excess hydrazine hydrate (6 ml) was heated under reflux for 2 h. The solid precipitate that formed was filtered off, washed with water and recrystallized from ethanol to give shiny yellow needles. M.p. 240-242°C, yield (5.20 g, 75%). IR: νmax cm-1: 3250, 3170 (NHNH2), 3055 (CHaromatic), 2899 (CH-aliphatic), 1666 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.74 (s, 3H, CH3), 4.25 (s, 2H, NH2), 7.30-8.09 (m, 11H, 10 H, Ar-H+ H-pyridine), 9.33 (s, 1H, NH); 13C-NMR (100 MHz, DMSO-d6), δ(ppm): 14.5 (CH3), 109.3 (C), 118.7 (2CH), 121.3 (C), 124.6 (CH), 126.2 (2CH), 127.6 (CH), 128.7 (2CH), 129.9 (2CH), 134.5 (C), 138.7 (C), 140.2 (CH), 143.2 (C), 150.1 (C), 158.9 (C), 166.3 (C=O). MS m/z: 343.41 [M+, 38]; Anal. Calcd. for C20H17N5O (343.38): C, 69.96; H, 4.99; N, 20.40. Found: C, 70.28; H, 4.89; N, 20.16.
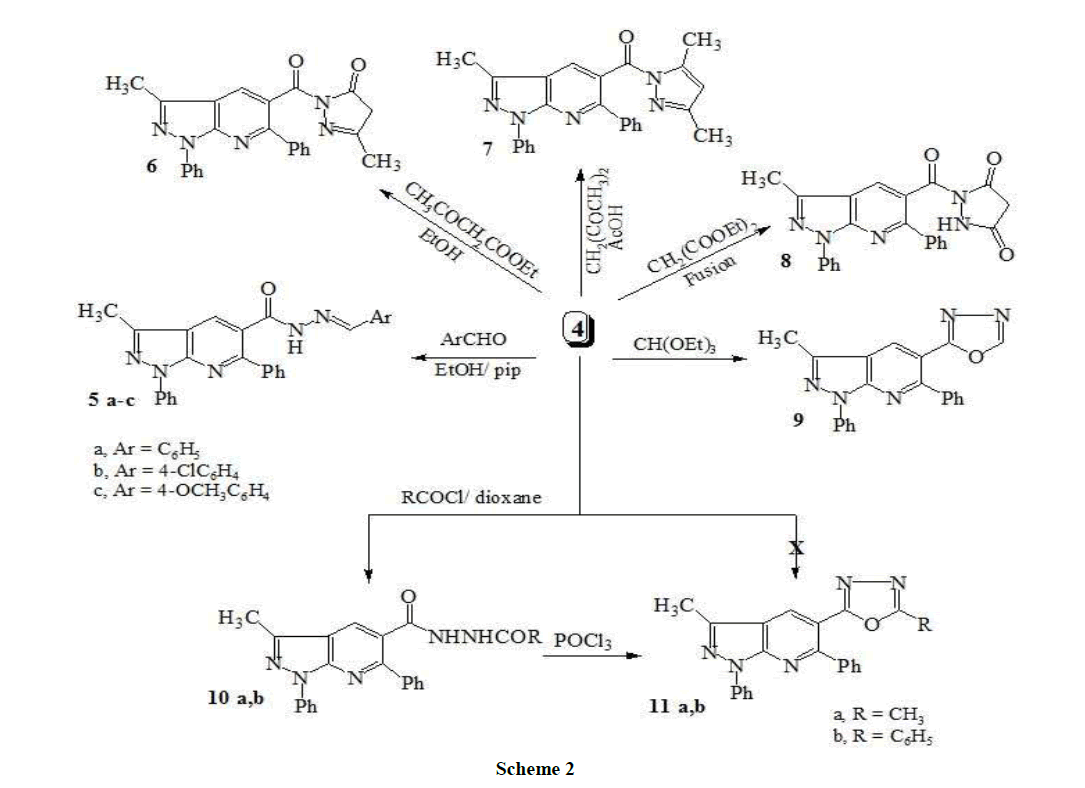
General procedure for the synthesis of N-arylidene-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (5a-c)
A mixture of 4 (0.69 g, 0.002 mol) the appropriate aromatic aldehyde (0.002 mol) was stirred under reflux in ethanol (30 ml) in the presence of a few drops of piperidine for 5 h. The reaction mixture was allowed to cool to room temperature, poured into water, whereby a solid formed that was filtered off and crystallized from an appropriate solvent.
N-benzylidene-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (5a): Yellow crystals. M.p. 182-184°C, yield (0.59 g, 66%). IR: νmax cm-1: 3280 (NH), 3047 (CH-aromatic), 2980, 2890 (CH-aliphatic), 1667 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.73 (s, 3H, CH3), 7.16-7.80 (m, 15H, Ar-H), 8.28 (s, 1H, pyridine-H), 8.45 (s, 1H, N=CH). Anal. Calcd for C27H21N5O (431.49): C, 75.16; H, 4.91; N, 16.23. Found: C, 74.94; H, 5.02; N, 16.61.
N-(4-chlorobenzylidene)-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (5b): Yellow crystals. M.p. 312-314°C, yield (0.68 g, 70%). IR: νmax cm-1: 3335 (NH), 3089 (CH-aromatic), 2976, 2849 (CH-aliphatic), 1674 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.76 (s, 3H, CH3), 7.20-7.90 (m, 14H, Ar-H), 8.11 (s, 1H, pyridine-H), 8.38 (s, 1H, N=CH). Anal. Calcd for C27H20ClN5O (465.93): C, 69.60; H, 4.33; Cl, 7.61; N, 15.03. Found: C, 69.91; H, 4.48; Cl, 7.49; N, 14.88.
N-(4-methoxybenzylidene)-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (5c): Yellow crystals. M.p. 202-204°C, yield (0.61 g, 64%). IR: νmax cm-1: 3365 (NH), 3077 (CH-aromatic), 2982, 2884 (CH-aliphatic), 1682 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.69 (s, 3H, CH3), 3.45 (s, 3H, OCH3); 7.23-8.12 (m, 15H, 14H, Ar-H+ 1H, pyridine-H), 8.42 (s, 1H, N=CH). Anal. Calcd. for C28H23N5O2 (461.51): C, 72.87; H, 5.02; N, 15.17. Found: C, 73.21; H, 5.20; N, 14.93.
5-Methyl-1-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbonyl)-2,4-dihydropyrazol-3-one (6): Ethyl acetoacetate (0.25 g, 0.002 mol) was added to a solution of the acid hydrazide 4 (0.69 g, 0.002 mol) in absolute ethanol (15 ml) and the reaction mixture was heated under for 4 h. Solvent was removed under reduced pressure and the residue obtained was collected and recrystallized from benzene to give pale yellow crystals. M.p. 177-179°C, yield (0.61 g, 69%). IR: νmax cm-1: 3055 (CH-aromatic), 2969 (CH-aliphatic), 1686 (C=O), 1665 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.35 (s, 3H, CH3), 2.68 (s, 3H, CH3), 3.15 (s, 2H, CH2), 7.08-7.95 (m, 10H, Ar-H), 8.32 (s, 1H, pyridine-H); Anal. Calcd for C24H19N5O2 (409.44): C, 70.40; H, 4.86; N, 17.10. Found: C, 70.73; H, 4.78; N, 17.38.
(3,5-Dimethyl-pyrazol-1-yl)-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-yl)-methanone (7): To a solution of acid hydrazide 4 (0.69 g, 0.002 mol) in glacial acetic acid (12 ml) was added acetylacetone (0.20 g, 0.002 mol). The reaction mixture was heated under reflux for 8 h and then allowed to cool. The solid product thus obtained was filtered, washed thoroughly with cold ethanol, dried and recrystallized from dioxane.
1-(1,6-Diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbonyl)-pyrazolidine-3,5-dione (8): A mixture of 4 (0.69 g, 0.002 mol) and diethyl malonate (0.48 g, 0.003 mol) was heated at 200 C in an oil bath for 2 h. After cooling, the solidified product was treated with cold diethyl ether, filtered, washed with diethyl ether, dried and recrystallized from ethanol to give plae light solid. M.p. 230-232°C, yield (0.63 g, 68%). IR: νmax cm-1: 3320 (NH), 3065 (CH-aromatic), 2964 (CH-aliphatic), 1710 (C=O), 1700 (C=O), 1688 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.70 (s, 3H, CH3), 6.15 (3.50 (s, 2H, CH2), 7.10-7.95 (m, 10H, Ar-H), 8.39 (s, 1H, pyridine-H), 10.12 (s, 1H, NH). Anal. Calcd. for C23H17N5O3 (411.41): C, 67.15; H, 4.16; N, 17.02. Found: C, 67.37; H, 4.42; N, 16.83.
3-Methyl-5-(1,3,4-oxadiazol-2-yl)-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine (9): A mixture of 4 (0.69 g, 0.002 mol) and triethylorthoformate (10 ml) was heated under reflux for 9 h. After cooling, the solvent was evaporated under reduced pressure and the solid product obtained was filtered off and recrystallized from ethanol to give colorless crystals. M.p. 219-221°C, yield (0.48 g, 68%). IR: νmax cm-1: 3050 (CH-aromatic), 2969 (CH-aliphatic), 1640 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.78 (s, 3H, CH3), 7.19-7.93 (m, 11H, 10 H, Ar-H + 1H, oxadiazole - H), 8.33 (s, 1H, pyridine-H). Anal. Calcd. for C21H15N5O (353.38): C, 71.38; H, 4.28; N, 19.82. Found: C, 71.84; H, 4.49; N, 19.51.
General procedure for the synthesis of N2-acetyl(benzoyl)-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (10a,b) To a solution of 4 (069 g, 0.002 mol) in dioxane (15 ml) acetyl or benzoylchloride (0.002 mol) was added. The reaction mixture was refluxed for 4 h, then the solvent was removed under reduced pressure and the residue was triturated with an ice-water mixture. The solid product obtained was filtered off and recrystallized from ethanol.
N2-acetyl-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (10a): White crystals. M.p. 190-192°C, yield (0.64 g, 79%). IR: νmax cm-1: 3300 (NH), 3030 (CH-aromatic), 2977 (CH-aliphatic), 1705 (C=O), 1679 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 1.95 (s, 3H, CH3), 2.75 (s, 3H, CH3), 7.27-7.97 (m, 10H, Ar-H), 8.19 (s, 1H, pyridine-H), 10.52 (s, 1H, NH), 10.30 (s, 1H, NH); Anal. Calcd. for C22H19N5O2 (385.42): C, 68.15; H, 4.97; N, 18.17. Found: C, 67.89; H, 5.11; N, 18.36.
N2-benzoyl-1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (10b): White crystals. M.p. 206-208°C, yield (0.74 g, 81%). IR: νmax cm-1: 3390 (NH), 3048 (CH-aromatic), 2988 (CH-aliphatic), 1692 (C=O), 1680 (C=O); 1 H NMR (DMSO-d6, 90 MHz): δppm: 2.66 (s, 3H, CH3), 7.17-7.98 (m, 15H, Ar-H), 8.27 (s, 1H, pyridine-H), 10.60 (s, 1H, NH), 11.20 (s, 1H, NH); Anal. Calcd. for C27H21N5O2 (447.49): C, 72.47; H, 4.73; N, 15.65. Found: C, 72.69; H, 4.88; N, 15.89.
General procedure for the synthesis of 1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-(5-substituted-[1,3,4]oxadiazol-2-yl) (11a,b) A solution of 10a,b (0.003 mol) in phosphorous oxychloride (10 ml) was heated under reflux at 100°C for 4 h. After cooling, the solvent was removed in vacuo and the residue was poured into an ice-water mixture and neutralized with ammonium hydroxide (20%). The solid product obtained was collected by filtration and recrystallized from ethanol.
1,6-Diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-(5-methyl-[1,3,4]oxadiazol-2-yl) (11a): white crystals. M.p. 116-118°C, yield (0.74 g, 81%). IR: νmax cm-1: 3017 (CH-aromatic), 2973 (CH-aliphatic), 1625 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.55 (s, 3H, CH3), 2.71 (s, 3H, CH3), 7.07-7.92 (m, 10H, Ar-H), 8.19 (s, 1H, pyridine-H); MS m/z: 367.19 [M+, 100%]; Anal. Calcd. for C22H17N5O (367.40): C, 71.92; H, 4.66; N, 19.06. Found: C, 71.76; H, 4.82; N, 19.31.
1,6-Diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-(5-phenyl-[1,3,4]oxadiazol-2-yl) (11b): white crystals. M.p. 98-100°C, yield (0.68 g, 78%). IR: νmax cm-1: 3035 (CH-aromatic), 2994 (CH-aliphatic), 1637 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.78 (s, 3H, CH3), 7.11-7.99 (m, 15H, Ar-H), 8.22 (s, 1H, pyridine-H); Anal. Calcd. for C27H19N5O (429.47): C, 75.51; H, 4.46; N, 16.31. Found: C, 71.70; H, 4.29; N, 16.53.
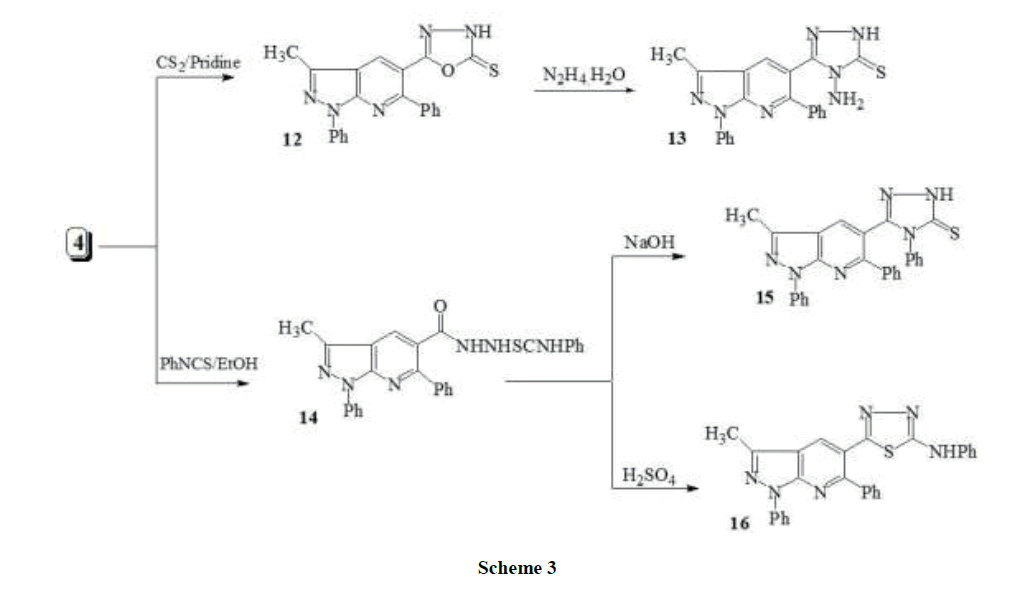
5-(3-Methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-1,3,4-oxadiazole-2(3H)-thione (12): A mixture of 4 (0.69 g, 0.002 mol) and carbon disulfide (6 ml) in pyridine (10 ml) was heated under reflux on water bath for 9 h. After cooling, the solvent was evaporated under reduced pressure and the residue was triturated with an ice-water mixture and neutralized with diluted HCl. The solid precipitate formed was filtered off and recrystallized from ethanol as pale yellow crystals. M.p. 262-264°C, yield (0.70 g, 91%). IR: νmax cm-1: 3019 (CH-aromatic), 2977 (CH-aliphatic), 1620 (C=N), 1210 (C=S). 1 H NMR (DMSO-d6, 90 MHz): δppm: 2.75 (s, 3H, CH3), 7.20-7.97 (m, 10H, Ar-H), 8.19 (s, 1H, pyridine-H), 10.33 (s, 1H, NH). Anal. Calcd. for C21H15N5OS (385.44): C, 65.44; H, 3.92; N, 18.17; S, 8.32. Found: C, 65.83; H, 4.11; N, 18.03; S, 8.10.
4-amino-5-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione (13): A suspension of 12 (0.38 g, 0.001 mol) and hydrazine hydrate (2 ml, 0.04 mol) in ethanol (10 ml) was refluxed for 6 h. After cooling, the precipitate formed was collected and recrystallized from ethanol to afford yellow crystals. M.p. 154-156°C, yield (0.30 g, 76%). IR: νmax cm-1: 3290, 3230, 3100 (NH, NH2), 3024 (CH-aromatic), 2993 (CH-aliphatic), 1631 (C=N), 1228 (C=S); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.70 (s, 3H, CH3), 5.25 (s, 2H, NH2), 7.13-7.95 (m, 10H, Ar-H), 8.21 (s, 1H, pyridine-H), 12.70 (s, 1H, NH). Anal. Calcd. for C21H17N7S (399.47): C, 63.14; H, 4.29; N, 24.54; S, 8.03. Found: C, 63.44; H, 4.02; N, 24.31; S, 8.29.
N-Phenyl-2-[(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridin-5-yl)carbonyl]hydrazine- Carbothioamide (14): A mixture of acid hydrazide 4 (1.37 g, 0.004mol) and phenyl isothiocyanate (0.54 g, 0.004 mol) in absolute ethanol (20 ml) was heated under reflux for 8 h. After cooling to room temperature, the reaction mixture was then poured into ice-cold water containing a few drops of acetic acid. The resulting precipitate was collected by filtration, washed with water, dried and recrystallized from diluted ethanol gave a yellow crystals. M.p. 177-179°C, yield (1.6 g, 79%). IR: νmax cm-1: 3438, 3398, 3336 (NH), 3092 (CH-aromatic), 2907 (CH-aliphatic), 1677 (C=O); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.65 (s, 3H, CH3), 7.10-7.92 (m, 15H, Ar-H), 8.33 (s, 1H, pyridine-H), 8.65 (s, 1H, NH), 9.50 (s, 1H, NH), 10.15 (s, 1H, NH); Anal. Calcd. for C27H22N6OS (478.57): C, 67.76; H, 4.63; N, 17.56; S, 6.70. Found: C, 68.09; H, 4.48; N, 17.28; S, 6.92.
5-(1,6-Diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-4-phenyl-2H-1,2,4-triazole-3(4H)-thione (15): Compound 14 (0.47 g, 0.001 mol), in an ethanolic sodium hydroxide solution 4% (10 ml), was heated under reflux for about 4 h. The solvent was concentrated, diluted with water and neutralized with diluted HCl at 0-5°C. The crude product was filtered off, washed with water and recrystallized from ethanol to give white crystals. M.p. 149-151°C, yield (0.35 g, 77%). IR: νmax cm-1:3345 (NH), 3076 (CH-aromatic), 2959 (CH-aliphatic), 1637(C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.69 (s, 3H, CH3), 7.11-7.98 (m, 15H, Ar-H), 8.41 (s, 1H, pyridine-H), 13.30 (s, 1H, NH); Anal. Calcd. for C27H20N6S (460.55): C, 70.41; H, 4.38; N, 18.25; S, 6.96. Found: C, 70.70; H, 4.56; N, 17.91; S, 6.73.
N-Phenyl-5-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-1,3,4-thiadiazol-2-amine (16): Compound 14 (0.47 g, 0.001 mol) was added gradually with stirring to an ice-cold concentrated sulphuric acid (5 ml) and the reaction mixture was further stirred for 4 h in an ice bath. It was then poured into crushed ice and the resulting solution was adjusted to pH 7-8 with ammonium hydroxide solution. The solid formed was filtered off and recrystallized from ethanol/dioxane (1:1) to give yellow crystals. M.p. 222-224°C, yield (0.35 g, 77%). IR: νmax cm-1: 3200 (NH), 3030 (CH-aromatic), 2991 (CH-aliphatic), 1635 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.781 (s, 3H, CH3), 7.23-8.03 (m, 15H, Ar-H), 8.29 (s, 1H, pyridine-H), 9.36 (s, 1H, NH); Anal. Calcd. for C27H20N6S (460.55): C, 70.41; H, 4.38; N, 18.25; S, 6.96. Found: C, 70.73; H, 4.19; N, 18.62; S, 76.14.
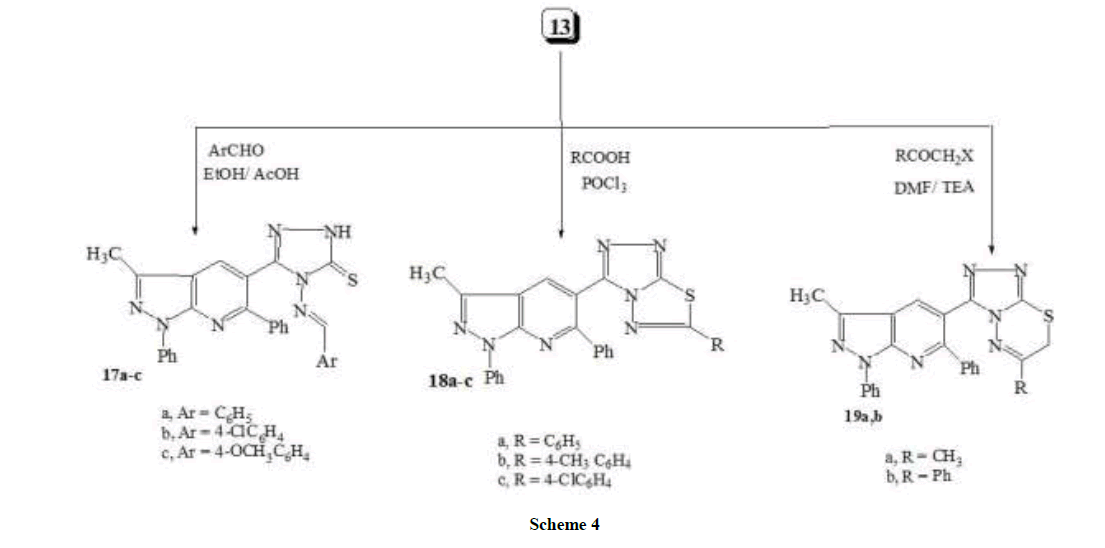
General procedure for the synthesis of 4-(arylideneamino)-5-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2H-1,2,4-triazole- 3(4H)-thione (17a-c)
To a mixture of 13 (0.80 g, 0.002 mol) and the appropriate aldehyde (0.002 mol), in EtOH (15 ml), few drops of glacial acetic acid were added. The reaction mixture was refluxed for 6 h then left to cool. The precipitated product was filtered off, washed with EtOH, dried and finally recrystallized from ethanol to afford the corresponding Schiff bases 17a-c.
4-(Benzylideneamino)-5-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2H-1,2,4-triazole-3(4H)-thione (17a): Pale yellow crystals. M.p. 245-247°C, yield (0.75 g, 77%). IR: νmax cm-1: 3275 (NH), 3010 (CH-aromatic), 2990 (CH-aliphatic), 1649 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.75 (s, 3H, CH3), 7.10-7.89 (m, 15H, Ar-H), 8.11 (s, 1H, CH-imine), 8.23 (s, 1H, pyridine-H); Anal. Calcd. for C28H21N7S (487.58): C, 68.97; H, 4.34;N, 20.11; S, 6.58. Found: C, 68.78; H, 4.44; N, 20.27; S, 6.43.
4-(4-Chlorobenzylideneamino)-5-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2H-1,2,4-triazole-3(4H)-thione (17b): Pale yellow crystals. M.p. 206-208°C, yield (0.66 g, 63%). IR: νmax cm-1: 3239 (NH), 3035 (CH-aromatic), 2982 (CH-aliphatic), 1636 (C=N); 1HNMR (DMSO-d6, 90 MHz): δppm: 2.68 (s, 3H, CH3), 7.08-8.0 (m, 14H, Ar-H), 8.10 (s, 1H, CH-imine), 8.19 (s, 1H, pyridine-H); Anal. Calcd. for C28H20ClN7S (522.02): C, 64.42; H, 3.86;Cl, 6.79; N, 18.78; S, 6.14. Found: C, 64.75; H, 3.77; Cl, 6.57; N, 19.13; S, 6.26.
4-(4-Methoxybenzylideneamino)-5-(1,6-diphenyl-3-methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2H-1,2,4-triazole-3(4H)-thione (17c): yellow crystals. M.p. 311-313°C, yield (0.84g, 81%). IR: νmax cm-1: 3315 (NH), 3048 (CH-aromatic), 2987 (CH-aliphatic), 1627 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.66 (s, 3H, CH3), 3.77 (s, 3H, O-CH3), 6.96-7.98 (m, 14H, Ar-H), 8.09 (s, 1H, CH-imine), 8.24 (s, 1H, pyridine- H); Anal. Calcd. for C29H23N7OS (517.60): C, 67.29; H, 4.48; N, 18.94; S, 6.19. Found: C, 67.66; H, 4.33; N, 18.67; S, 6.37.
General procedure for the synthesis of 3-methyl-1,6-diphenyl-5-(6-substiuted-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazol-3-yl)-1H-pyrazolo[ 3,4-b]pyridines (18a-c) A mixture of compound 13 (0.80 g, 0.002 mol) and aromatic carboxylic acid (0.002 mol) was added to dry phosphorous oxy chloride (10 ml) of and the solution was refluxed on water bath for 6 h. Excess of phosphorous oxychloride was removed under vacuum. The thick mass obtained was treated with water and left overnight. Solid thus obtained was filtered washed with 2% sodium carbonate solution, then with cold water, dried and recrystallized from ethanol.
3-Methyl-1,6-diphenyl-5-(6-phenyl-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazol-3-yl)-1H-pyrazolo[3,4-b]pyridines (18a): Yellow crystals. M.p. 212-214°C, yield (0.64g, 66%). IR: νmax cm-1: 3048 (CH-aromatic), 2914 (CH-aliphatic), 1578 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.69 (s, 3H, CH3), 6.89-8.10 (m, 15H, Ar-H), 8.25 (s, 1H, pyridine-H); Anal. Calcd. for C28H19N7S (485.56): C, 69.26; H, 3.94; N, 20.19; S, 6.60. Found: C, 69.50; H, 4.05; N, 19.94; S, 6.47.
3-Methyl-1,6-diphenyl-5-(6-(4-tolyl)-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazol-3-yl)-1H-pyrazolo[3,4-b]pyridines (18b): Yellow crystals. M.p. 287-289°C, yield (0.74 g, 74%). IR: νmax cm-1: 3070(CH-aromatic), 2955 (CH-aliphatic), 1601 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.45 (s, 3H, CH3), 2.71 (s, 3H, CH3), 6.95-8.00 (m, 14H, Ar-H), 8.20 (s, 1H, pyridine-H); Anal. Calcd. for C29H21N7S (499.59): C, 69.72; H, 4.24;N, 19.63; S, 6.42. Found: C, 70.07; H, 4.12; N, 19.44; S, 6.71.
3-Methyl-1,6-diphenyl-5-(6-(4-chlorophenyl)-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazol-3-yl)-1H-pyrazolo[3,4-b]pyridines (18c): Yellow crystals. M.p. 244-246°C, yield (0.75 g, 72%). IR: νmax cm-1: 3038(CH-aromatic), 2936 (CH-aliphatic), 1596 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.71 (s, 3H, CH3), 6.92-8.09 (m, 14H, Ar-H), 8.17 (s, 1H, pyridine-H); Anal. Calcd. for C28H18ClN7S (520.01): C, 64.67; H, 3.49;Cl, 6.82; N, 18.85; S, 6.17. Found: C, 64.38; H, 3.66; Cl, 7.04; N, 18.59; S, 5.89.
General procedure for the synthesis of 6-substituted-3-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-yl)-7H-[1,2,4]triazolo[3,4- b][1,3,4]thiadiazines (19a,b) A mixture of compound 13 (0.80 g, 0.002 mol), phenacyl bromide and/or chloroacetone (0.002 mol) and sodium acetate (0.4 g, 0.005 mol) in ethanol (25 ml) was refluxed for 5 h. The solvent was evaporated, the residue was poured onto water and the white solid product formed was filtered off and recrystallized from ethanol.
6-Methyl-3-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-yl)-7H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines (19a): yellow crystals. M.p. 166-168°C, yield (0.70 g, 80%). IR: νmax cm-1: 3040(CH-aromatic), 2966 (CH-aliphatic), 1633 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.71 (s, 3H, CH3), 3.15 (s, 3H, CH3), 5.35 (s, 2H, CH2), 6.90-7.90 (m, 10H, Ar-H), 8.31 (s, 1H, pyridine-H); Anal. Calcd. for C24H19N7S (437.52): C, 65.88; H, 4.38; N, 22.41; S, 7.33. Found: C, 66.17; H, 4.27; N, 22.22; S, 7.57.
6-Phenyl-3-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-yl)-7H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines (19b): yellow crystals. M.p. 113-115°C, yield (0.77 g, 77%). IR: νmax cm-1: 3065 (CH-aromatic), 2987 (CH-aliphatic), 1636 (C=N); 1H-NMR (DMSO-d6, 90 MHz): δppm: 2.75 (s, 3H, CH3), 4.45 (s, 2H, CH2), 6.90-8.05 (m, 15H, Ar-H), 8.27 (s, 1H, pyridine-H); Anal. Calcd. for C29H21N7S (499.59): C, 69.72; H, 4.24; N, 19.63; S, 6.42. Found: C, 69.39; H, 4.08; N, 19.85; S, 6.18.
Antimicrobial activity
Using the agar well-diffusion method 23, the antimicrobial activity of 22 new chemical compounds was tested in vitro against bacterial species obtained from contaminated soil, water and food substances such as Staphylococcus aureus [AUMC No. B-54] and Bacillus cereus [AUMC No. B-52], as Gram-positive bacteria and Escherichia coli [AUMC No. B-53], and Pseudomonas aeruginosa [AUMC No. B-73], as Gram-negative bacteria. They were also tested against four fungal species which are involved in human and animal diseases such as, man and animal diseases such as, Candida albicans [AUMC No.418], Trichophyton rubrum [AUMC No. 1804] and Aspergillus flavus [AUMC No. 3214], and or plant diseases (Fusarium oxysporum [AUMC No. 5119]. These strains are common contaminants of the environment in Egypt and some of all microbial strains were kindly provided by the Assiut University Mycological Centre (AUMC). To prepare inocula for bioassay, bacterial strains were individually cultured for 48 h in 100 ml conical flasks containing 30 ml nutrient broth medium. Fungi were grown for 7 days in 100 ml conical containing 30 mL Sabouraud's dextrose broth. Bioassay was done in 10 cm sterile plastic Petri plates in which microbial suspension (1 ml/plate) and 15 ml appropriate agar medium (15 ml/plate) were poured. Nutrient agar and Sabouraud's dextrose agar were respectively used for bacteria and fungi. After solidification of the media, 5 mm diameter cavities were cut in the solidified agar (4 cavities/plate) using sterile cork borer. Chemical compounds dissolved in DMSO at 2% w/v (=20 mg/ml) were pipetted in the cavities. The screening tests were carried out in triplicate and the results were expressed as a mean of three determinations.
Results and Discussion
This synthesis involves a cyclodehydration reaction of the readily available pyrazole aminoaldehyde [22] with benzoyl acetonitrile in refluxing pyridine to afford 3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile 1. The alkaline hydrolysis of 1 afforded the corresponding carboxylic acid derivative 2. The structure of synthesized compound 2 was established by 1H-NMR, IR, MS spectroscopes in combination with elemental analyses. The IR spectrum of 2 shows the following characteristic absorption bands at 3450-3250 (broad OH) and at 1715 (C=O, carboxyl) cm-1. In the 1H-NMR spectrum of compound 2, the CH pyridine ring is much deshielded and resonating at low field and appear at δ=8.30 ppm. The mass spectrum of compound 2 showed its correct parent ion peak at m/z 284.11 (M+-COOH), 67%). The corresponding methyl ester 3 was obtained via the treatment of the acid 2 with absolute methanol in the presence of concentrated sulfuric. The IR spectrum of compound 3 revealed the characteristic absorption band at 1738 cm-1 due to carbonyl ester. The synthesis of the key intermediate acid hydrazide 4 was achieved via the hydrazinolysis of the ester 3 by heating under reflux with ethanolic solution of hydrazine hydrate. The IR spectrum of compound 4 showed the presence of absorption bands at v=3250, 3170 cm-1, indicating the presence of NH2 and NH group. The MS spectrum revealed a signal at 343.41 (M+, 38), reinforced the formation of carbohydrazide 4 (Scheme 1).
Condensation of 4 was with different aromatic aldehydes in ethanol in the presence of piperidine to give the corresponding N-arylidene-3- methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-carbohydrazide (5a-c). The structure of compounds 5a-c was characterized by the disappearance of the NHNH2 group and revealed in each case band at 3425-3250 cm-1 assignable to NH groups. Also, their 1H-NMR spectra showed the presence of the azomethine and NH protons at 9.15-9.45 and 10.85-11.75 ppm, respectively. On the other hand, upon heating the acid hydrazide 4 with ethyl acetoacetate and acetylacetone, the N-pyrazolo derivatives 6 and 7 were produced. For example, the 1H-NMR spectrum for compound 7 revealed a singlet at 6.30 ppm due to the 4-H-pyrazole moiety. Its 13C-NMR spectral data displayed two characteristic singlets at 151.12 and 152.5 ppm for C3 and C5 of the pyrazole nucleus, respectively. Whereas, heating of 4 with diethylmalonate at 200°C afforded the targeted 1-[(3-methyl-1,6-dipheny-1H-pyrazolo[3,4-b]pyridine-5-yl)carbonyl]pyrazolidine-3,5-dione (8) (Scheme 2).
Compound 4 proved to be a versatile compound for the synthesis of a variety of oxadiazole, S-triazole and thiadiazole derivatives 9-16. Thus, the interaction of carbohydrazide 4 with triethylorthoformate yielded the oxadiazole derivative 9. On the other hand, the interaction of 4 with acid chlorides (acetyl- or benzoyl chloride) in boiling dioxane did not afford the expected 5-methyl or 5-phenyl[1,3,4]oxadiazole derivatives 11a,b, however the N-acetyl or N-phenyl carboxylic acid hydrazide 10a,b derivatives were shown to be the reaction products. The latter compounds 10a,b were subjected to cyclodehydration in boiling phosphorus oxychloride to give the corresponding oxadiazoles 11a,b (Scheme 2). IR spectra of compounds 11a-c, revealed the absence of absorption bans at 3300-3390 for NH and 1680-1705 for C=O groups in compounds 10a,b. Mass spectra of compound 11a showed its correct parent ion peak at m/z 367.19 (M+,100%). Also, the interaction of 4 with carbon disulfide in boiling pyridine gave the corresponding oxadiazolinethione 12. 4-Amino-5-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridin-5-yl)-2,4-dihydro-3H- 1,2,4-triazole-3-thione 13 was obtained by refluxing of 12 with hydrazine hydrate. The spectral data of the triazole 13 derivative was in agreement with the desired structure. In the IR spectra, N-H stretch was observed at about 3450, 3300 and 3270 cm-1. In the 1H-NMR spectra, the NH2 protons observed as broad singlet and resonated at 5.6 ppm, whereas, N-H of the triazole appeared as a singlet D2O exchangeable peaks at 13.6 ppm. On the other hand, The hydrazide 4 was allowed to react with phenyl isothiocyanate in boiling ethanol, giving N1-(1,3-diphenyl-3- methyl-1H-pyrazolo[3,4-b]pyridin-5-yl)carbonyl)-N4-phenylthiosemicarbazide (14). The Latter compound 14 underwent two different cyclization reactions. Thus, heating of compound 14 in an aqueous NaOH solution (5%) yielded, 1,2,4-triazole-3-thione 15. Whereas, treatment of 14 with cold concentrated H2SO4 resulted in dehydrocyclization giving the 2-phenylamino-1,3,4-thiadiazole derivative 16 (Scheme 3). The desired Schiff bases 17a-c were synthesized by refluxing the triazole (3) with corresponding aldehydes in glacial acetic acid (Scheme 3).
Compounds 17a-c were characterized by analytical and spectroscopic analysis. The chemical shifts of imine (CH=N) and N–H protons were observed as singlets at 8.45-8.90 and 14.08-14.15 ppm, respectively, in 1H-NMR spectra in the 13C-NMR spectra, the chemical shifts of carbon of imine group and C=S group showed resonance within the range of 159.70-161.2 and 168.1-169.4 ppm, respectively. Also in the IR spectra. The imine and C=S group stretching vibration in the Schiff bases 17a-c was indicated by 1618-1627 and 1247-1281 cm–1 band, respectively. On the other hand, refluxing of triazole 13 (Scheme 4) with aromatic carboxylic acids in the presence of phosphorus oxychloride, caused cyclization by elimination of H2O molecule to give the 6-Substituted triazolo[3,4-b]thiadiazole derivative 18a-c. Compounds 18a-c were, confirmed by IR as well as 1H-NMR spectra by the disappearance of signals at 3450, 3300 and 3270 cm-1 for NH, NH2. Finally, refluxing of triazole 13 with chloroacetone and/or phenacyl bromide in ethanol, produced, 6-substituted-3-(3-methyl-1,6-diphenyl-1H-pyrazolo[3,4-b]pyridine-5-yl)-7H- [1,2,4]triazolo[3,4-b][1,3,4]thiadiazines (19a,b). The latter products were established on the basis of their spectral data: For example, the lack of amino bands in their IR spectra and the appearance of the characteristic signals due to methylene protons of thiadiazine moiety in the region of 4.54-4.57 ppm in their 1H-NMR spectra.
Pharmacological evaluation
Antibacterial activity
Using the agar well-diffusion method [24], twenty two selected derivatives (compounds 1, 2, 3, 4, 5a, 5b, 5c, 9, 11a, 11b, 12, 13, 15, 16, 17a, 17b, 17c, 18a, 18b, 18c, 19a, and 19b) were evaluated for their antibacterial activities. Thus, these compounds were screened against Staphylococcus aureus, Bacillus cereus as a Gram-positive bacteria and Escherichia coli, Pseudomonas aeruginosa as Gram-negative bacteria using Streptomycin as control (Table 1). Compounds 5b, 16, 18c and 19a, were found to possess a remarkable antibacterial activity against the Gram-positive as well as the Gram-negative bacteria, The triazolothiadiazole derivative 18c (R=4-ClC6H4) was the more potent with zones of inhibition 32.7, 30.3, 25.8 and 28.7 against S. aureus (107% inhibition), B. cereus (102% inhibition), E. coli (96% inhibition) and P. aeruginosa (113% inhibition), respectively. Compounds 2 11b, 12, 13, 15, and 18a possessed good antibacterial activity against all of bacteria species except with P. aeruginosa. The thiadiazole derivative 16 is the most potent with zones of inhibition 26.5, 27.6 and 24.7 against S. aureus (87.7% inhibition), B. cereus (93.5% inhibition), E. coli (92.8% inhibition), respectively. On the other hand, compounds 17b and 18b, exhibited good antibacterial activity against S. aureus only with zones of inhibition 25.2 (83.2% inhibition) and 19.6 (64.7% inhibition), respectively. The rest of compounds under investigation possessed a weak activity against all of bacteria species.
| Compound | Inhibition zone (mm)a mean ± SD (n=3)b | |||
|---|---|---|---|---|
| Gram-positive bacteria | Gram-negative bacteria | |||
| S. aureus | B. cereus | E. coli | P. aeruginosa | |
| 1 | 10.9 ± 0.03 | NA | NA | NAc |
| 2 | 19.2 ± 0.51 | 21.2 ± 0.09 | 18.6 ± 0.23 | NA |
| 3 | NA | 8.6 ± 0.04 | NA | NA |
| 4 | 11.3 ± 0.35 | NA | NA | NA |
| 5a | 15.3 ± 0.07 | NA | 14.9 ± 0.21 | NA |
| 5b | 29.6 ± 0.03 | 23.7 ± 0.05 | 17.4 ± 0.21 | 20.3 ± 0.09 |
| 5c | 13.2 ± 0.33 | 11.3 ± 0.08 | NA | NA |
| 9 | 9.8 ± 0.32 | NA | NA | NA |
| 11a | 12.6 ± 0.24 | NA | NA | NA |
| 11b | 20.3 ± 0.06 | 26.2 ± 0.21 | 23.3 ± 0.51 | NA |
| 12 | 22.1 ± 0.08 | 15.4 ± 0.06 | 19.6 ± 0.22 | NA |
| 13 | 26.5 ± 0.09 | 27.6 ± 0.13 | 24.7 ± 0.05 | NA |
| 15 | 22.5 ± 0.06 | 16.1 ± 0.20 | 19.6 ± 0.08 | NA |
| 16 | 31.3 ± 0.53 | 28.8 ± 0.11 | 16.9 ± 0.13 | 22.1 ± 0.07 |
| 17a | NA | 10.1 ± 0.08 | NA | NA |
| 17b | 25.2 ± 0.16 | NA | NA | NA |
| 17c | NA | NA | 11.3 ± 0.23 | NA |
| 18a | 26.3 ± 0.11 | 21.5 ± 0.09 | 20.5 ± 0.60 | NA |
| 18b | 19.6 ± 0.16 | NA | NA | NA |
| 18c | 32.7 ± 0.08 | 30.3 ± 0.33 | 25.8 ± 0.39 | 28.7 ± 0.42 |
| 19a | 29.1 ± 0.50 | 23.5 ± 0.09 | 21.5 ± 0.33 | 20.7 ± 0.23 |
| 19b | 12.6 ± 0.09 | 14.6 ± 0.13 | NA | NA |
| Streptomycin | 30.3 ± 0.20 | 29.5 ± 0.4 | 26.6 ± 0.32 | 25.2 ± 0.10 |
aAll the test and the standard drug were tested at the concentration of 20 mg/ml; bEach result represents the average of triplicate readings; cNo activity observed
Table 1: Antibacterial activity of some pyrazolo[3,4-b]pyridines
Antifungal activity
The same compounds (1, 2, 3, 4, 5a, 5b, 5c, 9, 11a, 11b, 12, 13, 15, 16, 17a, 17b, 17c, 18a, 18b, 18c, 19a, and 19b) were screened for their antifungal activities against four fungal strains: Candida albicans, Trichophyton rubrum, Aspergillus flavus and Fusarium oxysporum using clotrimazole as control (Table 2). In general, all the compounds exhibited low to moderate antifungal activity except compounds 1, 5b, 13 and 18c which were effective against F. oxysporum with zones of inhibition 20.3 (74.9% inhibition), 23.5 (86.7% inhibition), 23.3 (85.9% inhibition) and 26.7 (98.5% inhibition), respectively. Also, compounds 16, 17b and 19a showed the highest activity against T. rubrum with zones of inhibition 21.4 (94.3% inhibition), 25.5 (112% inhibition) and 22.4 (98.6% inhibition), respectively. Other compounds exhibited none or weak activity against the tested fungi species.
| Compound | Inhibition zone (mm)a mean ± SD (n=3)b | |||
|---|---|---|---|---|
| C. albicans | T. rubrum | A. flavus | F. oxysporum | |
| 1 | 10.9 ± 0.03 | NA | NA c | 20.3 ± 0.42 |
| 2 | NA | NA | 10.6 ± 0.23 | NA |
| 3 | NA | 8.6 ± 0.04 | NA | NA |
| 4 | NA | NA | 11.2 ± 0.34 | NA |
| 5a | NA | 10.3 ± 0.55 | NA | NA |
| 5b | 11.2 ± 0.11 | NA | NA | 23.5 ± 0.51 |
| 5c | 11.6 ± 0.33 | NA | NA | NA |
| 9 | 13.3 ± 0.19 | NA | NA | 14.6 ± 0.31 |
| 11a | 12.8 ± 0.22 | 16.1 ± 0.33 | NA | NA |
| 11b | 15.1 ± 0.19 | 17.4 ± 0.14 | NA | NA |
| 12 | NA | 9.9 ± 0.18 | NA | NA |
| 13 | 11.3 ± 0.25 | 14.3 ± 0.23 | NA | NA |
| 15 | NA | NA | 11.6 ± 0.07 | NA |
| 16 | 13.1 ± 0.19 | 21.4 ± 0.17 | 15.5 ± 0.09 | NA |
| 17a | NA | 10.1 ± 0.08 | NA | NA |
| 17b | NA | 25.5 ± 0.44 | NA | 12.1 ± 0.33 |
| 17c | NA | NA | 11.3 ± 0.23 | NA |
| 18a | NA | NA | NA | 13. ± 0.13 |
| 18b | 11.6 ± 0.31 | NA | NA | NA |
| 18c | NA | NA | NA | 26.7 ± 0.35 |
| 19a | NA | 22.4 ± 0.12 | 11.7 ± 0.61 | NA |
| 19b | NA | 12.8 ± 0.51 | NA | NA |
| Clotrimazole | 19.5 ± 0. 19 | 22.7 ± 0.09 | 23.2 ± 0.22 | 27.1 ± 0.22 |
aAll the test and the standard drug were tested at the concentration of 20 mg/ml; bEach result represents the average of triplicate readings; cNo activity observed
Table 2: Antifungal activity of some pyrazolo[3,4-b]pyridines
Conclusion
The structures of all the new compounds have been established on the basis of their analytical and spectral data. Twenty two of the synthesized compounds were also evaluated for their antibacterial and antifungal activity using Streptomycin and Clotrimazole as the reference drugs
References
- M. Julino, M.F.G. Stevens, J. Chem. Soc. Perkin Trans., 1998, 1, 1677.
- M. Ibrahim Abdou, A.M. Saleh, H.F. Zohdi, Molecules., 2004, 9, 109.
- R. Filler, Chem. Technol., 1974, 4, 752.
- M.M. Ghorab, Z.H. Ismail, S.M. Abdel-Gawad, A. Abdel-Aziem, Heteroat. Chem., 2004, 15, 57.
- F.E. Goda, A.A.M. Abdel-Aziz, O.A. Attef, Bioorg. Med. Chem. Lett., 2004, 12, 1845.
- J. Witherington, V. Bordas, S.L. Garland, D.M.B. Hickey, R.J. Lfe, J. Liddle, M. Saunders, D.G. Smith, R.W. Ward, Bioorg. Med. Chem. Lett., 2003, 13, 1577.
- R. Lin, P.J. Connolly, Y. Lu, G. Chin, S. Li, Y. Yu, S. Huang, X. Li, S.L. Emanuel, S.A. Middleton, R. H. Gruninger, M. Adams, A.R. Fuentes-Pesquera, L.M. Greenberger, Bioorg. Med. Chem. Lett., 2007, 17, 4292.
- S. Schenore, G. Bruno, P. Fossa, A. Ranise, G. Menozzi, L. Mosti, F. Bondavalli, C. Martini, L. Trincavelli, Bioorg. Med. Chem. Lett., 2001, 11, 2529.
- L.P. Davies, S.C. Chow, J.H. Skerritt, D.J. Brown, G.A.R. Johuston, Life Sci., 1984, 34, 2117.
- M.A. Timoteo, L. Oliveira, E.C. Campesatto-Mella, A. Barroso, C. Silva, M.T. Magalhaes-Cardoso, W. Alves-do-Prado, P. Correiadesa, Neurochem. Int., 2008, 52, 834.
- M.A. Gaston, L.R. Dias, A.C. Freitas, A.L. Miranda, E.J. Barrero, Pharm. Acta. Helv., 1996, 71, 213.
- G.M. Shutask, J.E. Roehr, J. Heterocycl. Chem., 1997, 34, 787.
- J. Clark, G. Hitiris, J. Chem. Soc., 1984, 1, 2005.
- S.S. Parmar, B.R Pandey, C. Dwivedi, B. Ali, J. Med. Chem., 1974, 17, 1031.
- D.M. Helosia, E. Aurea, M.B. Alice, C.C. Marilene, L.L. Leonor, J. Med. Chem., 2004, 47, 5427.
- G. Yu, H.J Mason, X. Wu, J. Wang, S. Chong, G. Dorough, A. Henwood, R. Pongrac, L. Seliger, B. He, D. Normandin, L. Adam, J. Krupinski, J.E Macor, J. Med. Chem., 2001, 44, 1025.
- H.S. El-Kashef, T.I. El-Emary, J. Chem. Res. Synop., 1998, 20-21.
- T.I. El-Emary, E.A. Bakhite, Pharmazie., 1999, 54, 106.
- T.I. El-Emary, N. Al-Muaikel, O.S. Moustafa, Phosphorus Sulfur Silicon Relat. Elem., 2002, 177, 195.
- T.I. El-Emary, A. Khalil, G.A. El-Hag, M.A. El-Adasy, Phosphorus Sulfur Silicon Relat. Elem., 2005, 180, 19.
- S.H.A. Abdel-Mohsen, T.I. El-Emary, J. Adv. Chem., 2014, 10, 2901.
- J. Haeufel, E. Breitmaier, Angew. Chem. Internat. Edit., 1974, 13, 604.
- A.M. Hussein, T.I. El-Emary, J. Chem. Res.,1998, 20.
- K.J. Kwon-Chung, J.W. Bennett, Med. Mycol. Lea Febiger. Philadel., 1992, 81.