Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 2
Photovoltaic Energy Conversion and Optical Properties of Organic Molecules Based on Aceanthraquinoxaline
Taoufik Sahdane, Bousselham Kabouchi, Abbes Laghrabli and Brahim Azize
Abstract
In this investigation, we out theoretical study of electronic properties and geometry of four conjugated molecules based on aceanthraquinoxaline are used as donor layer for organic thin-film solar cells. The impact the substituted groups on the optoelectronic and structural properties, dipole moment, Highest Occupied Molecular Orbital (HOMO), Lowest Unoccupied Molecular Orbital (LUMO) and gap energies (Egap) are argue. The geometry at the ground state and the electronic structure of studied molecules are procure by Density Functional Theory (DFT) and density functional theory time-dependent (TD-DFT) with the B3LYP/6-31G (d). The results of this study exhibit that these compounds are good candidates for organic solar cells.
Keywords
Aceanthraquinoxaline, Electronic properties, DFT, HOMO, LUMO, Photovoltaic, Organic solar cells
Introduction
Solar panels on silicon are currently the most popular; this technology is limited by its high cost. An emerging technology that can address the current gaps in the field is organic photovoltaics in which semiconductors are carbon-based. These compounds are much less expensive to manufacture and require a lower initial investment [1,2]. They also exhibit the ability to be dissolved and incorporated into inks. They may benefit from the enormous expertise in printing technology, low cost and high speed of production. In addition, organic devices can be fabricated on flexible or semi-transparent substrates, and can benefit from new applications, such as solar textiles that wrap for easy transport, solar tinted windows [3,4]. The main limitation of organic photovoltaics is its low efficacy compared to its inorganic counterparts. To be economically viable and competitive, it is crucial to increase this effectiveness. To do this, we need to find organic compounds, which have the most relevant electronic properties, including optimum band gap and energy levels allowing heterojunction effective. The synthesis of all compounds is completely impossible, and it is crucial to probe how the theoretical properties for better effectiveness. Scharber et al. proposed a semi-empirical model in 2006 to explain the operation of the organic devices and provide an efficiency maximum of 11% based on this model [5]. It is useful from first principles and solving the Schrödinger equation. These calculation methods that are not based on experimental data are called ab-initio. An interesting technique to resolve the Schrödinger equation is the density functional theory [6].
Organic semiconductors have attracted the regard of researcher’s theorists and experimentalists in recent years, due to their excellent optical and electronic properties [7]. These materials can be used for producing such diverse devices as photovoltaic cells, field effect transistors, and light emitting diodes [8-13]. Most research of new conjugated molecules with specific applications has be one of the attractive topics in the fields of physical and chemical materials [14]. A necessary understanding of the relationship between structure and properties of these materials is desired to exploit their properties for photovoltaic cells.
In this study, four compounds based on aceanthraquinoxaline (AQ1, AQ2, AQ3, AQ4) are considered. These compounds were studied using DFT and TD-DFT calculations. HOMO, LUMO, Egap and open circuit voltage (Voc) of all molecules are investigated (Figure 1).
Materials and Methods
The calculations are take-out using the program GAUSSIAN 03 [15], and the simulation of current-voltage (J-V) characteristics, fill factor, open-circuit voltage, and conversion efficiency of organic photovoltaic cell was well established by gpvdm software. The geometry optimization of the studied compounds is evaluated by DFT/B3LYP using 6-31G (d) basis set [16]. The electronic absorption spectra are exploring using TD-DFT calculations with B3LYP/6-31G (d).
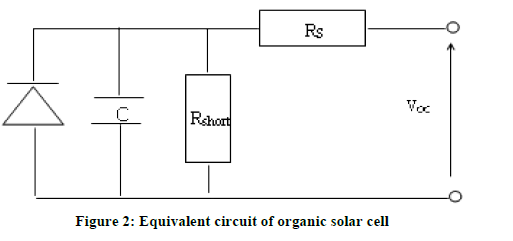
The circuit diagram has been given in Figure 2 [17].
Materials used for the design and bandwidth values data of materials are given in Table 1.

Conclusion
In this paper, the properties of the optical absorption and electronic structure of four compounds based on aceanthraquinoxaline were studied employing DFT and TD-DFT methods. We conclude the changes of several donors or acceptor groups do not change the geometric parameters and the energies of the orbital HOMO, LUMO and Egap showed that Egap studied molecules differ from 3.51-4.35 eV for the different structures. From these results, we can conclude that these compounds have shown significant optoelectronic properties, which leads to offer these materials for application of organic solar cells. This calculation process can be used as a model system for understanding the relationship between the electronic properties and molecular structure and can as be used to study their aptitude in electroluminescent devices. The results propose these materials as candidate for organic solar cells.
References
[1] F. Etzold, I.A. Howard, R. Mauer, M. Meister, T.D. Kim, K.S. Lee, N.S. Baek, F.J. Laquai, Am. Chem. Soc., 2011, 133, 9469-9479.
[2] J. Kalowekamo, E. Baker, Sol. Energ., 2009, 83, 1224-1231.
[3] M.R. Lee, R.D. Eckert, K. Forberich, G. Dennler, C.J. Brabec, R. A. Gaudiana, Science., 2009, 324, 232-235.
[4] R.R. Sondergaard, M. Höselet, F.C. Krebs, J. Polym. Sci., 2013, 51, 16-34.
[5] M.C. Scharber, D. Mühlbacher, M. Koppe, P. Denk, C. Waldauf, A.J. Heeger, C. Brabec, J. Adv. Mater., 2006, 18, 789-794.
[6] K. Capelle, Braz. J. Phys., 2006, 36, 1318-1343.
[7] S. Hotta, S.A. Lee, Synth. Met., 1999, 101, 551-552.
[8] Acc. Chem. Res., 1999, 32, 191.
[9] S. Miyata, H.S. Nalwa, Amsterdam, 1997.
[10] T. Skotheim, J. Reynolds, R. Elsenbaumer, Marcel Dekker, NY, USA, 1998.
[11] Bernier, Lefrant, Bidan, Elsevier., 1995.
[12] J. Pei, W.L. Yu, W. Huang, A.J. Hegger, Macromolecules., 2000, 33, 2462-2471.
[13] H. Yanagi, S. Okamoto, App. Phys. Lett., 1997, 71, 2563.
[14] T. Sahdane, A. Laghrabli, R. Benallal, H. Bougharraf, B. Azize, B. Kabouchi, JOCPR., 2016, 8(9), 130-135.
[15] M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, 2003.
[16] A.D. Becke, J. Chem. Phys., 1993, 98, 5648-5652.
[17] T. Sahdane, H. Bougharraf, A. Laghrabli, R. Benallal, B. Azize, B. Kabouchi, IJARCCE., 2016, 5(10), 283- 286.



