Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 3
Phytochemical Screening, Antioxidant Activity of the Various Extracts from Peganum harmala (Zygophyllaceae)
Hassina Guergour1,2*, Rima Allouni R2 and Abdelouahab Bouzidi A2
1Department of Biology, Faculty of Natural Sciences and Life, University Elbachir El brahimi, Bordj Bou Ariridj- 34000, Algeria
2Department of Biochemistry, Faculty of Natural Sciences and Life, University Ferhat Abbas, Setif-19000, Algeria
- *Corresponding Author:
- Hassina Guergour
Department of Biology
Faculty of Natural Sciences and Life
University Elbachir El brahimi
Bordj Bou Ariridj- 34000, Algeria
Abstract
The present study was to elucidate the phytochemical content and antioxidant activity (by DPPH and FRAP) of crude leaf extract (CLE), crude seeds extract (CSE) and alkaloid seeds extract (ASE) from Peganum harmala. Phytochemical screening revealed the presence of tannins, sterols and steroids, coumarins, anthocyanins and saponins. DPPH method specifies an IC50 equal to: 112.96 μg/mL, 35.83 μ/mL and 107.91 μg/mL respectively for leaf, seeds and alkaloids extracts. Standard ascorbic acid shows IC50 value of 4.24 μg/ml. Reducting power revealed that all extracts from Peganum present less significant effect and weaker than that of standard ascorbic acid (0.66, 0.30, 0.58 absorbance for seeds, leaf, alkaloids seeds at 800 μg/ml, ascorbic acid 3.82 absorbance at 200 μg/ml). The presence of secondary metabolites in leaves and seeds of P. harmala could be attributed for the antioxidant activity.
Keywords
Peganum harmala, Alkaloids extract, Crude extract, Photochemistry, Antioxidant activity
Introduction
Oxidative stress is an important risk factor in the pathogenesis of numerous chronic diseases. Free radicals and other reactive oxygen species are recognized as agents involved in the pathogenesis of sicknesses such as asthma, inflammatory arthropathies, diabetes, Parkinson's and Alzheimer's diseases, cancers as well as atherosclerosis. Reactive oxygen species are also said to be responsible for the human aging [1,2].
Natural antioxidants either in the form of raw extracts or their chemical constituents are very effective to prevent the destructive processes caused by oxidative stress [3]. They stabilize or deactivate free radicals, often before they attack targets in biological cells [4].
Peganum harmala L. is a wild-growing plant that belongs to the Zygophyllaceae family [1]. It is also called African rue, Syrian rue and wild rue. The plant is widely distributed in predesertic regions of south-east Morocco, North Africa and the Middle East [5]. The extracts of P. harmala exhibit fungicidal, bactericidal, anti-inflammatory and antitumor activity [6-9].
This work is to achieve a phytochemical screening and evaluating the antioxidant activity of various extracts of P. harmala plant.
Materials and Methods
Plant material
Seeds and leaves of P. harmala were collected in the month of August, in the region Bordj Bou Arreridj (north-east of Algeria). The plant was identified on the basis of its morphological characteristics.
Preparation of extracts
Methanol extracts
Seeds and leaves powder (50 g) was macerated during 24 h in 250 ml methanol, with continuous stirring at room temperature. The macerate was filtered, evaporated using rotary evaporator to obtain crude extracts preserved with 4°C until its use.
Total alkaloids extraction
The extraction of the total alkaloids from seeds of P. harmala is obtained by a liquid-liquid extraction. The powdered seed (100 g) was dilapidated with petroleum ether by maceration and with stirring at room temperature for 3 to 4 h. The defatted dry powder was alkalized with 40 ml of NH4OH (0.5 N) for 8 h. The total alkaloids are extracted by the Soxhlet extractor using dichloromethane for 6 h (At least 5 cycles are required for complete seed exhaustion). The crude extract is washed with aqueous sulphuric acid (0.5 N) the operation is repeated 3 times.
The solution obtained is treated with NH4OH (0.5 N) to pH 9 then separated three times by diethyl ether and then dehydrated with Na2SO4. The solvent layer was evaporated to dryness [10].
Thin layer chromatography (TLC)
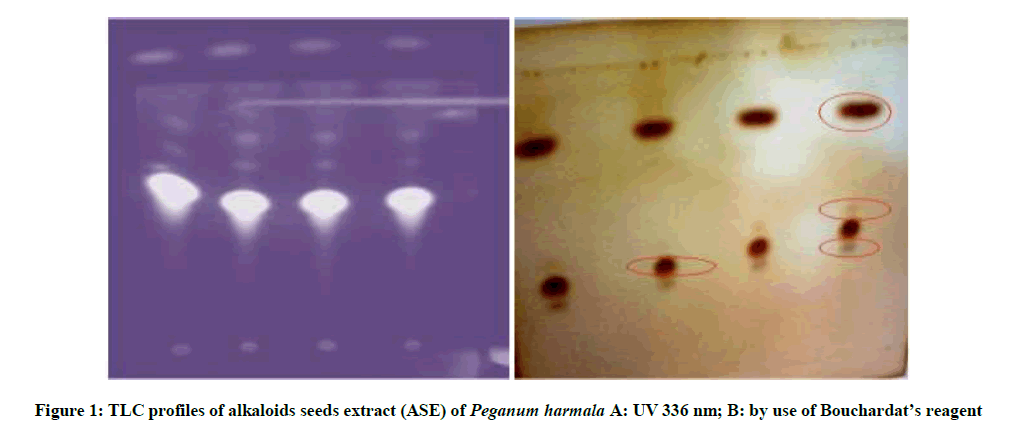
TLC was used for the conformation of presence of alkaloids in the ASE (alkaloid extract). We used plates of silica gel Macherey-Nagel. The solvent system contained methanol/chloroform/ammonia: 80/20/1, 5 (V/V/V). After dissolution of the sample in methanol, 10 μl of the extract were loaded on the analytical plate. The chromatograms were observed under visible light and photographed. Bouchardat’s reagent was used for the detection of alkaloids [11].
Phytochemical screening
Phytochemical properties were tested using the following chemicals and reagents according to the method of [12-14]. The powder of seeds and leaves were tested for the presence of steroids, tannins, saponins, coumarins and anthocyanins. The qualitative results are expressed as (+) for the presence and (-) for the absence of phytochemicals.
Antioxidant studies
DPPH free radical scavenging activity: DPPH free radical scavenging assay was performed to determine the antioxidant activity of different concentrations of extracts and ascorbic acid. DPPH (0.1 mM) was used as free radical (0.1 mM). The absorbance was measured at 517 nm that was determined after allowed to stand at room temperature for 30 min. The inhibition percentage of both standard and extracts was calculated for each concentration according to the following formula:
I% = [(AbsC- AbsS)/ AbsC] x 100
Where, AbsC is the absorbance of control reaction and AS the absorbance of test compounds. Ascorbic acid was used in a positive control. The data are presented as mean of triplicate and the concentration required for a 50% (IC50) reduction of DPPH radical was determined graphically.
Ferric reducing antioxidant power (FRAP) assay: The reducing power was determined according to the protocol described by Oyaizu [15]. Different concentrations of extracts were mixed with phosphate buffer and potassium ferricyanide [K3Fe(CN)6]. The mixture was incubated at 50°C for 20 min. A portion of trichloroacetic acid (10%) was added to the mixture, which was then centrifuged at 3000 rpm for 10 min. The upper layer of the solution was mixed with distilled water and FeCl3 and the absorbance measured at 700 nm. Increasing the absorbance of the reaction mixture indicated increased reducing power. Ascorbic acid was used as standards. All analyses were run in triplicate and results averaged.
Results and Discussion
Extraction yield
The percent extracts yielded from 13.44%, 10.52% and 2.11 % for CLE, CSE and ASE respectively. In a study by Benbott [16] on the same species, almost similar results were found by maceration of leaves and seeds in methanol. The extraction of the total alkaloids seeds of P. harmala enabled us to obtain a brick red extract which is almost the same yield found by Mahdeb [11]. TLC of alkaloids seeds extract of P. harmala revealed the presence of four distinct spots (Table 1 and Figure 1).
| Extracts | Yields |
|---|---|
| Crude leaf | 13.44% |
| Crude seeds | 10.52% |
| Alkaloids seeds | 2.11% |
Table 1: Yield of various extracts
Phytochemical screening
Qualitative analysis carried out of the plant showed the presence of most active constituents (Table 2).
| Secondary metabolites | Leaves | Seeds |
|---|---|---|
| Tannins | + + | + + + |
| Anthocyanins | - | + |
| Coumarins | - | + |
| sterols and steroids | + | + |
| Saponins | ++ | - |
Table 2: Result of phytochemical screening of Peganum harmala
The phytochimical screening showed the presence of three chemical groups tannins, alkaloids, sterols and steroids in the leaves and the seeds of P. harmala, these results are identical to that obtained by Behidj-Benyounes [17]. In the leaves of P. harmala we noted the presence of saponins. On the other hand, we noted the absence of the anthocyanins and coumarins. However, we note some differences between our results and those obtained by Behidj-Benyounes [17] which revealed the presence of anthocyanins and coumarins in leaves.
The phytochemical screening on qualitative analysis shows that the leaves and seeds of P. harmala are rich in alkaloids, steroids and tannins. These phytochemicals confer antioxidant and antimicrobial activities on the plant extract.
Antioxidant studies
Free radical scavenging activity
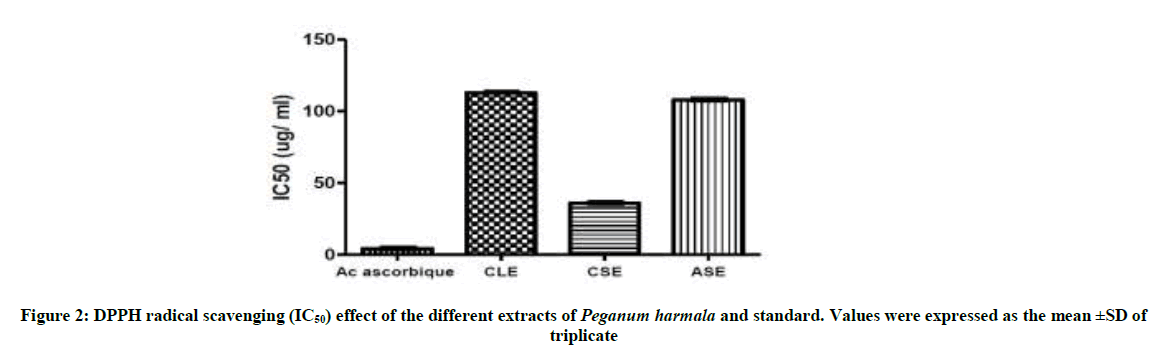
DPPH is a stable free radical that can accept an electron to become a stable molecule, commonly used as a substrate to evaluate antioxidant activity; it is a stable. The reduction of DPPH radical was determined by the decrease in its absorbance at 517 nm induced by antioxidants [18]. DPPH radicals are widely used in the model system to investigate the scavenging activities of several natural compounds. When DPPH radical is scavenged, the color of the reaction mixture changed from purple to yellow with decreasing of absorbance at wavelength 517 nm [19]. The values of the IC50 obtained for the whole of the extracts, are represented in the (Figure 2), whose values are expressed in final concentrations μg/ml.
The IC50 of the ascorbic acid 4.24 μg/ml is identical to that obtained from [20]. Free radical scavenging activity of CSE was highest having IC50 of 35.83 μ/ml. On other hand CLE and ASE demonstrated moderate free radical scavenging with the IC50 of 112.96 μ/ml and 107.91 μ/ml, respectively (Figure 2). The results of the antioxydant capacity of the extracts tested show that all our extracts exert a less significant antioxydant power with IC50 of about 35.83; 107.91; 112.96 μ/ml in comparison with the antioxydant capacity of standard (IC50= 4.24 μg/ml). In more they show that the CSE had the highest antioxidant activity in comparison to CLE and ASE because it is very rich in secondary metabolites.
Reducing power assay
In reducing power assay, the yellow colour of the test solution changes to green depending on the reducing power of the test specimen. It is an analysis of antioxidant activity that is fast and easy to perform based on the ability of molecules tested to reduce ferric iron Fe3+ in ferric chloride to ferrous Fe2+ [21].
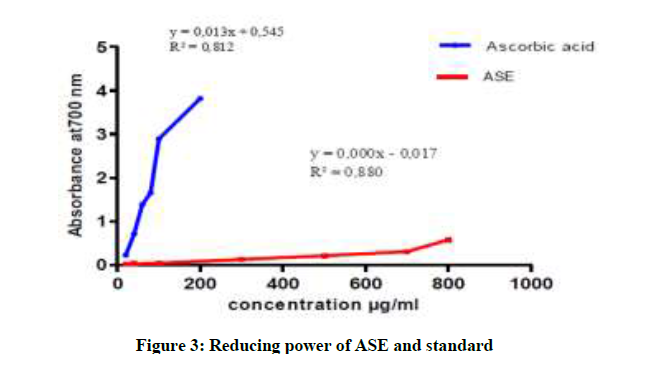
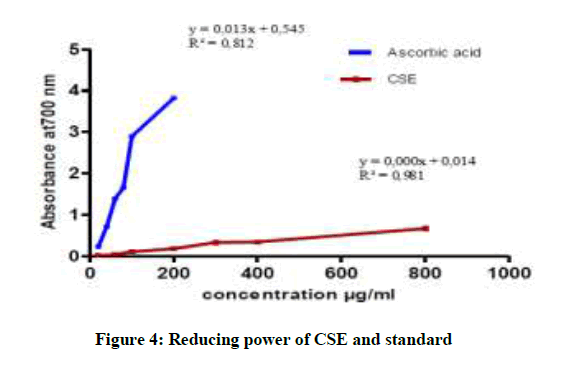
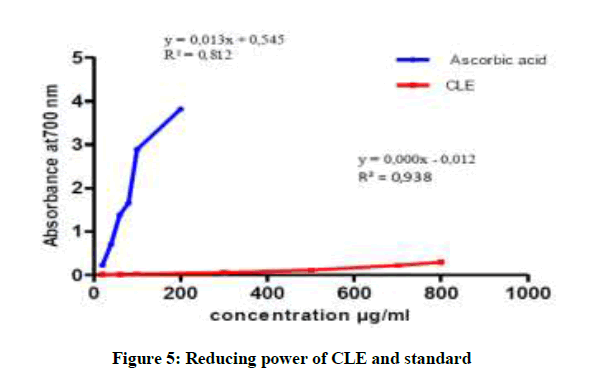
In more we note that the capacity to reduce iron is almost the same for CLE and ASE and it is lower than CES (Figures 3-5). The CES is most active with a maximum optical density of 0.667 nm for concentration 800 μg/ml. In addition all extracts of the plant present reducing ability definitely lower than Ascorbic acid [22,23].
Conclusion
In conclusion, our research allowed highlight the following families: alkaloids, tannins, sterols and steroids, coumarins, anthocyanins and of saponins in the species P. harmala. All extract from P. harmala present the average antioxidant capacity was observed in CLE and ASE with an IC 50 of the order 112.96 μg/ml, 107.91 μg/ml, on the other hand, the CSE possessing an important activity 35.83 μg/ml. But all extracts exert a lower antioxidant power compared to the standard antioxidant capacity (IC50 of 4.24 μg/ml). The reducing assay revealed that the P. extracts exhibited less activity than that of ascorbic acid.
References
- J.R. Kanwar, R.K. Kanwar, H. Burrow, S. Baratchi, Curr. Med. Chem., 2009, 16, 2373-2394.
- V. Chiavaroli, C. Giannini, S. De Marco, F. Chiarelli, A. Mohn, Redox. Rep., 2011, 16, 101-107.
- G. Zengin, Y.S. Cakmak, G.O. Guler, A. Aktumsek, Rec. Nat. Prod., 2011, 5, 123-132.
- P.X. Nunes, S.F. Silva, R.J. Guedes, Global Approaches to Their Role in Nutrition and Health, 2012, 3 p.
- M.R. Pradeep Kumar, D.J. Shrinivas, V.H. Kulkarni, C. Savant, J. App. Pharm. Sci., 2015, 5(5), 52-55.
- S. Bensalem, J. Soubhye, I. Aldib, L. Bournine, A.T. Nguyen, M. Vanhaeverbeek, A. Rousseau, K.Z. Boudjeltia, A. Sarakbi, J.M. Kauffmann, J. Neve, M. Prevost, C. Stevigny, F. Maiza- Benabdesselam, F. Bedjou, P. Van Antwerpen, P. Duez, J. Ethnopharmacol., 2014, 154, 361-369.
- F. Lamchouri, M. Zemzami, A. Jossang, A. Settaf, Z.H. Israili, B. Lyoussi, Pak. J. Pharm. Sci., 2013, 26, 699-706.
- A.M. Sobhani, S.A. Ebrahimi, M. Mahmoudian, J. Pharm. Pharm. Sci., 2002, 5, 19-23.
- Y.C. Song, D. Kesuma, H. Wang, Y. Deng, J. Duan, J.H. Wang, R.Z. Qi,. Biochem. Biophys. Res. Commun., 2004, 317, 128-132.
- J. Bruneton, Pharmacognosie. Phytochimie, Plantes médicinales. 3 èmeéd. Lavoisier Paris. 1999.
- N. Mahdeb, S. Mayouf, F. Boukhari, S. Souilah, A. Bouzidi, Asian. J. Plant Sci. Res., 2013, 3(6), 53-59.
- N. Dohou, K. Yamin, S. Tahrou, H.L.M. Idrissi, A. Badoc, N. Gmira, Bull. Soc. Pharm. Bordeaux., 2003, 142, 61-78.
- D. Diallo, R. Sanogo, 2015, ICS-UNIDO, INRSP. Ministry of Health, Mali.
- J. Bruneton, Pharmacognosie - Phytochimie, plantes médicinales, 4 ème éd., revue et augmentée, Tec et Doc. médicales internationales, Paris, 2009, 1288 p.
- M. Oyaizu M, Japan J. Nutrit., 1986, 44, 307-315.
- L. Benbott, L. Bahri, A. Boubendir, A. Yahia, J. Mater. Environ. Sci., 2013, 4 (4), 558-565.
- N. Behidj-Benyounes, T. Dahmane, F. Aknouche, K. Demmouche, Sci et Techno., 2013, 38, 27-37.
- Y.V. Ramshankar, P. Vinay, P. Vijayan, Pharmog. Mag., 2008, 4, 330-334.
- Benslama, S. Afissa, A. Saidi, A. Harrar, Int. J. Cur. Res., 2016, 8(12), 42675-42679.
- Erum, A. Kamariah, B.L.L. Linda, J. King Saud Univ, Sci., 2015, 27, 224-232.
- Karagozler, B. Erdag, Y. Calmaz Emek, Food Chem., 2008, 111, 400-407.
- M. Ozturk, F. Aydogmus-Ozturk, M.E. Duru, G. Topcu, Food Chem., 2007, 106, 1264-1270.
- L. Liuk, Y. Sun, T. Laura, X. Liang, H. Ye, X. Zeng, Food chem., 2009, 112, 35-40.