Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 1
Practical Aspects to Generate Cutaneous Experimental Burns in a Rat Model.
Abdeldjelil MC*, Messai A, Boudebza A and Beghoul SAbdeldjelil MC, PADESCA Research Laboratory, Institute of Veterinary Science, University of Constantine Algeria, Algeria, Email: a.mansour@etud.ensv.dz
Abstract
In order to evaluate the potential wound healing effect of new drugs, and synthetic dressings, it is essential to use reliable in vivo experimental models of burns. This review presents the key steps involved in generating cutaneous burns in a rat model. Besides presenting general consideration concerning the experimental design; the entire burn sequence, from burn up to postoperative care is described. First, major preoperative procedures (anaesthesia and, skin area preparation) are discussed. Then, a full description of the most current and widely used burn inflicting methods is presented. Finally, the postoperative care section discusses analgesia, fluid therapy, and wound care. By addressing all these issues, we provide new investigators with a solid background from which to begin their experimental studies on burns, especially those investigating the burn healing properties of new drugs.
Keywords
Experimental burns, Rat, Wound healing, Drug research
Introduction
Since man first learned to make fire, burns have become one of his most common and severe injuries [1]. The impact of major burns is likely to affect the patient’s physical, emotional, psychological, social, spiritual and vocational health [2]. Throughout the history of mankind, diverse remedies have been employed to treat burns [1]; however, most major advances in burn care occurred in the last 50 years, spurred on by wars and great fires [3]. The successful treatment of burns has been made possible by better knowledge of the pathophysiology of thermal injuries, and development of medical technology and surgical techniques [4]. All these advances have been facilitated by the development of in vivo experimental models of burns [5]. The use of these in vivo burn models is essential for burn research [6] especially experimental studies investigating the burn healing properties of new drugs [7]; given that new treatments must first be tested at the experimental level before clinical use [8].
In many published studies on experimental burns, the burn protocol is not fully described, leaving many important details incomplete or unmentioned. Such limitations make these studies not easily replicable [9]. In this article we present a detailed description of procedures used to generate cutaneous experimental burns in a rat model. This review is valuable for those interested in burn research. It allows new researchers to survey the methods used by their peers to create experimental burns within the context of studies on the burn healing properties of new drugs.
Pathophysiology of burn injury
A burn is a coagulative necrosis of the skin or other organic tissues, caused by the dissipation of thermal energy into it [10]. Burns occur following contact with hot liquids and steam (scalds), hot solids (contact burns), or flames (flame burns); injuries caused by radiation, radioactivity, electricity, friction or contact with certain chemicals are also considered as burns [11].
During burn trauma, prolonged exposure to high temperatures, leads to tissue damage via thermal denaturation of proteins, and loss of the plasma membrane integrity. These lesions cause cell death and various molecular structural alterations leading to the release of toxic metabolites as well as antigens and immunomodulatory agents [12]. Following the initial tissue damage, an inflammatory response takes place with a massive local influx of inflammatory cells that release various agents which coordinate the action of immuno-competent cells. The action of these cells is reflected by localised oedema, altered perfusion and systemic inflammatory response [13]. Local changes at the burn site can generally be divided into three concentric spheres of tissue damage: The centre of the burn is a zone of coagulation which is destroyed permanently (necrosis). The zone of coagulation is surrounded by a zone of stasis which is ischemic with poor vascular perfusion, and injured cells that are susceptible to necrosis. Beyond the zone of stasis, lies the zone of hyperaemia, which features increased blood flow resulting from the inflammatory response [14,15].
Based on the extent of damage caused to the different layers of skin, burns are classified as first, second and third degree burns. Firstdegree (superficial) burns affect only the epidermis, the outermost layer of skin. Second-degree (partial thickness) burns involve the epidermis and part of the dermis. Depending on the thickness of the damaged dermis, we distinguish second-degree superficial burns, reaching the papillary dermis; and second-degree deep burns, reaching the deep reticular dermis. Third-degree burns, also known as full thickness burns, destroy both epidermis and dermis with exposure of underlying tissues [13,14,16].
In extended burns (30% or more of total body surface area in humans), the release of cytokines and other inflammatory mediators at the site of injury, leads to a systemic response with cardiovascular, respiratory metabolic and immunological changes [17]. One potentially fatal example of these systemic responses is hypovolemic shock resulting from plasma loss [10].
Animals
Rats as burn models
In a number of mammalian species, experimental models of burn injury have been established, including for large mammals like pigs [18], sheep [19], rabbits [20]; and for small rodents like guinea pigs [21], hamsters [22], and in particular rats and mice, which by far, have been the most frequently used species in burn models [6]. Rats are also used to study other forms of skin injuries, as a popular model for excision and incision wounds [23-26]. The experimental rat is the Rattus norvegicus, of the family Muridae, genus Rattus. The most commonly stocks of rats used in burn healing experiments are two outbred albino rats: Wistar Kyoto and Sprague Dawley [27]. In burn research, rats have been used to investigate burn wound pathology [28,29], and also the influence of local and systemic drugs on burn healing [10,30-34]. With the completion of the sequences of the rat genome, and the creation of transgenic and knockout rats, the rat model of burn injury may offer the advantage of simultaneous investigations of inflammatory/ immunological and cardiovascular-metabolic perturbations occurring with burn injury [35]. Rats are attractive candidates for wound healing studies, because of their availability, low cost in terms of housing and maintenance, ease of handling, and their accelerated rate of healing [36,37].
The rat skin
The rat skin is composed of the major layers (epidermis, dermis) of human skin, however rat skin does not perfectly mimic the human skin; many discrepancies between human and rat skin exist. Compared to humans, rats are considered “loose skinned animals” their skin is elastic and not strongly adherent to the underlying structures [38]. Also the presence of the panniculus carnosus muscle in rodent skin, make it markedly different from human skin [36]. The panniculus carnosus, and the elasticity of the rats skin make contraction the major mechanism of wound healing in rats, whereas in humans reepithelialisation and granulation tissue formation are the major mechanisms involved [37]. Also, in rats, the thickness, vascularity and other skin characteristics are constantly undergoing cyclic, rapid and extensive changes, collectively referred to as the "skin cycle” [39] These changes are intimately associated with cyclic, wave-like patterns of hair growth: Areas of skin with growing hair (anagen skin) being markedly thicker than areas with resting hair (telogen skin) [39]. Because of the morphological and physiological contrast between anagen and telogen skin, the healing of experimental burns varies significantly with the phase of the skin.
Finally, rats have the ability to synthesize vitamin C in the liver [40], whereas in humans vitamin C source is exogenous, because humans have lost the capacity to synthesize it [41].
This metabolic difference is relevant, regarding the role played by vitamin C in wound healing, as an anti-oxidant and its role in collagen synthesis [42].
Determining the rat’s skin cycle
When using rats as burn models, the influence of the animal’s skin cycle, should be taken into consideration; therefore it is important to estimate the hair growth pattern before inflicting the burns [43]. By doing so, the investigator minimises variability in the depth of burns related to the skin cycle, and ensures the use of homogenous groups of animals at the same skin cycle [39]. As a practical method to achieve this goal, is the preparation of areas in identical hair growth phase by plucking or depilating telogen areas, using (barium sulphide) [39] or a wax/rosin mixture [44]. Removing all the hair shafts induces the anagen phase. As an alternative solution, investigators can use only younger rats which have synchronized hair follicle cycle [36].
Experimental design
Ethics approval
All experiments should be performed in accordance with current guidelines for the care and use of laboratory animals. Such guidelines intend to assist investigators to plan and conduct animal experiments in accord with the highest scientific, humane, and ethical principles [45]. In many countries, Animal Ethics Committee (AEC) is institutionalised and legislation requires an institutional and project license before testing on vertebrates is carried out [46].
Animal handling and housing
The animals used are of the same age group, approximately of similar weights. Regarding sex, in most studies, animals of either sex are used. However, the choice of male rats will prevent the variations in hormonal cycles that could intervene in the process of tissue repair when using female rats [47]. Before their use, newly received animals should be acclimatized to their new surroundings [48]. Knowing that rats are nocturnal animals, with most activity (eating, drinking, mating) are typically done at night [49], the acclimatization period is also used to implement a 12 h light/12 h dark cycle, permitting to rats and investigators to share peak activity periods. This 12 hr shift will require a 2-week accommodation period for the rat [48]. The acclimatization period will also be used to screen the animals’ health status. Rats are kept in individual clean cages and housed at a well-ventilated animal facility, under standard controlled conditions of temperature (22 ± 1ºC to 30 ± 1ºC) and relative humidity (35 ± 5% to 65 ± 5%) [7]. Because animals under stress are likely to be more variable than those kept in optimum conditions [50]; care should be taken to avoid unnecessary stress and discomfort to the rats. Also throughout the experiment, animals should have free access to water and feed (rodent chow).
Group assignments
Rats are randomly assigned to either control or experimental (treatment) groups.
In drug testing experiments, the control group is constituted of injured not treated rats. A sham group can also be added, this additional control group is constituted of animals exposed to all pre- and postoperative procedures (anaesthesia, shaving...), but instead of receiving a real burn, these animals receive a sham burn. For example in a scald burn model, rather than using hot water, the investigator uses water at room temperature [28]. Sham group animals receive no treatment and are kept undisturbed for the whole study protocol [31].
Treated (experimental) groups consist of burnt treated animals: besides the group treated with the drug being tested, two other treated groups are usually required: A vehicle control group and a comparative control group.
Animals receiving the treatment being tested can be assigned to more than one group in case we need to test different dosages of the treatment in question [31].
In the vehicle control group, rats receive a supposedly innocuous substance, to determine whether the vehicle alone has any effects when compared with the untreated control [51]. The vehicle drug can be saline [52], cold-cream [53,54] or any other blank ointment [32] used as a vehicle for the experimental compound [51]. In the comparative control group, animals are treated by a reference drug, used to treat burns, in order to make direct comparison with the drug being tested. Silver sulfadiazine (SSD) is frequently used as a comparative drug [30]. This silver compound is the mainstay of conventional topical treatment for burns, and a standard against which non-silver therapies are evaluated [10]. Other drugs used in the comparative control group include any burn healing drug like dexpanthenol [32], Nitrofurazone [55].
In some studies, no control group is used; instead a same animal serves as his own control. By doing so, investigators can reduce the number of used animals and ovoid the effect of interindividual variability [56]. In this model, small separated burn injuries are performed on each animal at different sites: Two or more burns are made along the head-to-tail axis [57,58]. One injury is used as control, and the other(s) as experimental.
Using a rat as his own control enables researchers to obtain comparable levels of information from fewer animals. Such practice is consistent with the principal of Reduction, one of the Three Rs (3Rs) (along with Replacement and Refinement) described first by Russell and Burch [50]. These three principles aim to improve animal welfare and promote a more ethical use of animals in testing. In many countries, these 3Rs are now explicit in legislation governing animal use.
Experimental group size: N factor
When choosing an experimental group size, two important elements should be taken into account: statistics and ethical considerations. The chosen group size must ensure the generation of statistically significant results [51] investigators may consult a statistician, computer software’s, or use on-line internet sites to calculate the figures [59].
For ethical reasons, it is important to use the minimum number of animals necessary to achieve the scientific objectives [60]. Other practical considerations determining experimental group size include costs and space [51].
Preoperative preparation
Anaesthesia
Since all anaesthetic drugs alter normal physiology in some way, their use during animal experimentations, may confound physiologic results [61]. Nevertheless, the use of anaesthetics is compulsory in animal burn models, due to the great amount of pain caused by this type of injury [13].
Proper general anaesthesia provides a state of unconsciousness, analgesia, muscle relaxation, and a-reflexia [62]. Due to their high metabolic rate and their inability to vomit, rats are not fasted routinely prior to anaesthetic events [63] Anaesthesia can be achieved using inhalational anaesthetics or injectable anaesthetics.
Inhalant anaesthesia enables regulation of anaesthetic depth in real time; they can be used for short or long durations of anaesthesia [62]. Furthermore, inhalant anaesthesia has the advantage of rapid recovery with a correspondingly short recovery monitoring period [64]. However, volatile anaesthetics do not provide perioperative analgesia, and their use requires specialized equipment which is not available in all facilities [65]. In burn experiments, currently-used inhalational anaesthetic include isoflurane [28,66] and halothane 4% [67]. Injectable anaesthetics are a convenient way to administer anaesthesia, because their use does not require specialized equipment. When used, doses administered depend on each animal’s body weight; therefore rats must be weighed on the day of anaesthesia.
Ketamine and xylazine combination is commonly used for the anaesthesia of rodents [63].
The combination Ketamine-xylazine provides a surgical anaesthesia plane and pain relief in rats [68]. Drugs are administered with Intraperitoneal (ip) injection which is generally the route used in laboratory rats. However, if the investigator lacks technique and experiment, error of targeted site may accur [69]. As an alternative route, drugs can be administered intramuscularly (im). Anaesthetic doses administered vary greatly in the literature (Table 1)
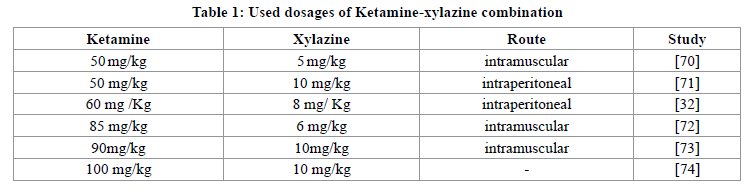
Skin preparation
The rat’s dorsum is the ideal site to create the burn, because this location is difficult for the animal to reach; which prevents further self-inflicted injuries to the wound area. Furthermore, this location facilitates the subsequent operations, like monitoring the evolution of the burn, taking photos, and applying treatments.
To ensure even burn wounding, the hair on the burn site is shaved off, using a sterile blade or a standard electric shaving machine. In order to obtain an absolutely smooth and hairless skin, any residual hair is removed with commercial depilatory crème [70-76]. The contact time with the cream, is made according to the manufacturer’s instructions, the cream is then removed and the skin is washed with water and then dried.
After hair removal, the skin can be disinfected with an antiseptic: povidon-eiodine 1% solution [72,77], 70% ethanol [78], savlon [74], 2% chlorhexidine scrub [79]. Since clipping and the depilation procedures produce irritation to the skin, it is recommended that 24 h should elapse before the bum is inflicted, allowing time for the skin irritation to disappear [20,21,70].
Burn infliction
Wound size
The size of the wound is expressed as % of Total Body-Surface Area (TBSA); the most commonly used formula to estimate bodysurface area in small experimental animals is the one postulated by Meet [80]. The Meeh's formula: A=k × W2/3, where: A=area in cm2; W=body weight in grams; k=a proportionality constant known as Meeh’s constant, in rats k=10 [6,80]. In burn experiments, the total area of the burn should be small enough to avoid a major systemic response in the animal subject, yet large enough to allow multiple biopsies and sampling [77]. According to Abdullahi et al. [81], inflicting a burn wound of greater than 60% TBSA, results in reduced survivability and is not sustainable for experimentation.
Burn infliction methods
Experimental cutaneous burns can be inflicted by various means, provided that the chosen method is simple, safe, and reproducible, i.e., able to produce burns of consistently similar extents and depths [77]. In studies on experimental burns, investigators using a particular burn model should present a detailed description of the method and instruments used for inflicting the burn injury. That includes: The size, location and depth of the burn, the temperature of the inflicting method, the duration of exposure, the pressure applied with the inflicting instrument. Many published studies fail to provide some of these detailed [9].
Two of the most used burn models in rats are: the scald burn model and the contact burn model using a hot metal instrument. Other less popular methods exist also.
The scald burn model
The most widely used scald burn model is the one proposed by Mason and Walker [82]. They developed a watertight burn apparatus made of a thin metal half-cylinder with an aperture of calculated dimensions corresponding to the size and location of the burn to be produced. The anesthetized rat is placed in supine position (back first) into the burn apparatus, then the burn apparatus is floated in a boiling water bath just far enough to submerge the exposed area of the animal’s skin. Time of exposure determines the burn’s depth: a 3 s exposure produced partial-thickness burn, and 10 s a full-thickness burn.
The Walker and Mason apparatus can be replaced by any simpler template permitting to immerse just a part of the rat’s body [83,84]. The possibility to vary water temperature, time of exposure, and the burned area make this method effective for reproducing almost any kind of thermal wound [85].
Table 2 shows different studies using different water temperatures and exposure time.

Once the exposure time is over, it is important to stop ongoing burning by placing the animal on its back on absorbent bench paper to remove any remaining hot water or applying ice-cold water to the scald [85-91]. The structure used to hold the rat must be watertight enough to prevent scalding of tissue beyond the intended site. Such requirement is one of the difficulties when using this scalding method [91]. Other disadvantages include the cumbersome nature of this technique and the significant potential risks to the investigator [77].
The contact burn model
In the direct contact method, a heated metal instrument is applied to the skin to induce the desired burn surface area. Inflicting burns by this method is both simple and safe for the investigator [77]. Instruments of different shapes and forms can be used: a bar, a plate, a rod, a block, and weights. The metal instrument is usually made of brass [34] aluminium [78] or stainless steel [36]. Thermal conductivity of the chosen materiel should permit efficient heat transfer by conduction [77]. For the same time of contact, metals with high thermal conductivity like brass would cause a greater depth of burn compared to metals with lower thermal conductivity like stainless steel [36].
The degree of the burn is determined by the temperature of the heated instrument, effective contact time and amount of pressure applied by the investigator or by the weight of the instrument. The shape and size of the injury depends on the shape (square, circle) and size of the contact surface of the metal instrument.
The most widely used method to heat the metal instrument is by immersion it into a hot water bath. Heating the metal instrument by using this source of thermal energy is both simple and convenient and allows investigators to reach temperatures up to boiling point (100ºC) [34].
To reach temperatures above 100ºC, the metal instrument can be heated with other sources of thermal energy like solar rays, burning inflammable liquids, electric energy or naked flame [92]. For the latter, a simple full-thickness burn model exists: a 2 × 2 cm brass plate is held for 2 min in the flame of a Bunsen burner and, subsequently, pressed against the prepared skin of the rat for 10 s [93].
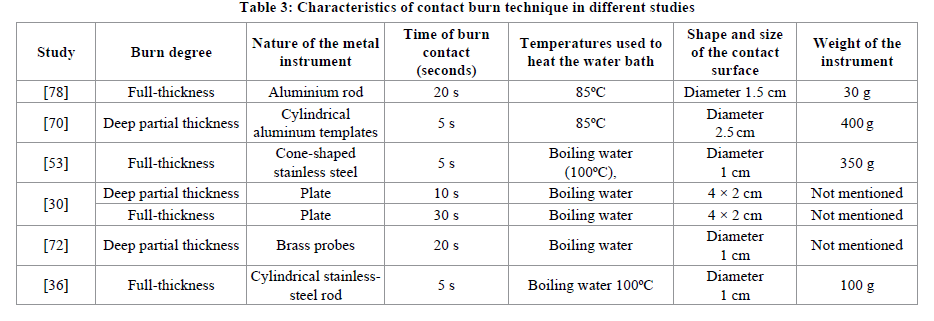
When using boiling water, some investigators do not measure the temperature of the water assuming that the boiling point of water is 100ºC [34]. They also do not directly monitor the temperature of the heated instrument, assuming that after a few minutes of immersion, the heated instrument had equilibrated with the temperature of the heated water [34]. To be more accurate, the temperature of the hot water bath can be measured conveniently using a thermometer [73] and to ensure that equilibration has indeed taken place the temperature of the metal instrument can be directly monitored using a thermometer [53], a digital multimeter [78] or a thermocouple [36].
Table 3 shows different studies using different water temperatures and exposure time.
As far as practicable, when inflicting the burns, the boiling water bath should be placed as close as possible to the rat, to prevent excessive heat loss when transferring the metal instrument from the bath to the skin [36]. For the same reason burns should be created immediately after removing the instrument from the water bath. Just prior to application, the heated instrument is rapidly blotted dry to prevent water droplets from creating a steam burn on the skin leading to irregular burn margins [77].
It would be convenient to provide more than one burning instrument for each group. By doing so, investigators can reduce the interval between the infliction of a burn and another within the same group, and thus avoiding large variations in the evaluation of healing time [73].
In order to standardize burn depth, when inflicting burns, the heated instrument should rest on its own weight on the rat’s skin without using additional pressure. The effect of a minimal pressure that can be used to ensure a perfect contact between the instrument’s surface and the underlying skin, can be neglected because according to Kaufman et al. [21], thermal energy transfer by conduction does not depend on the pressure employed, but primarily on the temperature gradient between the instrument and the skin and the path length through which the heat flows. Throughout the duration of burn infliction, the metal instrument should be held perpendicular to the skin, using minimal pressure [70], to ensure uniform distribution of pressure from the weight of the instrument [36]. The duration of contact is measured by an accurate stopwatch [78].
Other burn models
Modified scalding technique
This technique is proposed by Pfurtscheller et al. [85] to produce full-thickness scald burns.
In this technique the rat, in the prone position, is placed onto a specially designed template which is firmly pressed on the shaved back of the animal, exposing throught a rectangular aperture, approximately 20% of its body surface area (BSA). Scald burns are made by pouring boiling water from a small electric kettle into the rectangular aperture. After 10 s, hot water is allowed to trickle out into a washbasin.
Electric contact burn device
The original tip of an ordinary 40 W soldering iron is replaced with a square copper plate (10 × 10 mm). In order to allow precise temperature monitoring of the copper plate, an electronic temperature controller with a thermocouple-type feedback sensor is added. The desired stamp temperature (100, 150 and 200ºC) is reached 5 min after switching on the electric current. The device is applied to the skin under its own weight (85 g) during 9 s to create Partial-thickness burns [94,95].
Kistler’s apparatus
Kistler et al. [92] developed a burning apparatus for inflicting reproducible contact burns, in this apparatus, temperature, time, and pressure of the stamp can be individually set, controlled, and reproduced. The temperature is produced by an iron coil with a heat output of 200 Watts. However, the disadvantage of such apparatuses is that they have to be placed at a fixed inclination and the animals need to be kept in a fixed position.
Hot molten wax model
Partial thickness burns are created using a metal cylinder with a circular opening of 300 mm2 area and a capacity to hold 2-4 g of wax. Hot molten wax (at 80°C) is poured into the cylinder placed on the back of the rat. After solidification of the wax (8 min) the metal cylinder with wax is removed, leaving a circular burn wound [33,96].
Hot sponge model
Partial-thickness burn injury is made using a 3 × 3 × 3 cm3 sponge kept in boiling water. The hot sponge is placed over the rats shaved dorsum for 35 s, with no extra pressure but the sponges’ own weight [71].
Hot cloth model
In this model described by Bahar et al. [97] burns are created using a 2.5 × 2.5 cm, 5 mm-thick piece of absorbent lint cloth that had been immersed in boiling water (100ºC). The cloth is applied on the shaved dorsum and kept for 5 s to create superficial partialthickness dermal burns, and for 12 s to produce deep dermal burns. This method produced reliable superficial dermal burns, but the deep dermal burn wound model was more difficult to establish.
Hot glass chamber model
In this burn model developed by Suzuki et al. [98], the rat’s skin is in contact with a glass chamber through which heated water is pumped constantly. Water is heated in a large reservoir by an electric heater which can automatically control water temperature. A thermocouple needle is inserted into the chamber so that the temperature of the water flowing over the skin is accurately measured and maintained
Fractional CO2 laser burn creation
The rats are exposed to partial-thickness skin burns using a CO2 Fractional Laser System with the following setting parameters: density: (level: 20; line: 29 × 29; dot: 0841); energy level: 25 MJ; depth level: 4 [52].
Post burn recovery and monitoring
Fluid resuscitation
During burn, there is a generalized increase in capillary permeability due to heat effect and damage. This causes plasma to leak out from capillaries to interstitial spaces. This plasma loss is the cause of hypovolemic shock in burns [16]. Usually, partial or fullthickness burns of 20% or less TBSA are supported by rats, without any need of resuscitation [82,99]. However, in experimental burns involving large body surface area, rats could land up in hypovolemic shock if not adequately resuscitated. Fluid resuscitation requirements are determined using the Parkland formula (4 mL/% TBSA burn/kg) [100]. Immediately after burn infliction, rats can be fluid resuscitated intraperitoneally with lactated Ringer’s solution [72] or isotonic sodium chloride solution [29,85].
Monitoring anaesthetic recovery
During general anaesthesia, core body temperature decreases, therefore as part of anaesthesia management, it is essential to preserve the animal’s body heat as a protection from systemic hypothermia [62]; this can be achieved by providing heat through warming pads [85] or infrared lamps.
Scab excision
Within few days post burn, deep burns develop a thick scab that does not allow direct observation of the reepithelialisation underneath the scab. To allow the assessment of wound reepithelialisation, dead tissues can be excised using a sterile surgical blade [54,78].
Post-operative pain-relief
The use of analgesics in postoperative care to relief pain and suffering is an ethical requirement central to many laws that regulate animal experimentation [101]. However in many burn studies, the use of analgesics in postoperative care is not mentioned [9]. In full thickness burn models, some authors justify the lack of use of post-burn analgesics by the fact that nerve endings in the skin are destroyed and thus the skin becomes insensate [86]. The goal of pain relief in experimental animals is to achieve a balanced state during which an animal is neither significantly hindered by pain nor adversely affected by the side effects of analgesics. Objective pain assessment is required to determine the need for, or the appropriate dosage of an analgesic agent [102]. Unfortunately, detecting painful behaviour in these animals is challenging and requires training and skilled observation. Methods for grading severity of pain and distress in laboratory animals are available and are used in some countries’ legislation [103]. In postoperative care, opiate derived analgesics, are used to relief pain: strong opioids for extensive burns and weaker opioids for less extensive burns [9]. Drugs can be administered by injection (subcutaneously; intramuscularly) or orally administered in the drinking water supplied to animals. Among the most commonly used opioids for postoperative pain in laboratory animals, is Buprenorphine, mainly because of its long duration of action [104]. Other used opioids include piritramide [85], fentanyl citrate [71] and codeine phosphate hemihydrates [79]. Other used analgesics include non-steroidal anti-inflammatory drugs (NSAIDs), like dipyrone sodium [73]. Note that NSAIDinduced gastroenteropathy is a significant limitation to the use of this class of drugs [105].
Post burn monitoring and burn treatment and dressing
Animals are periodically weighed and wounds are inspected daily for signs of infection; rats that show signs of infection are separated and excluded from the study [7]. Depending on the experimental protocol and the tested drug, the administration of treatments can be either locally (topical) or systemically (oral or parenteral). Generally the surface of wounds are not covered by any dressing [30] because usually rats try vigorously to free themselves from anything that they consider foreign or irritating. Pfurtscheller et al. [85], has proposed an Armor-Like Wound Protector’’ that could be useful when the experimental protocol requires the use of a dressing.
References
[1] D.J. Hauben, E. Yanai, D. Mahler, Burns, 1981, 7, 6, 383-388.
[2] T.E. Abrams, R.J. Ogletree, D. Ratnapradipa, M.W. Neumeister, Burns., 2016, 42, 1, 152-162.
[3] K.C. Lee, K. Joory, N.S. Moiemen, Burns & Trauma., 2014, 2, 4, 169-180.
[4] M. Pećanac, Z. Janjić, A. Komarcević, M. Pajić, D. Dobanovacki, S.S. Misković, Med. Pregl., 2013, 66, 5-6, 263-267.
[5] P. Dahiya, Front Biosci., (Landmark Ed), 2009, 1, 14, 4962-4967.
[6] T.N. Meyer, A.L. Silva, Acta Cir. Bras., [serial online], 1999, 14, 4.
[7] R. Thakur, N. Jain, R. Pathak, S.S. Sandhu, Evid. Based. Compl. Alter. Med., 2011, 17.
[8] S. Asko-Seljavaara, Acta. Physiol. Scand. Suppl., 1986, 554, 209-213.
[9] J.K. Mitsunaga Junior, A. Gragnani, M.L. Ramos, L.M. Ferreira, Acta. Cir. Bras., 2012, 27, 6, 417-423.
[10] P.I. Jewo, I.O. Fadeyibi, O.S. Babalola, L.C. Saalu, A.S. Benebo, M.C. Izegbu, O.A. Ashiru. Ann. Burns. Fire. Disasters.,2009, 22, 2, 79-82.
[11] M.D. Peck, Burns, 2011, 37, 1087-1100.
[12] L.H. Evers, D. Bhavsar, P. Mailänder, Exp. Dermatol., 2010, 19, 9, 777-783.
[13] H. Laycock, J. Valente, C. Bantel, I. Nagy, Eur. J. Pharmacol., 2013, 716, 1-3, 169-178.
[14] R. Gómez, L.C. Cancio, Emerg. Med. Clin. North. Am., 2007, 25, 1, 135-146.
[15] D.K. Meyerholz, T.L. Piester, J.C.Sokolich, G.K. Zamba, T.D. Light, Int. J. Exp. Pathol., 2009, 90, 1, 26-33.
[16] V.K. Tiwari, Indian. J. Plast. Surg., 2012, 45, 2, 364-373.
[17] S. Hettiaratchy, P. Dziewulski, BMJ., 2004, 328, 7453, 1427-1429.
[18] M.J. Hoekstra, P. Hupkens, R.P. Dutrieux, M.M.C. Bosch, T.A. Brans, R.W. Kreis, British. J. Plastic. Surg., 1993, 46, 7, 585-589.
[19] J. Katahira, K. Murakami, F.C. Schmalstieg, R. Cox, H. Hawkins, L.D. Traber, Am. J. Physiol. Lung. Cell. Mol. Physiol., 2002, 283, 5, L1043-L1050.
[20] R.L. Sobec, C.N. Dobreanu, C.N. Magdas, A. Nagy, E. Dinte, M. Fodor, A. Somcutean, I. Tichil, M. Nastai, L.Fodor, Rev. Med. Chir. Soc. Med. Nat. Iasi., 2014, 118, 4, 1047-1053.
[21] T. Kaufman, S.N. Lusthaus, U. Sagher, M.R. Wexler, Burns, 1990, 16, 1, 13-16.
[22] S.H. Heo, K.B. Han, Y.J. Lee, J.H. Kim, K.H. Yoon, M.D. Han, K.S. Shin, W.J. Kim, Anim. Cell. Syst., 2012, 16, 3, 207-214.
[23] A. Pandurangan, R.L. Khosa, S. Hemalatha, Der. Pharmacia. Lettre., 2010, 2, 3, 444-449.
[24] P. Suruse, M.K. Kale, M. Gunde, N. Amnerkar, A.K. Pathak, Der. Pharmacia. Lettre., 2011, 3, 4, 200-206.
[25] K.N. Anitha, B. Sasivardhan Reddy, C. Velmurugan, M. Akmal Ali Baig, B. S. Ashok Kumar, Der Pharmacia Lettre, 2015, 7, 9, 310-319.
[26] S. Avinash, D.V. Gowda, J. Suresh, A.S. Aravind Ram, A. Srivastava, R.A.M. Osmani, Der Pharmacia Lettre, 2016, 8, 9, 52-63.
[27] F.X. Santos Heredero, C. Hamann, J.M. Obispo Martin, C. Rodriguez Arias, S. Coca Menchero, Ann. Burns. Fire. Disasters., 1996, 9, 2.
[28] L.A. Baer, X. Wu, J.C. Tou, E. Johnson, S.E. Wolf, C.E. Wade. Bone, 2013, 52, 2, 644-650.
[29] M. Goto, V. Samonte, M. Khan, F. Haque, A. Goyal, W. Al-Ghoul, S. Raziuddin, N. Fazal, T. Ravindranath, R.L. Reed, R.L. Gamelli, M.M. Sayeed, Shock., 2002, 18, 6, 523-528.
[30] M.R. Akhoondinasab, M. Akhoondinasab, M. Saberi, World. J. Plast. Surg., 2014, 3, 1, 29-34.
[31] N. Bhatia, G. Kaur, V. Soni, J. Kataria, R.K. Dhawan, Burns. Trauma., 2016, 4, 7.
[32] P. Khazaeli, M.K. Aramouzian, S.H. Rohani, B. Sadeghirad, N. Ghalekhani, Iranian. J. Pharma. Res., 2014, 13, 1, 243-251.
[33] M. Bharat, D.K. Verma, V. Shanbhag, R. Singh Rajput, D. Nayak, A. Amuthan, Int. J. Pharm. Sci. Rev. Res., 2014, 25, 2, 145-148.
[34] M. Hoşnuter, A. Gürel, O. Babucçu, F. Armutcu, E. Kargi, A. Işikdemir, Burns., 2004, 30, 2, 121-125.
[35] M.M. Sayeed, Shock, 2005, 1, 40-44.
[36] E.Z. Cai, C.H. Ang, A. Raju, K.B. Tan, E.C. Hing, Y. Loo, Y.C. Wong, H. Lee, J. Lim, S.M. Moochhala, C.A. Hauser, T.C. Lim, Arch. Plast. Surg., 2014, 41, 4, 317-324.
[37] R.D. Galiano, J. Michaels, M. Dobryansky, J.P. Levine, G.C. Gurtner, Wound Repair Regen., 2004, 12, 4, 485-92.
[38] W.A. Dorsett-Martin, Wound Repair Regen., 2004, 12, 6, 591-599.
[39] B.E. Zawacki, R.J. Jones, Br. J. Plast. Surg., 1967, 20, 4, 347-354.
[40] S.F. Djurašević, J. Djordjević, T. Drenca, N. Jasnić, G.C. Vijić, Arch. Biol. Sci., Belgrade, 2008, 60, 2, 169-173.
[41] G. Drouin, J.R. Godin, B. Pagé, Curr Genomics., 2011, 12, 5, 371-378.
[42] S. Chambial, S. Dwivedi, K.K. Shukla, P.J. John, P. Sharma, Indian. J. Clin. Biochem., 2013, 28, 4, 314-328.
[43] J.S. Knabl, G.S. Bayer, W.A. Bauer, I. Schwendenwein, P.F. Dado, C. Kucher, R. Horvat, E. Turkof, B. Schossmann, G. Meissl, Burns, 1999, 25, 3, 229-235.
[44] H.B. Chase, Physiol. Rev., 1954, 34, 113-126.
[45] https://grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf
[46] http://ec.europa.eu/environment/chemicals/lab_animals/legislation_en.htm
[47] D.C. Teves, A.C.V. Cabral, M.J. Simões, J.R.L. Kulay, J. Brasileiro. De. Medicina., 1986, 50, 39-44.
[48] P.E. Sharp, M.C. La Regina, The Laboratory Rat, CRC Press LLC, New York, US, 1998.
[49] S. Ambrish, Der Pharmacia Lettre, 2010, 2, 1, 396-407.
[50] W.M.S. Russell, R.L. Burch, Methuen, London, 1959, 238.
[51] P.D. Johnson, D.G. Besselsen, Oxford Journals: Science and Mathematics ILAR Journal, 2002, 43, 4, 202-206.
[52] S. Bardaa, N. Chabchoub, M. Jridi, D. Moalla, M. Mseddi, T. Rebai, Z. Sahnoun, J. Surg. Res., 2016, 201, 2, 464-472.
[53] G.D. Mogoşanu, F.C. Popescu, C.J. Busuioc, I. Lascăr, L. Mogoantă, Rom. J. Morphol. Embryol., 2013, 54, 1, 107-113.
[54] A.G. Pirbalouti, M. Yousefi, H. Nazari, I. Karimi, A. Koohpayeh, Electronic J. Biol., 2009, 5, 3, 62-66.
[55] B. Jaiprakash, Chandramohan, D. Narasimha Reddy, Ancient Science of Life, 2006, 25, 3-4, 16-18.
[56] D.C. Cambier, G.G. Vanderstraeten, M.J. Mussen, J.T. Van der Spank, Plast. Reconstr. Surg., 1996, 97, 3, 555-559.
[57] A. Ezzati, M. Bayat, A. Khoshvaghti, Photomed. Laser. Surg., 2010, 28, 5, 603-611.
[58] A. Golberg, M. Villiger, S. Khan, K.P. Quinn, W.C. Lo, B.E. Bouma, M.C. Jr Mihm, W.G. Jr Austen, M.L. Yarmush, J. Inves.t Dermatol., 2016, 136,11, 2297-2304.
[59] J.E. de Aguilar-Nascimento, Acta. Cir. Bras., 2005, 20, 1.
[60] M.F.W. Festing, D.G. Altman. Oxford. J. Sci. Mat. ILAR J., 2002, 43, 4, 244-258.
[61] J.E. Bazin, J.M. Constantin, G. Gindre, Ann. Fr. Anesth. Reanim., 2004, 23, 8, 811-188.
[62] S. Gargiulo, A. Greco, Gramanzini, S. Esposito, A. Affuso, A. Brunetti, G. Vesce, ILAR J., 2012, 53, 1, E55-69.
[63] M.B. Struck, K.A. Andrutis, H.E. Ramirez, A.H. Battles, J. Am. Assoc. Lab. Anim. Sci., 2011, 50, 3, 344-348.
[64] S.O. Hacker, C.E. White, I.H. Black, Contemp. Top. Lab. Anim. Sci., 2005, 44, 5, 7-12.
[65] H.N.C. Alves, A.L M. da Silva, I.A.S. Olsson, J.M.G. Orden, L.M. Antunes, J. Am. Assoc. Lab. Anim. Sci., 2010, 49, 4, 454-549.
[66] D.K.Meyerholz, T. L.Piester, J. C.Sokolich, G K. Zamba, TD. Int J Exp Pathol, 2009, 90, 1, 26-33.
[67] M.Y. Zhang, G.M. Wang, F.L. Li, L. Dong, Y.B. Xu, J.S. Chiang, Colombian J. Anesthesiol., 2013, 41, 2, 82-87.
[68] M.C. Giroux, P. Hélie, P. Burns, P. Vachon, Exp. Anim., 2015, 64, 3, 253-260.
[69] R.E. Lewis, A.L. Kunz, R.E. Bell, Lab. Anim. Care., 1966, 16, 6, 505-509.
[70] R. Mohd Zohdi, Z. Abu Bakar Zakaria, N. Yusof, N. Mohamed Mustapha, M.N. Abdullah, Compl. Alter. Med., 2012, 843025.
[71] U. Ozcelik, Y. Ekici, H.Y. Bircan, C. Aydogan, S. Turkoglu, O. Ozen, G. Moray, Med. Sci. Monit., 2016, 5, 22, 1903-1909.
[72] I. Yaman, A.S. Durmus, S. Ceribasi, M. Yaman, Vet. Med., 2010, 55, 12, 619-624.
[73] D. dos Santos Tavares Pereira, M.H.M. Lima-Ribeiro, N.T. de Pontes-Filho, A.M. dos Anjos Carneiro-Leão, M.T dos Santos Correia, J. Biomed. Biotechnol., 2012.
[74] N. Tanideh, M.H. Haddadi, M.H. Rokni-Hosseini, M. Hossienzadeh, D. Mehrabani, K. Sayehmiri, O. Koohi-Hossienabadi, World. J. Plast. Surg., 2015, 4, 1, 16-23.
[75] A.G. Pirbalouti, S. Azizi, A. Koohpayeh, Rev. Bras. Farmacogn. Braz. J. Pharmacogn, 2012, 22, 2, 397- 403
[76] K. Lee, B. Lee, M.H. Lee, B. Kim, K.S. Chinannai, I. Ham, H.Y. Choi. BMC Compl. Alter. Med., 2015, 15, 213.
[77] A.J. Singer, L. Berruti, H.C. Thode Jr, S.A. McClain, Acad. Emerg. Med., 2000, 7, 1, 1-6.
[78] N.K. Upadhyay, R. Kumar, M.S. Siddiqui, A. Gupta, Evid. Based. Compl. Alter. Med., 2011, 2011, 9.
[79] A.P. Campelo, M.W. Campelo, G.A. Britto, A.P. Ayala, S.B. Guimarães, P.R. Vasconcelos, Acta. Cir. Bras. 2011, 26 Suppl, 1, 38-42.
[80] E. Gouma, Y. Simos, I. Verginadis, E. Lykoudis, A. Evangelou, S. Karkabounas, Lab. Anim., 2012, 46, 1, 40-45.
[81] A. Abdullahi, S. Amini-Nik, M.G. Jeschke, Cell. Mol. Life. Sci., 2014, 71, 17, 3241-3255.
[82] H.L. Walker, A.D. Mason Jr, J. Trauma., 1968, 8, 6, 1049-1051.
[83] H.F.X. Santos, C. Hamann, M.J.M. Obispo, A.C. Rodriguez, M.S. Coca. Ann. Burns. Fire. Disasters., 1996, 9, 2, 96Y102.
[84] M. Trop, E.J. Schiffrin, E.A. Carter, Burns., 1990, 16, 1, 57-59.
[85] K. Pfurtscheller, T. Petnehazy, W. Goessler, I. Wiederstein-Grasser, V. Bubalo, M. Trop, J. Trauma. Acute. Care. Surg., 2013,
74, 3, 932-935.
[86] Q. Yang, M.A. Orman, F. Berthiaume, M.G. Ierapetritou, I.P. Androulakis, J. Surg. Res., 2012, 176, 2, 549-558.
[87] A.M. Al-Mousawi, G.A. Kulp, L.K. Branski, R. Kraft, G.A. Mecott, F.N. Williams, D.N. Herndon, M.G. Jeschke, Shock., 2010, 34, 3, 261-268.
[88] T. Xie, Y. Niu, K. Ge, S. Lu, J. Surg. Res., 2008, 1, 147, 1, 9-14.
[89] A.D. Niederbichler, L.M. Hoesel, K. Ipaktchi, L. Olivarez, M. Erdmann, P.M. Vogt, G.L. Su, S. Arbabi, M.V. Westfall, S.C. Wang, M.R. Hemmila, J. Surg. Res., 2011, 165, 1, 128-1235.
[90] M.R. Hemmila, A. Mattar, M.A. Taddonio, S. Arbabi, T. Hamouda, P.A. Ward, S.C. Wang, J.R. Jr Baker, Surg., 2010, 148, 3, 499-509.
[91] H.N. Paddock, G.S. Schultz, B.A. In: L.A. DiPietro, A.L. Burns (Ed.) Totowa, NJ 2003, 17-36.
[92] D. Kistler, B. Hafemann, K. Schmidt, Burns. Incl. Therm. Inj., 1988 14, 4, 297-302.
[93] O.L. Abbas, H. Borman, T. Bahar, N.M. Ertaş, M. Haberal, J. Burn. Care. Res., 2015, 36, 2, 47-54.
[94] A.P. Campelo, M.W. Campelo, G.A. Brito, F.V. Jamacaru, R.F. Leitão, P.R. Vasconcelos, Acta. Cir. Bras., 2015, 30, 6, 430-438.
[95] C.J. Mesquita, J.A. Leite, F.V. Fechine, C.J.L. Rocha, J.G. Leite, J.A. Leite Filho, R.A. Barbosa Filho. Burns., 2010, 36, 1, 97-108.
[96] P. Soman, P.K.M. Nagarathna, S.V. Harjubhai, R. Sarraf, S.S. Dani, Int. J. Curr. Pharm. Rev. Res. 2016, 7, 2, 106-109.
[97] T. Bahar, M.H.M.F.B. Bilezikci, T. Maral, H. Borman, Burns., 2007, 33, 1, S52-S53.
[98] T. Suzuki, T. Hirayama, K. Aihara, Y. Hirohata, Burns., 1991, 17, 6, 443-451.
[99] B.N. Bailey, S.R. Lewis T.G. Blocker Jr, Tex. Rep. Biol. Med, 1962, 20, 30-38.
[100] N. Tan, HC Jr Thode, AJ.Singer, Clin Exp Emerg Med, 2014, 30, 1, 1, 56-61.
[101] P. Hawkins, Lab. Anim., 2002, 36, 4, 378-395.
[102] P. Jablonski, B.O. Howden, K. Baxter, Lab. Anim., 2001, 35, 3, 213-222.
[103] FELASA (Federation of European Laboratory Animal Science Associations), Lab. Anim., 1994, 28, 2, 97-112.
[104] L.I. Curtin, J.A. Grakowsky, M. Suarez, A.C. Thompson, J.M. DiPirro, L.B.E. Martin, M.B. Kristal, Comp. Med., 2009, 59, 1, 60-71.
[105] R. Blackler, S. Syer, M. Bolla, E. Ongini, J.L. Wallace, PLoS One., 2012, 7, 4, e35196.



