Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 1
Regulatory Effects of Morus alba Aqueous Leaf Extract in Streptozotocin-Induced Diabetic Nephropathy
Abeer AA Salama1, Bassant MM Ibrahim1, Nemat A Yassin1, Sawsan S Mahmoud1, Amina A Gamal El-Din2 and Nermeen A Shaffie22Pathology Department, National Research Centre, Giza, Egypt
Abstract
This study aimed to investigate the effects of aqueous leaf extract of Morus alba on biochemical and histological changes in streptozotocin (STZ)-induced diabetic nephropathy. Male Wister albino rats were divided into five groups of six each: Group 1: Control group; received citrate buffer for 10 days. Group 2: Untreated diabetic rats injected intraperitoneally with 50 mg/kg of STZ; received distilled water for 10 days. Group 3-5: Diabetic rats; received Glimepiride and M. alba in 2 dose levels (250 and 500 mg/kg) for 10 days, orally. STZ-diabetic rats exhibited hyperglycemia accompanied with increased α-amylase levels, total cholesterol (TC) and kidney function, on the other hand, reduced high density lipoprotein cholesterol (HDL-C) and total antioxidant capacity (TAC) levels. Morus alba in its 2 dose levels (250 and 500 mg/kg) showed a significant lowering effect in serum glucose, α-amylase, TC levels and kidney function as well as increasing serum HDL-C and TAC levels comparable to STZ diabetic group. Microscopic examination of the pancreatic sections and kidney of diabetic rats treated with M. alba at higher dose revealed approximately normal structure, compared with STZ diabetic group. These results indicate that treatment with aqueous extract of M. alba may be effective in STZ- induced diabetic nephropathy as it showed higher hypoglycemic and hypolipidemic properties with more down-regulation of α-amylase and restore kidney function than, reference drug, Glimepiride potancy.
Keywords
Diabetic nephropathy, Morus alba, α-amylase, Total antioxidant capacity, Rats
Introduction
Nowadays, Diabetes Mellitus (DM) is a common chronic degenerative disease [1], about 250 million individuals worldwide suffer from diabetes and this number will increase by 2030 [2].
Pancreas, a vital endocrine-exocrine organ, helps in the digestion of carbohydrates, fats, and proteins via secreting several hormones and enzymes such as insulin which regulates carbohydrate metabolism in the body and maintains glucose passage across the cell membrane [3]. Therefore, the physiological function of the body may be directly affected by any change in the function of this organ [4].
Type 1 Diabetes Mellitus (DM) affects the ability of β-cells to secrete insulin which is required for blood glucose metabolism, leading to rise in blood glucose level and metabolic abnormalities. Hyperglycemia, retinopathy, neuropathy, nephropathy and cardiovascular disorders are associated with DM [5]. Moreover, inhibition of digestion and absorption of carbohydrates in small intestine are the most important measurements for diabetes treatment. That is achieved in two ways: the first is inhibition of alpha amylase, the enzyme that decomposes of carbohydrates, inducing inhibition of carbohydrates digestion and the second is inhibition of absorption via the sugar transporter systems, sodium/glucose co-transporters (SGLT1 and SGLT2) [6].
DM is a long life disease this push us toward searching for more effective and safer hypoglycemic plants origin which minimal toxic side effect [7]. Morus alba, known as white mulberry, is one of the Moraceae family [8]. It has long been used commonly in Ayurvedic medicine and many of traditional systems of medicine. It has a wide range of important pharmacological activities including antimicrobial, antioxidant, anticancer, immunomodulatory, hypocholesterolaemic, antidiabetic, nephroprotective, hepatoprotective effects. The plant contains ascorbic acid, folic acid, folinic acid, and also it is a very good source of carotene, vitamin B1 isoquercetin, quercetin, tannins, flavonoids and saponins [9].
Many Egyptian medicinal plants were tested in streptozotocin (STZ)-induced diabetic rat model and provided scientific validation to prove their antihyperglycemic activity. In view of supposed antidiabetic potential, the effects of aqueous extract of M. alba leaves on biochemical and histopathological changes in rats, pancreas in STZ-induced diabetic Nephropathy was investigated in this study.
Materials and Methods
Animals
Adult male albino Wistar rats, weighing 120-140 g were used in the current study. They were purchased from the National Research Centre (NRC; Giza, Egypt) animal house. Animals received human care in compliance with the guidelines of the animal care and use committee of the NRC. The animals were kept in a quiet place and were allowed free access to water and standard food pellets throughout the period of experiment. Experiments were performed according to the National Regulations of Animal Welfare and Institutional Animal Ethical Committee (IAEC).
Plant
Crude water extraction of Morus alba leaves
50 g dried Morus alba leaves were powdered and extracted with 50 m (w/v) of hot water (85ºC) for 3 h. The extract was filtered with Whatman No.1 filter paper and concentrated by heating at a non-boiling temperature near 100ºC, and then dried completely under vacuum at 25ºC. The dried extract (w/w=7.5 g, yield=15%) was used during experimentation and administered at dose of 250-500 mg/kg body weight.
Drugs
Glimepiride was obtained from Sanofi-Aventis, Egypt.
Chemicals
STZ, diethylether, sodium citrate and formaldehyde were obtained from Sigma Aldrich Chemical Co., USA, sterile saline was obtained from ADWIC Egypt.
Experimental Design
Type I diabetes was induced by a single intraperitoneal injection of STZ (50 mg/kg) dissolved in 0.1 M citrate buffer (pH 4.5) [10]. While, Group 1: Negative control group rats were treated with the same volume citrate buffer only without STZ for 10 days. Diabetes was confirmed at 48 h after STZ injection by measuring the glucose concentrations in blood samples obtained from the tail vein (One Touch SureStep Meter, LifeScan, Calif, USA). After diabetes was confirmed (>200 mg/ml), rats were assigned randomly into four groups: Group 2: Diabetic control rats (STZ). Group 3: Diabetic rats received, the reference drug, Glimepiride (0.5 mg/kg; p.o.) [11], for 10 days. Group 4 and 5: Diabetic rats received Morus alba (250 and 500 mg/kg; p.o.) for 10 days.
Methods
Preparation of blood samples
At the end of the period 10 days of treatment, the rats were anaesthetized with pentobarbital sodium and blood samples were collected for biochemical analyses. Three ml blood was withdrawn from the retro-orbital plexus vein of each rat for biochemical assays, Blood samples were left to clot at room temperature then centrifuged at 1500 rpm for 10 min for serum separation and serum samples were stored at -20ºC in order to determine blood glucose, α-amylase, Total Cholesterol (TC), High Density Lipoprotein (HDL) and Total Antioxidant Capacity (TAC).
Biochemical parameters
Determination of blood glucose and serum α-amylase levels were done [12,13] respectively, serum TC and HDL levels were estimated [14,15] respectively, and the serum level of TAC was determined [16], in addition, estimation of serum creatinine and urea were performed using Biodiagnostic commercial kits.
Histopathological study
At the end of the experiment; animals were sacrificed. Pancreas (splenic part), and kidneys were dissected and extracted from sacrificed animals. Organ tissues were fixed in 10% buffered formalin, processed through ascending grades of alcohol, cleared in xylene and prepared into paraffin blocks. Serial sections 5 microns thick were prepared from each block and stained with haematoxylin and eosin for routine histopathologic study. The sections were examined under an Olympus CX41 research microscope at the Pathology Department; National Research Centre. Slide tissue microphotography was done using CCD digital camera Olympus DP- 12 attached to the Olympus CX41 research microscope. Digital photomicrographic sections were taken at various magnifications.
Statistical analysis
Data are expressed as mean ± S.E. Data analysis was done using one way Analysis of Variance (ANOVA) followed by Least Significant Difference (LSD) test for multiple comparisons. Difference was considered significant when p is less than 0.05. SPSS (version 11) program was used to carry out these statistical tests.
Results
Effect of Morus alba on serum blood glucose and α-Amaylase levels
Animals treated with STZ (50 mg/kg, i.p) showed a significant increase in blood glucose and α-Amaylase levels after 10 days by 222 and 144% respectively, when compared to normal animals. Treatment to these diabetic animals with Morus alba (250 and 500 mg/kg, orally) induced a significant decrease in blood glucose levels after 10 days by 66% in both doses as well as in α-Amaylase levels by 13% and 48% respectively, also, treatment with reference drug Glimepiride caused a significant decrease in blood glucose levels by 69% as well as in α-Amaylase levels by 62%, after 10 days, when compared to STZ animals (Figure 1).
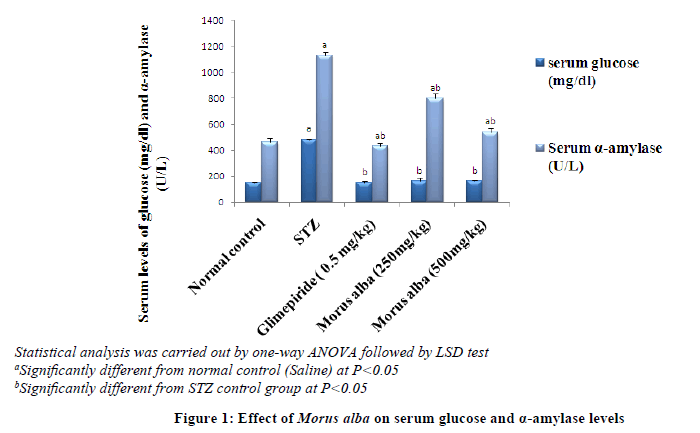
Effect of Morus alba on serum Total Cholesterol (TC) and high density lipoprotein cholesterol (HDL-C) levels
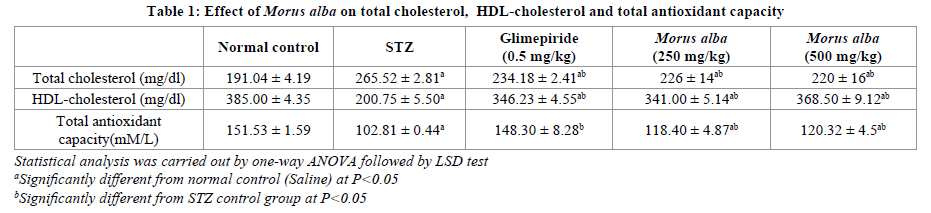
TC level was significantly increased in animals treated with STZ by 38.99% after 10 days but HDL-C level was significantly decreased by 47.86% when compared to normal animals. Treatment with both doses of M. alba caused a significant decrease in TC level by14.88% and 17.14% as well as showed a significant increase in HDL-C level after 10 days by 69.86% and 83.56% respectively, also Glimepiride caused a significant decrease in TC level by 11.80 % a significant increase in HDL-C level after 10 days by 72.47%when compared to STZ animals (Table 1).
Effect of Morus alba on serum Total antioxidant Capacity (TAC)
Animals treated with STZ showed a significant decrease in TAC after 10 days by 32.15% when compared to normal animals. While treatment with both doses of M. alba caused a significant increase in TAC after 10 days by 15.16% and 17.03% respectively, also Glimepiride caused a signiicant increase in TAC after 10 days by 44.25%when compared to STZ animals (Tables 1 and 2).
Effect of Morus alba on kidney function test
A significant elevation in serum creatinine and urea levels was observed in STZ-treated rats by 28.57% and 106.99% compared with those of normal group. Treatment of rats with both doses of M. alba restored the levels of serum creatinine by 15.56% and 18.89% and urea by 17.92% and 38.70%, respectively, also Glimepiride caused a significant decrease in serum creatinine and urea levels by 12.22% and 19.22% compared to those of STZ.
Effect of Morus alba on histopathological changes
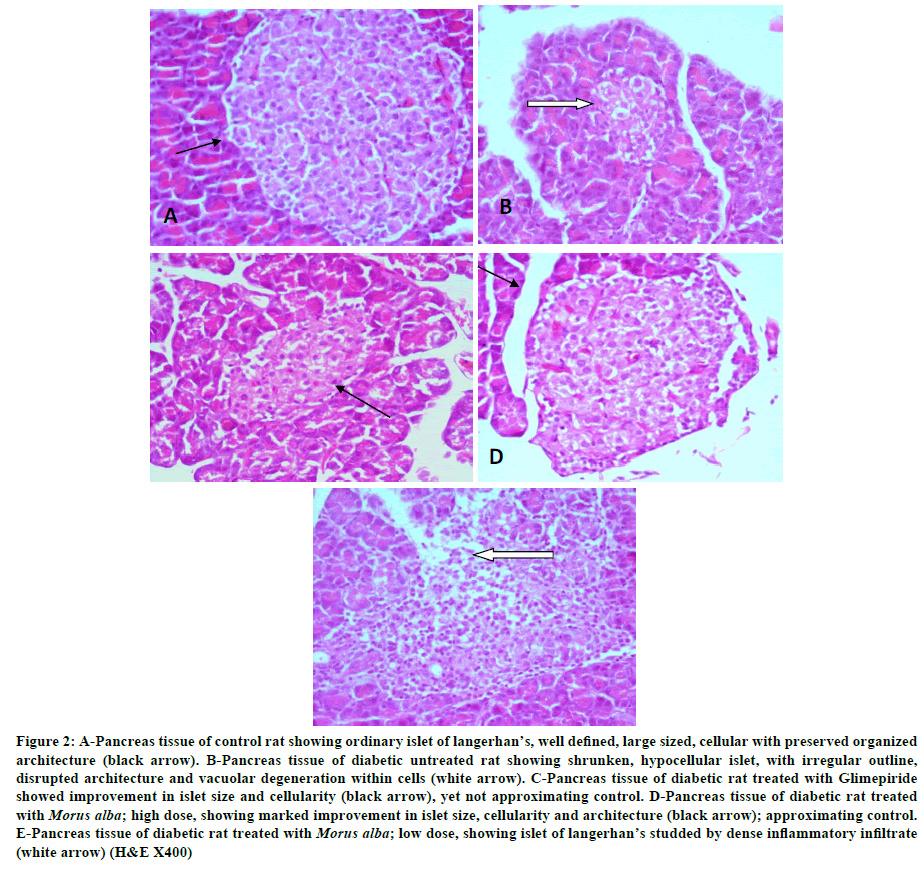
Pancreatic tissue of control rat showed islets of langerhan’s frequently distributed amidst pancreatic acini. Each Islet showed well defined borders, large size and abundant cellularity. Cells were arranged in organized manner with preserved architecture. Surrounding pancreatic acini were lined by pyramidal cells showing ample, apical cytoplasm and bland, basophilic, basoluminal nuclei (Figure 2A). Pancreatic tissue of diabetic untreated rats showed marked reduction in number and size of islets of langerhan’s, which appeared fewer and shrunken, much smaller in size. Islets showed ill-defined borders, some being stellate in shape. Beta cells within the islets of langerhan’s; as demonstrated by anti-insulin monoclonal antibody; showed reduction in number, as well as signs of degeneration and necrosis. Vacuolar degeneration was observed within islet cells especially at the center. Hyaline bodies were seen. Inflammatory cells were observed at the periphery of scattered islets (Figure 2B) Treatment with Glimepiride caused improvement in islet size, number, cellularity and architecture with no signs of degeneration (Figure 2C). Treatment with M. alba; high dose showed marked improvement; with restoration of islet size, cellularity and architecture; approximating control (Figure 2D). Treatment with M. alba; low dose showed improvement as well; but less than that noticed in high dose; with residual dense inflammatory aggregates infiltrating scattered islets of langerhan’s (Figure 2E).
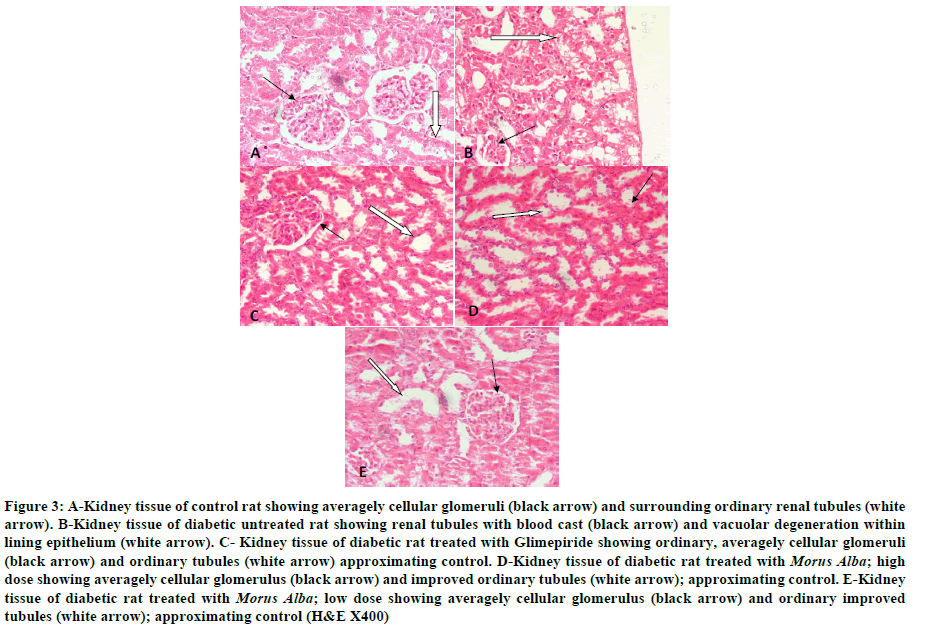
Kidney of control rat showed ordinary glomeruli with average overall cellularity. Renal tubules showed patent lumina, being lined by low cuboidal epithelium (Figure 3A). Kidney of diabetic rat showed markedly hypocellular, shrunken glomeruli. Renal tubules showed prominent vacuolar degeneration within lumina (Figure 3B). Blood casts were noticed within several renal tubules. Kidney of diabetic rat treated with Glimepiride showed marked improvement with averagely cellular glomeruli and ordinary tubules approximating control. No blood casts were seen within lumina of tubules. Also, the lining epithelium consisted of low cuboidal cells (Figure 3C). Kidney of diabetic rat treated with M. Alba; high dose showed marked improvement within both glomeruli and renal tubules (Figure 3D). Kidney of diabetic rat treated with M. Alba; low dose also showed markedly improved glomeruli and tubules (Figure 3E).
Discussion
Diabetes causes morbidity and mortality in all countries of the world (according to the literature). STZ is considered as the most used diabetogenic compound for provoking diabetes in experimental animal models [17]. One of diabetes complications is a diabetic nephropathy that may results from uncontrolled high blood sugar which leads to the development of kidney damage. Family history or genes plays a role in prevalence of diabetes. Plants to be considered hypoglycemic may act through different mechanisms. This study was designed biochemically and histologically to detect whether Morus alba plant had an effect on diabetes and its complications involving blood glucose, α- amylase levels, lipids, oxidative stress and renal function.
Our results described hypoglcaemic effect of aqueous M. alba extract through reducing serum glucose level in comparison with STZ-induced hyperglycaemic group. These results are in agreement with previous study [8]. The hypoglycemic activity of aqueous M. alba extract may be via the high fiber content, the presence of trigonelline bases, the presence of moran A and moranoline [18] that significantly increased the number of β-cells seen in specimens and inducing restoration of the islets diameter. The elevation of serum glucose level in this study supported with our microscopic examination of diabetic untreated group pancreatic sections which revealed marked vacuolar degeneration within cells inside islet of langerhan's with eosinophilic bodies, and residues of degenerated cells, while Morus alba in a high dose showing improved islet size and cell density concluding that the extract of this plant regenerates β cells reducing blood glucose level, which were also reported [19].
The activity of α-amylase in serum was increased by STZ, this result is in accordance with previous study [20]. While M. alba interfered with the activity of α-amylase that exhibited an effective strategy to lower the levels of hyperglycemia through the control of starch breakdown (as shown in this study). Another Indian study explored that M. alba act effectively as pancreatic α- amylase inhibitors [21]. The presence of α-glucosidase and pancreatic amylase enzyme inhibitors act by directly blocking the active center of the enzymes at various locations, these inhibitors reduce the glucose peak that can occur after meal slowing the speed with which amylase can convert starch to simple sugar so decreasing it in the blood. In addition, Bahman and Golboo [22] studied, in vitro, the starch breakdown by α-amylase and revealed α-amylase inhibitory effects of M. alba in a concentration-dependent manner.
In STZ-diabetic rats, in the current study, there was an impaired carbohydrate utilization inducing accelerated lipolysis, resulting in hyperlipidemia, evidenced by an elevation in serum TC level and a decrease in serum HDL level. Hyperglycemia provokes hyperlipidemia due to overproduction of TG-rich lipoprotein in the liver associated with a decrease of HDL cholesterol levels and a decrease of adipose tissue and muscle lipoprotein lipase (LPL) actively and the defective LPL decreases the catabolism of TG-rich lipoprotein reducing its clearance, in addition this enzyme also is affected by insulin deficiency [23]. Murti et al. [24] mentioned elevation in blood glucose concentration accompanied by increase in TC, triglycerides, LDL, VLDL and decrease in HDL-C in STZ-induced diabetic rats as compared to control animals. While we found fall in serum TC level and rise in serum HDL-C level after the treatment with both doses of M. alba extract as compared to STZ-treated rats, in addition high dose of M. alba is more potent than Glimepiride in restoring normal lipid levels. El-Beshbishy et al. [25] reported that M. alba root 70% alcohol extract may act as a potent hypocholesterolemic nutrient via the inhibition of LDL atherogenic modifications in hypercholesterolemic rats. Earlier studies showed the improvement in glycemic control after Morus alba treatment (600 mg/kg/day), followed by the reduction in VLDL production, that could be attributed to its dual mode of action: reduction in VLDL production and enhancement of VLDL removal [26], moreover, M. alba resulted in amelioration of the alterations of HDL, total cholesterol in another study [27]. These beneficial effects of M. alba may be due to the presence of phytochemical constituents, as fiber, fatty acids, phenolics, flavonoids, anthocyanins, vitamins and trace elements.
In our work, STZ produced reduction in serum TAC level as compared to normal control group. This result is in accordance with Andallu and Varadacharyulu [28] who observed decrease glutathione peroxidase activity in diabetic rats. In addition, Adewole et al. [29] reported that lipid peroxidation is usually associated with hyperglycemia. While Morus alba leaves extract, in this study, elevated the level of TAC level. Dkhil et al. [30] suggested that Morus alba leaves extract has a powerful antioxidant effect on kidney, testes, spleen and intestine of mice and Nazari et al. [31] indicated that the anti-hyperglycemic effect of M. alba leaves extract is mediated by increasing glutathione peroxidase (GPX) and glutathione reductase (GR) activities.
There is a damage in the capillaries of the filter of the kidneys (glomerules) which lead to a leak abnormal amount of protein into the urine and a reduction in the kidney's ability to function as expected from our results that showed elevation in serum levels of creatinine and urea in STZ-treated rats compared to normal rats, while both doses of Morus alba leaves extract have evolved the ability to restore the kidney function. These results supported by our microscopic examination that depictured that diabetic untreated group showed vacuolar degeneration within lining epithelium of renal tubule while Morus alba; in two doses showing ordinary improved tubules, in addition high dose of Morus alba is more potent than Glimepiride in restoring kidney function levels. Al- Janabi et al. [32] found that diabetic rats treated with alcoholic extract of M. alba leaves at 600 mg/kg decreased creatinine serum level as compared to STZ-treated rats.
So, the antidiabetic effect of Morus alba in digestive system provoking the reduction of glucose level that can occur via inhibition of alpha amylase and scavenging of ROS, and regenerating β-cell. It also exerts beneficial effects against diabetic, hyperlipidemic and nephropathy-induced by STZ.
Conclusion
From the above results, it may be concluded that Morus alba is a hypoglycemic herb, in addition to being a potent inhibitor of α- amylase, as well as having the ability to improve the redox balance and prevent the morphological destruction of the pancreas and kidney that is associated with diabetes. Therefore, long-term control of diabetes using Morus alba for treatment of the progression of diabetic complications in experimental animal models may be considered in further experiments prior to clinical trials.
Funding
The study was done under the project no: 10010306 and funded by National Research Centre, Dokki, Giza, Egypt.
References
[1] S.O. Ogbonnia, J.I. Odimegwu, V.N. Enwuru, Afr. J. Biotechnol., 2008, 7(15), 2535-2539.
[2] S. Wild, G. Roglic, A. Green, R. Sicree, H. King, Diabetes Care, 2004, 27, 1047-1053.
[3] A.K. Tiwari, J.M. Rao, Curr. Sci., 2002, 83, 30-38.
[4] J.E. Hall, 2006, Philadelphia, PA: Saunders, 791-807.
[5] G. Williams, J.C. Pickup, Cambridge Univ., 293 Press, Cambridge, UK, 1998.
[6] M. Najafian, M.Z. Jahromi, M.J. Nowroznejhad, Mol. Biol. Rep., 2011, 1(1), 1-8.
[7] G.G. Wang, X.H. Lu, W. Li, X. Zhao, C. Zhang, Evid. Based. Complement. Alternat. Med., 2011.
[8] S. Chaurasia, R.C. Saxena, I.D. Chaurasia, R. Shrivastava, Int. J. Chem. Sci., 2011, 9(2), 489-492.
[9] B. Devi, N. Sharma, D. Kumar, K. Jeet, Int. J. Pharm. Pharm. Sci., 2013, 5(2).
[10] M.M.A. Khalaf, G.A. El Sherbiny, H.A. AbdEllatif, A.A. Ain-shoka, M.E. El Sayed, British J. Pharmacol. Toxicol., 2012, 3(6):278-288.
[11] N.A. Mohamed, S.A. Zaitone, Y.M. Moustafa, IOSR J. Pharm., 2013, 3(9): 72-80.
[12] P. Trinder, Ann. Clin. Biochem., 1969, 6, 24-27.
[13] W. T Caraway, Am. J. Clin. Pathol., 1959, 32(1), 97-99.
[14] W. Richmond, Clin. Chem., 1973, 19(12), 1350-1356.
[15] M.F. Lopes-Virella, P. Stone, S. Ellis, J.A. Colwell, Clin. Chem., 1977, 23(5), 882-884.
[16] D. Koracevic, G. Koracevic, V. Djordjevic, S. Andrejevic, V. Cosic, J. Clin. Pathol., 2001, 54(5), 356-361.
[17] N.A. Qinna, A.A. Badwan, Drug. Design. Devel. Therapy., 2015, 9, 2515-2525.
[18] J. Mohammadi, P.R. Naik, Indian. J. Pharmacol., 2008, 40(1), 15-18.
[19] J. Mohammadi, P.R. Naik, Turk. J. Biol., 2012, 36, 211-216.
[20] M. Najafian, Zahedan J. Res. Med. Sci., 2015, 15.
[21] P. Sudha, S.S. Zinjarde, S.Y. Bhargava, A.R. Kumar, BMC Complement. Altern. Med., 2011, 20, 11-5.
[22] N. Bahman, M. Golboo, Iranian J. Pharma. Res., 2009, 8(2):115-119.
[23] I.J. Goldberg, J. Clin. Endocrinol. Metab., 2001, 86(3), 965-971.
[24] K. Murti, V. Lambole, M. Panchal, U. Kumar, Pharmacol., 2011, 1, 15-21.
[25] H. El-Beshbishy, A.B. Singab, J. Sinkkonen, K. Pihlaja, Life Sci., 2006, 78, 724-2733.
[26] M. Laakso, J. Intern. Med., 2001, 249, 225-235.
[27] H.I. El-Sayyad, M.A. El-Sherbiny, M.A. Sobh, A.M. Abou-El-Naga, M.A. Ibrahim, S.A. Mousa, Int. J. Biol. Sci., 2011, 7(6),715-728.
[28] B. Andallu, N. Varadacharyulu, Clin. Chim. Acta., 2003, 338, 3-10.
[29] S.O. Adewole, J.A. Ojewole, Afr. J. Tradit. Compl. Altern. Med., 2007, 4, 64-74.
[30] M.A. Dkhil, A.A. Bauomy, M.S.M. Diab, S. Al Quraishy, Pakistan J. Zool., 2015, 47(2), 393-397.
[31] M. Nazari, M.R. Hajizadeh, A. Eftekhar, S. Fattahpour, H. Ziaaddini, G. Hassanshahi, M. Mahmoodi, M. Rezaeian, Med. Chem.,2014, S1.
[32] O.S. Al-Janabi, M.S. Amer, M.H. Khayri, Int. J. Sci. Res., 2013, 4(3).



