Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 4
Screening Anti-Acne Potency of Microalgae: Antibacterial and Antioxidant Activities
Erismar Amri1,3, Abdi Dharma1, Armaini1 and Djong Hon Tjong22Biology Department, Universitas Andalas, Limau Manis, Indonesia
3Biology Education Department, STKIP PGRI Sumatera Barat, Padang, Indonesia
Abstract
Sixteen microalgae isolates were screened for their potency as the sources of anti-acne agents. The methanol and hexane extract of the isolates were assayed for their antibacterial bioactivity against Propionibacterium acnes and Staphylococcus aureus and theirantioxidant activities as free-radical-scavenging bioactivity by the 2,2-diphenyl-1-picryl-hydrazyl (DPPH) test. The results showed that the extract of Micractinium sp. Ehime was the most potential as the source of anti-acnes agent based on their combined activities: antibacterial against P. acnes (clear zone 28.6 mm) and antioxidant activity (IC50 361.23 μg/ml).
Keywords
Anti-acne, Antibacterial activity, Microalgae, Antioxidant
Introduction
Skin is the largest body organ that serves as an important environmental interface providing a protective envelope that is crucial for homeostasis. On the other hand, the skin is major target for toxic insult by a broad spectrum of physical (i.e., UV radiation) and chemical (xenobiotic) agents that are capable of altering its structure and function [1]. Acne vulgaris is extremely common skin disorder that effect virtually all individuals at least once during life. Acne is a disorder of the sebaceous follicles, which special pilosebaceous units located on face, chest, and back. They consist of sebaceous glands associated with small hair follicles. Several factors contribute to the pathogenesis of acne-sebum includingabnormal follicular differentiation, Propionibacterium acnes infection and inflammation. Each of these factors provides a potential target for treatment [2,3].
P. acnes, ananaerobic Gram-positive normally inhabits the skin and implicates in the inflammatory phase of acne [3,4]. This bacterium plays a central role in the current concept of acne pathogenesis [3,5] and appears to be the target of oral and topical antibiotic usage. The reduction of their numbers is a valid parameter for the therapeutic effectiveness of an antibiotic [3,6].
Recently it was reported that the most chronic medical conditions of acne are characterized by both oxidative stress and inflammation. It is likely that the blood levels of antioxidants are used up readily in those with acne because there is a greater demand to deal with free radicals. Compounds targeting acne, therefore, should be able to inhibit P. acnes growth and inhibit the oxidative stress. In other words, compounds or materials advocated for acne control should possess antibacterial and antioxidant activities [3].
Medicinal plants have been used in medicine for thousands of years. However, their efficacies proven with scientific methods, which can be employed to give a better understanding of their mechanisms of action, were only established in recent times. The antioxidant effects of antioxidant-rich polyphenols such as green tea, turmeric, and berries were reported to have good activity in reducing the oxidative stress of acne patients [3]. In this study, the antimicrobial activity against P. acnes, Staphylococcus aureus and antioxidant properties of some microalgae were investigated.
Materials and Methods
Microalgae materials
Sixteen microalgae isolates used in this study were collected from West Sumatera, Indonesia. Microalgae isolate Kili 1 and Kili 2 were collected from spring water Bukit Kili, Solok, Micractinium sp. CCAP, Micractinium sp. Ehime, and Mychonastes rotundus were collected from the palm oil mill effluent (POME) ofMutiara Agam Company, West Sumatera, Indonesia [7]. Other microalgae isolates were the collection of Biochemistry Laboratory, Faculty of Mathematic and Natural Science, Universities Andalas, Padang, Indonesia. The voucher specimens were collected at the Biomedicine Laboratory, Faculty of Medicine, Universitas Andalas, Padang.
Preparation of microalgae extracts
All samples were dried before being extracted with methanol and hexane. Briefly, the dried microalgae materials were extractedthree times with solvents (ratio of 1 g sample: 10 mL solvent), for 24 h. The extract were then dried in ovenat 40°C.
Antibacterial assay
The test organism used in this study were Propionibacterium acnes from Microbiology Laboratory, Faculty of Medicine, Indonesia University and Staphylococcus aureus from Bacteriology Laboratory, Balai Veteriner Bukittinggi. The medium consisted of blood agar with yeast extract for P. acnes and blood agar for S. aureus. Microalgae extract were diluted in dimethyl sulfoxide (DMSO) at concentration 10-15 mg/ml. P. Acnesn and S. Aureus were inoculated by equally distributed on the blood agar media, and then the paper discs were placed on the top of media. The DMSO sample solution (0.03 ml) were then dripped on paper disc. Incubation was performed in anaerobic condition for 72 h for P. acnes and in aerobic condition for 24 h for S. aureus. The zones of inhibition formed around the paper discs demonstrate the sample extract ability to inhibitthe growth of bacteria. The standard drug used was clindamycin.
Antioxidant assay
The antioxidant assay used in this study adopted a free-radical-scavenging activity using the 2,2-diphenyl-1-picryl-hydrazyl (DPPH) test [8]. Samples were diluted in methanol to make concentration of 62.5, 125, 250, 500 and 1000 ppm. An aliquot of sample (100 μl), 100 μl of 118 mg/L DPPH solution in methanol were added to each well of a 96-well plate. After 30 min incubation in dark room, the absorbance of the mixture was measured at 514 nm. The positive control was ascorbic acid, while methanol was used as the blank. The inhibitory activity was calculated according to the following equation:
Inhibition (%)=[1–(Asample–Acontrol)/(Ablank–Acontrol) × 100%
Where Asample is the absorbance of the sample, Acontrol is absorbance of ascorbic acid as control and Ablank is the absorbance of ethanol as the blank. Each sample concentration of the samples and positive control were tested in triplicate.
Preliminary phytochemical analysis
Preliminary analysis of phytochemical constituents of microalgae sample were carried out according to standard method described by Harborne [9]. The chemical tests to analyze the presence of steroid/terpen was by Salkowski; Libermann-Burchardand Libermann's test, phenol compounds were by ferric chloride, saponin were by frothing and haemolysis tests and flavonoids were by Shinoda test. The qualitative results are expressed as (+) for the presence and (-) for the absence of phytochemicals.
Result and Discussion
Sixteen microalgae were collected for anti-acne potency screening. The anti-acne potency of microalgae was analyzed based on antibacterial, and antioxidant activity. The data showthat all of methanol extracts were active to inhibit the growth of Propionibacterium acne and Staphylococcus aureus. On the other hand, most of the hexane extract were only active as growth inhibitor of most of P. acne but non to S. Aureus (Table 1). The most effective inhibitor for P. acne were the methanol extracts of Micractinium sp. Ehime, and Uncultured oscillatoria sp. IPOME4, while to the S. Aureus, all of the active extract shown only a minor inhibitor activity.
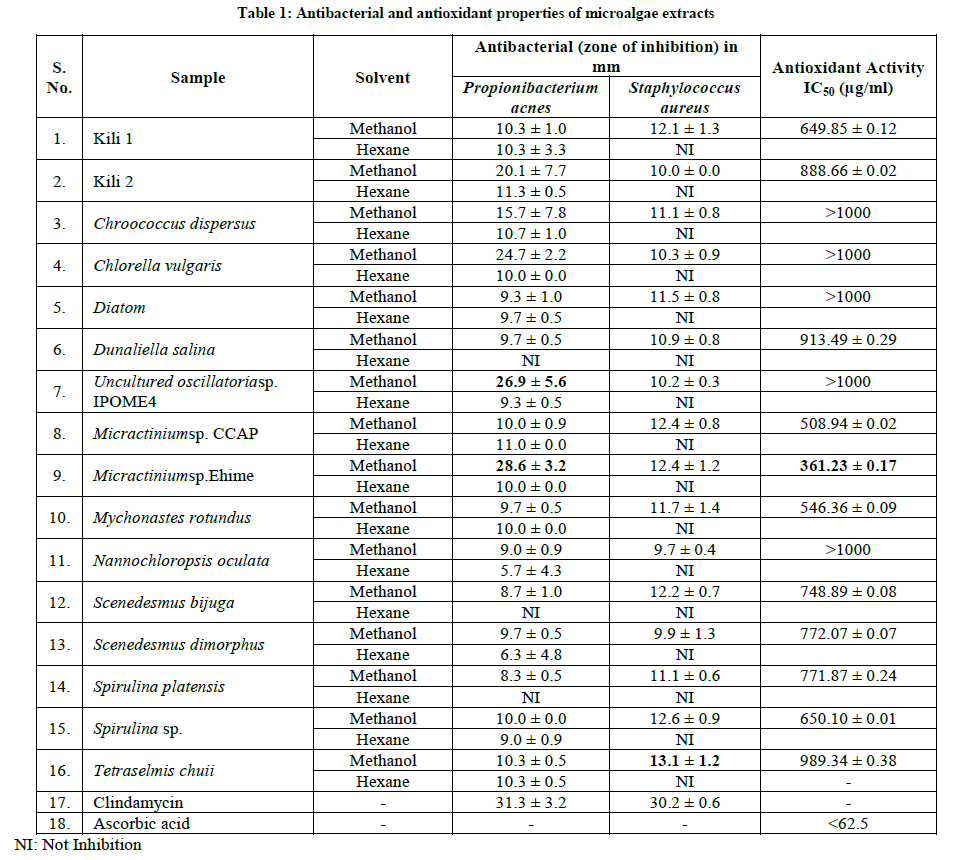
The diameter zone of inhibition values of the Micractinium sp. Ehime methanol extracts were 28.6 mm, which was closer to diameter zone of inhibition of the positive control clindamycin which was 31.3 mm. Several studies have shown that the compounds have high antibacterial ability is largely a polar compounds than non-polar compounds [10].
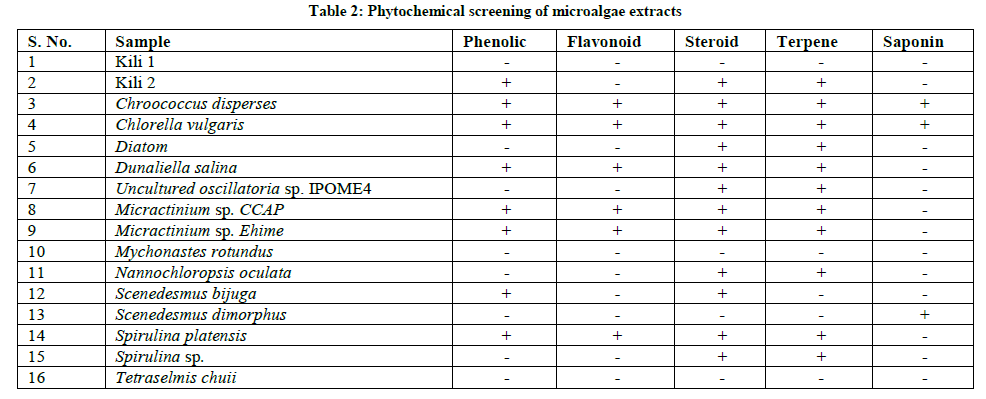
In this study, phytochemical screening shown that the methanol extracts of several microalgae contain flavonoids, steroids, terpenoids and phenolic compound (Table 2). The content of secondary metabolites is comparable with antibacterial capabilities. Generally, from microalgae methanol extract containing flavonoids, terpenoids, phenolic and steroids show good antibacterial abilities.
For antioxidant activity, 11 samples had IC50 values lower than 1000 μg/ml and Micractinium sp. Ehime had IC50 values 361.23 μg/ml, the lowest among all samples, but much higher than positive control, ascorbic acid (<62.5 μg/ml). On the other hand, 5 samples at a concentration of 1000 μg/ml could not inhibit the oxidation reaction of DPPH by 50% (Table 1). Although there is still the possibility of microalgae which has antioxidant potential is developed by isolating and purifying the active compound that has antioxidant activity.
From the data illustrated in Table 2. It was observed that phenolic, flavonoid, steroid, terpene and saponin were present in the extracts some microalgae samples studied.Almost all of microalgae which has high antibacterial and antioxidant activity contains phenolic and flavonoids compounds, and most of microalgae that contains no flavonoids and phenolic compounds haslow antioxidant and antibacterial activity.
Based on the two activities, Micractinium sp. Ehimeextracts (methanol) have the highest potential as an anti-acne agent. Among all samples, these extracts had the best antimicrobial activity and good antioxidant activity. It is important that the active anti-acne compounds from these species be isolated, purified and identified.
Conclusion
The data shown that from 16 microalgae isolate that were collected from Sumatera Barat, Indonesia, the sample with the best antibacterial activity were Micractinium sp. Ehime MeOH extracts to inhibit P. acnes. All of methanol extract shown their antibacterial activity against S. aureus, but the activity were very low compare to the control (clindamycin). The sample that had the best antioxidant activities was Micractiniumsp. Ehime. Based on the two activities, Micractinium sp. Ehime extracts (methanol) have the best potential as an anti-acne agent.
Acknowledgements
The authors would like to thank the Microbiology Laboratory, Faculty of Medicine, Indonesia University, for providing the P. acnes, the Bacteriology Laboratory, Balai Veteriner Bukittinggi for providing the S. aureus and laboratory facility for antibacterial tests; Direktorat Penelitian dan Pengabdian Masyarakat-Ditjen Dikti, Republic of Indonesia for financial support through Hibah tim pasca sarjana 2016.
References
[1] D.R. Bickers, M. Athar, J. Invest. Dermatol., 2006, 126, 2565-2575.
[2] S. Brown, A. Shalita, Lancet., 1998, 351, 1871-1876.
[3] I. Batubara, T. Mitsunaga, H. Ohashi, J. Wood. Sci., 2009, 55, 230-235.
[4] J. Strauss, D. Krowchuk, J. Leyden, A. Lucky, A. Shalita, E. Siegfried, D. Thiboutot, A. Van Voorrhees, K. Beutner, C. Sieck, and R. Bhushan, J. Am. Acad. Dermatol., 2007, 56, 651-663.
[5] L. Zane, Semin. Cutan. Surg., 2005, 24, 65-66.
[6] C. Burkhart, C. Burkhart, P. Lehmann, Postgr. Med. J., 1999, 75, 328-331.
[7] W. Sekatresna, A. Dharma, R. Zein, Z. Chaidir, J. Chem. Pharm. Res., 2015, 7, 222-231.
[8] Z. Chaidir, D. Nurakbari, M. Salim, R. Zainul, Der Pharmacia Lettre., 2016, 8, 73-78.
[9] J.B. Harborne, London: Chapman & Hall, 1998.
[10] M. Al-Ghorbani, V. Lakshmi Ranganatha, T. Prashanth, A. Bushra Begum, S.A. Khanum, Der. Pharma. Chem., 2013, 5, 269-273.



