Research Article - Der Pharma Chemica ( 2022) Volume 14, Issue 5
Standardization and Hypoglycemic Activity of Hydro-Alcoholic Leaves Extract of Morus Alba
Sunita Singh1, Aradhya Dev Srivastav2, Deepak Singh2, Bharat Mishra1, Abhishek Kumar Chandra3, Sudeep Yadav4 and Dhananjay Singh2*2Department of Chemical Engineering, IET Lucknow-226021, India
3Department of Chemical Engineering, UIET CSJM University, Kanpur-208024, India
4Department of Chemical Engineering, BIET Jhansi-284128, India
Dhananjay Singh, Department of Chemical Engineering, IET Lucknow-226021, India, Email: dsa768008@gmail.com
Received: 02-Apr-2022, Manuscript No. dpc-22-55903; Editor assigned: 05-Apr-2022, Pre QC No. dpc-22-55903; Reviewed: 19-Apr-2022, QC No. dpc-22-55903; Revised: 22-Apr-2022, Manuscript No. dpc-22-55903; Published: 02-May-2022, DOI: 10.4172/0975-413X.14.5.1-15
Abstract
The aim of this work is to carry out identification of some phytoconstituents of Morus alba which are responsible for antidiabetic activity and their complications as well as antioxidant activity. Phenolic compounds and flavonoidal compounds were estimated in the various extracts (chloroform, petroleum ether, ethanol and water). Phenolic compounds are determined by using a linear standard curve of gallic acid (y = 0.0025x+ 0.0334, R2 =0.9997) and found to be higher in water extract (67.32±0.50 mg/g GAE) in comparison to other extracts. Flavonoidal compounds estimated through a linear standard curve of quercetin (y=0.004x+ 0.0431, R2 =0.9985) and found to be higher in water extract (55.37±0.05 mg/g QE) than other extracts. HPLC was used for the determination of quantities of phytoconstituents present in M. alba. Chromatogram of HPLC shows the presence of quercetin (1.30 mg/g), chlorogenic acid (1.80 mg/g), rutin (1.20 mg/g), isoquercetin (0.35 mg/g) and kaempferol (0.80 mg/g). In vitro antioxidant activity was increase with increasing their concentration and it was found maximum in water extract than other extracts. The IC50 values have been estimated and it was found highest in petroleum ether extract than other extracts. Normal healthy rats treated with hydro-alcoholic leaves extracts; they did not show any harmful effect during 14 days up to dose level 600mg/kg. Metformin improved glucose tolerance up to 14.6% at a dose 100 mg/kg while plant drug 11.38 % at a dose 600 mg/kg. STZ treated rats caused induction of hyperglycemia. The reduction of glucose level 21.9% by metformin at a dose of 100 mg/kg while Morus alba shows 13.2 % at a dose of 600 mg/kg. Treatment with hydro-alcoholic leaves extracts of various dosage shows the reduction in all biochemical parameters, but HDL was increased. The present investigation on the hydro-alcoholic extract of Morus alba leaves shows improvement in glucose tolerance and significant hypoglycemic activity in STZ induced diabetic SD male rats by regeneration of β cells in the pancreas that secrete insulin. This result gives opportunity to develop novel oral hypoglycaemic drugs in advance pharmaceutical therapies.
Keywords
Morus alba; Phytochemical analysis Antidiabetic activity; Antioxidant activity; Streptozotocin; Metformin
Introduction
Diabetes is a metabolic disorder of endocrine gland. The β-cells of pancreatic gland produce insulin for breakdown of carbohydrate, fat and protein for producing the energy. Lacking of insulin in the blood the cells cannot produce sufficient energy from the nutrients and increase blood sugar level. Due to increase blood glucose level, immune system of body attacking on pancreas for producing of insulin from β-cells [1]. The pancreas becomes damage and β- cells stop insulin production. Damage of pancreas is known as pancreatitis. It occurs due to heavy alcohol consumption, heredity, high lipid levels, cystic fibrosis, viral infections, hormonal abnormalities, certain medications, gallstones and some autoimmune conditions [2]. Type-I diabetes is known as insulin dependent diabetes mellitus (IDDM) and Type 2 diabetes is known as noninsulin dependent diabetes mellitus (NIDDM). The ability for production of insulin can recovered by type 1 diabetic patient [3].
Streptozotocin (STZ) is a glucosamine–nitrosourea compound which is obtained from Streptomyces achromogenes. It is used for inducing diabetes by damaging islet β- cells of pancrease, results is decrease in insulin level and increase glucose level in the blood. The mode of action of STZ can induce diabetes by selectivity for β- cells is connected with special addition of the chemical in β- cells after entrance through the GLUT2 glucose transporter receptor, similarity of chemical structural with glucose is allow STZ to bind to this receptor. At high dose, STZ targets β- cells by its alkylating property related to that of cytotoxic nitrosourea compounds [4,5]. Several synthetic oral hypoglycaemic drugs such as sulfonylurea, biguanides, meglitinides, α- glucosidase inhibitors, thiazolidinediones and metformin are available in market to reduce sugar level in diabetic patients but these synthetic drugs causes adverse effects.
Herbal medicines are widely used for treatment of various diseases because they exhibit more therapeutic effects and minor or no side effects. While the synthetic drugs are more expensive and produced more side effects in comparison to herbal drugs. Generally, herbal medicines are very effective but their standardization is important for confirmation to its quality, purity, safety and biological properties [6,7]. Medicines are used for saving the lives from various diseases and manufactured by using chemical components on industrial scale or it can be extracted from natural sources. Plants are the major source of phytoconstituents, It can be derived from the different parts of plant such as rhizomes, leaves, roots, stems, barks, flowers, fruits and peels [8].
Various drugs are reported in Ayurveda for treatment of hyperglycemia but we have taken Morus alba (MA) for present study. It belongs to family moraceae and also known as white mulberry. It is medium sized tree, their height about 17 m and width of trunk up to 1.5 meter. It appears rounded bushy due to more diffused branching. Stem barks are greyish brow with rough vertical fissures [9]. It is cultivated in the rainy season seeds, cuttings and grafts. For cultivation loamy and sandy soil are preferred. Seeds are very small and dispersed by animals, birds and human beings [10]. It is a composite fruit and consisting of many drupes. The size of fruits about 2 cm long, ovoid or ellipsoid, white to pinkish in color when ripe. The leaves of MA contains various active phytoconstituents such as ascorbic acid, flavones, vitamin D, steroids, carotene, amino acids, folic acid, triterpenes, minerals, and folic acid. It is used in treatment of various diseases such as diabetes, cancer, asthma, obesity, microbial infection, inflammation, hypertension & rheumatism. It is widely cultivated for feeding the silkworm (bombyx mori) for sericulture [11,12].
Material and Methods
Chemicals
The standard compounds chlorogenic acid, quercetin, isoquercitin, rutin, kaempferol, gallic acid, metformin and 2,2 Diphenyl -1-Picrylhydrazyl (DPPH) were purchased from Sigma- Aldrich (St.Louis, MO, USA). Folin-Ciocalteau reagent, aluminium chloride, chloroform, petroleum ether and ethanol,gum acacia and other chemicals were procured from Merck chemical suppliers (Darmstadt, Germany).
Plant material
Morus alba (white mulberry) have been found in rich amount in the vicinity of Institute of Engineering & Technology, Lucknow, Utter Pradesh. It has been collected in March, 2017 from the campus of this Engineering College. The authentification of MA has been done with the help of CSIR-NBRI, Lucknow, Utter Pradesh.
Experimental animals
Male rats were taken for the antidiabetic study which was 7–8 weeks old and their weights about 160±20 grams. Experimental rats were housed in cages of polypropylene and maintained their temperature23±2ºC, intensity of light 300 lux, 12 hrs dark and light cycle and 55% humidity. Provide drinking water and standard pellet diet ad libitum to experimental animals [13].
Standardization of plant material
Analysis of physicochemical Parameters
Physicochemical parameters were analyzed with the help of air-dried crude drugs. It is important for confirmation of quality, purity and safety of crude drugs (IP, 1996). Some important physico-chemical parameters like moisture content, extractive value, ash value, foreign organic matters, foaming index and pH of extract have been determined [14].
Evaluation of Ash value
1.0 gm powdered drug was ignited at temperature 200ºC for the evaluation of ash value. The quality of a crude drug was analyzed through the inorganic substances (carbonates, silicates, oxalate and phosphates) left after ignition. The ash value is important characteristics to detect the adulteration and secure the quality and purity of the crude drug [14].
Evaluation of total ash
2.0 g crude powered drug was heated in a silica crucible at 200ºC temperature to obtained carbon free material. For evaluation of total ash, the ignited powdered drug was cooled and weight [15].
Evaluation of acid insoluble ash
Acid insoluble ash was residues obtained by boiling the total ash with 25ml dilute HCl (6N) for 5 min. It was filtered, washed with hot water, ignited at 200ºC in porcelain dish to its constant weight and then cooled.
Evaluation of water soluble ash
Total ash was boiled with 25ml of distilled water then filtered it and collects the insoluble ash. It was ignited at 200ºC to its constant weight and then cooled in desiccators [15].
Evaluation of extractive value
Determination of extractive value helps in evaluation of crude drugs and provides an idea regarding the nature of phytochemicals.
Evaluation of alcohol soluble extractive value
About 5 gm dried crude drug was macerated into a closed flask with 100 ml of 95% of ethanol for one day. It was shaken for 6 hrs and filtered. The filtrate was evaporated by using rotary evaporator at 105ºC to s weight. The amount of ethanol soluble extractive value was calculated with reference.
Evaluation of water soluble extractive value
The coarsely dried powdered drug was extracted with distilled water for 24 hours. It was stirred at different time intervals and filtered. The remaining material was dried (105ºC) and weighed.
Evaluation of foreign matter
The herbal drug should be free from adulterants and contaminants. It may be different parts of others plants, stones, dust, soil, insects, moulds and animal excreta. 100gm of coarsely powered crude drug sample uniformly spread on a plate (glass). It was separated by examine the magnifying glass or nude eyes and weighed [14,15].
Evaluation of moisture content
Determination of moisture as well as volatile content present in particular sample was done by loss on drying method. 5 gm pulverized drug was taken in silica crucible then it was dried in hot air oven for 5 hrs at 105ºC temperature and weighed. The process was continues until two synchronized evaluation have been achieved.
Evaluation of foaming index
1 gm powdered drug boiled with 100 ml distilled water at 90ºC for 30min. It was cooled and filtered. 10 test tubes have been marked with 1 to10 and decoction poured in test tubes from 1ml, 2ml, 3ml, and 4ml, up to 10 ml were marked 1to10. By adding distilled water the volume of test tubes was adjusted to 10ml. The test tubes were shaken length wise for 15 seconds and for 15 minutes. The height of the foam was measured in all test tubes.
Determination of pH
pH (10%) solution: 10 gm crude drug dissolved in 10 ml distilled water and then it was filtered. pH of filtrate was checked by using standardized glass electrode. pH (1%) solution: About 1 gm coarsely powdered drug was dissolved in 10ml distilled water and filtered it. Then check the pH of filtrate by standardized glass electrode.
Elemental analysis
AAS was used for the estimation of heavy metals and minerals by wet digestion method. About 2 gm of powdered drug were treated with nitric acid, percloric acid and sulphuric acid. This solution was used for the analysis of heavy metals and minerals. Plant materials should be free from heavy metals as well as hazardous materials because heavy metals cause cancers [16].
Fluorescence analysis
The powdered drug material was treated with certain organic solvents. The herbal drug molecules absorb some specific wavelengths range of UV light and they emit radiations. This radiations was examined under UV light (254 nm and 366 nm) as well as in day light. This process can be useful in certain cases for identification of the crude drugs.
Phytochemical analysis
Secondary metabolites of the MA extract was isolated and quantified by using HPLC technique. Individual components of MA extract can be purified by this technique [17,18]. MA leaves was dried in shed at room temperature and crushed it to powdered form by using mechanical pulveriser.The sample was subjected to organic solvents (water: methanol 20:80, v/v) in ultrasonic exposure at 40ºC. The un-dissolved material was separated by using centrifugation and leaves extract was concentrated through vacuum evaporator. Chromatograms were developed by HPLC and it is consisting of binary pumps, rheodyne manual injector, PDA detector and column oven. Methanol and acidified water were used to obtain acceptable isolation of phytoconstituents through Phenomenax Luna (250x4.6mm, 5μm) C18 column [19, 20].
The column temperature was set 25ºC and injection volume was 10 μl. The detection range of Photo-diode array was 200-800nm and data attainment was done at 254nm to build up fingerprints [21]. The chromatograms of sample were analyzed by comparing both retention times (standard compound with MA compound). All constituents were confirmed by retention time and quantified by external standard method [22].
Total phenolic compound
Folin’s reagent, phenol and distilled water were used for the preparation of Folin-Ciocalteau reagent. It was used for the analysis of total phenolic compound in different extracts (chloroform, petroleum ether, ethanol and water) of MA. Total Phenolic compound obtained in the leaves extract of MA, has been represented as mg/g GAE (gallic acid equivalent). Gallic acid was used as standard compound .The absorbance of test compound and standard compound was calculated at 760nm through UV spectrophotometer [23-25].
Total flavonoid compound
Aluminium chloride colorimetric method was used for the determination of total flavonoid compound present in different organic leaves extracts (petroleum ether, chloroform, ethanol and water) of MA. Total flavonoidal compound present in leaves extracts of MA was represented in mg of quercetin equivalents per gm of extracts (mg QE/g extract). Quercetin was used as standard compound [26]. The absorbance of test component and standard component was calculated at 760nm through UV spectrophotometer [27].
Pharmacological Study
In vitro antioxidant activity
MA leaves extracts (chloroform, petroleum ether and ethanolic) can be analyzed for determination of their scavenging capacity by 2,2 Diphenyl -1- Picrylhydrazyl (DPPH) assay. DPPH is a stable free radical. The colour of DPPH is purple when it is reduced to form diphenyl-picrylhydrazine which is yellow in colour due to the action of an antioxidant component or a free radical scavenging species [28,29]. Change in the colour of DPPH is calculated by the spectrophotometer at 517 nanometer. Quercetin was used as standard compound and various concentrations (20, 40, 60 and 80 μg/ml) of quercetin were prepared by methanol solution. Concentration (100 μg/ml) of MA leaves extract was also prepared by methanol solution [30,31].
The amount of DPPH about 0.2 milligram was dissolved in 15.3 ml methanol solution for preparation of stock solution. The combination of 0.1 ml MA leaves extract, DPPH solution 1.8 ml and methanol solution 0.1 ml, allowed to stand for 30 minutes at room temperature in dark and absorbance taken at 517 nanometer against a blank (DPPH in methanol) [32,33]. The DPPH ability was expressed as IC50 (inhibitory concentration). It is the capability of the plant extract essential to decrease 50% of the DPPH in the mixture [34-35]. Results are shown as average % scavenging activity (% inhibition)
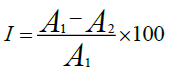
I is the percentage inhibition, A1 is the absorbance of blank (DPPH in methanol solution), A2 is the absorbance of the plant extract.
Preparation of hydro-alcoholic extract for in vivo studies
MA leaves were cleaned with running tap water and dried in the shade at room temperature. Plant material was powdered by using mechanical pulverizer and consequently sieved. Then measured amount of powdered leaves were packed in soxhlet apparatus. Extraction was done with distilled water (60%) and ethanol (40%) at 400C temperature [36,37]. The extraction process was continuous approx 2 days when the solvent of the siphon tube becomes colorless. Concentrated the leaves extract by using rotary evaporator and then it preserved in suitable vials in the freeze [38].
Acute oral toxicity study in SD male rats
For the evaluation of acute oral toxicity of hydro-alcoholic Morus alba leaves extract (MALE) was carried out in SD male rats to explore its safety profile according to the OECD test guideline no. 432 (1987). Rats were divided in to 3 groups and every group has 5 rats [39]. MALE given orally to overnight starved normal healthy SD male rats in single dose (group 1, MALE at 500 mg/ kg body weight; group 2, MALE at 1000 mg/ kg body weight and group 3, MALE at 2000 mg/ kg body weight). One more group of 5 rats was termed as normal control group and administered only distilled water [40]. The animals were monitored for abnormal symptoms on the day of administration until the end of experiment (14 days).
Estimation of OGTT in normal SD male rat
After fasting of 6 hrs, total rats were divided into five groups and every group has 5 rats. 1% gum acacia used as a vehicle for preparing 100mg/kg b. wt dose of standard metformin. Suspension of MALE (150, 300 and 600 mg/ kg body) and metformin (100mg/kg b. wt) were administered to SD male rats. The same quantity (100mg/kg b. wt) of gum acacia was given orally to rats of normal control (NC) group [41].
After half an hour of the test drug or vehicle glucose load (2 g/kg) was orally administration to each group of SD rats. The level of blood glucose of every rat was measured at 0 min, 15 min, 30 min, 60 min, 90 min, and 120 min. After administration of glucose, glucometer was used to measure the level of blood glucose. Throughout the examination, only distilled water has been provided to SD male rats [42].
The decreasing level of blood glucose between 0-2 hrs was determined by graph among glucose level and time. Level of blood glucose was plotted on y- axis and time was plotted on x- axis. Prism Software was used for examination of area under curve (AUC) of all groups. The percentage enhancement on OGTT after loading the glucose can be estimated by comparing the percentage lowering in AUC of test group (MA treated group) and vehicle treated normal control (NC) group [43].
% hypoglycemic activity = (AUC of test group/ AUC of normal control group) × 100
Group designed for OGTT study as follows:
Group I was termed as normal control group and orally administered with 1 % gum acacia
Group II was termed as positive control group and orally administered with metformin to rats
Group III was orally administered with 150 mg/ kg b. wt of MALE to SD male rats
Group IV was orally administered with 300 mg/ kg b. wt of MALE to SD male rats
Group V was orally administered with 600 mg/ kg b. wt of MA leaves extract to SD male rats
Induction of diabetes by STZ
Overnight starved SD male rats were made diabetic by intraperitoneal injecting a single dose (55 mg/kg b. wt) of freshly prepared STZ solution. STZ was prepared by dissolving it in the 0.1M citrate buffer (pH 4.5). After 2 days, the blood sample was taken from the pinched tail of each SD male rats to check the glucose level in blood by using glucometer to confirm diabetes. Rats taken for experiment when their blood glucose level is higher than 280 mg/dl.
Assessment of hypoglycemic effect in STZ-induced diabetic rat
The STZ induced diabetic rats has been divided into six groups with five rats in each group. The different doses (150, 300 and 600 gm/ kg b. wt) of MALE and metformin (100mg/kg b. wt) were made in 1.0% gum acacia. It was administered orally through gastric catheter to SD r ats of each group in the suspension form. For dissolving the samples, gum acacia has been used as vehicle. The same quantity (100mg/kg b. wt) of 1.0% gum acacia also given orally to rats in the normal control group. The level of glucose in the blood of all SD male rats was measured at 0 hr, 0.25 hr, 0.5 hr, 1hr, 1.5 hrs, 2 hrs, 3 hrs, 4 hrs and 5 hrs after orally administration of samples. Only distilled water was providing to test animals during the experimentation [44].
For the evaluation of lowering blood glucose level between 0 hr to 5 hrs, graph plotted between time and blood glucose level. During this time period the percentage decreasing in level of glucose can be analyzed by comparing the percentage lowering in area under curve of experimental group and normal control group.
Group designed for study of STZ (streptozotocin) induced diabetic action as follows:
Group I was termed as normal control group and orally administered with only simple distilled water.
Group II was termed as diabetic control group and orally given gum acacia (1%) later than 2nd day of intraperitoneal injection of streptozotocin
Group III was termed as positive control group and orally administered with metformin later than 2nd day of intraperitoneal injection of streptozotocin
Group IV was orally administered with150 mg/ kg b. wt of MALE than 2nd day of intraperitoneal injection of streptozotocin
Group V was orally administered with 300 mg/ kg b. wt of MALE later than 2nd day of intraperitoneal injection of streptozotocin
Group VI was orally administered with 600mg/ kg b. wt of MALE later than 2nd day of intraperitoneal injection of streptozotocin
Assessment of biochemical parameters
SD rats were allowed to carry on nourish according to their standard diets until the end of experimentation. 2-4 ml blood was withdrawn from during the night starved all rats, after end of the experiment. Glass capillary tube was used for withdrawing the sample of blood from venous plexus of retro-orbital under ether anesthesia of every group. For clotting of blood it allowed to stand at room temperature for 10 minutes and centrifuged at 2,500 rpm to isolate the serum. Isolated serum was used for assessment of different biochemical parameters by using standard method. Various biochemical parameters were estimated, which are as follows:
Assessment of serum lipid profile
Low-density lipoprotein, high-density lipoprotein, cholesterol, triglycerides and total cholesterol were assessed from separated blood serum.
Assessment of liver function test
SGOT and SGPT (serum glutamate oxaloacetic acid transferase and serum glutamate phosphor transferase) were analyzed from separated blood serum.
Assessment of kidney function
The creatinine and urea were also assessed from separated blood serum.
Statistical analysis
The statistical analysis was performed by Dunnetts test using “GraphPad Prism software (version 6, GraphPad, San Diego, CA, USA)”. The significance of mean difference between different groups was done by Tukey, s post hoc test. Results are expressed as mean ± SD. The results were considered statistically significant if P value is less than 0.05.
Mean (average) and SD (standard deviation) calculated which are as follows:
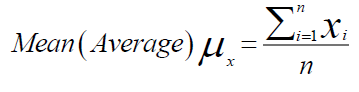
x= variable parameters
i=1…….n, denote the no. of samples
Results and Discussions
Physicochemical parameters and leaf constants
The results of Physico-chemical parameters such as total ash, water-soluble ash, acid insoluble ash, Alcohol soluble extractive value, water-soluble extractive value, foreign organic matter, moisture content, foaming index and pH of extract are summarized in Table1.
Elemental analysis
Atomic absorption spectrophotometer (AAS) used for assessment of heavy metals and minerals in the leaves of MA. The quantity of Sodium (Na), potassium (K), calcium (Ca), magnesium (Mg), phosphorus (p), sulphur (S), iron (Fe), copper (Cu), aluminium (Al), zink (Zn), arsenic (Ar), cadmium (Cd), lead (Pb) and nickel (Ni) are summarized in Table1.
| Physico-chemical parameters | Values ± SD | Elemental analysis | Values±SD (µg/g) |
|---|---|---|---|
| Total ash | 8.56 ±0.02 (%w/w) | Sodium (Na) | 0.55±0.27 |
| Water-soluble ash | 6.16±0.07 (%w/w) | Potassium (K) | 0.36±0.12 |
| Acid insoluble ash | 3.27±0.01 (%w/w) | Calcium (Ca) | 0.11±0.32 |
| Alcohol soluble extractive value | 11.23±0.03(%w/w) | Magnesium (Mg) | 0.43±0.42 |
| Water-soluble extractive value | 7.34 ±0.08 (%w/w) | Phosphorus (p) | 0.27±0.36 |
| Foreign organic matter | 0.61 ±0.02 (%w/w) | Sulphur (S) | 0.53±0.28 |
| Moisture content | 8.30±0.05 (%w/w) | Iron (Fe) | 1.74±0.35 |
| Foaming index | 100±0.06 (%w/w) | Copper (Cu) | 0.51±0.18 |
| pH of 1% aqueous solution | 6.23±0.04 | Aluminium (Al) | 0.82±0.35 |
| pH of 10% aqueous solution | 7.52±0.03 | Zink (Zn) | 0.69±0.22 |
| - | - | Arsenic (Ar) | 0.013±0.01 |
| - | - | Nickel (Ni) | 0.019±0.38 |
| - | - | Cadmium (Cd) | 0.021±0.06 |
| - | - | Lead (Pb) | 0.014±0.12 |
Fluorescence investigation
Fluorescence study was done by using different chemical reagents on powdered leaves of MA in visible light as well as in UV l ight (254 nm and 366 nm). The results are summarized in Table 2.
| Visible light | Powder and used Chemicals | UV (254nm) Lamp | UV(366nm) Lamp |
|---|---|---|---|
| Green (dark) | Dried powder | Green (light) | Brown (blackish) |
| Green (yellowish) | KOH (5%)+Powder | Brown | Green (dark) |
| Green (light) | NaOH (5%) + Powder | Brown (greenish) | Brown (dark) |
| Brown (blackish) | H2SO4 + Powder | Green (brownish) | Brown |
| Green (blackish) | Iodine solution +Powder | Brown (reddish) | Brown (blackish) |
| Red (light) | Conc.HNO3+ Powder | Green | Greenish brown |
| Green (light) | Conc. HCl+ Powder | Green(yellowish) | Brownish-yellow |
| Brown (dark) | dil. Ammonia +Powder | Brown (greenish) | Green (dark) |
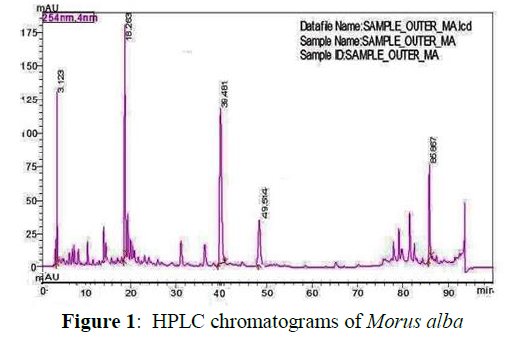
HPLC analysis of Morus alba
Retention time of quercetin 3.123, chlorogenic acid 18.263, rutin 39.481, isoquercitin 49.514 and kaempferol was observed at 85.867 min. The value of all constituents was estimated at 254 nanometer, 4 nanometer and chromatogram of MA shown in Figure 1. Analysis shows the presence of quercetin 1.30 mg/g, chlorogenic acid 1.80 mg/g , rutin 1.20 mg/g, isoquercetin 0.35 mg/g and kaempferol 0.80 mg/g were found in MA leaves extract.
Total phenolic compound of Morus alba leaves extracts
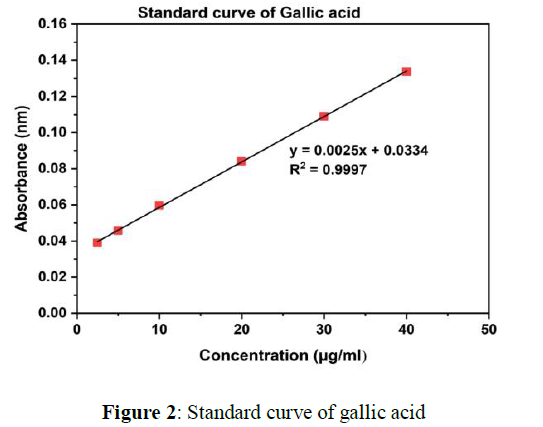
Absorbance of MA extracts (chloroform, petroleum ether, ethanol and water) and standard compound (gallic acid) has been taken by using UV-vis spectrophotometer at 760nm. Quantity of total phenolic contents in various extracts of MA were calculated by a linear curve of gallic acid (y = 0.0025x+ 0.0334, R2 =0.9997). The linear gallic acid standard curve shown in the Figure 2.
Total phenolic compound were determined in water extract 67.32±0.50 milligram/gram GAE, in extract of ethanol 61. 37±0.24 milligram/gram GAE, in extract of chloroform 53.22±0.86 milligram/gram GAE and in extract of petroleum ether 47.44± 0.29 milligram/gram GAE.
Total flavonoid compound of Morus alba leaves extracts
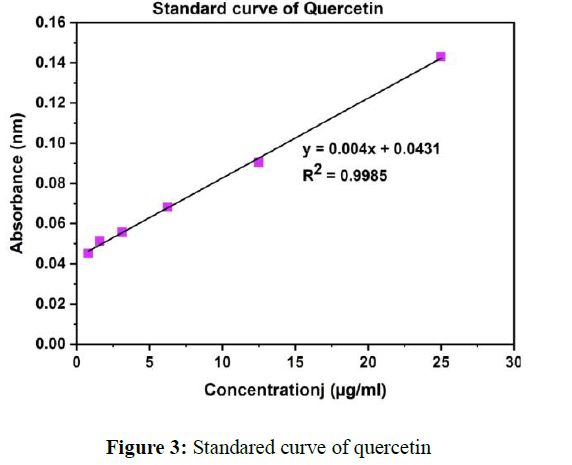
Total flavonoidal contents of MA in various organic solvents (chloroform, petroleum ether, ethanol and water) were analyzed with the help of standard compound. The amount of total flavonoids in various extracts was estimated by a linear regression curve of quercetin (y = 0.004x+ 0.0431, R2= 0.9985) and standard compound has been used for estimation of flavonoids and represented as mg/g QE (Figure 3).
Total contents of flavonoid in MA were estimated 55.37±0.05 milligram/gram QE in extract of water, 47.62±0.29 milligram/gram QE in extract of ethanol, 42.35± 0.21 milligram/gram QE in extract of chloroform and 35.51± 0.24 milligram/gram QE in extract of petroleum ether through quercetin regression curve.
In-vitro antioxidant activity
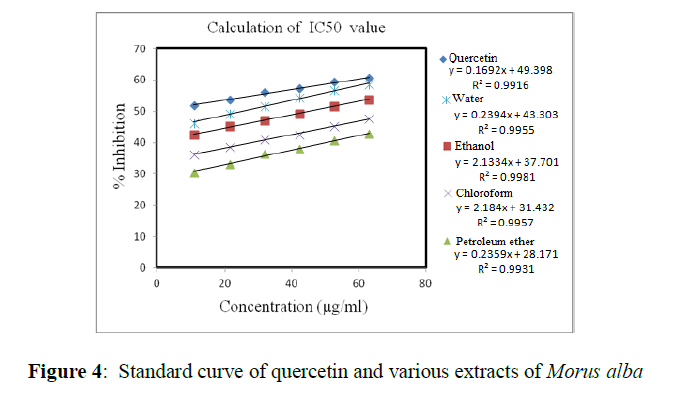
IC50 value of various MA extracts and standard compound (quercetin) was calculated by plotting a graph between the concentration and % inhibition of standard compound as well as various extracts of MA (Figure 4). This graph indicates the antioxidant activity increased with their increase in concentrations.
Antioxidant capacity of water extract was higher than ethanol, chloroform and petroleum ether extract. The values of IC50 is inversely proportional to antioxidant capacity and expressed in μg/ml. IC50 values of quercetin, water extract, ethanol extract, chloroform extract and petroleum ether extract of MA are summarized in Table 3.
| Plant extracts | IC50 values( µg/ml) |
|---|---|
| Quercetin (std. compound) | 3.16±0.05 |
| water | 4.12±0.01 |
| ethanol | 7.19±0.12 |
| chloroform | 9.23±0.09 |
| petroleum ether | 12.18±0.03 |
Acute toxicity
A normal healthy rat, up to dose level 2000 mg/kg b. wt does not show any harmful effect when treated with hydro-alcoholic leaves extract of MA, during the examination time of 14 days. The finding of this study indicates that hydro-alcoholic leaves extract of MA is non-toxic up to 2000 mg/kg body weight and can be considered as safe for consumption.
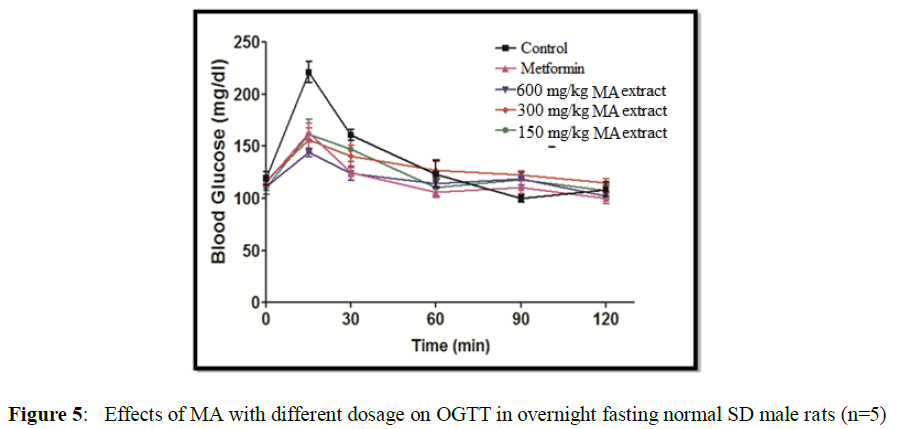
Effect of MALE on OGTT in SD male rats
The glucose level in the blood of all male was recorded at a time15 min, 30 min, 60 min, 90 min and 120 min after administration of MA leaves extract (Figure 5).
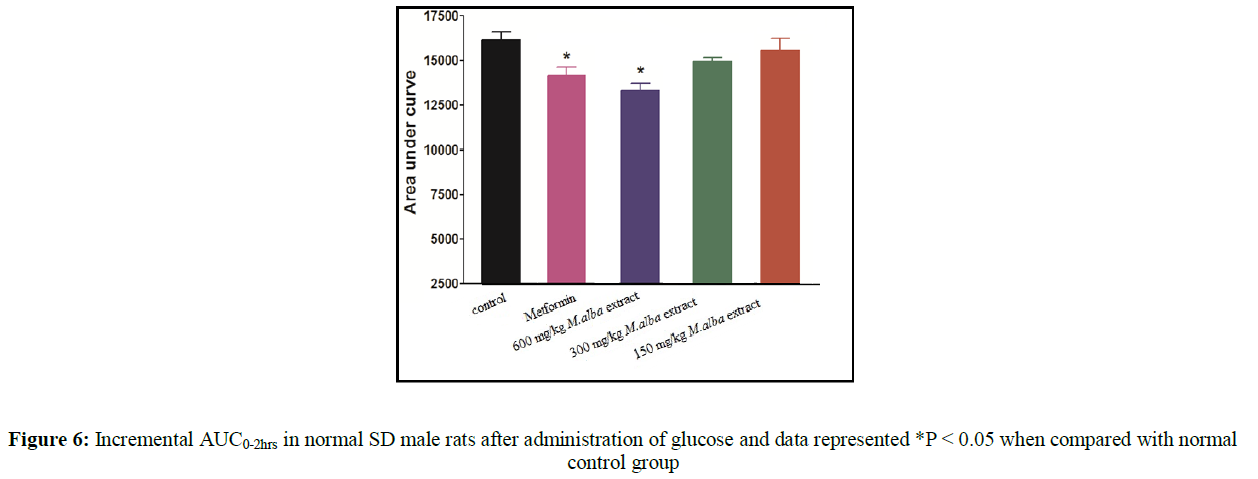
The area under curve (0 hr to 2 hr) shows significant improvement in oral glucose tolerance test after glucose treatment in normal rats (Figure 6). The % anti-hyperglycemic activity was determined by comparing the AUC of MA treated group with vehicle treated control group. Metformin improved glucose tolerance up to 14.6 percent at a dose 100 mg/kg body weight while MA improve 3.45% at a dose 150 mg/kg body weight, 8.12% at a dose 300 mg/kg body weight and 11.38 % at a dose 600 mg/kg body weight.
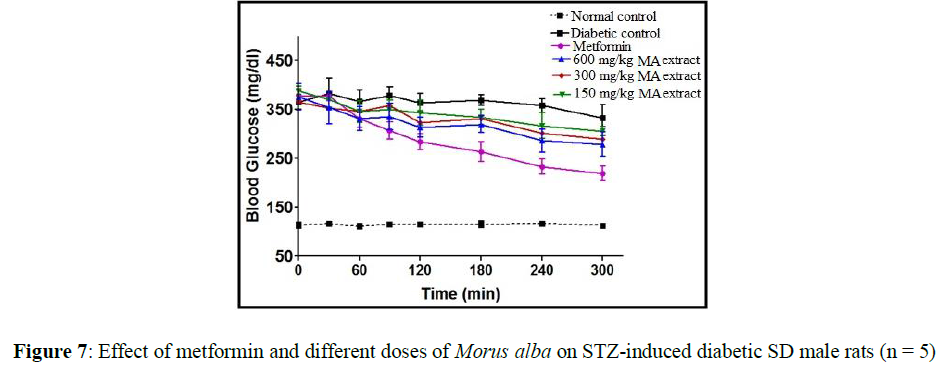
Effect of anti-hyperglycemic activity of Morus alba in streptozotocin-induced SD male rats
STZ treated male rats caused hyperglycemia. After the oral administration of metformin and MA extract in diabetic male rats, there is significant reduction of blood glucose level have been observed. MA leaves extract causes time dependent decrease in blood glucose level in diabetic male rats at different dose of 150 mg/kg body weight, 300 mg/kg body weight and 600 mg/kg body weight (Figure 7).
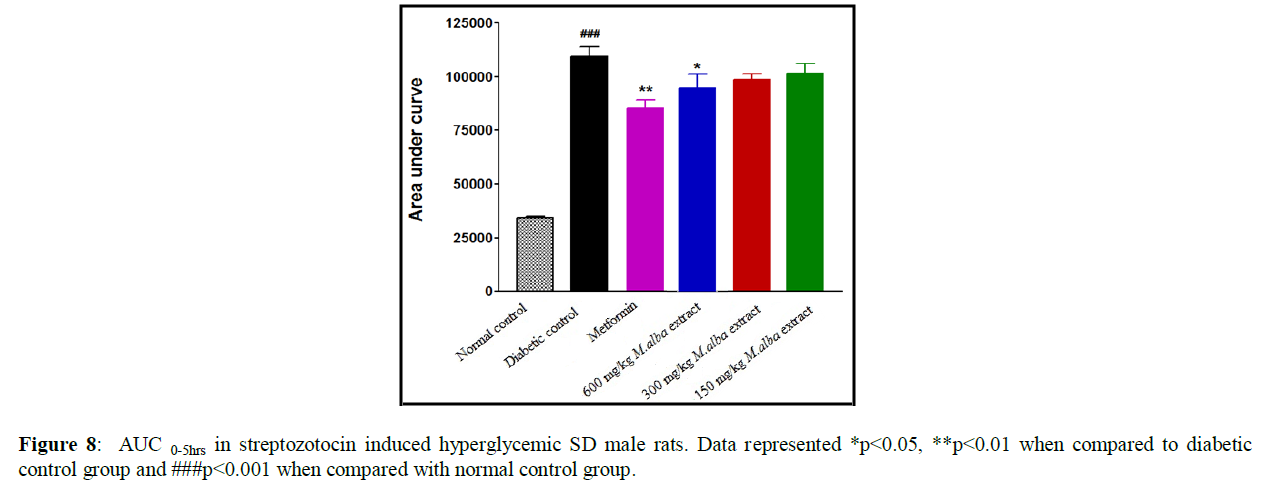
After administration of MA extract and metformin in diabetic male rats, the area under curve (AUC0-5hrs) show the major decline in glucose level (Figure 8). Throughout testing with different dosage of MA extract, 13.2 % decline in glucose level at a dose of 600 mg/kg body weight, 9.11% reduction in blood glucose level at a dose of 300 mg/kg b. wt. and 7.23% reduction in level of blood glucose at a dose 150 mg/kg b. wt. was obtained. The decline in blood glucose was obtained 21.9% throughout testing with metformin at a dose of 100 mg/kg body weight. The % anti-hyperglycemic activity was determined by comparing, AUC of experimental animals with vehicle treated control group.
After administration of MA extract and metformin in diabetic male rats, the area under curve (AUC0-5hrs) show the major decline in glucose level (Figure 8). Throughout testing with different dosage of MA extract, 13.2 % decline in glucose level at a dose of 600 mg/kg body weight, 9.11% reduction in blood glucose level at a dose of 300 mg/kg b. wt. and 7.23% reduction in level of blood glucose at a dose 150 mg/kg b. wt. was obtained. The decline in blood glucose was obtained 21.9% throughout testing with metformin at a dose of 100 mg/kg body weight. The % anti-hyperglycemic activity was determined by comparing, AUC of experimental animals with vehicle treated control group.
Assessment of biochemical parameters
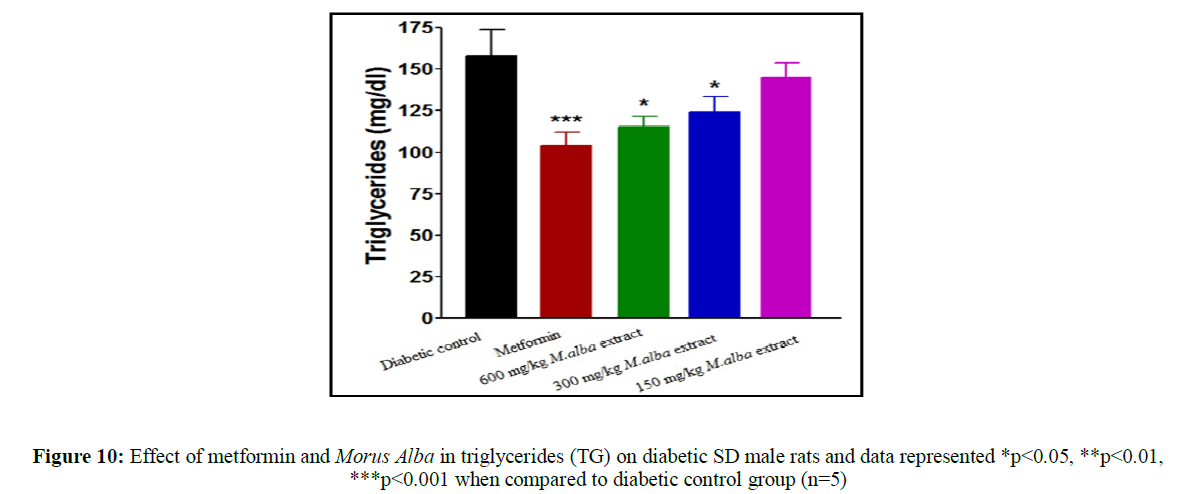
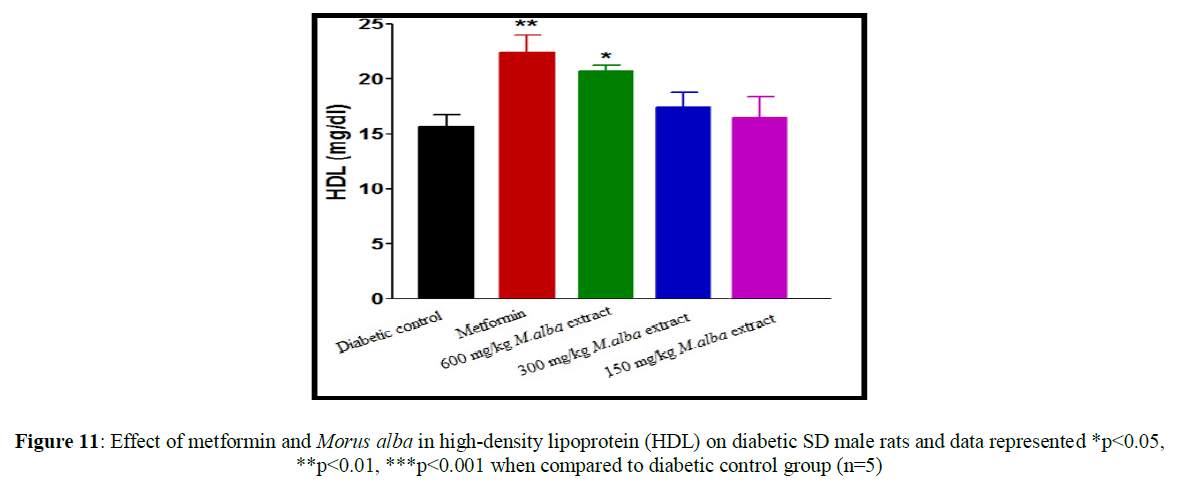
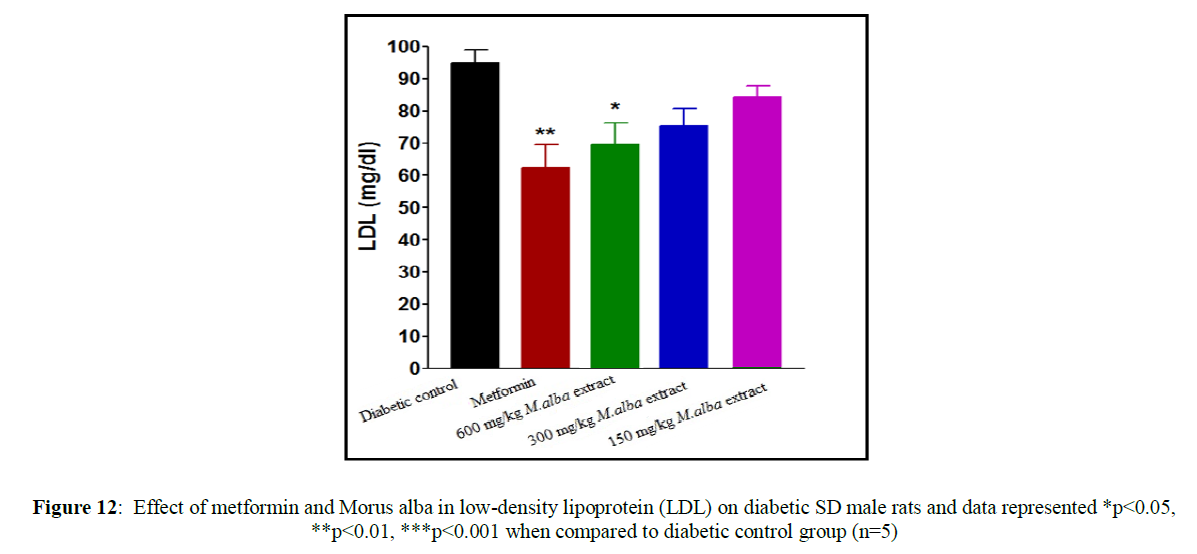
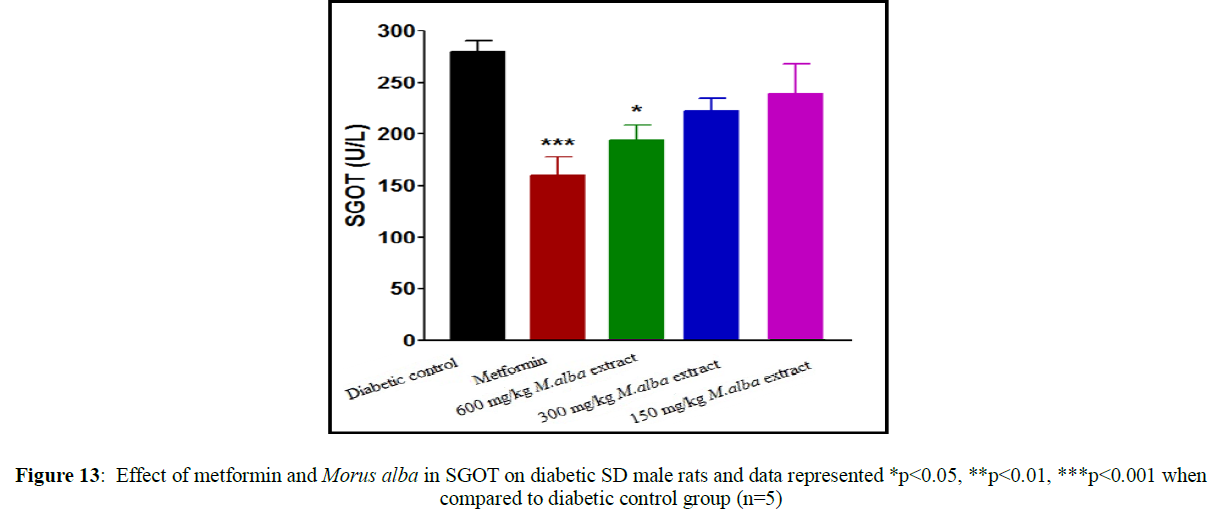
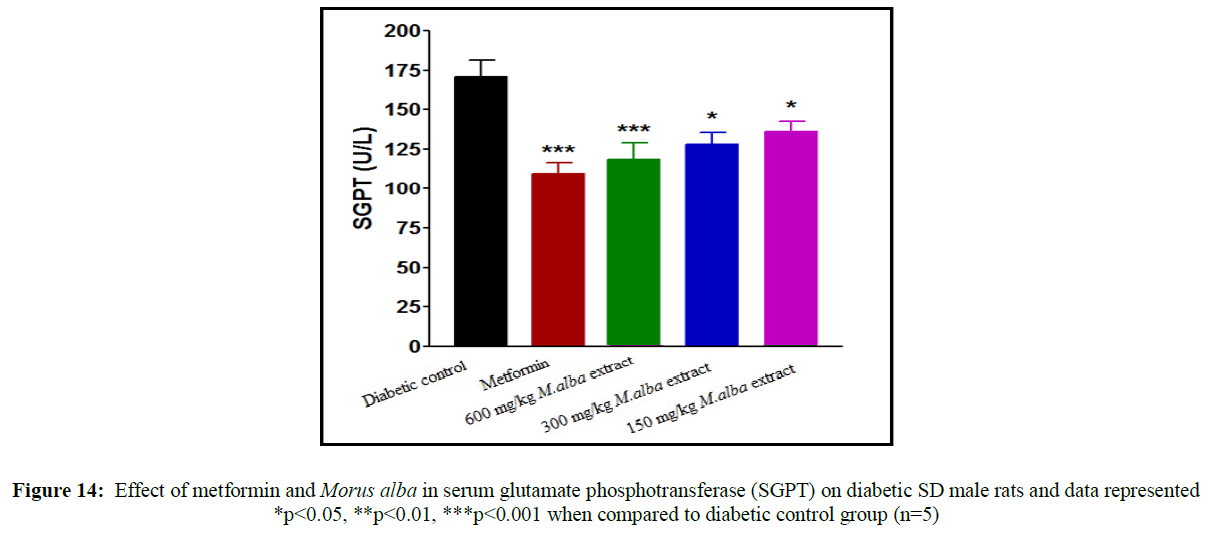
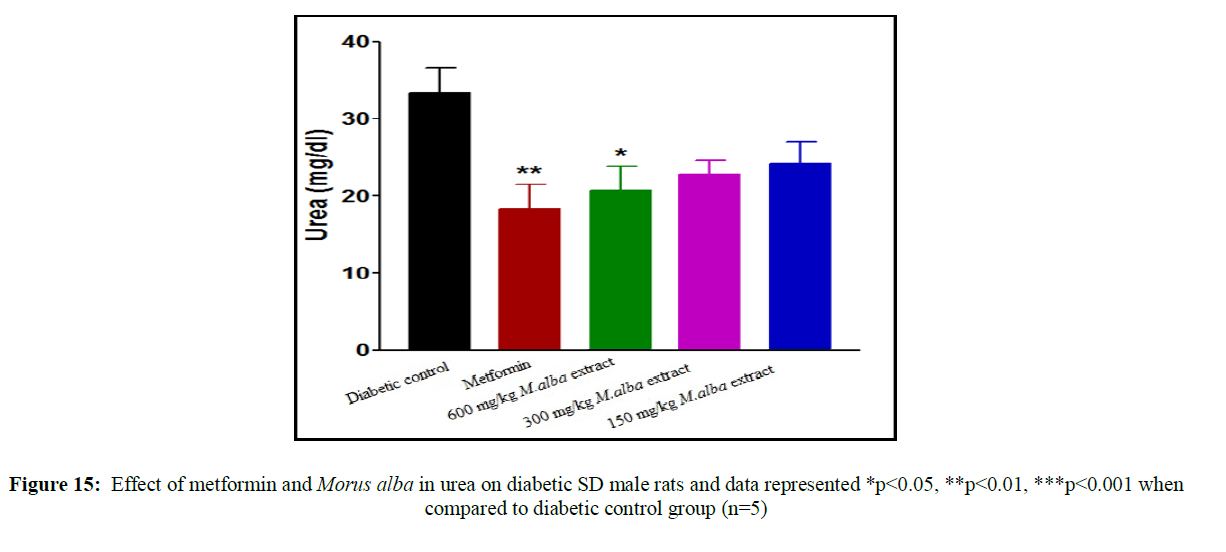
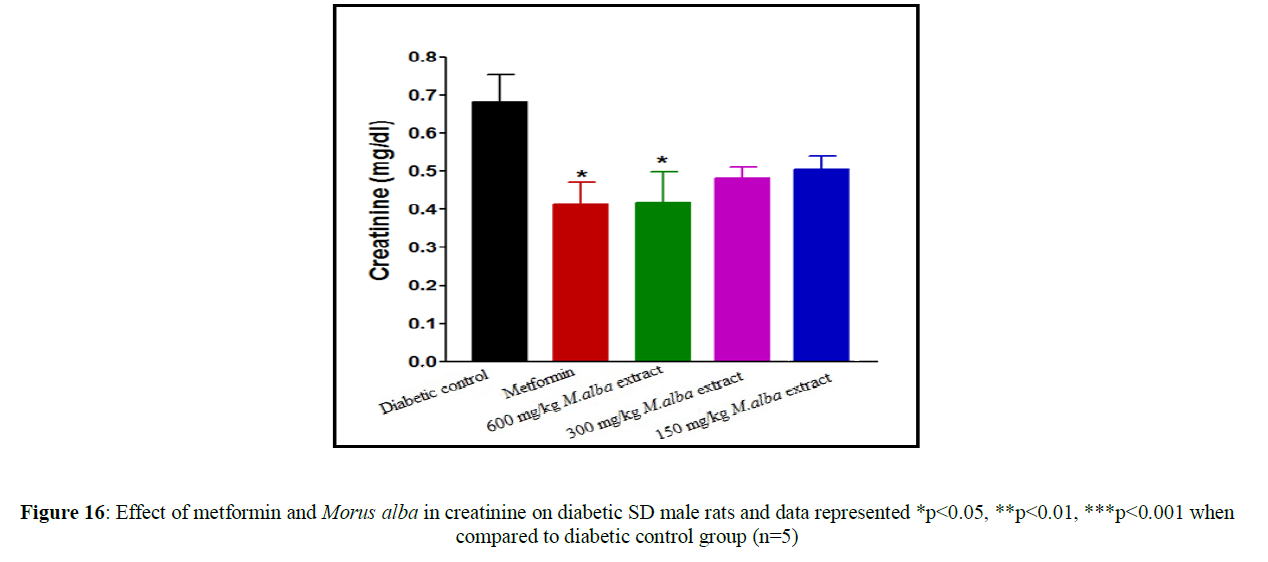
Treatment with metformin and MA have been shown significant change in their biochemical parameters of like urea, triglycerides (TG), creatinine, low-density lipoprotein (LDL), total cholesterol (TC), high-density lipoprotein (HDL), SGPT and SGOT (Figures 9-16).
Total cholesterol (142.17±1.25), triglycerides (160.12±8.33), low-density lipoprotein (95.37±2.95), serum glutamate oxaloacetic acid transferase (285.73±2.38), serum glutamate phosphotransferase (174.12±5.42), urea (34.22±5.20) and creatinine (0.68±0.05) were obtained to be considerably raised in streptozotocin-induced diabetic SD male rats as compare to normal control group, although high-density lipoprotein (16.32±2.59) was significantly decreased.
Total cholesterol (TC) in SD male rats, significant reduced by metformin was found up to (75.28±8.14) while up to (105.27±1.14) at dose of 600 mg/kg body weight, reduction in triglycerides (TG) by metformin, was found up to (103.13±3.18) while up to (106.37±4.11) at dose of 600 mg/kg body weight, reduction in low-density lipoprotein (LDL) by metformin, was found up to (62.82±2.63) while up to (71.12±2.85) at dose of 600 mg/kg b. wt, decline in SGOT by metformin, was obtained up to (160.45±2.10) whereas up to (198.92±2.20) at dose of 600 mg/kg b. wt, decline in SGPT by metformin, was obtained up to (116.08±3.52) although up to (122.75±3.36) at dose of 600 mg/kg b wt, decline in urea by standard drug (metformin), was obtained up to (19.02±2.36) although up to (23.12±3.38) at dose of 600 mg/kg b. wt, reduction in creatinine by metformin, was obtained up to (0.40±0.03) whereas up to (0.41±0.02) at dose of 600 mg/kg b. wt. HDL was improved by standard drug up to (23.14±3.11) whereas up to (22.1±1.42) at dose of 600 mg/kg b. wt.
Treatment with hydro-alcoholic leaves extracts of various dosage of M. alba (600 mg/kg, 300 mg/kg and 150 mg/kg body weight) show the reduction in all biochemical parameters, but high-density lipoprotein was increased.
Discussion
From the literature survey, we can see that there are major opportunities in the extraction of bioactive components derived from leaves of Indian medicinal plants and their animal testing. Our country is very rich in natural resources like plants and marine drugs [45]. Plants are always a natural source for drug development for treatment of various diseases. Various bioactive phytoconstituents are obtained from plants have a number of therapeutic activities. several plants are known in folk medicine to be used for their hypoglycemic activity by different mechanism such as stimulating the pancreas for releasing of insulin from β- cells, declining hepatic glucose production, Glucose-6-phosphatase as well fructose-1, 6-biphosphatase inhibition in the liver, reduction of the absorption of glucose from intestine, improves glucose tolerance, increase plasma insulin level and maintains the homeostasis of glucose by controlling the carbohydrate metabolizing enzyme [46,47].
Previous studies have been examined the total Phenolic contents, total flavonoid contents and anthocyanins in methanol, water and methanol-water (70-30 v/v %). The highest Phenolic contents was found in methanol-water (70-30 v/v %) extract was 580 mg GAE/100g, while water extract exhibited lowest Phenolic contents was 330 mg GAE/100g (kutlu et al.,2011). In our study total phenolic compound were determined in water extract 67.32±0.50 milligram/gram GAE, in extract of ethanol 61. 37±0.24 milligram/gram GAE, in extract of chloroform 53.22±0.86 milligram/gram GAE and in extract of petroleum ether 47.44± 0.29 mg/g GAE. The highest Phenolic content was found in water extract, while petroleum ether extract exhibited lowest Phenolic contents. Differences in total amount of phenols are depends on the collection of the plants materials from different regions, use of extraction method, polarity of the extracting solvents, duration of extraction can be influence the total Phenolic contents [48].
The present study was designed to investigate the in-vitro antioxidant and in- vivo anti-hyperglycemic activity of leaves extracts of M. alba. In a previous work, methanol was used for extraction of the crude drug. While in our study Hydro-alcohol was used instead of methanol due to the toxicity in animals. Hydro-alcoholic leaves extract is good solvent for extraction of polyphenol and is safe for consumption [49]. It shows great improvement in oral glucose tolerance in normal rats and decrease in level of blood glucose in STZ-induced diabetic male rats. These results prove their potential as hypoglycemic agent that may be triggered by increasing the production of insulin from pancreatic β- cells. Some authors have investigated ethanolic extracts of three plants Pongamia pinnata fruits, Ficus bengalensis aerial roots, Tinospora cardifolia stem and aqueous extracts of Withania coagulance fruits, Pterocarpus marsupium heartwood to isolate their antidiabetic compound. All isolated compounds were evaluated for in-vivo antidiabetic activity in STZ induced β- cell damaged diabetic male SD rats and activities were compared with metformin. All isolated components shows good antidiabetic activity [50,51].
Streptozotocin cause toxic effect on β- cells of pancreas, leaving less functional cells which consequences in hyperglycemia. Blood glucose lowering effect of M. Alba has also been reported in alloxane induced diabetic rats. In most studies alloxane or Streptozotocin was used for inducing diabetes mellitus. Improvement in oral glucose tolerance (OGTT) and decreased in level of blood glucose by hydro-alcoholic extract of M. Alba may be their regenerative property of pancreatic β- cell due to the presence of various phytoconstituents [52]. These phytoconstituents are quercetin, chlorogenic acid, rutin and isoquercetin reported by Hunyadi et al 2012 [53]. Several previous investigations confirmed that different aspects of the controlling effects on free radicals and diabetes by active compounds obtained from the Morus alba leaf extract. Some authors have reported hypoglycemic efficacy of Lagenaria siceraria which are normally used as vegetables.
Methanolic leaves extract of M. alba was used for isolation and quantification of the occurrence of rutin, chlorogenic acid, isoquercetin, quercetin and kaempferol through HPLC analysis. These phytoconstituents shows strong antioxidant activity. Antioxidant compounds are scavenges the free radicals and protects our body from damaging cells. DPPH (2,2 Diphenyl -1-Picrylhydrazyl) assay was used for determination of their scavenging capacity of free radicals [54]. Generation of free radicals is the intracellular phenomena which is responsible for cytotoxicity. M. alba was analyzed for physicochemical study to detect heavy metals because they cause cancer and other serious side effects.
Acute oral toxicity was performed in normal SD male rats during the study, which does not show any harmful effect. Most common complications during diabetes are abnormalities in serum lipid profile, liver function and kidney function. Insulin deficiency causes increase free fatty acid, due to decrease lipoprotein lipase enzyme activity [55,56]. Present research article reported increase in biochemical parameters such as, triglycerides (TG) level, low-density lipoprotein (LDL) level, total cholesterol (TC) level, SGPT level, SGOT level along with creatinine and urea in STZ induced diabetic male rats comparison to normal rats while high-density lipoprotein (HDL) level was decrease.
Insulin deficiency in the blood is related to hypertriglyceridemia and hypercholesterolemia, which is responsible for coronary heart disease and atherosclerosis. M. alba leaves extract decrease triglyceride (TG) level, total cholesterol (TC) level along with low-density lipoprotein (LDL) level in streptozotocin-induced diabetic SD rats while it increase high-density lipoprotein (HDL) level.
Liver is an insulin dependent organ which play important role in lipid and carbohydrate metabolism. In present study increase in SGPT level and SGOT level due to the damage of hepatic tissues. Decrease in SGOT level and SGPT level by curative effect of hydro-alcoholic extract of M. alba in STZ induced diabetic SD male rats.
Renal dysfunction is a common complication of diabetic patient. Urea and creatinine level is increased in STZ induced diabetic SD male rats comparison to normal rats. The STZ induced diabetic rats treated with leaves extract showed significant decrease in urea and creatinine level. Our study indicates the Morus alba leaves is very valuable natural drug for improving glucose tolerance, decreasing the raised lipids level and increased anti-oxidant enzyme activities.
Conclusion
M. alba leaves extract consists of a huge amount of Phenolic and flavonoidal compounds which works as anti-hyperglycemic agent, free radical scavenging agent, lipid lowering agent, liver protective agent, and kidney protective agent. The results of the present study that hydro-alcoholic leaves extract of Morus alba useful in glycemic control and reducing the complications related to diabetes mellitus. Present study gives us insight to develop novel herbal medicines to cure diabetes.
Acknowledgements
We are grateful to Director NBRI Lucknow for plant authentication and Director CDRI, Lucknow for giving us opportunity for animal testing. The kind support of Institute of Engineering & Technology Lucknow is greatly acknowledged.
References
- Figueredo KC, Guex CG, Reginato FZ, et al., J. Ethnophar. 2018, 224: p. 290-296.
- Gebremeskel L, Tuem KB, Teklu T. Diab Met Synd Obes. 2020, 13: p. 1481-1489.
- Bajpai S, Bhaskara Rao AV, Muthukumaran M, et al., J. Pharm, 2012, 2(4): p.13-16.
- Chan EW, Lye PY and Wong SK. J Nat Med. 2016, 14(1): p. 17-30.
- Awasthi AK, Nagaraja GM, Naik GV, et al., BMC Genet, 2004, 5(1): p. 1471-2156.
- Devi B, Sharma N, Kumar D, et al., Int J Pharm. 2013, 5(2): p. 14-18.
- Gryn-Rynko A, Bazylak G, Olszewska-Slonina D. Biomed Pharmacother. 2016, 84(1), p. 628-636.
- Gundogdu M, Tuncturk M, Berk S, et al., Indian J Pharm Educ Res. 2018, 52(4): p. 96-101.
- Sanchez SEM, Tassotti M, Del RD, et al., Food Chem. 2016, 212: p. 250-255.
- Danijela AK, Danica SD, Snezana SM, et al., Ana J Pharm Res. 2013, 12: p. 105-110.
- Butt MS, Nazir A, Sultan TS, et al., Tren Food Sc Tech. 2008, 19(10): p. 505-512.
- Yang ZG, Matsuzaki K, Takamatsu S, et al., Molecules. 2011, 16(7): p. 6010-6022.
- Khan D, Sharif A, Zafar M, et al. Awan Curr Phar Biot. 2020, 21: p.1059-1069.
- Ansari SH, Essentials of Pharmacognosy, 2008, 2nd Edn, Birla Publishing Pvt. Ltd., New Delhi, p. 9-11.
- Pharmacopoeia I. Ministry of health and family welfare. Govt. of India Publication. New Delhi, 1996, p. 947-950.
- Paul V, Pandey R, Ramesh KV, et al., ICAR Manual. 2017, p. 84-86.
- Ilesanmi OB, Olaleye TM, Akinmoladun AC, et al., Afri J Biotech. 2016, 15: p. 320-330.
- Garcia AB, Berrueta LA, Lobato GS, et al., J Chrom, 2009, 1216(28): p. 5398-415.
- Singh S, Tripathi A, Lal VK, et al., Asian J Chem. 2019, 31: p. 1801-1804.
- Supe U, Daniel P. J Agric Enviro Sci. 2015, 15: p. 2196-2200.
- Wang XY, Liang Q, Chen HG, et al., Chem Pape. 2018, 72: p. 2255-2264.
- Hameed SSA, Bazaid SA, Shohayeb MM. Chem Pape. 2014, 68: p. 1358-1367.
- Vuong QV, Hirun S, Roach PD, et al., J Herb Med. 2013, 3: p. 104-111.
- Seeram NP, Lee R, Scheuller HS, et al., Food Chem. 2006, 97(1): p. 1-11.
- Sudjaroen Y, Hull WE, Erben G, et al., Phytochemistry. 2012, 77(1): p. 226-37.
- Kassima M, Achoui M, Mustafa MR, et al., Nutr Res. 2010, 30(9): p. 650-59.
- Singh S. Asian J Bioch Pharm Res. 2012, 2: p. 388-392.
- Thiele FB, Marina Z, Mariana P, et al., Molecules. 2018, 18: p. 8342-8357.
- Kutlu T, Durmaz G, Tes B, et al., Turk J Biol, 2011, 35: p. 103-110.
- Singh S, Tripathi A, Lal VK, et al., Der Pharm Chem. 2019, 11: p. 25-30.
- Uddin R, Saha MR, Subhan N, et al., Adv Phar Bulleti. 2014, 4(3): p. 273-281.
- Kaur S and Mondal P. J Microbiol Exp. 2014, 1(1): p. 23-28.
- Pham HNT, Vuong QV, Bowyer MC, et al., Chem Pape. 2017, 71: p. 2233-2242.
- Bhuyan DJ, Vuong QV, Chalmers AC, et al., Chem Paper. 2016, 70(5): p. 567-75.
- Arfan M, Khan R, Rybarczyk A, et al., Int J Mol Sci. 2012, 13: p. 2472-2480.
- Fernandes A, Sousa A, Mateus N, et al., Food Chem. 2011, 125(4): p. 1398-1405.
- Goldsmith CD, Vuong QV, Sadeqzadeh E, et al., Molecules. 2015, 20(7): p. 12992-3004.
- Maurya S and Singh D. Int J Pharm Tech Res. 2010, 2: p. 2403-6.
- Elkotby D, Hassan AK, Emad R, et al., Int J Pure Appl Zoo. 2018, 6: p. 1-8.
- Mohammad ST, Mohammad ST, Mahboubeh M. Biomed Res. 2019, 11: p. 115-120.
- Hussain F, Rana Z, Shafique H, Malik A, et al., Asian Pac J Trop Biomed. 2017, 7(10): p. 950-956.
- Mohammadi J and Naik RP. Ind J Pharm. 2008, 40(1): p. 15-18.
- Tamrakar AK, Tiwari P, Ahmad R, et al., Med Chem Res. 2008, 17: p. 62-73.
- Singh S, Maurya DP, Shoaib A, et al., J Ind Chem Soc. 2021, 98: p. 100170.
- Ionica ME, Nour V and Trandafir I. J Hortic Biol Environ. 2018, 8(2): p. 79-88.
- Li Y, Bao T, Chen W. Food Chem. 2018, 243(1): p. 65-73.
- Kusano G, Orihara S, Tsukamato D, et al., Chem Pharm Bull. 2002, 50(2): p. 185-192.
- Sunitha P, Sathyanarayana N, Suresh VC. Ind J Pharm Sci. 2018, 80(1): p. 192-198.
- Park KT, Kim JK, Hwang D, et al., Food Chem Toxicol. 2011, 49(12): p. 3038-45.
- Sharma SB, Gupta S, Rini AC, et al., J Pharm and Pharm. 2010, 62(2): p. 247-55.
- Chaurasia S, Saxena RC, Chaurasia ID, et al., Int J Chem Sci. 2011, 9(2): p. 489-492.
- Ajiboye BO, Shonibare MT, Oyinloye BE. J Diab Met Diso. 2020, 19: p. 343-352.
- Hunyadi A, Martins A, Hsieh TJ, et al., PLOS One. 2012, 7: p. 619-625.
- Supritha P and Radha KV. Int J Pharm Edu Res. 2018, 52(2): p. 321-326.
- Hamdy SM. J of Appl Sc Res. 2012, 8(1): p. 10-16.
- Mohammadi J and Naik PR. Turk J Bio. 2012, 36(2): p. 211-216.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref