Letter - Der Pharma Chemica ( 2023) Volume 15, Issue 4
Study of Electrical and Mechanical Properties of Polyester and Graphene Composites
Ghufran M Shabeeb1*, Zainab J Sweah2, Ahmed J Mohammed1 and Ibrahim Kadim A12Department of Chemistry and Polymer Technology, Polymer Research Center, Basra, Iraq
Ghufran M Shabeeb, Department of Material Sciences, Polymer Research Center, University of Basra, Iraq, Email: ghufran.shabeeb@uobasrah.edu.iq
Received: 29-Mar-2023, Manuscript No. dpc-23-93547; Editor assigned: 31-Mar-2023, Pre QC No. dpc-23-93547; Reviewed: 14-Apr-2023, QC No. dpc-23-93547; Revised: 17-Apr-2023, Manuscript No. dpc-23-93547; Published: 24-Apr-2023, DOI: 10.4172/0975-413X.15.4.1-4
Abstract
The exotic physical properties of graphene have led to intense research activities on the synthesis and characterization of graphene during the last decade. In this article, the methods developed for the preparation of such materials and the different application areas are reviewed. The composites discussed are of two types, via; graphene/polymer. The weight percent of graphene was 1%, 3%, 5% and 7% wt percent of the total weight of polyester were investigated. Electrical and mechanical properties were also investigated for all prepared ratios.
Keywords
Polyester; Graphene; Electrical Properties; Mechanical Properties
INTRODUCTION
Graphene is an atom-thick layer of two-dimensional carbon atoms that attracted many researchers because of its physical and chemical nature. Graphene and its derivatives are nanomaterials that significantly improve the performance of the polymer and change its mechanical, electrical, and thermal properties [1,2]. Because of their light weight, low cost, ease of manufacture, and desirable qualities, polymers have been employed in numerous studies. Examples include polyester (PS) [3,4], polyvinyl chloride (PVC) [5,6], and polyvinyl alcohol (PVA) [7,8] and PMMA [9,10]. There are four main classes of such polyester polyols (Randall, 2002): linear or lightly branched aliphatic polyester polyols with terminal hydroxyl groups, low molecular weight aromatic polyester polyols for rigid foam applications, polycaprolactones and polycarbonate polyols. The linear or lightly branched polyester polyols may be aliphatic or aromatic. Aromatic polyester polyols are produced from phthalic acid, isophthalic acid, or terephthalic acid. While the manufacture from phthalic or isophthalic acids is generally based on the monomers terephthalic acid oligomers may as well be produced from monomeric esters such as the dimethyl ester of terephthalic acid (DMT) or polyesters such as polyethylene terephthalate (PET), polybutylene terephthalate (PBT) or polypropylene terephthalate (PPT). Polyesters are an important class of high-performance and engineering polymers, which find use in many applications, the role of the polymer resin is primarily to bind the fibres Together, and the compound gives a beautiful surface appearance as well as the environment tolerance and provide overall durability [11-13].
Many studies have evidenced that graphene/polymer nanocomposites can be used in different fields by chemical activation of graphene used as filler. The manufacturing industry cannot be neglected. The use of graphene (or graphene-like materials) in commercial products. This is expected to happen it will turn from a typical material into a high-performance fabric that can provide the best solutions for the industry and consumers. Therefore, the commercial effect of graphene/polymer nanocomposites are constantly increasing [14].
Graphene was mixed with ethylene-vinyl alcohol (EVOH) Nano composites developed by melt mixing. With four different entries checked. Two graphene fillers and two graphite fillers displaying different morphologies were selected [15]. The addition of GnP to the polymer foam also resulted in significant improvements in its electrical conductivity, and the final value depends on the microstructure of the Nano composite [16]. The effect of GO on the mechanical properties of LLDPE composites has been studied through experiments. Samples were made by melt mounting and injection moulding. A series of experiments, including a tensile test, improved the tensile properties of the LLDPE polymer with the addition of GO [17]. The aim of this study was to using grapheme as fillers to Increase efficiency of commercial polyesters and study the electrical and mechanical properties of the prepared samples and work to use them in appropriate and important applications.
MATERIALS AND METHODS
The polyester used in this study was purchased as a basic material by (Henkel A.S.). Use graphene platelet nano powder from skyspring nano material with a thickness of (6.8 nm).
For the preparation of polyester / graphene samples, different proportions of graphene 1%, 3%, 5%, 7% w/w were mixed with fixed proportions of polyester, and study their electrical and mechanical properties. A set of samples was prepared in this study by manual mixing of polyester in a constant proportion with different percentages of graphene, at room temperature, and then the samples are poured on a glass plate and left to dry for 24 hours.
RESULTS AND DISCUSSION
Electrical conductivity
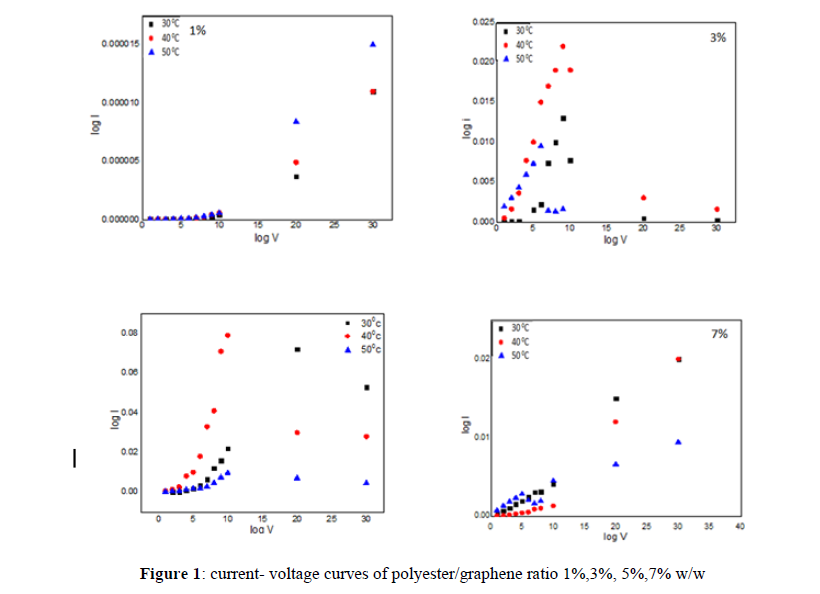
The polyester/graphene feature of current and voltage was measured at various temperatures (293-303 K). As the relationship between the current and voltage is subject to ohmic behavior in the low voltage range, which runs between 1 and 10 volts, the current passing through the sample is dependent on temperature and the applied voltage. Since the transfer of charge carriers is influenced by the voltage barrier generated at the interface between the metal electrode and the polymer, or by the voltage barrier generated inside the polymeric material between the electronic levels, the concentration of charge carriers generated in this region as a result of thermal irritation is significantly higher than the charges injected from the electrodes identifying the conduction mechanisms that regulate the transmission [18]. Figure (1) show current- voltage curves of polyester/graphene ratio 1%,3%, 5%,7% w/w .
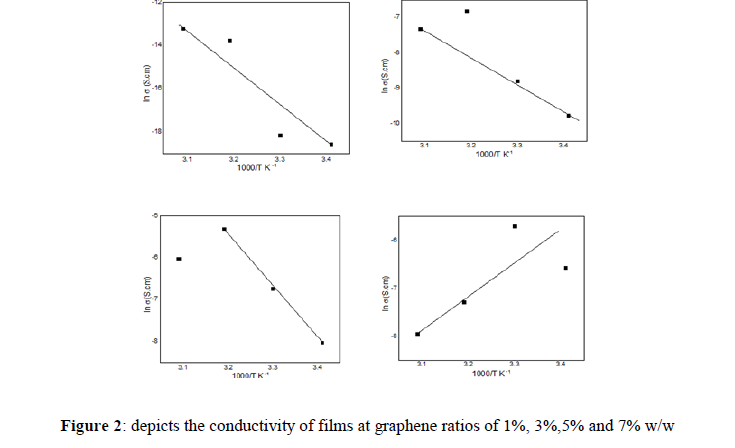
Figure 2 shows the relationship between 1000/T and ln σdc for polyester/ graphene for which the activation energy Ea could be estimated from the slope of the straight line using the following [19].
Ea=2.303*8.6*10-5*slope
Mechanical properties
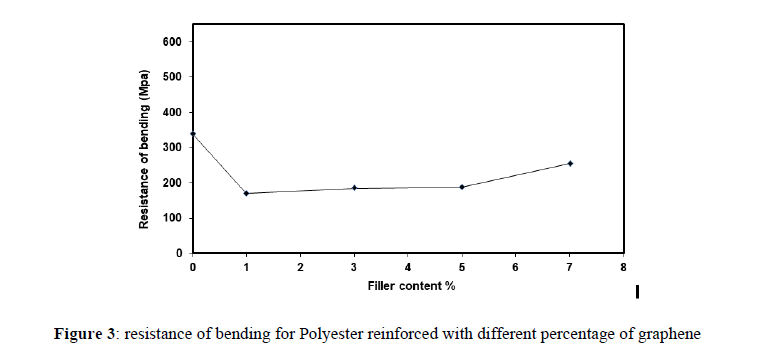
Figure (3) shows the relation between the bending resistance and the weight ratios for the (polyester / graphene). Bending resistance behavior starting with a strong effect when (0%) value of (338) MPa. When the polymer is pure without any addition, the sample elasticity is lower and has a high hardness than the other samples that have added fillers. The bending resistance behavior of the measurement model decreases at the weight ratio of the fillers (3%) at a value of 185 MPa. While the behavior increases with the increase in the weight ratio of the fillers (7%) at a value of 255 MPa. This result occurred because polyester and graphene received most of the pressure (the distribution of the additive to the base material is inhomogeneous). Thus, given the distribution and pressures on all sides of the sample, no pressure is concentrated in one area. The result is a failure and less resistance; thus, maximum stress is needed to achieve the loss in bending resistance at this ratio of the polymer with fillers. A strong relationship may exist between the graphene and polyester, creating a good surface. Thus, failure to bending resistance requires maximum stress.
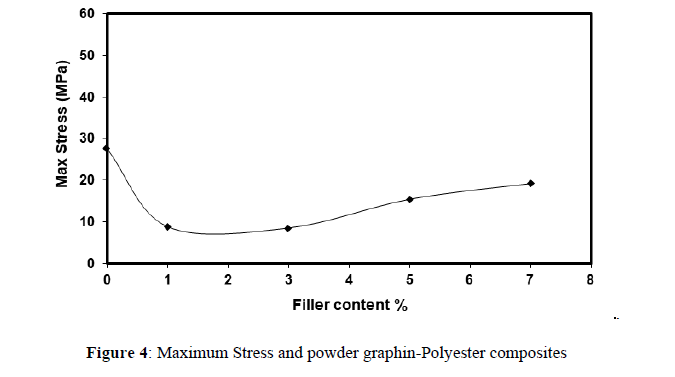
The behavior of the stress begins the low effect when the percentage of (3 %), of the additive, and then increases to (15.4 Mpa) when the percentage is (5 %) for tensile strength, and stress increases when increase the ratio of adding graphene when the percentage is (7 %). This shows that filler (graphene) works to improve the hardness property at a percentage, when the polymer hardness extends at this rate of the effect of the distribution of homogeneous material nature solid. The relation between stresses with percentages added to the polymer shows in figure 4.
CONCLUSION
In this study, different proportions of graphene were mixed with polyester, and the electrical and mechanical properties were studied at different temperatures. The results showed that the electrical conductivity of polyester/graphene at room temperature for the ratios 1%, 3%, 3% and 7% is (8.6 ×10-9, 5.7× 10-5,3.2×10-4 and 1.4 ×10-3 ) S/cm respectively. The mechanical properties have been studied for polyester/ graphene ratios. When the polyester is pure without any addition, the sample elasticity is lower and has a high hardness than the other samples that have added fillers. The graphene works to improve the hardness property at a percentage when the polymer hardness extends at this rate of the effect of the distribution of homogeneous material nature solid.
REFERENCES
- Brownson DAC, Kampouris DK, Banks CE. Chemical Society Reviews. 2012, 41(21): p. 6944-6976.
- Liu J, Chen Y, Guo Y, et al., J Nanomaterials. 2013, vol. 2013, Article ID 632809, 6 pages, 2013.
- Liao K and Li S. Applied Physics Letters. 2001, 79 (25): p. 4225.
- Fang M, Wang K, Lu H, et al., Materials Chemistry. 2009, 19: p. 7098-7105.
- Abbaspour A and Izadyar A. Talanta. 2007, 71(2): p. 887-892.
- Hasan M and Lee M. Progress in Natural Science: Materials International. 2014, 24(6): p. 579-587.
- Liu L, Barber AH, Wagner HD. Advanced Functional Materials. 2005, 15(6): p. 975-980.
- Zhao X, Zhang Q, Chen D, et al., Macromolecules. 2010, 43(5): p. 2357-2363.
- Stephan C, Nguyen TP, Chapelle MLd, et al., Synthetic Metals. 2000, 108: p. 139-149.
- Villar-Rodil S, Paredes JI, Martínez -Alonso A. J Materials Chemistry. 2009, 19(22): p. 3591.
- Fadhil AH, Mohammed SH, Reham RA. Eng &Tech Journal. 2016, 34: p. 9.
- Ayesha K. J Plastic Film & Sheeting. 2019, 35(1): p. 22-44.
- Davallo M, Pasdar H, Mohseni M. Int J ChemTech Res.2010, 2(4): p. 2113-2117
- Se J, Seo J, In-Yup. Polymers. 2022, 14: p. 4733.
- Anthony B, Fabrice G, Eliane E. Polymers. 2021, 13: p. 3546.
- Marcelo A, Gabriel G, Hooman A, et al., Materials Today: Proceedings. 2016, p. S233 – S239.
- JuntaoL, EbruG, Imad B. J Composite Materials. 2019,19: p.2761-2773.
- Hashem, Muhammad Abbas Abdel-Muttalib. Master Thesis, College of Science, University of Basra, 1989.
- Ullmann’s Encyclopedia of Industrial Chemistry: Sixth, Completely Revised Edition. J American Chemical Society. 2003, 125(35): p. 10768-10768.
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref