Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 3
Synthesis and Anti-Convlsant Evaluation of Some Novel 4-Phenyl-6-Methyl-5-[(2'-Substituted-Phenyl) 1,3,4-Oxadiazole)]-3,4-Dihydropyrimidin-2(1h)-One Derivatives
Bharat SH*
Department of Pharmaceutical Chemistry, S.V.N.H.T'S College of B. Pharmacy, Shrishivajinagar (Rahuri factory), Tal-Rahuri, Dist-Ahmednagar-413706, India
- *Corresponding Author:
- Bharat SH
Department of Pharmaceutical Chemistry
S.V.N.H.T'S College of B. Pharmacy
Shrishivajinagar (Rahuri factory), Tal-Rahuri
Dist-Ahmednagar-413706, India
Abstract
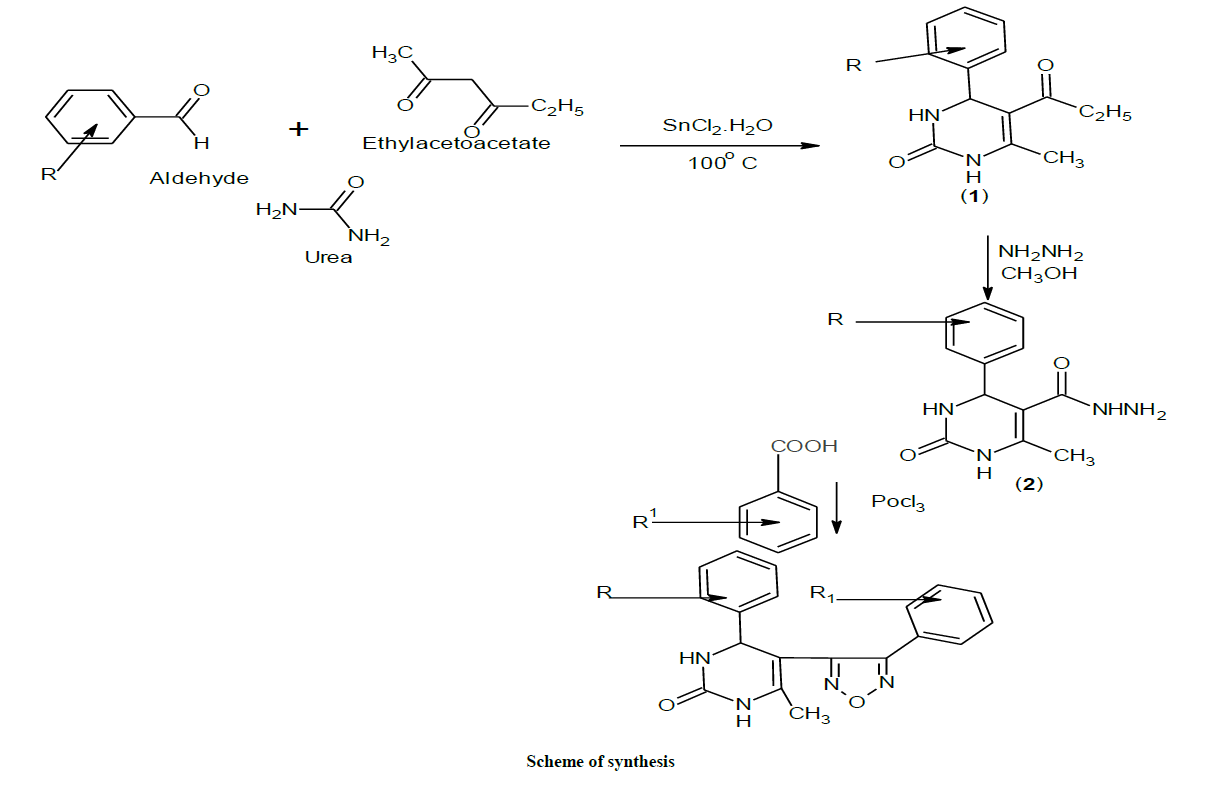
The reaction of substituted aromatic aldehydes with ethylacetoacetate in presence of urea yielded 5-Ethoxy carbonyl-6- methyl-4-phenyl-3, 4- dihydropyrimidin-2 (1H)-one.(1), which on treatment with hydrazine hydrate produced 4-phenyl-6-methyl-2-pyrimidinone 5-carbohydrazide.(2), cyclization with substituted benzoic acids in presence of phosphorous oxychloride produced 4-phenyl-6-methyl-5-[(2'- substituted-phenyl) 1,3,4- oxadiazole)] - 3, 4-dihydropyrimidin-2(1H)-one. 3(a-i). Purity was checked by Thin Layer Chromatography (TLC) and the chemical structures of synthesized compounds were elucidated by their Infra-Red (IR), Proton nuclear magnetic resonance (1H-NMR) analysis data. The synthesized compounds were screened for Anti-convulsant activity.
Keywords
3,4-dihydropyrimidin-2 (1H)-one, Oxadiazole, Anti-convulsant activity
Introduction
The presence of pyrimidine ring in cytosine, thymine and urea, which are the essential binding blocks of nucleic acids, DNA and RNA is one possible reason for their activity [1]. Additionally, the structurally related marine alkaloids batzelladine A and B were shown to be the first low molecular weight natural products to inhibit the binding of HIV gp-120 to CD-4 cells, so disclosing new vistas towards the development of AIDS therapy [2]. These ring systems are often incorporated into drugs designed as anticancer, antiviral, antihypertensive, analgesic, antipyretic, anti-inflammatory, antipsoriasis agents. Some of them are active on the blood circulatory system and can stimulate the skin preparative regeneration and increase the efficacy of antibiotic therapy [3].
Materials and Methods
Melting points of all the synthesized compounds were determined by open capillary tubes using paraffin bath and are uncorrected. Proton nuclear magnetic resonance (1H-NMR) spectra were recorded on a Varian-NMR-mercury 300 MHz spectrophotometer in CDCl3 using TMS as an internal standard.
Experimental
Synthesis of 5-Ethoxy carbonyl-6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-one:
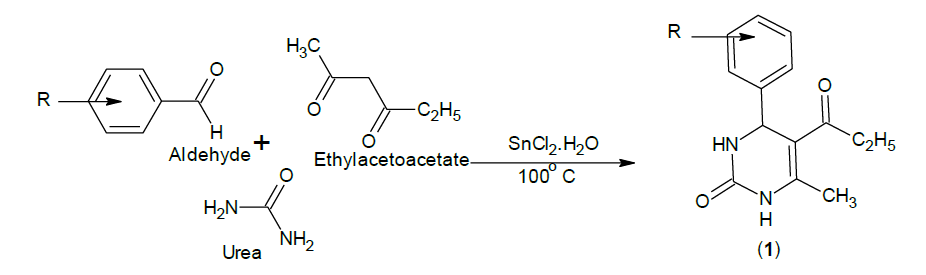
1a: IR (KBr): 3349 (N-H), 3044 (aromatic CH stretching), 1720 (C=O ester), 1468 (unsaturated aromatic C-N) and 2875 cm-1 (CH3).
Synthesis of 4-phenyl-6-methyl-2-pyrimidinone 5-carbohydrazide:
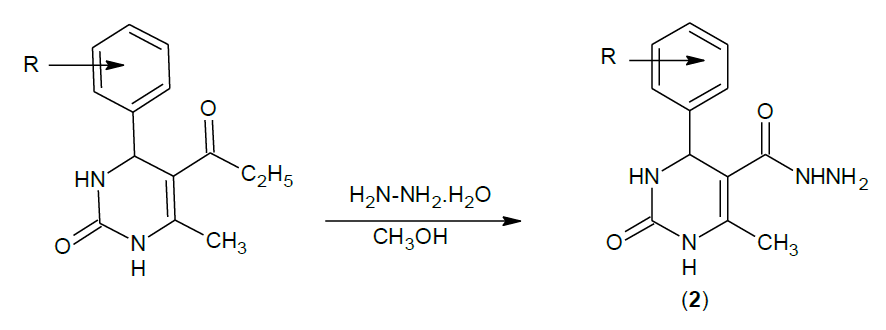
2a: IR (KBr): 3354 (N-H), 3134 (unsaturated aromatic C=C), 1600 (C=O amide), 2855 (CH3 Asymmetric), 1455 (C-N).
Synthesis of 4-phenyl-6-methyl-5-[(2'-substituted-phenyl)1,3,4-oxadiazole)]-3,4-dihydropyrimidin 2(1H)-one. 3(a-i):
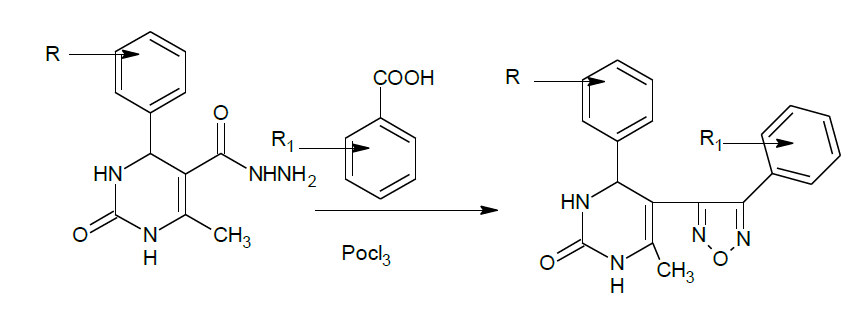
3a: IR (KBr): 3345 (N-H), 3160 (unsaturated aromatic C=C), 1647 (C=N), 1133 cm-1 (C-O-C), and 740 cm-1 (C-Cl); 1H-NMR: δ=5.4-5.54 (m, 8H, Pyrimidine ring); 7.26-7.31(m, 5H, Chloro phenyl), and 2.01 (d, 6H, methyl).
3b: IR (KBr): 3422 (N-H), 3345 (unsaturated aromatic C=C), 1149 (C-O-C).
3c: IR (KBr): 3348 (N-H), 3167 cm-1 (unsaturated aromatic C=C), 1600 (N=C stretching), 1592 (C=N), and 1109 (C-O-C).
3d: IR (KBr): 3380 (N-H), 3160 (unsaturated aromatic C=C), 1680 (C=N), 1197 (C-O-C), 850 (C-Cl).
3e: IR (KBr): 3343 (N-H stretching), 3215 (unsaturated aromatic C=C), 1181 (C-O-C), 1599 (C-N), 774 (C-Cl).
3f: IR (KBr): 3343 cm-1 (N-H), 2989 (unsaturated aromatic C=C), 1545 (C-O-C), 695 (C-Cl).
3g: IR (KBr): 3243 cm-1 (N-H), 3193 (unsaturated aromatic C=C), 1149 (C-O-C), 730 (C-Cl), 1684 (C=N).
3h: IR (KBr): 3167 (unsaturated aromatic C=C), 1232 (C-O-C), 1545 (NO2), 1690 (C=N).
3i: IR (KBr): 3275 (N-H), 3089 (unsaturated aromatic C=C), 1109 (C-O-C), 1535 (NO2), 1700 (C=N).
Anticonvulsant activity
Anticonvulsant is an agent that blocks experimentally produced seizures in laboratory animals and an antiepileptic drug is a drug used medically to control the epilepsies, not all of which are convulsive in humans [4-10]. Male albino mice of weight range 20-25 g were selected and grouped into a group of 6 animals. Before study food was withdrawn but animal had free access to water. Test compounds were administered orally at a dose of 20 mg/kg body weight suspended in sterile water for injection. Standard group received diazepam at a dose of 5 mg/kg body weight intraperitonally. After 30 min an electric shock of 50 ma, 60 Hz was delivered over a period of 0.2 sec by using corneal electrodes. The duration of tonic hind limb extension (Tonic convulsion) was noted. The same procedure was repeated after 90 min of test solution administration. The duration of hind limb extension was noted as protection against induced convulsion. The statistical analysis was done by using One way ANOVA followed by Dunnet test (Table 1) [11,12].
| Compound Code | R | R1 | Molecular formula | Molecular weight | Melting point | Yield (%) |
|---|---|---|---|---|---|---|
| 3a | H | 2-Cl | C18H15N4O2Cl | 354.5 | 128-120 | 70.25 |
| 3b | H | H | C18H16N4O2 | 320 | 120-122 | 74.33 |
| 3c | H | 4-NH2 | C18H17N5O2 | 335 | 228-230 | 82.85 |
| 3d | 4-Cl | 2-Cl | C18H14N4O2Cl2 | 389 | 180-182 | 78.71 |
| 3e | 4-Cl | H | C18H15N4O2Cl | 354.5 | 158-160 | 90.23 |
| 3f | 4-Cl | 4-NH2 | C18H16N5O2Cl | 369.5 | 212-214 | 90 |
| 3g | 3-NO2 | 2-Cl | C18H14N5O4Cl | 399.5 | 166-168 | 78.18 |
| 3h | 3-NO2 | H | C18H15N5O4 | 365 | 156-158 | 70.83 |
| 3i | 3-NO2 | 4-NH2 | C18H16N6O4 | 380 | 122-124 | 65.82 |
Table 1: Physical data of synthesized 3,4-dihydropyrimidin-2(1H)-one compounds
Results and Discussions
The experimental protocol was approved by the Institutional Animal Ethical Committee (IAEC) and conducted according to the guidelines for the use and care of experimental animals. All the synthesized compounds were subjected to anticonvulsant activity at a 20 mg/kg, b.w. dose using electroshock method in mice. Many of the synthesized compounds showed significant anticonvulsant activity as compared to diazepam as standard. The results of the anticonvulsant activity are presented below (Table 2 and Figure 1).
| Group | Dose | Duration at 30 min. in seconds. | Duration at 90 min. in seconds |
|---|---|---|---|
| Control | 0.1 ml/10 g | 69 ± 2.082 | 78 ± 3.021 |
| Standard | 5 mg/kg, b.w. | 22.66 ± 2.182** | 18 ± 0.157** |
| 3a | 20 mg/kg, b.w. | 28.33 ± 1.002** | 21 ± 1.045** |
| 3b | 20 mg/kg, b.w. | 55.63 ± 1.028* | 70 ± 2.096NS |
| 3c | 20 mg/kg, b.w. | 41 ± 1.212** | 48 ± 1.13** |
| 3d | 20 mg/kg, b.w. | 24 ± 1.053** | 23 ± 1.6** |
| 3e | 20 mg/kg, b.w. | 65 ± 4.159** | 49 ± 2.16** |
| 3f | 20 mg/kg, b.w. | 47 ± 3.018** | 51 ± 2.12** |
| 3g | 20 mg/kg, b.w. | 29 ± 3.018** | 27 ± 2.33* |
| 3h | 20 mg/kg, b.w. | 38 ± 1.528** | 42 ± 2.6** |
| 3i | 20 mg/kg, b.w. | 50 ± 3.528** | 54 ± 3.78** |
Results are in comparison to control; **p ≤ 0.01, * p ≤ 0.05, NS Non significant, n=6; Data expressed as Mean ± SEM; Data was analyzed by one-way ANNOVA followed by Dunnett’s test
Table 2: Duration of hind limb extensor at 30 and 90 min of synthesized substituted 3,4-dihydropyrimidin-2(1H)-one derivatives
All the synthesized compounds exhibited significant anticonvulsant activity against maximal electroshock induced convulsions in mice at the 30 and 90 min time interval. The hind limb extension time for compounds 3a, 3d and 3g were 28, 24 and 29 sec respectively, and all compounds possessing o-chloro substituents.
All the compounds have also shown significant activity at the 90 min time interval. Compound 3a, 3d and 3g have showed the hind limb extensor time 21, 23 and 27 seconds at 90 min time interval respectively, and all compound possessing o-chloro substituents. The compounds 3a, 3d and 3g have shown anticonvulsant activity at both the time intervals. Amongst all the synthesized derivatives, compound 3d (at 20 mg/kg, b.w. dose) was found to be most active (at 5 mg/kg, b.w. dose).
Conclusion
The 3 4-dihydropyrimidin-2(1H)-one was synthesized by solvent free Biginelli reaction using stannous chloride as catalyst. The synthesized pyrimidinone was reacted with hydrazine hydrate to yield carbohydrazide which on further reaction with various substituted benzoic acid yielded the final 4-phenyl-6-methyl-5-[(2'- substituted-phenyl) 1,3,4-oxadiazole)] - 3, 4-dihydropyrimidin-2(1H)-one. All the synthesized compounds were screened for anticonvulsant. The anticonvulsant activity was carried out using maximal electroshock method and measuring the duration of hind limb extension as protection criteria against induced convulsions. The ability of the compounds to prevent convulsion was screened at 30 minutes and 90 minutes time interval after oral administration of compounds. All the synthesized compounds exhibited significant anticonvulsant activity against maximal electroshock induced convulsions in mice. The compounds 3a, 3d and 3g possessing o-chloro, substituents showed very good activity at the 30 minutes time interval, while remaining compounds were found to possess moderate activity as compared to standard.
• The compounds 3a, 3d, and 3g having o-chloro substitutions have shown good activity at 90 minutes time interval.
• The compounds 3a, 3d, and 3g have shown anticonvulsant activity at both the time interval. Amongst all the synthesized derivatives, compound 3d (at 20 mg/kg, b.w. dose) was found to be most active.
References
- M. Amir, S.S. Javed, Harish Kumar, Indian J. Pharmaceutical Sci., 2007, 337.
- A. Mithun, S.H. Bantwal, S.K. Nalilu, Eur. J. Medicinal Chem., 2007, 42, 380.
- C.R. Kodihalli, M.V. Hosadu, P.V. Vijayvithal, ARKIVOC, 2008, xi, 1.
- Asif Husain, F.J. Ahmed, J. Serbian Chem Society., 2008, 73, 789-791.
- R. Iqbal, K.M. Khan, J. Chem. Society Oark., 2006, 28, 165-168.
- B.S. Kittur, B.S. Sastry, S.R. Pattan, P.A. Pabara, 2009, 46(4), 287-290.
- M. Shahar Yar, A. Ahmed Siddiqui, M. Ashraf Ali, J. Chinese Chem. Society., 2009, 54-58.
- P.J. Das, A. Baruah, Indian J. Chem., 2008, 47, 1568-1571.
- H.G. Vogel, Drug Discovery and Evaluation Pharmacological Assays., 487.
- M.M. Foreman, T. Hanania, S.C. Stratton, K.S. Wilcox, H.S. White, J.P. Stables, M. Eller, Pharmacology, Biochemistry and Behavior., 2008, 89, 523.
- S.D. Ambawade, V.S. Kasture, S.B. Kasture, Indian J Pharma., 2002, 34,251.
- Chi Yuoh-fong, Wu Yuan-Liu, Scientia Sinica.s, no.5, Vol.VI, 853.



