Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 8
Synthesis and Anti-Inflammatory Activity of New Substituted 4,5- Dihydro-3-(Naphthalene-6-Yl)-5-Phenyl Pyrazole Carbothio Aminde Derivatives
Poonam K, Priya M, Anshuman S, Bhumika Y* and Sujeet KG
Department of Pharmaceutical Chemistry, Hygia Institute of Pharmaceutical Education and Research, Lucknow-226021, India
- *Corresponding Author:
- Bhumika Y
Department of Pharmaceutical Chemistry
Hygia Institute of Pharmaceutical Education and Research
Lucknow-226021, India
Abstract
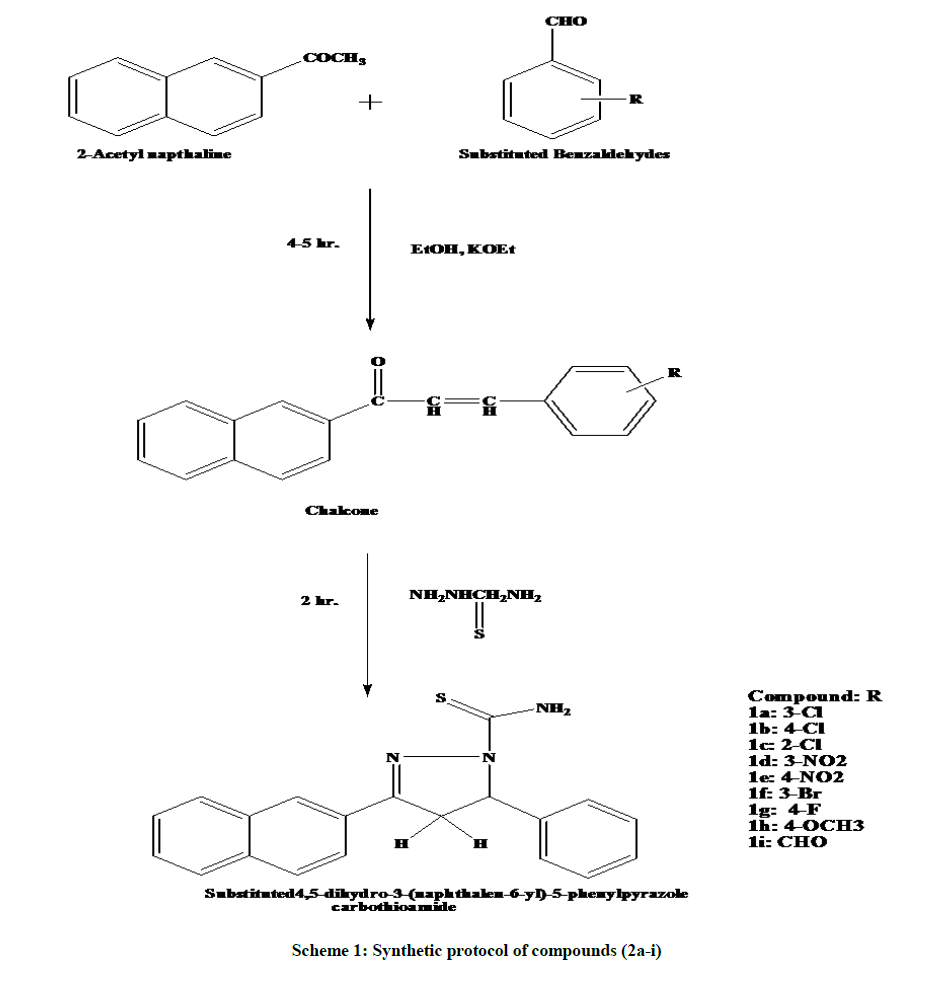
In the present study we have made an attempt to synthesize substituted 4,5-dihydro-3-(naphthalene-6-yl)-5-phenyl pyrazole carbothioamide derivatives (2a-i) and evaluate them for anti-inflammatory activity using carrageenan induced rat paw edema model. In the first step, 2-acetylnaphthalene (1) was allowed to react with substituted benzaldehyde (2) in equimolar amount with sodium hydroxide to form various substituted derivatives (1a-i). Further substituted chalcone derivatives on treatment with thiosemicarbazide in the presence of ethanol corresponding substituted 4,5-dihydro-3-(naphthalene-6-yl)-5-phenyl pyrazole carbothioamide (2a-i). The structure of the final analogues has been confirmed on the basis of elemental analysis, Fourier Transform Infrared (FTIR), Proton Nuclear Magnetic Resonance (1H-NMR). All the values of elemental analysis, FTIR, 1H-NMR were found to be prominent. Among all synthesized compounds for anti-inflammatory activity compounds 2d and 2h were found to be most potent in comparison with standard diclofenac.
Keywords
Pyrazole, Anti-inflammatory, Chalcone, Carrageenan induced rat paw edema model.
Introduction
The anti-inflammatory properties of nonsteroidal anti-inflammatory drugs (NSAIDs) have been attributed to their ability to inhibit the enzyme cyclooxygenase (COX) enzymes, which catalyzes the formation of arachidonic acid (AA) to prostaglandins H2 (PGH2) [1-4]. Many of (NSAIDs) have a wide clinical use in the treatment of acute or chronic inflammation [5,6]. There are two isoforms of cyclooxygenase (COX); COX-1 and COX-2 [7]. These isoforms are poorly distinguishable by most of the classical NSAIDs. They actually inhibit COX-1 extensively; COX-1 has housekeeping functions, including low-level production of gastro protective PGs, besides COX-2, leading to gastrointestinal injury suppression of thromboxane A2 (TXA2) formation and platelet aggregation. The combination of these interactions is probably the reason for gastrointestinal bleeding as the most serious complication of these drugs [8]. To prevent or decrease these aspect effects, a current strategy consists of planning selective cyclooxygenase-2 inhibitors with associate improved internal organ safety profile. The improved safety profile of cyclooxygenase-2 inhibitors could permit the utilization of those new agents in the field of medicinal chemistry [9]. Pyrazole derivatives were found to exhibit anti-inflammatory [10-12], analgesic [13], anticancer [14,15], antiviral [16,17], antiepileptic [18] and antimicrobial activities [19]. Herein we have a tendency to synthesis of new pyrazole derivatives and screen them for anti-inflammatory activity.
Materials and Methods
Experimental
All chemicals were purchased from common commercial suppliers and used without further purification. The melting points were determined in open capillary tubes and were uncorrected. The purity of all the synthesized compounds were checked by TLC on percolated silica gel-G aluminum sheets (Type 60 GF 254, Merck) and the spots were detected by exposure to iodine vapors. The Fourier Transform Infrared (FT-IR) spectra were recorded on 470-Shimadzu infrared spectrophotometer using the KBr disc prepared by pressed pellet technique and vmax is expressed in cm-1. Nuclear Magnetic Resonance (NMR) spectra were measured in Dimethyl sulfoxide (DMSO-d6) as solvent at 300 MHz (1H-NMR). Elemental analyses were carried elemental vario EL III carlo Erba 1108, and the values were within (± 0.4%) of the theoretical values. All the solvent were distilled and dried with usual desiccant.
General procedure for synthesis of substituted chalcone (1a-i)
A mixture of aromatic aldehyde (0.01 mol, 2.8 g) and 2-acetylnaphthaline (0.01 mol, 3.4 g) was dissolved in ethanol (30 ml). Sodium hydroxide (5 g) was added to alkaline the reaction mixture. The reaction mixture was stirred for 4-5 h. at room temperature. Left the reaction mixture in a refrigerator overnight and the reaction mixture was washed with water and recrystallized from ethanol to give corresponding chalcone derivatives (1a-i).
General procedure for synthesis of substituted 4,5-dihydro-3-(naphthalene-6-yl)-5-phenyl pyrazole carbothioamide derivatives (2a-i)
A mixture of thiosemicarbazide (0.012 mol, 2.18 g) with chalcone (0.02 mole, 2 g) and sodium hydroxide (0.025 mol. in 5 ml water) in ethanol 100 ml was heated under reflux for 2 h. The final product was poured into an ice water and the crude product was filtered, washed and recrystallised from ethanol (2a-i) [20].
5-(3-chlorophenyl)-4,5-dihydro-3-(naphthalene-6-yl) pyrazole-1-carbothioamide (2a)
Yellow crystal; M. p. 175°C-176°C; Yield 20.62%; FTIR (KBr: v cm-1): 667 (Ar-C-Cl, str.); 756 (Al- N-N, str.), 825(C=S str.), 1216 (Al-C-N, str.), 1525 (C-N str.), 1625 (C=N, str.), 3261 (Ar- C-H, str.); 1H-NMR (300 MHz, DMSO-d6): δ ppm: 2.01-(s, 2H, NH2), 1.22 (s, 1H, CH), 1.23-1.25 (d, 2H, CH2); 6.72-6.98 (m, 7H, Ar-H), 7.62-7.68 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H16ClN3S: C, 65.65; H, 4.41; N; 11.48; Found: C, 65.61; H, 4.37; N, 11.44%.
5-(4-chlorophenyl)-4,5-dihydro-3-(naphthalene-6-yl)pyrazole-1-carbothioamide (2b)
Yellow crystal; M. p. 173°C-175°C; Yield 66.87%; FTIR (KBr: v cm-1): 600 (Ar, C-Cl str.), 756 (Al, N-N str.), 829 (Al, C=S str.), 1230 (Al, C-N str.), 1676 (Al, C=N str.), 3025 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ ppm: 1.31 (s, 1H, CH), 1.32-1.35 (d, 2H, CH2), 2.06 (s, 2H, NH2), 6.97- 6.99 (m, 7H, Ar-H), 7.52-7.62 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H16ClN3S: C, 66.65; H, 4.41; N, 11.42; Found: C, 66.63; H, 4.42; N, 11.46%.
5-(2-chlorophenyl)-4, 5-dihydro-3-(naphthalene-6-yl) pyrazole-1-carbothioamide (2c)
Yellow crystal; M. p. 173°C-175°C; Yield 57%; FTIR (KBr, v cm-1): 600.20 (Ar, C-Cl str.), 760.14 (Al, N-N str.), 830 (Al, C=S str.), 1218.24 (Al, C-N str.), 1525.60 (Al, C-N str.), 1600.12 (Al, N-N str.), 3124.12; 1H-NMR (300 MHz, DMSO-d6): δ ppm: 1.10 (s, 1H, CH), 1.20-1.22 (d, 2H, CH2), 2.05 (s, 2H, NH2), 6.98-7.55 (m, 7H, Ar-H), 7.63-7.69 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H16ClN3S: C, 65.65; H, 4.41; N, 11.48; Found: C, 65.64; H, 4.44; N, 11.39%.
4,5-dihydro-3-(naphthalene-6-yl)-5-(3nitrophenyl) pyrazole-1-carbothioamide (2d)
White crystal; M. p. 172°C-173°C; Yield 50%; FTIR (KBr, v cm-1): 765 (Al, N-N str.), 831 (C=S str.), 1228 (Al, C-N str.), 1326 (N-O sym str.), 1530 (N-O asym str.), 1628 (Al, C=N str.), 3125 (Ar-C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ ppm: 1.02 (s, 1H, CH), 1.35-2.02 (d, 2H, CH2), 2.05 (s, 2H, NH2), 7.15-7.35 (m, 7H, Ar-H), 7.75-7.56 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H16N4O2S: C, 63.81; H, 4.28; N, 14.88; Found: C, 63.82; H, 4.22; N, 14.84%.
4,5-dihydro-3-(naphthalene-6-yl)-5-(4nitrophenyl) pyrazole -1-carbothioamide (2e)
Brown crystal; M. p.178°C-180°C; Yield 47.20%; FTIR (KBr, v cm-1): 760 (Al, N-N str.), 820 (Al, C=S str.), 1211 (Al, C-N str.), 1322 (N-O sym str), 1522 (N-O asym str.), 1526 (Al, C-N str.), 1632 (Al, C=N str.), 3123 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=1.25 (s, 1H, CH), 2.04 (s, 2H, NH2), 2.41-2.45 (d, 2H, CH2), 6.22-6.32 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H16N4O2S: C, 63.81; H, 4.28; N, 14.88; Found: C, 63.78; H, 4.25; N, 14.81%.
5-(3-bromophenyl)-4, 5-dihydro-3-(naphthalene-6-yl)pyrazole-1-carbothioamide (2f)
Brown crystal; M. p. 173°C-175°C; Yield 40.49%; FTIR (KBr, v cm-1): 550 (Ar, C-Br str.), 759 (Al, N-N str.), 829 (Al, C=S str.), 1218 (Al, C-N str.), 1523 (Al, C-N str.), 1638 (Al, C=N str.), 3065 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=1.56-2.01 (d, 2H, CH2), 1.75 (s, 1H, CH), 2.04 (s, 2H, NH2), 6.99-7.26 (m, 7H, Ar-H), 7.26-7.52 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal calcd for C20H16BrN3S: C, 58.54; H, 3.93; N, 10.24; Found: C, 58.51; H, 3.94; N, 10.22%.
5-(4-fluorophenyl))-4, 5- dihydro-3-(naphthalene-6-yl)pyrazole-1-carbothioamide (2g)
White crystal; M. p. 172°C-174°C; Yield 37.71%; FTIR (KBr, v cm-1): 776 (Al, N-N str.), 836 (Al, C=S str.), 1200 (Ar, C-F str.), 1232 (Al, C-N str.), 1503 (Al, C-N str.), 1635 (Al, C=N str.), 3078 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=1.01-2.5 (d, 2H, CH2), 1.05 (s, 1H, CH), 2.06 (s, 2H, NH2), 7.25-7.35 (m, 7H, Ar-H), 7.75-7.91 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal calcd for C20H16FN3S: C, 68.78; H, 4.62; N, 12.03; Found: C, 68.76; H, 4.66; N, 12.00%.
4,5-dihyro-5-(4-methoxyphenyl)-3(naphthalene-6-yl)pyrazole-1-carbothioamide (2h)
Light Brown crystal; M. p. 172°C-174°C; Yield 37.71%; FTIR (KBr, v cm-1): 770 (N-N str.), 845 (Al, C=S str.), 929.12 (Ar, C-O str.), 1028 (C-O-C sym. str.), 1230 (C-O-C asym. str.), 1250 (Al, C-N str.), 1645 (Al, C=N str.), 3145.16 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=1.19 (s, 1H, CH), 1.48-1.53 (d, 2H, CH2), 2.05 (s, 2H, NH2), 3.48 ( s, 3H, OCH3), 6.46-6.53(m, 7H, Ar-H), 7.55-7.60 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+, Anal. calcd for C21H19N3OS: C, 69.78; H, 5.30; N, 11.63; Found: C, 69.81; H, 5.32; N, 11.61%.
4, 5-diyhro-3-(naphthalene-6-yl)-5-phenylpyrazole-1-carbothioamide (2i)
Yellow crystal; M. p. 173°C-175°C; Yield 78.73%; FTIR (KBr, v cm-1): 769 (Al, N-N str.), 869 (Al, C=S str.), 1200 (Al, C-N str.), 1503 (Al, C-N str.), 1633 (Al, C=N str.), 3175 (Ar, C-H str.); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=1.43 (s, 1H, CH), 2.05 (s, 2H, NH2), 2.73-2.75 (d, 2H, CH2), 6.55-6.96 (m, 7H, Ar-H), 7.64-7.67 (m, 4H, Ar-H); EIMS (m/z): 327.14 [M]+, 328.18 [M+1]+; Anal. calcd for C20H17N3S: C, 72.48; H, 5.17; N, 12.68; Found: C, 72.45; H, 5.16; N, 12.64%.
Anti-inflammatory activity
Anti-inflammatory activity was performed by carrageenan induced inflammation in rat paw edema model. This project has been approved by the institutional animal ethical committee at Hygia Institute of Pharmaceutical Education and Research, (Ref NO. HIPER/ IAEC/02/17/04). Adult albino rats (wister strain) of either sex weighing 150-200 g were used. The animals were divided into three group (control, standard and test) and each group comprising of three rats. Animal were deprived of food for 12 h prior to experiment and only water is given. First group was used as a control and received 1 ml of 1% w/v CMC suspension in saline, the second group received CMC suspension of Diclofenac sodium (20 mg/kg) orally. Orally and the third group received 1ml of 1% w/v sodium CMC suspension in saline, the second group CMC suspension of test compounds at a dose of 20 mg/kg orally. One hour after the administration of the compounds, carrageenan suspension (0.1ml of 1% w/v suspension in 0.9% saline solution) was volume was measured using digital vernier calliper (initial paw thickness, vc). Thereafter, the paw volume was measured after 0 h, 1 h, 3 h, and 4 h after carrageenan administration. The difference between initial and subsequent readings gave the change in edema volume for the corresponding time [21].
Results and Discussion
The desired pyrazole derivatives were prepared by multistep reaction summarized in Scheme 1. In the first step, 2-acetylnaphthalene and substituted benzaldehyde in prensence of ethanol and sodium hydroxide afforded the corresponding chalcone derivatives (1a-i). Chalcone derivatives were allowed to react with thiosemicarbazide in ethanol and sodium hydroxide to gives corresponding pyrazole derivatives (2a-i). Infrared (IR) spectra of all final pyrazole derivatives (2a-i) showed an intense peak in the region 1676-1600 cm-1 due to the C=N stretching vibration which indicate the presence of C=N in pyrazole ring. A strong, characteristic band in the region 1230-1200 cm-1 due to the C-N stretching vibration, peak appeared at 869-820 cm-1 due to C=S stretching. Band for aromatic C-H stretching vibrations was observed at 3261-3025 cm-1. Peak for aromatic C-H are generally appeared at longer wavelength than the aliphatic C-H. It is due to the higher stretching of pi electrons present in aromatic ring.
For compound 2h C-O-C asymmetric and symmetric stretching vibrations was appeared between 1230 cm-1 and 1028 cm-1 respectively. Vibration characteristic of C-O-C system would not be expected to differ greatly from the C-C-C system, however vibrations involving oxygen atom results in greater dipole moment changes than those involving carbon atoms, more intense IR bands are observed for ether. For compound 2d and 2e N-O asymmetric and symmetric stretching vibrations was appeared between 1530-1522 cm-1 and 1326-1322 cm-1 respectively. These two compounds show two intense bands due to asymmetric and symmetric stretching vibrations of the highly polor nitrogen- oxygen bonds. A stretching vibration of C-Br was appeared at 550 cm-1 for compound 2c and stretching vibration of C-F was appeared at 1200 for compound 2 g. An intense peak was observed in the region of 3261-3322 cm-1 due to N-H stretching vibration which confirms the conversion of substrates into the expected products.
The 1H-NMR spectra of the compound 2h a characteristic singlet of three protons for methoxy group was appeared at δ 3.48 ppm. Multiplet for seven naphthalene protons was appeared between δ 6.22-7.69 ppm. In the 1H-NMR spectra of the compounds (2a-i) showed, a singlet of two proton for amino group between δ 2.06-2.01 ppm which was found to be D2O exchangeable. Multiplet for four aromatic protons was appeared between δ 6.22-7.69 ppm. In 1H-NMR spectra of the compounds, a doublet of two methylene proton was appeared between δ 1.35-2.75 ppm. All final derivatives (2a-i) show multiplet peak in prominent region due to aromatic protons. In the mass spectra of all compounds (2a-i), [M+1]+ peak were observed. All compounds gave satisfactory elemental analysis.
Pharmacological evaluation
Anti-inflammatory activity
The structure activity relationship revealed that compounds having different substitution on pyrazole moiety exhibited significant anti-inflammatory activity. Compounds 2a, 2b, 2c, 2d, 2e, 2f, 2g, 2h, and 2i at 20 mg/kg exhibited singnificant anti-inflammatory activity at all-time intervals as compared to control group. The entire compounds (2a-i) exhibited varing degree of anti-inflammatory activity (21-46%). Compound 2d in which nitro group at 3-Position has shown maximal activity (46%) whereas when compound 2a benzaldehyde chloro group has shown minimal activity (21%) as shown in Table 1.
| Compounds code | 0 h | % Inhibition | 1 h | % Inhibition | 2 h | % Inhibition | 4 h | % Inhibition |
|---|---|---|---|---|---|---|---|---|
| 2a | 1.54 ± 0.01 | 1% | 1.39 ± 0.09 | 9% | 1.14 ± 0.02 | 20% | 1.12 ± 0.21 | 22% |
| 2b | 1.52 ± 0.03 | 3% | 1.38 ± 0.01 | 10% | 1.15 ± 0.19 | 19% | 1.08 ± 0.07 | 21% |
| 2c | 1.53 ± 0.02 | 2% | 1.40 ± 0.08 | 8% | 1.18 ± 0.12 | 16% | 1.05 ± 0.05 | 27% |
| 2d | 1.46 ± 0.09*** | 9% | 1.25 ± 0.13*** | 23% | 0.98 ± 0.05*** | 36% | 0.86 ± 0.06*** | 46% |
| 2e | 1.49 ± 0.06 | 6% | 1.32 ± 0.06 | 16% | 1.10 ± 0.07 | 24% | 1.06 ± 0.02 | 26% |
| 2f | 1.53 ± 0.02 | 2% | 1.36 ± 0.04 | 12% | 1.15 ± 0.19 | 19% | 1.02 ± 0.3 | 30% |
| 2g | 1.51 ± 0.04 | 4% | 1.38 ± 0.06 | 10% | 1.14 ± 0.20 | 20% | 1.03 ± 0.24 | 29% |
| 2h | 1.48 ± 0.07** | 7% | 1.26 ± 0.02** | 22% | 1.07 ± 0.07** | 27% | 0.99 ± 0.13** | 33% |
| 2i | 1.52 ± 0.03 | 3% | 1.37 ± 0.11 | 11% | 1.16 ± 0.18 | 18% | 1.01 ± 0.31 | 31% |
| Control | 1.55 ± 0.03 | 1.48 ± 0.04 | 1.34 ± 0.04 | 1.32 ± 0.21 | ||||
| Standard Dicloenac | 1.46 ± 0.09 | 9% | 1.18 ± 0.3 | 30% | 0.92 ± 0.41 | 41% | 0.79 ± 0.53 | 53% |
Table 1: Anti-inflammatory activity of titled compounds 20 mg/kg (Dose)
Data of % inhibition shown that amongst all the tested compound having nitro and methoxy groups as substitution showed highest activity. % Inhibition in anti-inflammation test compounds (2a,2b) shown mild to-moderate anti- inflammatory activity.
Conclusion
The structures of all the new compounds have been established on the basis of their analytical and spectral data. All of the synthesized compounds were also evaluated for their anti-inflammatory activity using carrageenan induced rat paw edema model and Diclofenac as the reference drug.
Acknowledgement
Authors would like to thank management of Hygia Institute of Pharmaceutical Education and Research, Lucknow for providing research facilities. CDRI, Lucknow is acknowledged for providing the spectral data of the synthesized compounds.
References
- S.H. Ferreira, S. Moncada, J.R. Vane, Nat. New Biol.,1971, 231, 237.
- G. Dannhardt, W. Kiefer, Eur. J. Med. Chem.,2001, 36, 109.
- J.S carter Expert opinion on therapeutic patents, 2000, 10, 1011.
- J.J. Talley, Prog. Med. Chem., 1999, 36, 201.
- V.A. Skoutakis, C.A. Carter, T.R. Mickle, V.H. Smith, C.R. Arkin, Drug Intell. Clin. Pharm., 1988, 22, 850.
- J.G. Lombardino, Nonsteroidal antiinflammatory drugs, Wiley Interscience, New York, 1985, 251-431.
- D.A. Kujubu, B.S. Fletcher, B.C. Varnum, R.W. Lim, H.R.Herschman, J. Biol. Chem., 1991, 266, 12866.
- C.C. Chan, S. Boyce, C. Brideau, S. Charleson, W. Cromlish, J. Pharmacol. Exp. Ther., 1999, 290, 551.
- M.V. Patel, R. Bell, S. Majest, R. Henry, T. Kolasa, J. Org. Chem., 2004, 69, 7058.
- R. Tabassum, N. Mohammad, P. Kumari, R. Bharatiya, N. Srivatava, B. Yogi, A. Sinha, S. Gupta, Indian J. Heterocy. Ch., 2017, 27, 1, 107.
- S. Gupta, A. Mishra. Adv. Biores., 2016, 7, 5, 27.
- S. Gupta, A. Mishra. Indian J. Heterocy.Ch., 2016, 25, 3, 263.
- A. kumar, S. Sharma, K. Bajaj, D. Bansal, D. Sharma, Indian J. chem. Sect. B, 2003, 42, 1979.
- K.L. Khanduja, S.C. Dogra, S. Kaushal, R.R. Sharma, Biochem. Pharmacol., 1984, 33, 449.
- M. Mahmoud, R. Abdel-Kader, M. Hassanein, S. Saleh, S. Botros, Eur. J. Pharmacol., 2007, 569, 222.
- A. Evstropov, V. Yavorovskaya, E. Vorob'ev, Z. Khudonogova, L. Gritsenko, Pharm. Chem. J., 1992, 26, 426-430.
- V. Michon, C.H. Penhoat, F. Tombret, J. Gillardin, F. Lepage, Eur. J. Med. Chem., 1995, 30, 147.
- E. Ispir, S. Toroglu, A. Kayraldiz, Transition Met. Chem., 2008, 33, 953.
- R.N. Comber, R.J. Gray, J.A. Secrist, Carbohydr. Res., 1991, 216, 441.
- O.Zuhal, A.H. zdemir, B.B. Kandilci, Bu¨lentGu¨mu¨sxel, Unsal C¸ A. alısx a, A. Bilgin, Eur. J. Med. Chem.,2007, 42, 373.
- S. Gupta, A. Mishra. Med. Chem.,2016, 15.