Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 3
Synthesis and Characterization of Maleimide-Epoxy Resins and Composites Formation
Jyoti Chaudhary1*, Swati Purohit1, Supriya Dadhich1 and Radha Chaudhary22Department of Chemistry, M.L. Sukhadia University, Udaipur, India
Jyoti Chaudhary, Department of Polymer Science, M.L. Sukhadia University, Udaipur, India, Tel: 9828626594, Email: jyotichaudhary13@gmail.com
Abstract
Maleimide modified epoxy compound was prepared by reacting copolymer of Acrylic acid (AA) of N-[(4-acetyl)phenyl]maleimide (APMI) maleimide APMI-co-AA with epichlorohydrin in presence of Triethylamine (TEA). The resulting compound possessed both the oxirane ring and maleimide group. Synthesized epoxy maleimide resins characterized by FT-IR, 1H-NMR spectroscopy. Thermal stability of uncured resins was evaluated by TGA and DSC analysis. Glass fiber reinforced composites was also prepared and their chemical and mechanical properties were checked out. Resultant cured sample have good thermal stability, chemical resistance (acid/alkali/solvent) and water absorption resistance properties.
Keywords
Epoxy-maleimide, Epichlorohydrin, TGA, Mechanical properties, Glass fiber reinforced composites
Introduction
Maleimide epoxy resins design and synthesis is continuous growing interest area for their application in research and industry such as thermal and thermo-oxidative resistance, chemical resistance and mechanical strength are provided by the resins and maleimide possess unique properties of chemicals and electrical resistance, good adhesion and cohesion to different kind of materials and heat resistance. Maleimide are prime candidates as matrix resins for fiber reinforced composites. This is because of their stability at elevated temperature and hot/wet environment [1]. Epoxy resins have various applications in coating, composites and as structural adhesives. Epoxy resins modified with maleimide due to similar curing condition and processing properties and modified epoxy maleimide show appropriate properties between epoxy and maleimide [2]. Maleimide based epoxy resins have been proposed as manufacturing materials in the automotive industry [3] and aerospace industries in such applications as hypersonic aircraft, reusable space planes, and missiles [4]. However, such composites are highly susceptible to environmental conditions, primarily due to the degradation of epoxy matrix [5].
In the present work first maleimide was synthesized and maleimide epoxy resins were synthesized through reaction between hydroxyl group of maleimide and oxirane ring of epichlorohydrin. We studied the synthesis, characterization, composites formation and their chemical and mechanical properties.
Materials and Method
All chemicals were used as received. 4-Amino acetophenone, Maleic anhydride, Acrylic acid (AA), Epichlorohydrin, Triethylamine (TEA) were perchesed from loba chamicals Mumbai. DDM (Sigma Aldrich), E type glass woven fabric (PI compatible) of 0.25 mm thick (Unnati Chemicals, India.
Copolymer APMI-co-AA was synthesized according to reported method [5]. APMI-co-AA (5.10 g, 0.02 mole) and THF (20 mL) were placed in a 250 mL two-neck round bottom flask equipped with a reflux condenser. This was heated to reflux until the solution became homogenous. Epichlorohydrin (1.85 mL, 0.02 mole) and TEA (0.55 mL, 0.04 mole) were added.
Upon addition of these reagents, the solutions color was changed from yellow to light orange. After 6 h, the solution became brown red than removed from heat. Upon cooling, the viscous, dark brown red solution was stirred with 20 mL 5% HCl solution (Scheme 1). Structure was confirmed by FT-IR and 1H-NMR.
Composite fabrication
The glass fiber reinforced composites were prepared by a mixture of maleimide epoxy resins and DDM, AIBN stirred for 5 mints. The suspension was then applied with a brush on a 150 × 150 mm glass cloth. The 10 [S1] and 15 [S2] dried prepreges prepared by this way were then stacked one on top of another and pressed between the steel plates coated with a Teflon film release sheet and compressed in a flat platen under pressure of about 70 psi. The prepreges stacks were cured by heating at 150ºC for 10 hrs in an air circulated oven. The composite so obtained was cooled to 50ºC before the pressure was released. The specimens were made by cutting the composites and machining them to final dimension.
Results and Discussions
Characterization
Fourier Transform Infrared (FTIR) spectra was recorded on a FTIR Perkin-Elmer spectrophotometer model RX-I. The sample was prepared in KBr pellets, and the spectrum was obtained in the range 250-4000 cm−1. Nuclear Magnetic Resonance (NMR) spectra of newly synthesized monomer, homopolymer and copolymer have been scanned on Bruker Avance II 400 MHz NMR Spectrometer.
TMS was use as a reference.
The IR spectra of the maleimide epoxy resin (S1) shows the characteristic bands, at 912 cm-1 due to oxirane ring stretching and 3042 cm-1 attributed to the C-H Str of aromatic CH=CH and at 1415, 1512, 1597, 1676 cm-1 attributed to the Stretching of C=C of aromatic CH=CH, at 2958 due to C-H str of CH2, C=O stretch of acetyl group at 1676 cm-1. The characteristic bands, at 1708 and 1745 attributed to the C=O sym. and asym. Stretching of imide ring, at 1382 cm-1 attributed to the C-N-C stretch of N–substituted maleimide.
Important chemical shift values of the maleimide epoxy resin (S1) at 7.0-8.2 ppm due to phenyl proton of ortho and meta to N of imide, at 2.2-2.3 ppm due to –COCH3. In addition to above all observed signal in the region 6.8-7.74 ppm due to aromatic ring was also found. –CH protons of oxirane ring attached to phenyl ring at 3.4-3.6, –CH2 protons of oxirane ring attached to phenyl ring 2.5- 2.7.
Molecular weight
The molecular weight of the synthesized maleimide epoxy resin was measure by Gel permeation chromatography (GPC) analysis. GPC is a reliable and fast technique to determine the polydispersity index (PDI) and molar mass averages of polymers. The number average and weight average molecular weights (Mn, Mw) and polydispersity index of epoxy resin were summarizes in Table 1.
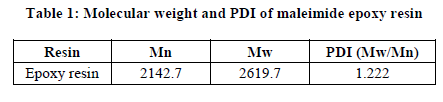
TGA analysis
Data derived from TG curves are furnished in Table 2 where the thermal stability in terms of temperature of 10% weight. Thermogram of maleimide epoxy resin is given in Figure 1, depicts one step decomposition reaction. Examination of TGA data reveals that the maleimide epoxy starts degradation around 100ºC and remains 47.43% around 700ºC. Activation energy Ea, order of reaction (n) were also calculated and summarized in Table 3.


Composite physical and chemical resistance test
The physical properties such as resin content, water absorption and boiling water absorption were evaluated. Their results are summarized in Table 4.
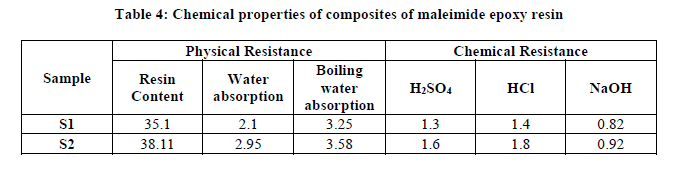
The chemical resistant test for the entire composite sample was performed according to ASTM D 543 method. The composite sample dimensions were 2 × 2 cm. The samples were immersed in 25% H2SO4, 25% HCl 25% NaOH for seven days at room temperature. After seven days the samples were taken out from the reagents and were analyzed for their percentage change in their weight. Their results are summarized in Table 4.
Mechanical test
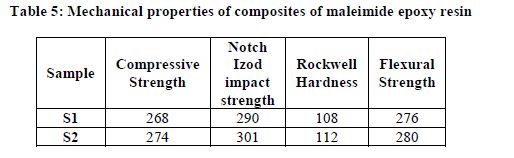
All the mechanical tests were performed using three specimens and their average results are summarized in Table 5. The compressive strength was measured on a Universal Instron testing machine model no. A-7437, at the room temperature according to ASTM D 695. The notched izod impact strength of the composites was measured on a Zwick Model no. 8900 Impact machine at the room temperature according to ASTM D256.
Rockwell Hardness was measured on a Rockwell hardness tester model no. RAS/Saro Engg. Pvt. Ltd. India according to ASTM D 785 at room temperature. The measurement of Flexural strength was carried out with Universal Instron Testing Machine model no. A-7437, at room temperature according to method of ASTM D790. Mechanical test are summarized in Table 5.
Conclusion
This work focused on the development of good adhesion to maleimide epoxy resin and their glass fiber composites with good chemical and mechanical resistance. Synthesized maleimide epoxy showed good thermal stability and degraded in one step. The total heat of combination of resin changes linearly. Based on the thermal analysis of resin. These polymers could be promising materials for as an adhesive and coating materials in industry.
References
[1] G. Liang, G. Aijiuan, Polymer Comp., 1997, 18, 2.
[2] B. Lakshmi, K.N. Shivananda, G.A. Prakash, A.M. Isloor, K.N. Mahendra, Bull. Korean. Chem. Soc., 2012, 33, 2473.
[3] N. Abacha, M. Kubouchi, K. Tsuda, T. Sakai, Express. Polym. Lett., 2007, 1, 364-369.
[4] J.K. Kim, C. Hu, R.S Woo, M.L.Sham, Compo. Sci. Technol., 2005, 65, 805-813.
[5] J. Chaudhary, S. Purohit, R. Chaudhary, Int. J. Eng. Sci. Res. Tech., 2016, 5(8).



