Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 2
Synthesis Antimicrobial and Anti-inflammatory Activities of 3,5-Diphenyl-1H-pyrazole Substituted Azepine-5-carboxamide dérivatives
Satyanarayana KVVV1*, Venkata Swamy Tangeti2,3*, Eswar Kumar K4
1Coastal Chemical Research Society, Visakhapatnam, India
2Raghu Engineering College, Visakhapatnam, Andhra Pradesh-531162, India
3Vector Control Research Centre (ICMR), Puducherry, India
4School of Pharmacy, Andhra University, Vishakhapatnam, India
- *Corresponding Author:
- Satyanarayana KVVV
Coastal Chemical Research Society
Visakhapatnam, India
Venkata Swamy Tangeti
Raghu Engineering College
Visakhapatnam, Andhra Pradesh-531162, India
Abstract
The versatile synthons N-(4-acetylphenyl)-10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (4) and aryl aldehyde with different substituents on aryl group were used as precursors for the synthesis of a series of 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide. The anti-microbiological evaluation of were carried out in vitro assays for antifungal, antibacterial and anti-inflammatory activities. Antibacterial, antifungal and anti-inflammatory screening showed that the newly synthesized compounds 5a to 5k and 6a to 6k were display impressive antimicrobial and anti-inflammatory activity.
Keywords
Pyrazole, Antimicrobial activity, Anti-inflammatory activity
Introduction
Pyrazoles have become a major class of antimicrobial agents, which are under extensive clinical development. Antimicrobial resistance is a major problem in many hospitals as well as in our community settings. In the current generations, pathogenic microbes are evolving resistance to currently used antimicrobial agents. Gram-positive pathogens such as Staphylococcus aureus, Enterococcus faecalis, Enterococcus faecium and Streptococcus pneumoniae are becoming resistant to most of the existing antibiotics and fungal species like Candida spp., Cryptococcus neoformans and Aspergillus spp. are among the leading fungi which show resistance to most of the existing antifungal drugs (J.L. Kane). Hence, the design of new compounds against emerged resistant bacteria has become one of the most important challenges in the field of antimicrobials.
Present day studies are conducted to produce new drugs against these microbes and Pyrazole and related fused heterocycles which are budding as potential bioactive molecules due to their biological and chemotherapeutic importance. Pyrazole and their derivatives exhibit a broad spectrum of biological activities such as antibacterial [1-3], anti-inflammatory [4-6] and antitumoral activities [7,8]. Synthesis of variety of phenyl pyrazoles for biological and pharmacological evaluation is reported [9].
Significant attention is given to the antimicrobial tricyclic structures, such as dihydro-dibenzo[b,e]azepines but show effect on the Central Nervous System (CNS). Among these compounds, l-alkylamino-5,6-dihydro-dibenzo[b,e]azepin-6-ones have been known to possess anti-convulsing properties [10]. 11-alkyloxy derivatives of 5,6-dihydro-dibenzo[b,e]azepin-6-ones show anti-convulsant activity [11]. The bioactivity of these compound have claimed attention of the chemists and biologist, execute research of pyrazole coupled with dibenzo[b, f]azepin. Recently we have developed two new methodologies for synthesis of biologically active furopyrazoles, furocoumarins. [12-19]. In continuation of this efforts we are motivated by these research’s, we report the synthesis and antimicrobial evaluation of a series of 10-methyl-N-(4-(3-arylacryloyl) phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide and 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[ b,f]azepine-5-carboxamide against the most common pathogens, both bacterial and fungal.
Materials and Methods
Melting points were determined in open capillaries and are uncorrected. The IR spectra were recorded on Nicolet Impact 410 FTIR spectrophotometer using KBr pellets. Varian 400 MHz spectrometer instrument using Deuterated Chloroform (CDCl3) as solvent using Tetramethylsilane (TMS) as internal standard: the chemical shifts are reported in ppm. Signal multiplicities are represented by s (singlet), d (doublet), t (triplet), dd (double doublet), m (multiplet) and brs (broad singlet). Mass spectrum was recorded on FinniganMAT (ModelMAT8200) spectrometer The purity of the compounds were checked on Merck precoated silica gel 60 F-254 plate using hexane and ethyl acetate.
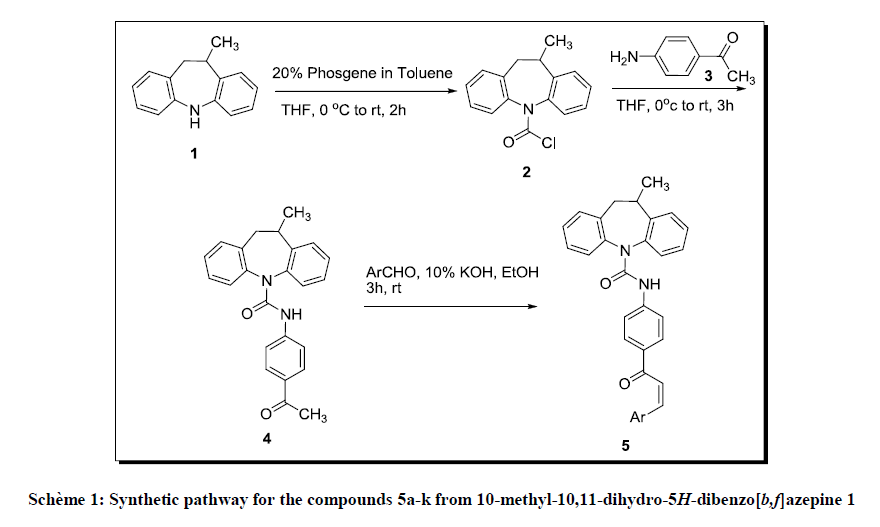
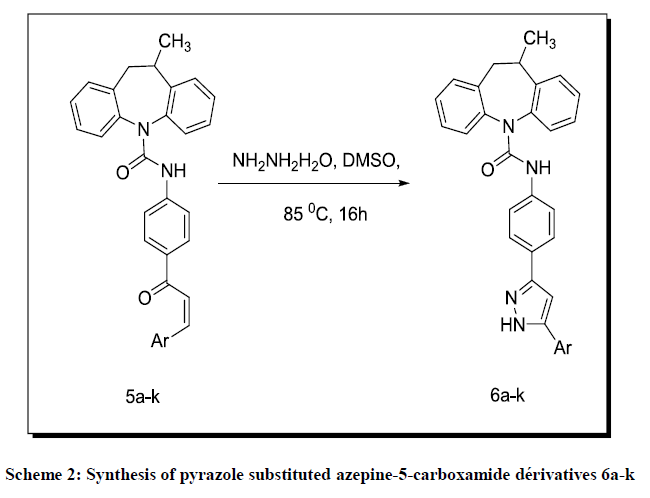
10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbonyl chloride (2) required for the present work has been prepared by the reaction of the 10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine (1) and 20% phosgene solution in toluene at 0°C. N-(4-acetylphenyl)-10-methyl-10, 11- dihydro-5H-dibenzo [b, f] azepine-5-carboxamide (4) formed from 10-methyl-10,11-dihydro-5H-dibenzo[b, f] azepine-5-carbonyl chloride (2) in anhydrous THF and 1-(4-aminophenyl) ethanone (3) in THF, which upon condensation with aromatic aldehydes in ice cold KOH solution affords 10-methyl-N-(4-(3-arylacryloyl) phenyl)-10, 11-dihydro-5H-dibenzo [b, f] azepine-5-carboxamide (5a-k) in good yields, Scheme 1. This is treated with hydrazine hydrate in DMSO forms 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl) phenyl)-10,11-dihydro-5H-dibenzo[b, f]azepine-5- carboxamide (6a-k), Scheme 2.
General experimental procedure for the synthesis of 10-methyl-10, 11-dihydro-5H-dibenzo [b,f] azepine-5-carbonyl chloride 2
Scheme 1, step 1
A solution of 10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine (1) (0.1 mmol) (purchased from Saffire Chemicals, Mumbai) in anhydrous THF (3.5 ml) was added drop-wise to a stirred 20% phosgene solution in toluene (1.1 ml) at 0°C. The reaction mixture was then allowed to warm to room temperature and stirred for 1 h. The reaction mixture was concentrated in vacuo, redissolved in ethyl acetate (10 ml) and filtered through a pad of silica gel. The solvent was removed in vacuo to give the 10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbonyl chloride (2). M.P. 171-176°C; 89% Yield; color-pale brown.
General experimental procedure for the synthesis of preparation of N-(4-acetylphenyl)-10-methyl-10,11-dihydro-5H-dibenzo [b,f] azepine-5-carboxamide 4
Scheme 1, step 2
In 50 ml of anhydrous THF, (0.2 mmol) of 1-(4-aminophenyl)ethanone (3) was dissolved, and under cooling below 0°C with stirring, a solution prepared by dissolving (0.1 mmol) of 10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbonyl chloride (2) into 50 ml of anhydrous THF was added drop-wise. The reaction was continued at a room temperature for 3 h, the solvent was then removed by distillation under a reduced pressure. To the residue thus obtained was added water and the precipitate thus formed was separated by filtration and dried, and re-crystallized from ether-petroleum ether to obtain N-(4-acetylphenyl)-10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (4). M.P. 194-198°C; 80%Yield; color-off white.
General experimental procedure for the synthesis of (Z)-10-methyl-N-(4-(3-arylacryloyl) phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine- 5-carboxamide (5a-k)
Scheme 1, step 3
Aromatic aldehyde (0.1 mmol) was added to iced KOH solution prepared by dissolving 1.32 g of KOH in 5 ml of water and 30 ml of MeOH. The solution was allowed to react. After 10 min, N-(4-acetylphenyl)-10-methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (4) (0.1 mmol) was added thereto, stirred in ice bath for 3 h and subjected to filtration to obtain the residue. The residue was washed with 40 ml cold methanol and dried in vaccuo to obtain (Z)-N-(4-(3-arylacryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide. The progress of the reaction and the purity of compounds were routinely checked on TLC (Aluminium sheet, silica gel; 60F245; E. Merck) and were observed under UV chamber.
10-methyl-N-(4-(3-phenylacryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (5a): The compound obtained as a white colour crystalline solid by using benzaldehyde. (Yield-66%), M.P. 145-148°C. IR (KBr in cm-1): C-H (2950), Ar (842), C=O (1690), C-C (1270), NH (3370), C-CH3 (1380). 1H-NMR (CDCl3, 400 MHz), δ=1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 5H, C20-C24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13) 459: Anal. C31H26N2O2 (Mol. Wt. 458.55).
10-methyl-N-(4-(3-(4-N,N,dimethylphenyl)acryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f] azepine-5-carboxamide (5b): The compound obtained as a buff white colour crystalline solid by using 4-N, N dimethyl benzaldehyde.(Yield-68%), M.P. 157-160°C. IR (KBr in cm-1): C-H (2970), Ar (822), C=O (1670), C-C (1240), NH (3350), C-CH3 (1375). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C- 11), 2.85 (s, 6H, N-Me2), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 6.6 (dd, 2H, C-21 & 23), 7.3 (dd, 2H, C-20 & 24), 7.5- 7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 &19), 8.9 (s, 1H, NH at 13). m/z: (M+) 502: Anal. C33H31N3O2 (Mol. Wt. 501.62)
10-methyl-N-(4-(3-(2-hydroxyphenyl)acryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5c): The compound obtained as a buff white colour crystalline solid by using 2-hydroxy benzaldehyde. (Yield-70%), M.P. 125-130°C. IR (KBr in cm-1): C-H (2945), Ar (824), C=O (1664), C-C (1251), NH (3365), C-CH3 (1371), C-OH (3263). 1 H-NMR (CDCl3, 400 MHz), δ(ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 4.9 (s, 1H, OH), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14- C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 475: Anal. C31H25N3O2 (Mol. Wt. 474.55).
10-methyl-N-(4-(3-(2-chlorophenyl)acryloyl)phenyl)-10,11-dihydro-5Hdibenzo[b,f]azepine-5-carboxamide (5d): The compound obtained as a greyish colour crystalline solid by using 2-chlorobenzaldehyde. (Yield-45%), M.P. 141-145°C. IR (KBr in cm-1): C-H (2941), Ar (832), C=O (1683), C-C (1264), NH (3365), C-CH3 (1376), C-Cl (770).1 H-NMR (CDCl3, 400 MHz), δ: 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20-C23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 &19), 8.9 (s, 1H, NH at 13) ppm. m/z: (M+) 494: Anal. C31H25ClN2O2 (Mol. Wt. 493.00).
10-methyl-N-(4-(3-(4-methylphenyl)acryloyl)phenyl) -10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5e): The compound obtained as a Beige colour crystalline solid by using 4-methyl benzaldehyde. (Yield-78%), M.P. 171-175°C. IR (KBr in cm-1): C-H (2939), Ar (838), C=O (1677), C-C (1256), NH (3365), C-CH3 (1364). 1 H-NMR (CDCl3, 400 MHz) δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C- 11), 2.7 (s, 3H, CH3 at C-22), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 5H, C20-C24), 7.5-7.7 (m, 4H, C14- C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 473, 306, 267, 127, 110: Anal. C32H28N2O2 (Mol. Wt. 472.58).
10-methyl-N-(4-(3-(2,4,6-trichlorophenyl)acryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5f): The compound obtained as a white colour crystalline solid by using 2,4,6-trichloro benzaldehyde. (Yield-63%), M.P. 134-139°C. IR (KBr in cm-1): C-H (2933), Ar (824), C=O (1675), C-C (1256), NH (3357), C-CH3 (1368), C-Cl (775). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2 (s, 2H, C21 & 23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 562: Anal. C31H23Cl3N2O2 (Mol. Wt. 561.89).
10-methyl-N-(4-(3-(3,5-dinitrophenyl)acryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]-azepine-5-carboxamide (5g): The compound obtained as a yellowish colour crystalline solid by using 3,5-Dinitro benzaldehyde. (Yield-75%), M.P. 127-132°C. IR (KBr in cm-1): C-H (2925), Ar (836), C=O (1667), C-C (1248), NH (3349), C-CH3 (1360), C-NO2 (1651).1 H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.2-8.4 (m, 3H, C20, 22 & 24), 8.9 (s, 1H, NH at 13). M/z: (M+) 549: Anal. C31H24N4O6 (Mol. Wt. 548.55).
10-methyl-N-(4-(3-(4-triflouromethylphenyl)acryloyl)-4-phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5h): The compound obtained as a white colour crystalline solid by using 4-trifluro methyl benzaldehyde. (Yield-74%), M.P. 135-139°C. IR (KBr in cm-1): C-H (2938), Ar (827), C=O (1676), C-C (1265), NH (3354), C-CH3 (1363), C-F (580). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 526, 510, 488, 432, 388, 344, 311, 243, 157.: Anal C32H25F3N2O2 (Mol. Wt. 526.55).
10-methyl-N-(4-(3-(4-chlorophenyl)acryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5i): The compound obtained as a buff colour crystalline solid by using 4-chloro benzaldehyde. (Yield-61%), M.P. 142-147°C. IR (KBr in cm-1): C-H (2932), Ar (813), C=O (1674), C-C (1255), NH (3346), C-CH3 (1357), C-Cl (765). 1H-NMR (CDCl3, 400 MHz) δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 494: Anal. C31H25ClN2O2 (Mol. Wt. 493.00).
10-methyl-N-(4-(3-(3-nitrophenyl)acryloyl)-phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5j): The compound obtained as a yellowish colour crystalline solid by using 3-nitrobenzaldehyde. (Yield-45%), M.P. 152-155°C. IR (KBr in cm-1): C-H (2939), Ar (830), C=O (1671), C-C (1252), NH (3363), C-CH3 (1364), C-NO2 (1510). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2 (d, 1H, C20), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C- 18 & 19), 8.2 (m, 2H, C2 & C 24), 8.4 (s, 1H, C-21), 8.9 (s, 1H, NH at 13). m/z: (M+) 504: Anal. C31H25N3O4 (Mol. Wt. 503.55).
10-methyl-N-(4-(3-(2,4-dimethylphenyl)acryloyl)-phenyl)-10,11-dihydro-5H-dibenzo[b,f]-azepine-5-carboxamide (5k): The compound obtained as a buff colour crystalline solid by using 2,4-dimethyl benzaldehyde. (Yield-48%), M.P. 168-172°C. IR (KBr in cm-1): C-H (2941), Ar (835), C=O (1657), C-C (1235), NH (3354), C-CH3 (1368). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.3 (s, 6H, Me2), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 3H, C20, 21 & 23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.1 (m, 2H, C-18 & 19), 8.9 (s, 1H, NH at 13). m/z: (M+) 487: Anal. C33H30N2O2 (Mol. Wt. 486.60).
General experimental procedure for the synthesis of 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (6a-k)
A mixture of (Z)-10-methyl-N-(4-(3-arylacryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (0.55 mmol) and of hydrazine hydrate (0.50 ml, 0.55 g, 5.1 mmol) in DMSO (5 ml) was heated in air in a pre-equilibrated oil bath at 85°C for 16 h. The reaction solution was cooled to room temperature and partitioned with 15 ml of diethyl ether and 5 ml of water. The organic layer was separated and washed with four 5 ml portions of water. The aqueous layers were combined, extracted with three 10 ml portions of diethyl ether. The organic layers were combined, dried (MgSO4) and concentrated in vacuo. The residue was chromatographed over a 1 × 8 cm silica gel column (eluted with ethyl acetate-hexanes, 1:8) to give 96 mg (59%) of the desired 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide.
10-methyl-N-(4-(5-phenyl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6a): The compound obtained as a white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-phenylacryloyl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5- carboxamide(5a). (Yield-46-%), M.P. 132-136°C. IR (KBr in cm-1): C-H (2948), Ar (840), C=O (1688), C-C (1268), NH (3372), C-CH3 (1378). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 5H, C20-C24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z:(M+) 471: Anal. C31H26N4O (Mol. Wt. 470.56).
10-methyl-N-(4-(5-(4-N,N,dimethylphenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6b): The compound obtained as a buff white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(4-N,N,dimethyphenyl)acryloyl)phenyl)-10,11- dihydro-5H-dibenzo[b,f]azepine-5-carboxamide(5b). (Yield-58%), M.P. 141-146°C. IR (KBr in cm-1): C-H (2967), Ar (819), C=O (1668), C-C (1237), NH (3346), C-CH3 (1372). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 2.85 (s, 6H, N-Me2), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 6.6 (dd, 2H, C-21 & 23), 7.3 (dd, 2H, C-20 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+) 514, 255, 159, 157: Anal. C33H31N5O (Mol. Wt. 513.63).
10-methyl-N-(4-(5-(2-hydroxyphenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6c): The compound obtained as a white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(2-hydroxyphenyl)acryloyl)phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide(5c). Yield-26%), M.P. 157-162°C. IR (KBr in cm-1): C-H (2943), Ar (821), C=O (1661), C-C (1248), NH (3362), C-CH3 (1368), C-OH (3260). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 4.9 (s, 1H, OH), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+)487: Anal. C31H26N4O2 (Mol. Wt. 486.56).
10-methyl-N-(4-(5-(2-chlorophenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6d): The compound obtained as a beige colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(2-chlorophenyl)acryloyl)phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide (5d). (Yield-47%), M.P. 114-118°C. IR (KBr in cm-1 C-H (2938), Ar (829), C=O (1679), C-C (1262), NH (3361), C-CH3 (1372), C-Cl (768).). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C- 18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20-C23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+) 506: Anal. C31H25ClN4O (Mol. Wt. 505.01).
10-methyl-N-(4-(5-(4-methylphenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide(6e): The compound obtained as a Buff colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(4-methylphenyl)acryloyl)phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide (5e). (Yield-56%), M.P. 129-134°C. IR (KBr in cm-1): C-H (2936), Ar (835), C=O (1674), C-C (1253), NH (3362), C-CH3 (1361). 1H-NMR (CDCl3, 400 MHz) δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 2.7 (s, 3H, CH3 at C-22), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 5H, C20-C24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+) 485: Anal. C32H28N4O (Mol. Wt. 484.59).
10-methyl-N-(4-(5-(2,4,6-trichlorophenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6f): The compound obtained as a white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(2,4,6-trichlorophenyl)acryloyl)phenyl)-10,11-dihydro- 5H-dibenzo[b,f]azepine-5-carboxamide (5f). (Yield-64%), M.P. 142-147°C. IR (KBr in cm-1): C-H (2930), Ar (822), C=O (1673), C-C (1253), NH (3354), C-CH3 (1365), C-Cl (771). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2 (s, 2H, C21 & 23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+)575: Anal. C31H23Cl3N4O (Mol. Wt.573.90).
10-methyl-N-(4-(5-(3,5-dinitrophenyl)-1H-pyrazol-3-yl)3,5-dinitrophenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6g): The compound obtained as a Brownish colour Crystalline solid by using (Z)-10-methyl-N-(4-(3-(3,5-Dinitrophenyl)acryloyl)phenyl)-10,11- dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5g) (Yield-18%), M.P. 138-143°C. IR (KBr in cm-1): C-H (2922), Ar (833), C=O (1665), C-C (1246), NH (3347), C-CH3 (1357), C-NO2 (1647). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.2-8.4 (m, 3H, C20, 22 & 24), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+) 561: Anal. C31H24N6O5 (Mol. Wt. 560.56)
10-methyl-N-(4-(5-(4-triflouromethylphenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6h): The compound obtained as a creamish colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(4-triflouro methyl phenyl)acryloyl)-4-phenyl)-10,11- dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5h). (Yield-45%), M.P. 162-166°C. IR (KBr in cm-1): C-H (2935), Ar (824), C=O (1673), C-C (1262), NH (3351), C-CH3 (1360), C-F (581). 1H-NMR (CDCl3, 400 MHz), δ (ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19). m/z: (M+) 539.: Anal C32H25F3N4O (Mol. Wt. 538.56).
10-methyl-N-(4-(5-(4-chlorophenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6i): The compound obtained as a white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(4-chlorophenyl)acryloyl)phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide (5i) (Yield-26%), M.P. 128-131°C. IR (KBr in cm-1): C-H (2929), Ar (811), C=O (1672), C-C (1253), NH (3343), C-CH3 (1354), C-Cl (762). 1H-NMR (CDCl3, 400 MHz), δ: 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8- 7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 4H, C20, 21, 23 & 24), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.9 (s, 2H, NH at 13 & 19) ppm. m/z: (M+) 506: Anal C31H25ClN4O (Mol. Wt. 505.01).
10-methyl-N-(4-(5-(3-nitrophenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6j): The compound obtained as a pale yellowish colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(3-nitrophenyl)acryloyl)-phenyl)-10,11-dihydro-5Hdibenzo[ b,f]azepine-5-carboxamide (5j) (Yield-48%), M.P. 119-123°C. IR (KBr in cm-1): C-H (2936), Ar (826), C=O (1668), C-C (1249), NH (3360), C-CH3 (1361), C-NO2 (1507). 1 H-NMR (CDCl3, 400 MHz) δ(ppm): 1.9 (d, 2H, C-5), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C- 18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2 (d, 1H, C20), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C10), 8.2 (m, 2H, C2 & C 24), 8.4 (s, 1H, C-21), 8.9 (s, 2H, NH at 11 & 17). m/z: (M+) 516: Anal. C31H25N5O3 (Mol. Wt. 515.56).
Synthesis of 10-methyl-N-(4-(5-(2,4-dimethylphenyl)-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carbox-amide (6k): The compound obtained as a white colour crystalline solid by using (Z)-10-methyl-N-(4-(3-(2,4-dimethylphenyl)acryloyl)-phenyl)-10,11- dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (5k). (Yield-65%), M.P. 131-134°C. IR (KBr in cm-1): C-H (2938), Ar (832), C=O (1653), C-C (1232), NH (3352), C-CH3 (1361). 1H-NMR (CDCl3, 400 MHz), δ: 1.9 (d, 2H, C-5), 2.3 (s, 6H, Me2), 2.5 (d, 3H, C-11), 3.3 (m, 1H, C-6), 6.5 (s, 1H, C-18), 6.8-7.0 (m, 6H, C2, C3, C4, C7, C8 & C9), 7.2-7.4 (m, 3H, C20, 21 & 23), 7.5-7.7 (m, 4H, C14-C17), 7.9 (d, 2H, C1 & C 10), 8.9 (s, 2H, NH at 13 & 19) ppm. m/z: (M+) 499, 470, 426: Anal. C33H30N4O (Mol. Wt. 498.62).
Test microorganisms
Salmonella typhi, Vibrio cholera and Shigella dysenteriae and are gastrointestinal pathogens. Klebsiella pneumoniae is causative agent of pulmonary infections. C. albicans are causative agents of skin infections. S. typhi, V. cholera and S. dysenteriae are clinical isolates and remaining cultures are purchased from IMTEC, Chandigarh, India and NCL, Pune, India.
Antimicrobial assay by agar well diffusion method
The bacteria were grown in Muller-Hinton media (HiMedia Pvt. Ltd., Mumbai, India) at 370°C while fungi were grown in Sabouraud Dextrose Agar media at 280°C and maintained on nutrient agar slants at 40°C and stored at -200°C. Inoculum of bacteria was prepared by growing pure isolate in nutrient broth at 370°C for overnight. The overnight broth bacterial cultures was sub-cultured in fresh nutrient broth and grown for 3 h to obtain log phase culture. 21 days old grown fungi culture was scraped with sterile scalpel and dissolved in sterile saline solution to make different dilutions. The diluted suspension which has the absorbance of 0.600 at 450 nm determined spectroscopically (Electronics India) then it was used as inoculums for fungi. The agar plates were prepared by pour plate method using 20 ml of agar medium. The sterile agar medium is cooled to 450×C and mixed thoroughly with 1 ml of growth culture of concerned test organism (1 × 108 cells) and then poured into the sterile petri dishes and allowed to solidify. Wells of 6 mm size were made with sterile cork borer and test extracts were added. The agar plates were incubated at for 4 days at 280°C for fungi wile 24 h at 370 for bacteria. The diameter of inhibition zones was measured in mm using HiMedia zone reader. Ciprofloxacin (Antibiotic) used as standard while solvent (DMSO) used for control (K. Eswar Kumar).
Carrageenan induced rat hind paw edema
Albino rats of either sex weighing between 150-200 g were used during the study. They were purchased from a local vendor i.e., Sri Sai Enterprises, HYD. The study had permission from the Animal Ethical Committee. All the animals were fasted overnight and allowed water. Anti-Inflammatory activity of synthesized compounds was determined by carrageenan induced rat hind paw edema method.
Anti-Inflammatory activity of synthesized compounds was determined by carrageenan induced rat hind paw edema method. The drugs were prepared as a suspension by triturating with 1% tween 80. The suspension of test compounds (10 mg/kg) were administered orally in the 15 treated groups and after 30 min, inflammation was induced by injecting 0.1 ml of 1% carrageenan solution in the intraplantar region. The standard group received 10 mg/kg of diclofenac, test group received 10 mg/kg of synthesized compounds and the control group received 1% w/v of tween 80. The percentage inhibition of inflammation after 4 h was calculated using following formula (D. Rosa).
% Inhibition of edema = (1-Et/Ec) × 100
Where, Et represents the average value of the edema in ml in treated groups in 1-4 h after carrageenan injection, while, Ec represents the average value of the edema in mL in control group in 1-4 h after carrageenan injection.
Results and Discussion
The formation of 10-methyl-N-(4-(3-arylacryloyl) phenyl)-10,11-dihydro-5H-dibenzo [b,f] azepine-5-carboxamide (5a-k) by the reaction between 10-methyl-10,11-dihydro-5H-dibenzo[b, f]azepine-5-carbonyl chloride (2) and 1-(4-aminophenyl) ethanone (3) was confirmed by 1H-NMR spectra displayed a peak at δ=8.1 (m, 2H) and δ=7.2-7.4 (m, 5H). The structures of the so obtained 10-methyl-N-(4-(5-aryl-1H-pyrazol-3- yl)phenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide (6a-k) from 10-methyl-N-(4-(3-arylacryloyl) phenyl)-10, 11-dihydro-5H-dibenzo[ b,f]azepine-5-carboxamide (5a-k) were confirmed by 1H-NMR spectra displayed a peak at δ=8.9 (s, NH) and δ=7.2-7.4 (m, 5H) and also confirmed by the IR spectra at NH (3372). The IR, 1H-NMR and mass spectral data finally ascertained the structures of all the compounds. The investigation revealed that some of the tested compounds showed moderate to good bacterial inhibition. With reference to the ciprofloxacin standard used against bacteria; compounds 5a, 5b, 5c, 6a, 6b and 6c has shown good activity against S. typhi, V. cholera, S. dysenteriae, K. pneumoniae and C. albicans at a dose of both 50 and 100 μg. The results indicate both 3-aryl and 5-aryl group substituent’s are act against gastrointestinal, pulmonary infections and skin infections. The results were summarized in Tables 1 and 2 explain that the phenyl, methyl and hydroxyl group substituent’s showed marked inhibition against tested microorganisms.
| Compound | Concentration (µg) | Zone of inhibition at various doses (mm) | ||||
|---|---|---|---|---|---|---|
| Salmonella typhi | Vibrio cholera | Shigella dysenteriae | Klebsiella pneumoniae | Candida albicans | ||
| 5a | 50 | 6 | 9 | 8 | 9 | 7 |
| 100 | 11 | 10 | 12 | 11 | 13 | |
| 5b | 50 | 5 | 7 | 8 | 9 | 7 |
| 100 | 10 | 12 | 11 | 11 | 10 | |
| 5c | 50 | 6 | 7 | 7 | 5 | 8 |
| 100 | 8 | 11 | 12 | 11 | 13 | |
| 5d | 50 | 4 | 3 | 5 | 4 | 4 |
| 100 | 8 | 7 | 6 | 5 | 7 | |
| 5e | 50 | 4 | 5 | 4 | 3 | 5 |
| 100 | 8 | 6 | 7 | 6 | 7 | |
| 5f | 50 | 5 | 3 | 4 | 4 | 5 |
| 100 | 8 | 7 | 9 | 8 | 7 | |
| 5g | 50 | 5 | 6 | 3 | 4 | 5 |
| 100 | 8 | 7 | 5 | 6 | 7 | |
| 5h | 50 | 3 | 5 | 4 | 4 | 5 |
| 100 | 5 | 6 | 6 | 5 | 8 | |
| 5i | 50 | 4 | 3 | 5 | 4 | 4 |
| 100 | 6 | 7 | 7 | 6 | 6 | |
| 5j | 50 | 3 | 5 | 4 | 3 | 5 |
| 100 | 5 | 6 | 6 | 5 | 7 | |
| 5k | 50 | 5 | 4 | 4 | 6 | 6 |
| 100 | 7 | 6 | 7 | 8 | 8 | |
| Ciprofloxacin | 30 | 10 | 10 | 12 | 11 | 13 |
Table 1: Antimicrobial activity of 3 aryl substituted series 5a to 5k
| Compound | Concentration (µg) | Zone of inhibition at various doses (mm) | ||||
|---|---|---|---|---|---|---|
| Salmonella typhi | Vibrio cholera | Shigella dysenteriae | Klebsiella pneumoniae | Candida albicans | ||
| 6a | 50 | 8 | 7 | 6 | 7 | 9 |
| 100 | 12 | 10 | 11 | 12 | 14 | |
| 6b | 50 | 8 | 7 | 7 | 8 | 10 |
| 100 | 13 | 12 | 14 | 13 | 15 | |
| 6c | 50 | 7 | 8 | 9 | 11 | 12 |
| 100 | 11 | 12 | 11 | 13 | 14 | |
| 6d | 50 | 4 | 3 | 5 | 4 | 5 |
| 100 | 8 | 6 | 7 | 6 | 8 | |
| 6e | 50 | 3 | 4 | 3 | 5 | 5 |
| 100 | 5 | 6 | 6 | 8 | 9 | |
| 6f | 50 | 4 | 5 | 4 | 3 | 4 |
| 100 | 7 | 7 | 8 | 6 | 8 | |
| 6g | 50 | 6 | 4 | 5 | 5 | 4 |
| 100 | 8 | 6 | 8 | 9 | 8 | |
| 6h | 50 | 5 | 3 | 4 | 3 | 5 |
| 100 | 8 | 6 | 7 | 7 | 8 | |
| 6i | 50 | 4 | 5 | 3 | 4 | 4 |
| 100 | 6 | 7 | 5 | 7 | 9 | |
| 6j | 50 | 3 | 5 | 4 | 5 | 6 |
| 100 | 5 | 8 | 7 | 8 | 10 | |
| 6k | 50 | 4 | 5 | 6 | 5 | 7 |
| 100 | 7 | 7 | 8 | 6 | 10 | |
| Ciprofloxacin | 30 | 13 | 12 | 11 | 12 | 15 |
Table 2: Antimicrobial activity of 5 aryl substituted series 6a to 6k
All the compounds were subjected to anti-inflammatory activity by paw edema method using indomethacin as standard. All the 3,5-diphenyl- 1H-pyrazole substituted azepine-5-carboxamide dérivatives 5a-k and 6a-k showed promising anti-inflammation activities, as represented in Tables 3 and 4 and the results were compared against control. The dose of 50 μg of all tested compounds exhibited increase in anti-inflammatory activity, but 7 compounds showed decrease in activity in 4th h whereas 3 compounds demonstrated further increase in anti-inflammatory activity even in the 4th h. Seven compounds displayed greater than 50% paw edema protection in the 4th h and three compounds in the 3rd h of the study. The study indicates the phenyl, methyl and hydroxyl substituents showed significant reduction in carrageenan induced paw edema compared to other halogen substituent groups.
| Compound | % Inhibition at different intervals (h) | |||
|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | |
| 5a | 28.23 ± 2.31* | 38.10 ± 1.38** | 47.23 ± 2.34*** | 60.44 ± 3.45*** |
| 5b | 29.13 ± 1.65* | 38.96 ± 2.46** | 51.49 ± 1.89*** | 61.33 ± 2.65*** |
| 5c | 32.44 ± 2.15* | 41.26 ± 2.11*** | 52.36 ± 2.66*** | 63.46 ± 3.10*** |
| 5d | 21.23 ± 3.64* | 32.12 ± 3.10* | 49.23 ± 1.56*** | 51.10 ± 2.44** |
| 5e | 25.16 ± 1.47* | 27.64 ± 1.64* | 39.16 ± 3.16** | 35.64 ± 2.81* |
| 5f | 28.47 ± 2.21* | 29.94 ± 1.82* | 35.46 ± 2.46* | 33.94 ± 1.56* |
| 5g | 27.10 ± 3.12* | 31.46 ± 3.24* | 36.82 ± 1.46** | 34.77 ± 1.97* |
| 5h | 30.99 ± 2.65* | 39.29 ± 2.46** | 42.33 ± 2.22** | 43.46 ± 2.66** |
| 5i | 26.65 ± 2.10* | 33.79 ± 2.76* | 37.47 ± 1.94* | 35.30 ± 2.31* |
| 5j | 24.64 ± 3.23* | 34.34 ± 2.40* | 45.06 ± 2.33** | 42.14 ± 2.44** |
| 5k | 27.13 ± 1.46* | 33.29 ± 1.66* | 41.19 ± 2.46** | 40.06 ± 1.98** |
| Indomethacin | 29.11 ± 1.98* | 36.13 ± 1.48** | 44.66 ± 1.56** | 59.44 ± 4.23*** |
Results expressed in mean ± SEM. (n=6). ANOVA followed by Dunnett’s test. ***p < 0.001, **p < 0.01, *p < 0.05 when compared to control group
Table 3: Anti-inflammatory activity of 3 aryl substituted series 5a to 5k and indomethacin in carrageenan induced inflammation
| Compound | % Inhibition at different intervals (h) | |||
|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | |
| 6a | 31.43 ± 2.31* | 37.81 ± 3.12** | 47.98 ± 3.21*** | 57.13 ± 2.39*** |
| 6b | 28.79 ± 1.49* | 33.25 ± 3.16* | 45.69 ± 2.46*** | 58.66 ± 2.46*** |
| 6c | 33.66 ± 2.46* | 42.57 ± 2.46** | 51.32 ± 2.56*** | 64.69 ± 2.64*** |
| 6d | 23.46 ± 2.61* | 28.69 ± 2.16* | 34.56 ± 68* | 43.26 ± 2.51** |
| 6e | 20.13 ± 1.65* | 24.65 ± 2.94* | 31.26 ± 1.64* | 30.16 ± 3.16* |
| 6f | 24.46 ± 1.94* | 29.68 ± 2.768 | 38.21 ± 1.94** | 36.11 ± 3.46* |
| 6g | 22.16 ± 2.33* | 27.46 ± 1.64* | 34.56 ± 3.16* | 33.65 ± 1.61* |
| 6h | 24.65 ± 1.16* | 30.16 ± 1.67* | 40.16 ± 3.16** | 39.24 ± 1.49** |
| 6i | 21.47 ± 0.94* | 26.68 ± 3.16* | 33.19 ± 2.64* | 34.12 ± 1.84* |
| 6j | 23.60 ± 2.16* | 29.16 ± 2.31* | 38.17 ± 2.49** | 37.05 ± 2.16** |
| 6k | 22.35 ± 2.35* | 27.95 ± 1.19* | 38.72 ± 3.16** | 37.00 ± 1.94** |
| Indomethacin | 26.45 ± 1.65* | 32.16 ± 1.94* | 44.95 ± 2.44*** | 54.33 ± 4.23*** |
Results expressed in mean ± SEM. (n=6). ANOVA followed by Dunnett’s test. ***p < 0.001, ** p < 0.01, *p < 0.05 when compared to control group
Table 4: Anti-inflammatory activity of 5 aryl substituted series 6a to 6k and indomethacin in carrageenan induced inflammation
Conclusion
Novel 10-methyl-N-(4-(5-aryl-1H-pyrazol-3-yl)phenyl)-10,11-dihydro-5Hdibenzo[b,f]azepine-5-carboxamide and N-(4-acetylphenyl)-10- methyl-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide were synthesized. The results of antibacterial screening reveal that among all the newly synthesized compounds six of the newly synthesized compounds showed good bacterial inhibition and anti-inflammatory activity.
Acknowledgement
We thankful to Dr. P. Uma Devi and M. Venkata Ramana, Visakhapatnam, India for helping in bringing out the above literature and experimentation.
References
- L. Xin-Hua, Z. Jing, Z. An-na, S. Bao-An, Z. Hai-Liang, Bioorg. Med. Chem., 2009, 17, 1207.
- M.F. Ahmad, S.M. Abdelrahman, E.B. Saber, H.B. Ashraf, Bioorg. Med. Chem., 2008, 16, 4569.
- J.F. Chimenti, B. Bizzarri, F. Manna, A. Bolasco, D.P. Secci, Bioorg. Med. Chem. Lett., 2005, 15, 603.
- E.R. Aymn, I.H. Mohamed, E.A.M. Randa, A.M. Jehan, M.E. Farouk, Bioorg. Med. Chem., 2008, 16, 7102.
- R.R. Ranatunge, M. Augustyniak, U.K. Bandarage, R.A. Earl, Med. Chem., 2004, 47, 2180.
- D. Fancelli, J. Moll, M. Varasi, R. Bravo, R. Artico, D. Berta, Med. Chem., 2006, 49, 7247.
- P.G. Baraldi, G. Balboni, M.G. Pavani, G. Spalluto, M.A. Tabrizi, J. Med. Chem., 2001, 44, 2536.
- P. Pevarello, M.G. Brasca, R. Amici, P. Orsini, G. Traquandi, L. Corti, C. Piutti, P. Sansonna, M. Villa, J. Med. Chem., 2004, 47, 3367.
- A.M. Farag, A.S. Mayhoub, S.E. Barakat, A.H. Bayomi, Bioorg. Med. Chem.,2008, 16, 881.
- W.S. Waring, B.A. Whittle, J. Pharm. Pharmacol., 1969, 21, 520.
- P. Vittorio, V. Giovanni, N. Rossan, B. Franc, F. Martin, L. Alessandro, V. Giovanna, M. Albert, Eur. J. Med. Chem., 1988, 23, 473.
- S. Tangeti, G.V. Siva Prasad, J. Panda, K.R. Varma, Syn. Commun. 2016, 46, 878.
- H.S.P. Rao, V.S. Tangeti, J. Chem. Sci., 2013, 125, 777.
- H.S.P. Rao, V.S. Tangeti, A. Laxminarayana, Res. Chem. Intermed., 2016, 42, 7285.
- V.S. Tangeti, K.R. Varma, G.V. Siva Prasad, K.V.V.V. Satyanarayana, Syn. Commun., 2016, 46, 613.
- H.S.P. Rao, V.S. Tangeti, Lett. Org. Chem., 2012, 9, 218.
- H.S.P. Rao, V.S. Tangeti, Proceedings of National Academy of Sciences-India Sec-a., 2015, 85, 41.
- S.T. Nishith..; H.S.P. Rao, V.S. Tangeti. and R. Krishna, Acta Cryst., 2013, E6, o284.
- V.S. Tangeti, D. Vasundhara, K.V.V.V. Satyanarayana, K.S.P. Kumar, Asian J. Chem., 2017, 29, 527.