Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 11
Synthesis, Characterization and Antibacterial Activity of Some Novel C-7-Substituted-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine Derivatives
Ashok Reddy Ankireddy1, Gundla Rambabu1*, Tuniki Balaraju2, Venkanna Banothu3, Kalyani Paidikondala3 and Manohar Mantipally4
1Department of Chemistry, School of Science, GITAM University, Hyderabad, India-502329
2Department of Chemistry, Birla Institute of Technology, Mesra, Jharkhand, India-835215
3Centre for Chemical Sciences and Technology, Institute of Science and Technology, Jawaharlal Nehru Technological University Hyderabad, Kukatpally, Hyderabad-500085, India
4Center for Biotechnology, IST, JNTUH, Kukatpally, Hyderabad-500085, India
- *Corresponding Author:
- Gundla Rambabu
Department of Chemistry
School of Science
GITAM University
Hyderabad, India-502329
Abstract
A number of pre-designed C-7-substituted-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine were synthesized for the first time using Palladium catalyzed suzuki reactions in microwave with a ligand X-PHOS. A key intermediate 7-chloro-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (7) was synthesized and treated with 20 different boronic acids to give 20 derivatives in good to excellent yields. All these compounds were tested for their antibacterial properties against four strains of bacterial microorganisms including two gram-positive and two gram-negative species. Out of twenty compounds, five compounds, compound 8, 16, 23, 24, 25 showed better activity against gram negative as well as gram positive strains.
Keywords
Quninazoline derivatives, Antibacterial activity, Escherichia coli (-ve), Klebsiella pneumoniae (-ve), Pseudomonas species (+ve), Staphylococcus aureus.
Introduction
Many substituted quinazoline derivatives possess a wide range of bioactivities such as antimalarial, anticancer, antimicrobial, antifungal, antiviral, antiprotozoan, anti-inflammatory, diuretic, muscle relaxant, antitubercular, antidepressant, anticonvulsant, acaricidal, weedicide, and many other biological activities. Quinazoline and quinazolinone compounds are also used in preparation of various functional materials for synthetic chemistry and also present in various drugs molecules. This review is an attempt to expand the huge potentiality and ocused on the various biological activities of quinazolines. Quinazoline derivatives, which belong to the N-containing heterocyclic compounds, have caused universal concerns due to their widely and distinct biopharmaceutical activities. Researchers have already determined many therapeutic activities of quinazoline derivatives, including insecticidal [1], analgesic [2], antifungal [3], antibacterial [4], anticancer [5], anti-inflammatory [5], anticonvulsant [6], antiviral [7], diuretic [7], antihypertensive [8] and α-Glucosidase inhibitors [9] activities. Medicinal chemists synthesized a variety of quinazoline compounds with different biological activities by installing various active groups to the quinazoline moiety using developing synthetic methods. And the potential applications of the quinazoline derivatives in fields of biology, pesticides and medicine have also been explored. Quinazoline nucleus is found in many bioactive natural products. So, because of these reasons much attention is being paid for the synthesis of quinazoline derivatives. Looking at the biological significance of quinazoline nucleus it was thought to design and synthesize new quinazoline derivatives and screen them for their antibacterial activity [1-4].
Materials and Methods
Melting points were determined by open glass capillary method on a Cintex melting point apparatus. Proton Nuclear Magnetic Resonance (1H-NMR) and Carbon-13 Nuclear magnetic Resonance (13C-NMR) spectra were recorded on a Varian 300 or 400 MHz spectrometer using CDCl3 and DMSO-d6, with reference to Tetramethylsilane (TMS) as an internal reference. Mass spectra were recorded on a Jeol JMC D-300 instrument by using Electron ionization at 70 eV. All reactions were monitored by Thin Layer Chromatography (TLC) on pre-coated silica gel plates. Column chromatography was performed by using silica gel (100-200 mesh, SRL, India) (10-20 times (by weight)) of the crude product.
Synthesis
7-chloroquinazoline-2,4(1H,3H)-dione (2)
Compound 2 prepared by following the earlier reported procedure ‘Organic process research & development, 2003, 7, 700-706’ [5].
2,4,7-trichloroquinazoline (3)
Compound 3 prepared by following the earlier reported procedure: Bioorganic & medicinal chemistry, 2003, 11, 2439-2444 [6].
2,7-dichloro-N-(pyridin-2-ylmethyl)quinazolin-4-amine (5)
To the solution of 2,4,7-trichloroquinazoline (5 g, 21.41 mmol, 1.0 equiv) in THF (50 mL) was added pyridin-2-ylmethanamine (2.31 g, 21.41 mmol, 1.0 equiv). The reaction mixture was stirred at room temperature for 12 h. Solvent was evaporated, and the reaction mixture was diluted with water (100 ml) and the mixture was extracted with EtOAc (2 × 50 ml). The organic layer was dried (Na2SO4), filtered and concentrated in vacuo. The crude compound was purified by combi flash chromatography (10 to 20% EtOAc/Hexane) to give pure compound 5 (3.00 g, 9.83 mmol). Color: Yellow solid. Yield: 45%. TLC (0.4 Rf: 40%EtOAc/Hexane). LCMS (MM): m/z=306 [M + H]+, HPLC area >99%.
7-chloro-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (7)
To the solution of 2,7-dichloro-N-(pyridin-2-ylmethyl)quinazolin-4-amine (5) ( 10.00 g, 32.76 mmol, 1.0 equiv) and morpholine (6) (5.70 g, 65.53 mmol, 2.0 equiv) in Isopropyl alcohol (50 ml) was added DIPEA (12.70 g, 98.30 mmol, 3.0 equiv); The reaction mixture was stirred at 100°C for 12 h. solvent was evaporated, and the reaction mixture was diluted with water (100 ml) and the mixture was extracted with EtOAc (2 × 50 ml). The organic layer was dried (Na2SO4), filtered and concentrated in vacuo. The crude compound was purified by combi flash chromatography (10 to 20% EtOAc/Hexane) to give pure compound 7 (5.00 g, 14.05 mmol). Color: Yellow solid; Yield: 42%; TLC (0.4 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.84 (t, J=5.6 Hz, 1H), 8.50 (dd, J=0.8 Hz, 4.8 Hz, 1H), 8.11 (d, J=8.8 Hz, 1H), 7.71 (td, J=1.6 Hz, 7.6 Hz, 9.6 Hz, 1H), 7.33 (d, J=8.0 Hz, 1H), 7.28 (d, J=2.0 Hz, 1H), 7.28-7.17 (m, 1H), 7.13 (d, J=2.0 Hz, 8.8 Hz, 1H), 4.75 (d, J=5.6 Hz, 2H), 3.62 (brs, 4 H), 3.53 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.48, 158.90, 158.80, 148.73, 137.14, 136.50, 124.89, 123.63, 122.42, 121.91, 121.06, 120.75, 109.34, 65.99, 46.00, 43.88. LCMS (MM): m/z = 356 [M + H]+. HPLC area >99%.
General procedure for the preparation of 8 to 27
To the solution of compound 7 (1 mmol, 1.0 equiv) in THF (6.0 ml) were added boronic acid (2 mmol, 2.0 equiv) and purged with nitrogen for about 5 min and then added Pd(OAc)2 (0.1 mmol, 0.1 equiv), X-Phos (0.2 mmol, 0.2 equiv), K3PO4 (2 mmol, 2.0 equiv), H2O (1.0 ml). The reaction mixture was irradiated under CEM microwave (120°C, 200 psi, 200 watts) for 30 min to 60 min. The progress of the reaction was monitored by checking TLC at a regular interval. Reaction mixture was diluted with water (10 ml) and the mixture was extracted with EtOAc (2 × 10 ml). The organic layer was dried (Na2SO4), filtered and concentrated in vacuo. The obtained crude compound was purified by combi flash chromatography (EtOAc/Hexane) to afford compound 8 to compound 23.
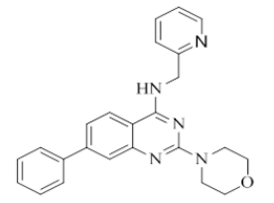
2-morpholino-7-phenyl-N-(pyridin-2-ylmethyl)quinazolin-4-amine (8)
Color: Off white solid. Yield: 80%. Melting point: 155°C-161°C; TLC (0.4 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.85 (brs, 1H), 8.50 (d, J=3.6 Hz, 1H), 8.20 (d, J=8.4 Hz, 1H), 7.77 (d, J=7.2 Hz, 2H), 7.71 (t, J=7.6 Hz, 1H), 7.57 (s, 1 H), 7.50-7.45 (m, 3 H), 7.42-7.39 (m, 1 H), 7.35 (d, J=8.0 Hz, 1H), 7.24 (t, J=5.6 Hz, 1H), 4.79 (d, J=5.2 Hz, 2H), 3.64 (brs, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.66, 159.21, 158.65, 151.66, 148.69, 145.56, 140.90, 136.46, 134.61, 130.32, 129.25, 127.54, 126.09, 125.91, 124.85, 122.66, 122.12, 121.85, 120.98, 109.47, 66.06, 46.01, 44.05, 20.08. LCMS (MM): m/z calcd for C24H23N5O [M + H]+ 397.47; found 398. HPLC area >98%.
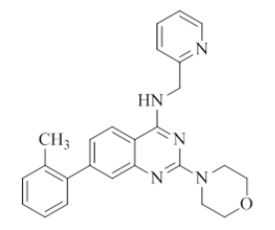
2-morpholino-N-(pyridin-2-ylmethyl)-7-(o-tolyl)quinazolin-4-amine (9)
Color: Brown gummy solid. Yield: 65%. Melting point: 142°C-144°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.79 (brs, 1H), 8.50 (d, J=4.0 Hz, 1H), 8.15 (d, J=8.4 Hz, 1H), 7.71 (t, J=6.8 Hz, 1H), 7.35 (d, J=8.0 Hz, 1H), 7.30-7.22 (m, 5 H), 7.18 (s, 1 H), 7.09 (d, J=7.6 Hz, 1H), 4.79 (d, J=5.2 Hz, 2H), 3.62 (brs, 4 H), 3.52 (brs, 4 H), 2.26 (s, 3 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.63, 159.15, 158.68, 152.13, 148.71, 142.96, 136.49, 136.01, 135.98, 129.11, 129.01, 128.92, 123.58, 122.30, 121.87, 120.99, 119.62, 116.14, 115.93, 115.83, 115.61, 109.79, 66.05, 46.00, 44.03. LCMS (MM): m/z calcd for C25H25N5O [M + H]+ 411.50; found 412. HPLC area >98%.
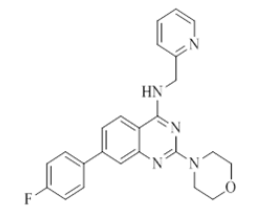
7-(4-fluorophenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (10)
Color: Brown solid. Yield: 77%. Melting point: 181°C-183°C; TLC (0.4 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.78 (brs, 1H), 8.50 (d, J=4.0 Hz, 1H), 8.18 (d, J=8.4 Hz, 1H), 7.82 (dd, J=5.6 Hz, 2.8 Hz, 2H), 7.71 (t, J=7.2 Hz, 1H), 7.52 (s, 1 H), 7.43 (d, J=7.2 Hz, 1H), 7.35-7.28 (m, 3 H), 7.24 (t, J=6.4 Hz, 1H), 4.78 (d, J=5.2 Hz, 2H), 3.63 (brs, 4 H), 3.53 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.62, 159.09, 158.36, 148.72, 144.13, 139.48, 136.50, 128.96, 128.03, 126.90, 123.60, 122.04, 121.89, 121.01, 119.88, 109.79, 66.02, 46.03, 44.08. LCMS (MM): m/z calcd for C24H22FN5O [M + H]+ 415.46; found 416. HPLC area >98%.
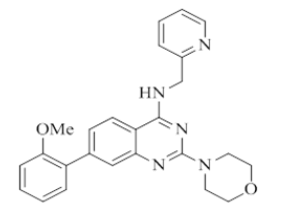
7-(2-methoxyphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (11)
Color: Brown solid. Yield: 62%. Melting point: 133°C-135°C; TLC (0.4 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.72 (t, J=5.6 Hz, 1H), 8.50 (d, J = 4.4 Hz, 1H), 8.09 (d, J=8.4 Hz, 1H), 7.71 (t, J=7.6 Hz, 1H), 7.39-7.28 (m, 4H), 7.23 (d, J=8.4 Hz, 2H), 7.13 (d, J=8.0 Hz, 1H), 7.05 (t, J=7.2 Hz, 1H), 4.78 (d, J=5.2 Hz, 2H), 3.78 (s, 3 H), 3.61-3.60 (m, 4 H), 3.54-3.53 (m, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.60, 159.24, 158.55, 156.18, 151.54, 148.70, 142.41, 136.49, 130.37, 129.31, 129.28, 125.19, 122.56, 122.21, 121.86, 120.91, 120.80, 111.81, 109.37, 66.05, 55.51, 45.96, 44.05. LCMS (MM): m/z calcd for C25H25N5O2 [M + H]+ 427.50; found 428. HPLC area >98%.
7-(4-methoxyphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (12)
Color: Brown solid. Yield: 66%. Melting point: 191°C-194°C; TLC (0.3 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.68 (brs, 1H), 8.51 (s, 1H), 8.14 (d, J=8.4 Hz, 1H), 7.72 (d, J=8.0 Hz, 3H), 7.49 (s, 1H), 7.41 (d, J=8.0 Hz, 1H), 7.34 (d, J=7.2 Hz, 1H), 7.24 (brs, 1H), 7.04 (d, J=8.4 Hz, 2H), 4.78 (d, J=4.4 Hz, 2 H), 3.81 (s, 3H), 3.64 (brs, 4 H), 3.55 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.60, 159.24, 158.55, 156.18, 151.54, 148.70, 142.41, 136.49, 130.37, 129.31, 129.28, 125.19, 122.56, 122.21, 121.86, 120.91, 120.80, 111.81, 109.37, 66.05, 55.51, 45.96, 44.05. LCMS (MM): m/z calcd for C25H25N5O2 [M + H]+ 427.50; found 428. HPLC area >98%.
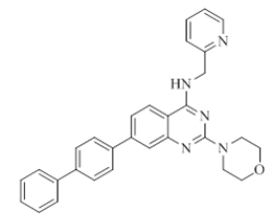
7-([1,1'-biphenyl]-4-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (13)
Color: Brown solid. Yield: 67%. Melting point: 177°C-179°C; TLC (0.3 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.50 (d, J=4.0 Hz, 1H), 8.18 (d, J=8.4 Hz, 1H), 7.85 (d, J=8.4 Hz, 2H), 7.77 (d, J=8.0 Hz, 2H), 7.71 (d, J=7.6 Hz, 3H), 7.60 (s, 1H), 7.48 (t, J=7.6 Hz, 4H), 7.39-7.35 (m, 2H), 7.24-7.21 (m, 1H), 4.80 (s, 2H), 3.67 (brs, 4 H), 3.58-3.57 (m, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.53, 159.10, 158.65, 152.09, 148.54, 143.44, 139.73, 139.56, 138.58, 136.30, 128.79, 127.39, 127.30, 127.08, 126.48, 123.46, 122.24, 121.70, 120.93, 119.47, 79.06, 78.73, 78.40, 66.09, 47.98, 47.77, 47.56, 47.35, 47.14, 44.01. LCMS (MM): m/z calcd for C30H27N5O [M + H]+ 473.57; found 474. HPLC area >98%.
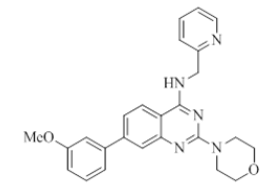
7-(3-methoxyphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (14)
Color: Brown solid. Yield: 75%. Melting point: 201°C-204°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.78 (brs, 1H), 8.50 (d, J=3.2 Hz, 1H), 8.18 (d, J=8.4 Hz, 1H), 7.71 (t, J=7.2 Hz, 1H), 7.54 (s, 1H), 7.45 (d, J=8.0 Hz, 1H), 7.41-7.24 (m, 5H), 6.97 (d, J=7.6 Hz, 1H), 4.78 (d, J=4.8 Hz, 2 H), 3.84 (s, 3H), 3.64 (brs, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.72, 159.62, 159.18, 158.64, 152.07, 148.72, 143.96, 141.08, 136.50, 129.99, 123.47, 122.47, 121.88, 120.98, 119.87, 119.24, 113.86, 112.10, 109.92, 66.05, 55.13, 45.99, 44.02. LCMS (MM): m/z calcd for C25H25N5O2 [M + H]+ 427.50; found 428. HPLC area >98%.
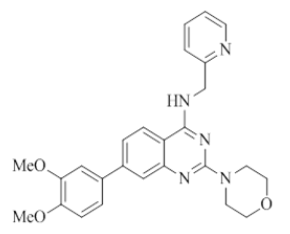
7-(3,4-dimethoxyphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (15)
Color: Brown solid. Yield: 65%. Melting point: 198°C-201°C; TLC (0.3 Rf: 60%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.73 (t, J=4.8 Hz, 1H), 8.50 (d, J=4.0 Hz, 1H), 8.15 (d, J=8.4 Hz, 1H), 7.71 (t, J=7.2 Hz, 1H), 7.53 (s, 1H), 7.46 (d, J=8.4 Hz, 1H), 7.35-7.31 (m, 3H), 7.24 (t, J=6.0 Hz, 1H), 7.04 (d, J=8.8 Hz, 1H), 4.78 (d, J=5.2 Hz, 2 H), 3.88 (s, 3H), 3.80 (s, 3H), 3.64 (brs, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.62, 159.23, 158.64, 152.15, 149.05, 148.96, 148.71, 143.93, 136.49, 132.12, 123.31, 121.87, 121.69, 120.99, 119.65, 119.20, 112.05, 110.40, 109.40, 66.06, 55.54, 55.53, 45.98, 44.05. LCMS (MM): m/z calcd for C26H27N5O3 [M + H]+ 457.52; found 458. HPLC area >98%.
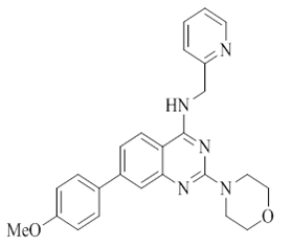
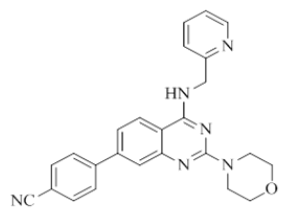
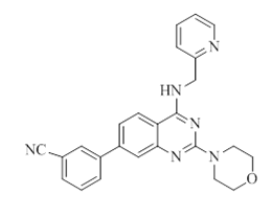
4-(2-morpholino-4-((pyridin-2-ylmethyl)amino)quinazolin-7-yl)benzonitrile (16)
Color: Brown solid. Yield: 75%. Melting point: 176°C-181°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.84 (brs, 1H), 8.50 (d, J=3.2 Hz, 1H), 8.22 (d, J=8.4 Hz, 1H), 7.98-7.87 (m, 4H), 7.71 (t, J=6.8 Hz, 1H), 7.61(s, 1H), 7.49 (d, J=8.0 Hz, 1H), 7.34 (d, J=7.6 Hz, 1H), 7.23 (t, J=5.6 Hz, 1H), 4.78 (d, J=4.8 Hz, 2 H), 3.64 (brs, 4 H), 3.53 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.58, 159.05, 158.69, 152.07, 148.73, 144.03, 142.05, 136.51, 134.20, 132.79, 131.16, 128.15, 127.85, 127.41, 123.85, 123.13, 121.90, 121.01, 119.57, 118.77, 116.36, 110.58, 110.51, 66.03, 46.02, 43.99. LCMS (MM): m/z calcd for C25H22N6O [M + H]+ 422.48; found 423. HPLC area >98%.
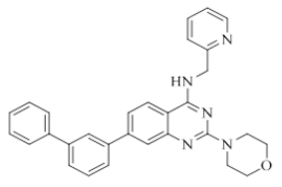
7-([1,1'-biphenyl]-3-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (17)
Color: Brown solid. Yield: 67%. Melting point: 188°C-192°C; TLC (0.3 Rf: 40%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=9.00 (brs, 1H), 8.69 (brs, 1H), 8.40 (d, J=8.0 Hz, 1H), 8.18 (s, 1H), 7.98-7.84 (m, 6H), 7.75 (brs, 2H), 7.67 (brs, 2H), 7.57-7.52 (m, 2H), 7.42 (brs, 1H), 4.98 (s, 2H), 3.83 (brs, 4 H), 3.72 (m, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.65, 159.18, 158.64, 152.11, 148.72, 143.97, 140.93, 140.26, 140.02, 136.50, 129.60, 128.89, 127.57, 126.96, 126.41, 126.06, 125.30, 123.56, 122.57, 121.88, 120.99, 119.98, 109.95 66.06, 46.02, 44.04. LCMS (MM): m/z calcd for C30H27N5O [M + H]+ 473.57; found 474. HPLC area >98%.
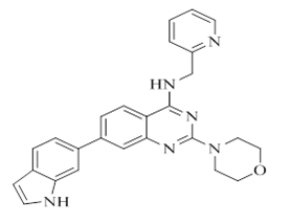
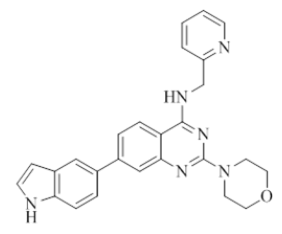
7-(1H-indol-6-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (18)
Color: Brown solid. Yield: 81%. Melting point: 166°C-169°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=11.20 (s, 1H), 8.71 (brs, 1H), 8.51 (d, J=3.6 Hz, 1H), 8.17 (d, J=8.4 Hz, 1H), 7.75-7.70 (m, 2H), 7.64 (d, J=8.4 Hz, 1H), 7.54 (s, 1H), 7.48 (d, J=8.4 Hz, 1H), 7.42 (brs, 2H), 7.35 (d, J=8.0 Hz, 1H), 7.25-7.22 (m, 1H), 6.47 (s, 1 H), 4.79 (d, J=4.8 Hz, 2 H), 3.65 (brs, 4 H), 3.56-3.55 (m, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.65, 159.26, 158.65, 152.15, 148.71, 145.59, 136.50, 136.42, 132.60, 127.59, 126.54, 123.34, 122.01, 121.87, 120.97, 120.44, 120.08, 118.33, 109.74, 109.23, 100.97, 66.31, 66.07, 45.98, 44.06. LCMS (MM): m/z calcd for C26H24N6O [M + H]+ 436.51; found 437. HPLC area >98%.
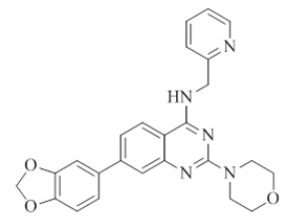
7-(benzo[d][1,3]dioxol-5-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (19)
Color: Brown solid. Yield: 88%. Melting point: 211°C-213°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=8.75 (t, J=5.1 Hz, 1H), 8.51 (d, J=4.2 Hz, 1H), 8.15 (d, J=8.4 Hz, 1H), 7.71 (t, J=7.5 Hz, 1H), 7.49 (s, 1 H), 7.42-7.33 (m, 3H), 7.28-7.22 (m, 2H), 7.01 (d, J=8.1 Hz, 1H), 6.08 (s, 2H), 4.79 (d, J=5.1 Hz, 2 H), 3.64 (brs, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.60, 159.21, 158.65, 152.11, 148.70, 148.00, 147.29, 143.74, 136.48, 133.76, 123.39, 121.88, 121.86, 120.97, 120.68, 119.66, 109.52, 108.62, 107.25, 101.20, 66.05, 45.98, 44.03. LCMS (MM): m/z calcd for C25H23N5O3 [M + H]+ 441.48; found 442. HPLC area >98%.
7-(1H-indol-5-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (20)
Color: Brown solid. Yield: 81%. Melting point: 173°C-175°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.87 (brs, 1H), 8.51 (d, J=4.0 Hz, 1H), 8.29 (d, J=9.2 Hz, 1H), 7.79 (d, J=3.2 Hz, 1H), 7.74-7.66 (m, 3H), 7.40-7.35 (m, 3H), 7.26-7.22 (m, 2H), 7.16 (t, J=7.2 Hz, 1H), 6.74 (d, J=3.2 Hz, 1H), 4.80 (d, J=5.6 Hz, 2 H), 3.65 (brs, 4 H), 3.55-3.54 (m, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.51, 159.10, 158.86, 152.91, 148.75, 142.63, 136.53, 134.75, 129.43, 128.36, 124.79, 122.54, 121.92, 121.04, 121.01, 120.56, 117.54, 116.46, 110.71, 108.68, 104.20, 66.04, 46.00, 43.97. LCMS (MM): m/z calcd for C26H24N6O [M + H]+ 436.51; found 437. HPLC area >98%.
3-(2-morpholino-4-((pyridin-2-ylmethyl)amino)quinazolin-7-yl)benzonitrile (21)
Color: Brown solid. Yield: 75%. Melting point: 135°C-13°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.82 (t, J=5.2 Hz, 1H), 8.50 (d, J=4.0 Hz, 1H), 8.26 (s, 1 H), 8.21 (d, J=8.4 Hz, 1H), 8.12 (d, J=7.6 Hz, 1H), 7.85 (d, J=8.0 Hz, 1H), 7.73-7.62 (m, 3H), 7.51 (d, J=7.2 Hz, 1H), 7.34 (d, J=8.0 Hz, 1H), 7.23 (t, J=6.4 Hz, 1H), 4.79 (d, J=5.2 Hz, 2 H), 3.64 (brs, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.59, 159.10, 158.67, 152.10, 148.73, 141.77, 140.63, 136.50, 131.69, 131.52, 130.54, 130.09, 123.79, 122.87, 121.89, 121.01, 119.54, 118.70, 112.12, 110.37, 66.03, 46.02, 44.00. LCMS (MM): m/z calcd for C25H22N6O [M + H]+ 422.48; found 423. HPLC area >98%.
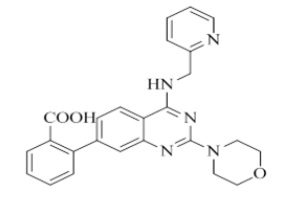
2-(2-morpholino-4-((pyridin-2-ylmethyl)amino)quinazolin-7-yl)benzoic acid (22)
Color: Brown solid. Yield: 88%. Melting point: 159°C-163°C; TLC (0.3 Rf: 50%EtOAc/Hexane); 1H-NMR (300 MHz, DMSO-d6): δ (ppm)=10.38 (s, 1H), 8.63 (brs, 1H), 8.50 (d, J=3.6 Hz, 1H), 8.06 (d, J=9.0 Hz, 1H), 7.98 (d, J=6.9 Hz, 1H), 7.88 (d, J=7.2 Hz, 1H), 7.74-7.70 (m, 1H), 7.64-7.45 (m, 4H), 7.34 (d, J=7.8 Hz, 1H), 7.26-7.22 (m, 1H), 4.77 (d, J=5.1 Hz, 2 H), 3.62-3.61 (m, 4 H), 3.54 (brs, 4 H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=165.91, 159.35, 159.27, 158.66, 152.49, 148.69, 142.76, 136.49, 134.85, 131.69, 131.17, 128.40, 128.15, 127.72, 127.40, 123.20, 121.87, 121.03, 114.41, 113.95, 106.94, 66.05, 61.99, 45.99, 44.01, 21.55. LCMS (MM): m/z calcd for C25H23N5O3 [M + H]+ 441.48; found 442. HPLC area >98%. TLC (0.3 Rf: 50%EtOAc/Hexane).
2-morpholino-7-(naphthalen-2-yl)-N-(pyridin-2-ylmethyl)quinazolin-4-amine (23)
Color: Brown solid. Yield: 75%. Melting point: 129°C-131°C; TLC (0.4 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.84 (t, J=5.2 Hz, 1H), 8.52 (d, J=4.8 Hz, 1H), 8.39 (brs, 1H), 8.25 (d, J=8.8 Hz, 1H), 8.05 (d, J=2.4 Hz, 1H), 8.03 (d, J=8.4 Hz, 1H), 7.97 (brs, 1 H), 7.95 (d, J=2.4 Hz, 1H), 7.75-7.70 (m, 2H), 7.63 (dd, J=1.6 Hz, 8.4 Hz, 1H), 7.58-7.52 (m, 2H), 7.37 (d, J=7.6 Hz, 1 H), 7.26-7.23 (m, 1H), 4.81 (d, J=5.6 Hz, 2 H), 3.66-3.65 (m, 4H), 3.56-3.55 (m, 4H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.66, 159.19, 158.66, 152.16, 148.74, 143.78, 136.74, 136.52, 133.27, 132.51, 128.47, 128.37, 127.44, 126.41, 126.35, 125.82, 125.01, 123.64, 122.64, 121.90, 121.02, 119.90, 109.92, 66.06, 46.03, 44.05. LCMS (MM): m/z calcd for C28H25N5O [M + H]+ 447.53; found 448. HPLC area >98%. TLC (0.4 Rf: 50%EtOAc/Hexane).
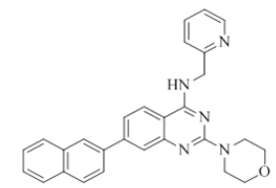
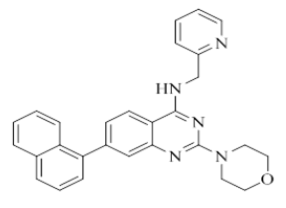
2-morpholino-7-(naphthalen-1-yl)-N-(pyridin-2-ylmethyl)quinazolin-4-amine (24)
Color: Brown solid. Yield: 71%. Melting point: 162°C-165°C; TLC (0.4 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.88 (t, J=5.6 Hz, 1H), 8.52 (d, J=4.8 Hz, 1H), 8.26 (d, J=8.4 Hz, 1H), 8.00 (d, J=8.0 Hz, 1H), 7.96 (d, J=8.0 Hz, 1H), 7.88 (d, J=8.4 Hz, 1H), 7.72 (td, 1.6 Hz, 7.6 Hz, 9.2 Hz, 1H), 7.60-7.46 (m, 4H), 7.39 (brs, 1H), 7.37 (d, J=1.2 Hz, 1 H), 7.26-7.22 (m, 2H), 4.84 (d, J=5.6 Hz, 2 H), 3.65-3.63 (m, 4H), 3.54-3.53 (m, 4H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.73, 159.20, 158.70, 151.74, 148.73, 144.25, 139.05, 136.49, 133.38, 130.61, 128.35, 127.92, 126.76, 126.41, 125.96, 125.70, 125.54, 125.21, 122.93, 122.81, 121.88, 121.01, 109.86, 66.07, 46.05, 44.04. LCMS (MM): m/z calcd for C28H25N5O [M + H]+ 447.53; found 448. HPLC area >98%.
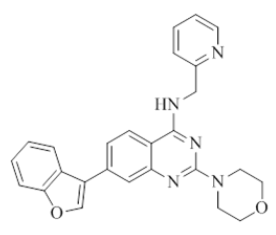
7-(benzofuran-3-yl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (25)
Color: Brown solid. Yield: 69%. Melting point: 171°C-173°C; TLC (0.4 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.81 (t, J = 5.6 Hz, 1H), 8.55 (s, 1 H), 8.51 (d, J=4.4 Hz, 1H), 8.23 (d, J=8.4 Hz, 1H), 7.99 (dd, J=2.0 Hz, 6.8 Hz, 1H), 7.75-7.66 (m, 3H), 7.53 (dd, J=1.2 Hz, 8.4 Hz, 1H), 7.44-7.35 (m, 3H), 7.26-7.23 (m, 1H), 4.80 (d, J=6.0 Hz, 2 H), 3.66-3.65 (m, 4H), 3.56-3.55 (m, 4H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.58, 159.18, 158.66, 155.27, 152.18, 148.74, 143.84, 1336.51, 135.65, 125.34, 124.84, 123.70, 123.53, 122.30, 121.90, 121.02, 120.68, 120.46, 119.87, 111.87, 109.76, 66.06, 46.01, 44.02. LCMS (MM): m/z calcd. for C26H23N5O2 [M + H]+ 437.49; found 438. HPLC area >98%.
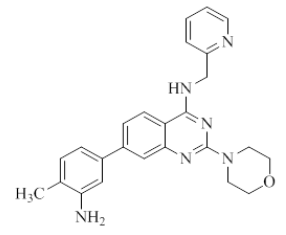
7-(3-amino-4-methylphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (26)
Color: Brown solid. Yield: 75%. Melting point: 188°C-190°C; TLC (0.5 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.73 (brs, 1H), 8.50 (d, J=4.0 Hz, 1H), 8.14 (d, J=8.4 Hz, 1H), 7.71 (t, J=7.2 Hz, 1H), 7.45 (brs, 1H), 7.36-7.30 (m, 2H), 7.24 (t, J=5.6 Hz, 1H), 7.05 (s, 1H), 7.02 (d, J=7.6 Hz, 1H), 6.87 (d, J=7.6 Hz, 1H), 4.97 (brs, 2H), 4.78 (d, J=4.8 Hz, 2 H), 3.63 (brs, 4H), 3.54 (brs, 4H), 2.10(s, 3H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=159.6, 159.2, 158.6, 152.0, 148.7, 147.0, 144.7, 137.7, 136.4, 130.4, 123.2, 121.8, 121.6, 121.2, 120.9, 119.5, 114.6, 112.2, 109.4, 66.0, 54.8, 45.9, 44.0, 40.0, 39.8, 39.6, 39.4, 39.2, 39.0, 38.8, 31.2, 29.0, 22.0, 17.1, 13.9. LCMS (MM): m/z calcd for C25H26N6O [M + H]+ 426.51; found 427. HPLC area >98%.
7-(2,4-dimethoxyphenyl)-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (27)
Color: Brown solid. Yield: 70%. Melting point: 175°C-178°C; TLC (0.4 Rf: 50%EtOAc/Hexane); 1H-NMR (400 MHz, DMSO-d6): δ (ppm)=8.73 (brs, 1H), 8.50 (d, J = 4.4 Hz, 1H), 8.07 (d, J=8.4 Hz, 1H), 7.71 (td, J=1.6 Hz, 7.6 Hz, 9.2 Hz, 1H), 7.34-7.22 (m, 4H), 6.69-6.62 (m, 2H), 6.57 (s, 1H), 4.78 (d, J=5.6 Hz, 2 H), 3.82 (s, 3H), 3.79 (s, 3H), 3.61-3.60 (m, 4H), 3.54-3.53 (m, 4H); 13C-NMR (400 MHz, DMSO-d6): δ (ppm)=160.4, 159.5, 159.2, 157.2, 148.7, 142.3, 136.5, 131.0, 122.6, 122.1, 121.8, 120.9, 108.9, 105.3, 98.9, 66.0, 55.5, 55.2, 45.9, 44.0, 28.9. LCMS (MM): m/z calcd for C26H27N5O3 [M + H]+ 457.52; found 458. HPLC area >98%.
Pharmacology
Test of antibacterial activity
A collection of three organisms including Gram-positive and Gram-negative organisms were used for this study of clinical isolates such as Escherichia coli (-ve), Klebsiella species (+ve), Pseudomonas species (+ve) were obtained from Microbiology laboratory of Global Hospital, Hyderabad. All strains were tested for purity by standard microbiological methods. The bacterial stock cultures were maintained on Mueller Hinton Agar (MHA) slants and stored at 4°C.
Determination of antibacterial activity
An agar-well diffusion method was employed for evaluation of antibacterial activity. The bacterial strains were reactivated from stock cultures by transferring into Mueller Hinton Broth (MHB) and incubating at 37°C for 18 h. A final inoculum containing 106 colony forming units (1 × 106 CFU/ml) was added aseptically to MHA medium and poured into sterile Petri dishes. Different test compounds at a concentration of 1.0 mg/50 μl were added to wells (8 mm in diameter) punched on agar surface. Plates were incubated overnight at 37°C and diameter of inhibition zone (DIZ) around each well was measured in mm. Experiments were performed in triplicates. Antibiotic such as ciprofloxacin at a concentration of 1 mg/50 μl were used as positive reference to determine sensitivity of microorganisms tested. DMSO was used as a negative control.
Results and Discussions
7-chloro-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine (compound 7) was chosen as a key starting material for the study. Compound 7 was modified at C-7 position by utilizing Suzuki–Miyaura cross-coupling reaction conditions in microwave to obtain novel substituted quinazolinones [7-9].
Synthesis of 7-substituted quinazoline compounds
The initial experiments were performed without using any ligand and were unsuccessful to prepare compound 8 to 27 (Scheme 1 and Table 1). The development of easy synthetic strategies for the construction of carbon-carbon bonds with palladium catalyzed Suzuki-Miyaura cross-coupling reactions remains an important challenge in organic synthesis. 7-Chloro-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine were chosen for the present study. The overall aim was not only to determine the best conditions for Suzuki-Miyaura cross coupling reactions, but also to find the optimal conditions that can be applied for the synthesis of a broad range of 7-aryl, heteroaryl, and biphenyl quinazolin-4-amine. Optimization experiments were carried out using Pd(PPh3)4, PdCl2(PPh3)2, PdCl2(dcpf), PdCl2(dtbpf), PdCl2(dppf), Pd(OAc)2 and Pd2(dba)3 as metal sources, ligands L1–L5 (structures were included in supporting information) were selected for combination with the metal. After trying several combinations of catalyst and ligand in different bases K2CO3, Na2CO3, Cs2CO3, K3PO4, we optimized the conditions in microwave Pd(OAc)2, X-PHOS K3PO4, THF, H2O (3: 1), 120°C, 30 min to 60 min, and prepared compound 8 to 27 in 62 to 88% yield. Experimental details and 1H-NMRs and 13C-NMRs are provided in Supplementary Information.
| S. No. | Compound ID |  |
Name of Compounds |
|---|---|---|---|
| 1 | 8 |  |
 |
| 2 | 9 | 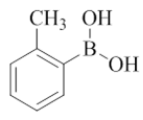 |
 |
| 3 | 10 | 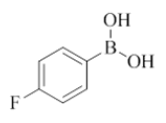 |
 |
| 4 | 11 | 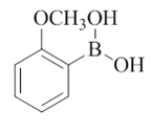 |
 |
| 5 | 12 | 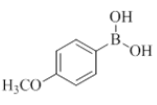 |
 |
| 6 | 13 | 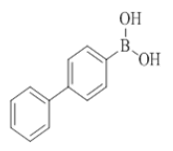 |
 |
| 7 | 14 | 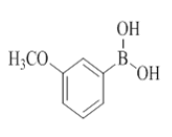 |
 |
| 8 | 15 | 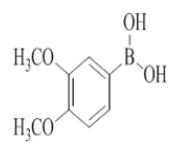 |
 |
| 9 | 16 | 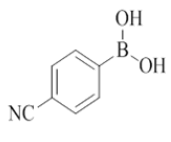 |
 |
| 10 | 17 | 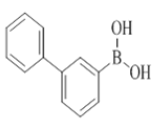 |
 |
| 11 | 18 | 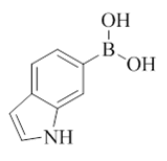 |
 |
| 12 | 19 | 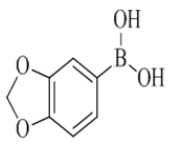 |
 |
| 13 | 20 | 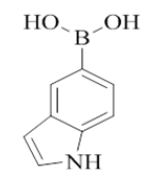 |
 |
| 14 | 21 | 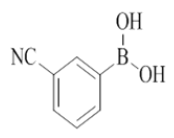 |
 |
| 15 | 22 | 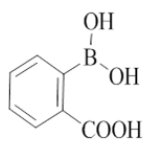 |
 |
| 16 | 23 | 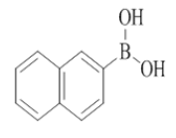 |
 |
| 17 | 24 | 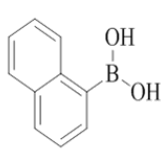 |
 |
| 18 | 25 | 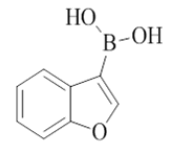 |
 |
| 19 | 26 | 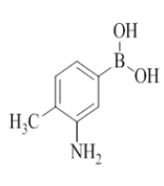 |
 |
| 20 | 27 | 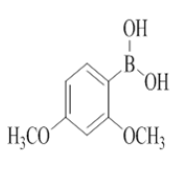 |
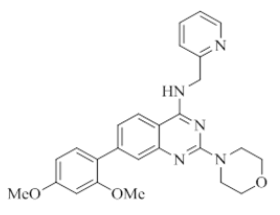 |
Table 1: List of the derivatives (8-27)
Antibacterial activity
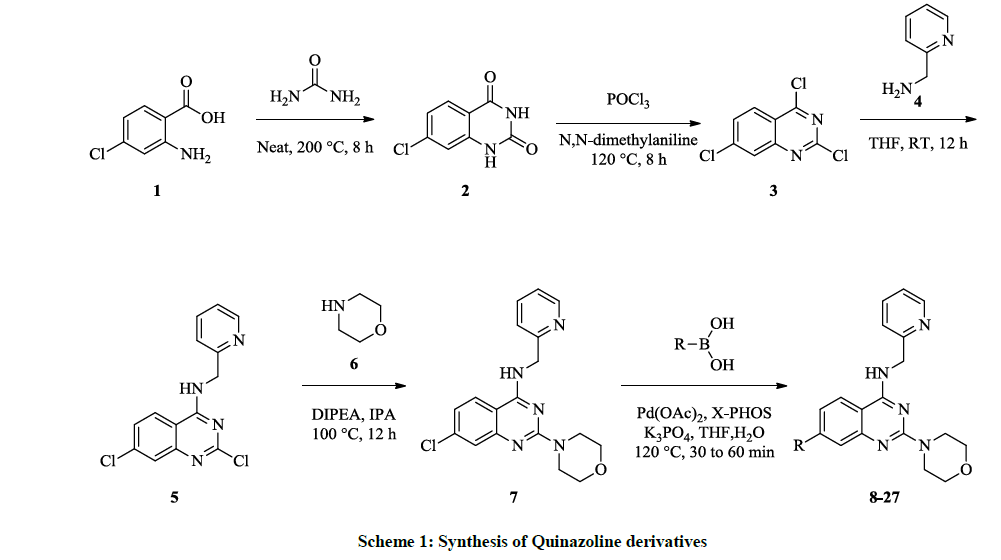
All the synthesized compounds exhibited mediocre antibacterial activity against both Gram-positive, Gram-negative bacteria and the results are summarised in Table 2 (Figure 1). Compounds 8, 16, 23, 24 and 25 displayed best activity against all the Gram-positive and Gram-negative bacterial strains. Compound 25 exhibited significant activities against all the Gram-positive, Gram-negative bacterial strains and S. aureus.
| S. No. | Compound ID | Diameter of Zone of Inhibition (mm) at Concentration of 1.0mg/50µl | |||
|---|---|---|---|---|---|
| Escherichia coli (-ve) |
Klebsiella pneumoniae (-ve) |
Pseudomonas species (+ve) |
Staphylococcus aureus | ||
| 1 | 8 | 15 | NI | 21 | 26 |
| 2 | 9 | NI | NI | 19 | 17 |
| 3 | 10 | 18 | 20 | NI | 16 |
| 4 | 11 | 13 | 15 | 13 | 20 |
| 5 | 12 | NI | NI | 9 | 19 |
| 6 | 13 | 18 | 22 | 23 | NI |
| 7 | 14 | 12 | NI | 11 | 16 |
| 8 | 15 | NI | NI | 15 | 16 |
| 9 | 16 | 25 | 24 | 22 | 28 |
| 10 | 17 | 16 | 12 | 11 | 10 |
| 11 | 18 | 13 | NI | 18 | 14 |
| 12 | 19 | 12 | NI | 11 | 16 |
| 13 | 20 | 21 | 22 | NI | 24 |
| 14 | 21 | NI | NI | 11 | 18 |
| 15 | 22 | 19 | 18 | 16 | 17 |
| 16 | 23 | 21 | 24 | 29 | 28 |
| 17 | 24 | 25 | 26 | 28 | NI |
| 18 | 25 | 30 | 33 | 34 | 36 |
| 19 | 26 | 14 | 11 | NI | 10 |
| 20 | 27 | NI | 10 | 10 | 12 |
| 21 | CPF | 32 | 35 | 33 | 37 |
| 22 | NRF | 30 | 31 | 29 | 33 |
NI: No Inhibition, CPF: Ciprofloxacin, NRF: Norfloxacin, Data are means (n=3) ± Standard deviation of three replicates
Table 2: Antibacterial activities of the compounds 8 -27
Conclusion
C-7-substituted-2-morpholino-N-(pyridin-2-ylmethyl)quinazolin-4-amine were prepared by palladium catalyzed Suzuki-Miyaura cross-coupling reactions in excellent yields and are evaluated for antibacterial activity against a panel of cell lines. Among the screened, compounds 8, 16, 23, 24 and 25 demonstrated good activity. Compound 25 is promising candidate for a broad spectrum antibacterial they can be considered as good lead compound for antibacterial agents.
Acknowledgement
We acknowledge DST (Department of Science and Technology) India and GITAM (Gandhi Institute of Technology And Science) University School of Science, departmet of chemistry,Hyderabad
References
- F. Kavanagh, Analytical Microbiology, Academic Press, New York and London, 1972, 2, 11-23.
- A.A. Miles, S.S. Misra, J. Hyg., 1938, 38, 732.
- S. Satish, K.A. Raveesha, G.R. Janardhana, Lett. Appl. Microbiol., 2002, 22, 145.
- S. Senthil Kumar, M. Kamaraj, Am. Eur. J. Agric Environ. Sci., 2010, 7, 176.
- S. Goto, H. Tsuboi, M. Kanoda, K. Mukai, K. Kagara, Org. Proc. Res. Develop., 2003, 7, 700-706.
- V.J. Ram, B.K. Tripathi, A.K. Srivastava, Bioorg. Med. Chemi., 2003, 11, 2439-2444.
- S. Goto, H. Tsuboi, M. Kanoda, K. Mukai, K. Kagara, Org. Proc. Res. Develop., 2003, 7, 700-706.
- V. Gurram, R. Garlapati, C. Thulluri, N. Madala, K.S. Kasani, P.K. Machiraju, R. Doddapalla, U. Addepally, R. Gundla, B. Patro, Med. Chem. Res., 2015, 24, 2227-2237.
- A. Ankireddy, R. Gundla, T. Balaraju, V. Banothu, K.P. Gundla, U. Addepally, J. Chimakurthy, Eur. J. Chem., 2018, 9(4), 322-330.