Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 9
Synthesis, Characterization, and In vitro Anticancer Evaluation of 7-Piperazin-Substituted [1,3]Oxazolo[4,5-D]Pyrimidines
Yevgeniya S Velihina, Maryna V Kachaeva, Stepan G Pilyo, Victor V Zhirnov and Volodymyr S Brovarets*
Department of Chemistry of Bioactive Nitrogen-Containing Heterocyclic Bases, Institute of Bioorganic Chemistry and Petrochemistry, NAS of Ukraine, 1, Murmanskaya str, Kyiv, 02094, Ukraine
- *Corresponding Author:
- Volodymyr S Brovarets
Department of Chemistry of Bioactive Nitrogen-Containing Heterocyclic Bases
Institute of Bioorganic Chemistry and Petrochemistry
NAS of Ukraine, 1, Murmanskaya str, Kyiv, 02094, Ukraine
Abstract
A novel series of five 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines have been synthesized and characterized by Infrared (IR), Proton Nuclear Magnetic Resonance (1H-NMR), Carbon-13 Nuclear Magnetic Resonance (13C-NMR) spectroscopy, elemental analysis and chromato-mass-spectrometry. The anticancer activities of the all the newly synthesized compounds were valuated via single high dose (10-5M) against 60 cancer cell lines by the National Cancer Institute according to its own screening protocol. In the next phase, the compounds have been selected for five-dose assay. Among these compounds 5-phenyl-7-piperazin-1-yl-2-p-tolyl[1,3]oxazolo[4,5-d]pyrimidine displayed the most growth inhibitory (GI50 was in range of 0.2−2.0 μM), cytostatic (TGI − 0.3-4.2 μM) and cytotoxic (LC50 − 0.6-7.8 μM with the exception of Leukemia CCRF-CEM cell line, LC50>100 μM) activities against all cancer cell lines. The most selectivity 5-phenyl-7-piperazin-1-yl-2-p-tolyl[1,3]oxazolo[4,5-d]pyrimidine demonstrated against Leukemia and Colon Cancer subpanels. These results provided evidence that compound could be useful for developing new anticancer drugs.
Keywords
7-Piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines, Synthesis, Anticancer activity, Selectivity.
Introduction
Cancer is a general term for malignant diseases characterized by uncontrolled abnormal cell growth, and the second leading cause of death after cardiovascular diseases worldwide [1,2]. The current chemotherapy of cancer produces side effects all of which are a result of drug toxicity to the normal cells. Furthermore, some of these chemotherapeutics are ineffective due to developing resistance and because of their insoluble, unstable, and low bioavailability. Anticancer drug development aims the generation of chemical structures that can control the growth of cancerous cells efficiently. Developing new effective anticancer drugs is an important strategy in cancer treatment. The most drugs belong to a class of hetero-genius structures. Heterocycles are also key structural components of many of the anticancer drugs available on the market today [3]. Heterocyclic compounds have been reported as treatments for a number of cancer and cancer related conditions [4,5]. Many of these structures, which demonstrated anticancer activity, include piperazine [6-10], and pyrimidine [11,12] pharmacophores. Compounds bearing oxazole backbone also have pharmacological applications as anticancer agents [13-17]. The idea of combining two or more potentially bioactive substructures to make the new fused heterocyclic ring systems with a higher anticancer activity is conceptually successful in the context of tumor diseases that require effective treatment.
In this paper we described the syntheses and anticancer activity of a novel class of oxazole derivatives such as 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines. The synthesized compounds were screened for their anticancer activities against full NCI 60 cell line panel.
Material and Methods
All the chemicals and solvents used for the synthesis work acquired from commercial sources, were of analytical grade, and used without further purification. Melting points were measured on a Fisher-Johns apparatus. IR spectra were recorded on a Vertex-70 spectrometer from KBr pellets. Proton Nuclear Magnetic Resonance (1H-NMR) spectra were recorded on Varian VXR-300 spectrometer (300 MHz), Varian Mercury 400 (400 MHz) or Bruker Avance DRX 500 (500 MHz) spectrometers in DMSO-d6. 13C NMR spectra for compounds 3b, 5d and 5e were obtained on a Bruker Avance DRX 500 (150 MHz) spectrometer in DMSO-d6. LC-MS analysis was conducted on an Agilent 1200 Series system equipped with a diode array and a G6130A mass-spectrometer (atmospheric pressure electrospray ionization). Combustion elemental analysis was made in the Institute of Bioorganic Chemistry and Petrochemistry analytical laboratory.
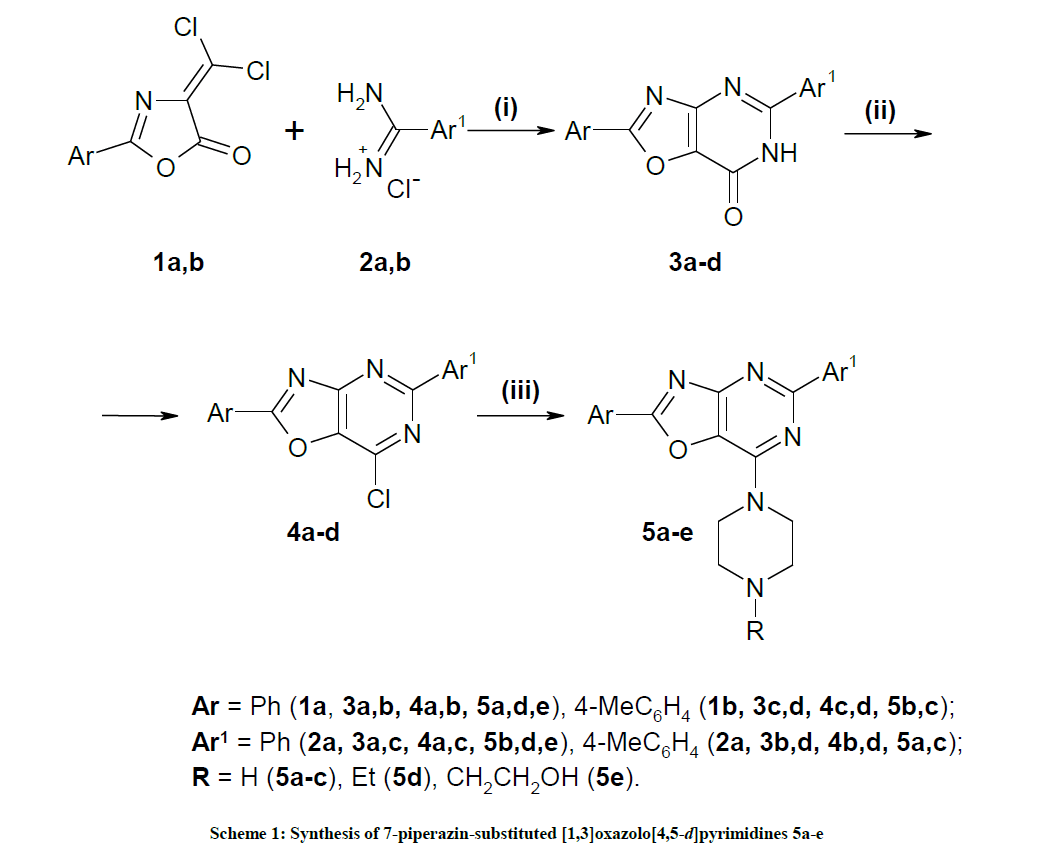
General procedure for the synthesis of 2,5-diaryl[1,3]oxazolo[4,5-d]pyrimidin-7(6H)-ones 3a-d
To a soln of 1,3-oxazol-5(4H)-one 1a,b (40 mmol) [18] in dry THF (100 ml) amidine hydrochloride 2 (40 mmol) was added followed by Et3N (5.74 ml, 41 mmol). The mixture was stirred at r.t. for 72 h. The precipitate formed was filtered off, washed with H2O, dried, dissolved in pyridine (60 ml) and refluxed for 10 h. The solvent was removed in vacuo. The residue was treated with H2O, filtered off, dried, and recrystallized from DMF.
2,5-Diphenyl[1,3]oxazolo[4,5-d]pyrimidin-7(6H)-one (3a)
Color: White solid; Yield 83%; M. P. 319-320°C; spectral and elemental analysis data are identical to literature reports [19].
2-Phenyl-5-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidin-7(6H)-one (3b)
Color: Light yellow solid; Yield 84%; M. P. 327°C-328°C; IR (KBr, cm-1) νmax: 3328-2725 (NH, Ar-CH), 1693, 1537, 1516, 1485, 1339, 918, 825, 776, 716, 685; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=12.82 (br s, 1H, NH), 8.22 (d, 2H, Ar-H), 8.07 (d, 2H, Ar-H), 7.73-7.66 (m, 3H, Ar-H), 7.40 (d, 2H, Ar-H), 2.40 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz): δ (ppm)=164.6, 158.6, 155.7, 152.0, 141.5, 132.5, 129.3, 129.2, 128.9, 127.8, 127.4, 127.3, 125.5, 20.9; MS, m/z: 304 [M+1]+; Anal. Calcd. for C18H13N3O2: C, 71.28; H, 4.32; N, 13.85. Found: C, 71.24; H, 4.30; N, 13.92%.
5-Phenyl-2-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidin-7(6H)-one (3c)
Color: Light yellow solid; Yield 82%; M.P. 343°C-345°C; IR (KBr, cm-1) νmax: 3280-2600 (NH, Ar-CH), 1691, 1541, 1493, 1339, 923, 822, 771, 742, 686; 1H-NMR (DMSO-d6, 500 MHz): δ (ppm)=13.06 (br s, 1H, NH), 8.13-8.07 (m, 4H, Ar-H), 7.61-7.56 (m, 3H, Ar-H), 7.45 (d, J = 7.0 Hz, 2H), 2.42 (s, 3H, CH3); MS, m/z: 304 [M+1]+; Anal. calcd. for C18H13N3O2: C, 71.28; H, 4.32; N, 13.85. Found: C, 71.25; H, 4.33; N, 13.77%.
2,5-Di-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidin-7(6H)-one (3d)
Color: Light yellow solid; Yield 80%; M. P. 349°C-350°C; IR (KBr, cm-1) νmax: 3271-2653 (NH, Ar-CH), 1692, 1537, 1491, 1337, 919, 824, 776, 734, 690; 1H-NMR (DMSO-d6, 500 MHz): δ (ppm)=12.97 (s, 1H, NH), 8.07 (d, 2H, Ar-H), 8.03 (d, 2H, Ar-H), 7.45 (d, 2H, Ar-H), 7.36 (d, 2H, Ar-H), 2.42 (s, 3H, CH3), 2.39 (s, 3H, CH3); MS, m/z: 318 [M+1]+; Anal. calcd. for C19H15N3O2: C, 71.91; H, 4.76; N, 13.24. Found: C, 71.88; H, 4.74; N, 13.30%.
General procedure for the synthesis of 2,5-diaryl-7-chloro[1,3]oxazolo[4,5-d]pyrimidines 4a-d
A mixture of compound 3a-d (10 mmol), POCl3 (30 ml), and Me2NPh (2.42 g, 20 mmol) was refluxed for 3 h. After evaporation of POCl3 excess the residue was recrystallized from 1,4-dioxane.
7-Chloro-2,5-diphenyl[1,3]oxazolo[4,5-d]pyrimidine (4a)
Color: White solid; Yield 87%; M. P. 219°C-220°C; spectral and elemental analysis data are identical to literature reports [19].
7-Chloro-2-phenyl-5-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (4b)
Color: White solid; Yield 86%; M. P. 237°C-239°C; IR (KBr, cm-1) νmax: 1601, 1540, 1482, 1374, 1325, 1046, 987, 784, 710, 690; 1H-NMR (DMSO-d6, 300 MHz): δ (ppm)=8.30-8.23 (m, 4H, Ar-H), 7.80-7.63 (m, 3H, Ar-H), 7.25 (d, 2H, Ar-H), 2.39 (s, 3H, CH3); MS, m/z: 322 [M+1]+; Anal. calcd. for C18H12 ClN3O: C, 67.19; H, 3.76; Cl, 11.02; N, 13.06. Found: C, 67.15; H, 3.77; Cl, 11.10; N, 13.02%.
7-Chloro-5-phenyl-2-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (4c)
Color: White solid; Yield 84%; M. P. 197°C-199°C; IR (KBr, cm-1) νmax: 1608, 1544, 1497, 1373, 1319, 1049, 984, 771, 693; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.43-8.40 (m, 2H, Ar-H), 8.20 (d, 2H, Ar-H), 7.54-7.47 (m, 5H, Ar-H), 2.48 (s, 3H, CH3); MS, m/z: 322 [M+1]+; Anal. calcd. for C18H12ClN3: C, 67.19; H, 3.76; Cl, 11.02; N, 13.06. Found: C, 67.14; H, 3.78; Cl, 11.08; N, 13.00%.
7-Chloro-2,5-di-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (4d)
Color: White solid; Yield 81%; M. P. 288°C-290°C; IR (KBr, cm-1) νmax: 1598, 1541, 1372, 1318, 1049, 990, 787, 727; 1H-NMR (DMSO-d6, 500 MHz): δ (ppm)=8.30 (d, 2H, Ar-H), 8.22 (d, 2H, Ar-H), 7.49 (d, 2H, Ar-H), 7.37 (d, 2H, Ar-H), 2.40 (s, 3H, CH3), 2.36 (s, 3H, CH3); MS, m/z: 336 [M+1]+; Anal. calcd. for C19H14 ClN3: C, 67.96; H, 4.20; Cl, 10.56; N, 12.51. Found: C, 67.93; H, 4.19; Cl, 10.61; N, 12.45%.
General procedure for the synthesis of 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines 5a-e
A mixture of compound 4 (2 mmol), appropriate piperazine derivative (2 mmol), and Et3N (0.28 ml, 2 mmol) in dioxane (15 ml) was refluxed for 6 h. After removal of the solvent, the residue was triturated with water, filtered off, dried, and recrystallized from DMF/MeCN (1: 3).
2-Phenyl-7-piperazin-1-yl-5-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (5a)
Color: White solid; Yield 77%; M. P. 256°C-258°C; IR (KBr, cm-1) νmax: 3412-2707 (NH, Ar-CH), 1611, 1548, 1371, 1307, 1053, 1021, 776, 710; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.26-8.21 (m, 4H, Ar-H), 7.70-7.61 (m, 3H, Ar-H), 7.27 (d, 2H, Ar-H), 3.98 (s, 4H, CH2 (piperazine)), 2.94-2.93 (m, 4H, CH2 (piperazine)), 2.37 (s, 4H, CH3, NH); MS, m/z: 372 [M+1]+; Anal. calcd. for C22H21N5O: C, 71.14; H, 5.70; N, 18.85. Found: C, 71.10; H, 5.67; N, 18.92%.
5-Phenyl-7-piperazin-1-yl-2-(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (5b)
Color: White solid; Yield 74%; M. P. 273°C-275°C; IR (KBr, cm-1) νmax: 3320-2690 (NH, Ar-CH), 1613, 1552, 1370, 1307, 1061, 1024, 771, 707, 695; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.38-8.36 (m, 2H, Ar-H), 8.12 (d, 2H, Ar-H), 7.49-7.43 (m, 5H, Ar-H), 3.98 (s, 4H, CH2 (piperazine)), 2.92 (s, 4H, CH2 (piperazine)), 2.43 (s, 4H, CH3, NH); MS, m/z: 372 [M+1]+; Anal. calcd. for C22H21N5O: C, 71.14; H, 5.70; N, 18.85. Found: C, 71.09; H, 5.68; N, 18.94%.
7-Piperazin-1-yl-2,5-di(4-tolyl)[1,3]oxazolo[4,5-d]pyrimidine (5c)
Color: Light yellow solid; Yield 73%; M. P. 279°C-281°C; IR (KBr, cm-1) νmax: 3380-2707 (NH, Ar-CH), 1611, 1551, 1368, 1310, 1163, 1057, 1020, 939, 783, 727; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.25 (d, 2H, Ar-H), 8.09 (d, 2H, Ar-H), 7.42 (d, 2H, Ar-H), 7.27 (d, 2H, Ar-H), 3.98 (s, 4H, CH2 (piperazine)), 2.95 (s, 4H, CH2 (piperazine)), 2.43 (s, 3H, CH3), 2.38 (s, 3H, CH3), 2.30 (s, 1H, NH); MS, m/z: 386 [M+1]+; Anal. calcd. for C23H23N5O: C, 71.67; H, 6.01; N, 18.17; Found: C, 71.64; H, 5.99; N, 18.10%.
7-(4-Ethylpiperazin-1-yl)-2,5-diphenyl[1,3]oxazolo[4,5-d]pyrimidine (5d)
Color: White solid; Yield 72%; M. P. 215°C-217°C; IR (KBr, cm-1) νmax: 3108-2622 (Ar-CH), 1618, 1547, 1375, 1311, 1164, 1056, 1018, 927, 770, 696; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.36-8.34 (m, 2H, Ar-H), 8.19 (d, 2H, Ar-H), 7.66-7.58 (m, 3H, Ar-H), 7.46-7.45 (m, 3H, Ar-H), 4.01 (s, 4H, CH2 (piperazine)), 2.58 (s, 4H, CH2 (piperazine)), 2.42 (q, 2H, CH2(ethyl)), 1.06 (t, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz): δ (ppm)=188.2, 176.2, 164.1, 161.6, 137.8, 132.7, 129.9, 129.3, 128.2, 127.8, 127.7, 127, 6, 125.6, 52.1, 51.6, 44.7, 11.9; MS, m/z: 386 [M+1]+; Anal. calcd. for C23H23N5O: C, 71.67; H, 6.01; N, 18.17. Found: C, 71.61; H, 5.98; N, 18.24%.
2-[4-(2,5-Diphenyl[1,3]oxazolo[4,5-d]pyrimidin-7-yl)-piperazin-1-yl]-ethanol (5e)
Color: White solid; Yield 75%; M. P. 220°C-222°C; IR (KBr, cm-1) νmax: 3261 (OH), 3099-2615 (Ar-CH), 1619, 1578, 1547, 1474, 1448, 1378, 1313, 1216, 1054, 1006, 935, 770, 700; 1H-NMR (DMSO-d6, 400 MHz): δ (ppm)=8.36-8.34 (m, 2H, Ar-H), 8.21-8.19 (m, 2H, Ar-H), 7.68-7.59 (m, 3H, Ar-H), 7.46-7.45 (m, 3H, Ar-H), 4.32 (br s, 1H, OH), 4.04 (s, 4H, CH2 (piperazine)), 3.63 (t, 2H, CH2CH2OH), 2.73 (s, 4H, CH2 (piperazine)), 2.58 (t, 2H, CH2CH2OH); 13C-NMR (DMSO-d6, 125 MHz): δ (ppm)=190.4, 177.7, 164.1, 161.5, 159.2, 137.7, 132.6, 129.9, 129.2, 128.2, 127.7, 127.6, 125.5, 60.0, 58.3, 52.7, 44.5; MS, m/z: 402 [M+1]+; Anal. calcd. for C23H23N5O2: C, 68.81; H, 5.77; N, 17.44. Found: C, 68.77; H, 5.75; N, 17.34%.
In vitro anticancer screening of the synthesized compounds
One doses full NCI 60 cell panel assay
Synthesized compounds 5a-e was submitted to National Cancer Institute NCI, Bethesda, Maryland, U.S.A. under the Developmental Therapeutic Program DTP. The cell line panel engaged a total of 60 different human tumor cell lines derived from nine cancer types, including lung, colon, melanoma, renal, ovarian, brain, leukemia, breast and prostate.
Primary in vitro one dose anticancer screening was initiated by cell inoculating of each 60 panel lines into a series of standard 96-well microtiter plates at 5000-40000 cells/well in RPMI 1640 medium containing 5% fetal bovine serum and 2 mm L-glutamine (day 0), and then preincubated in absence of drug at 37°C and 5% CO2 for 24 h. Test compounds were then added into the plates at one concentration of 10-5 M (day 1) followed to incubation for a further 48 h at the same conditions. Then the media were removed, the cells were fixed in situ, washed, and dried (day 3). The sulforhodamine B assay was used for cell density determination, based on the measurement of cellula r protein content. After an incubation period, cell monolayers were fixed with 10% (wt/vol) trichloroacetic acid and stained for 30 min, after which the excess dye was removed by washing repeatedly with 1% (vol/vol) acetic acid. The bound stain was resolubilized in 10 mm Tris base solution and measured spectrophotometrically on automated microplate readers for OD determination at 510 nm.
Five doses full NCI 60 cell panel assay
Cells of all 60 lines, representing nine cancer subpanels, were incubated at five different concentrations (0.01, 0.1, 1, 10 and 100 μm) of the tested compounds. The outcomes were used to create log10 concentration versus percentage growth inhibition curves and three response parameters (GI50, TGI and LC50) were calculated for each cell line. The GI50 value (growth inhibitory activity) corresponds to the concentration of the compound causing 50% decrease in net cell growth. The TGI value (cytostatic activity) is the concentration of the compound resulting in total growth inhibition. The LC50 value (cytotoxic activity) is the concentration of the compound causing net 50% loss of initial cells at the end of the incubation period of 48 h.
The three dose response parameters GI50, TGI and LC50 were calculated for each experimental compound. Data calculations were made according to the method described by the NCI/NIH Development Therapeutics Program (https://dtp.cancer.gov/discovery_development/nci-60/default.htm).
The % growth curve is calculated as:
[(T-T0)/(C-T0)] × 100
Where,
T0 is the cell count at day 0,
C is the vehicle control (without drug) cell count (the absorbance of the SRB of the control growth).
T is the cell count at the test concentration at day 3.
The GI50 and TGI values are determined as the drug concentrations result in a 50 % and 0% growth at 48 hr drug exposure. Growth inhibition of 50% (GI50) is calculated from:
[(T-T0)/(C-T0)] × 100 = 50.
The TGI is the concentration of test drug where:
100 × (T - T0)/(C - T0) = 0.
Thus, the TGI signifies a cytostatic effect.
The LC50, which signifies a cytotoxic effect, is calculated as:
[(T-T0)/T0] ×100= -50,
when T< T0.
Selectivity index (SI) of the compounds is calculated as:
SI = MIDp/MIDsp,
Where, MIDp – the average sensitivity of all cell lines towards the test agent,
MIDsp – the average sensitivity of all cell lines of a particular subpanel towards the test agent.
Results and Discussion
Chemistry
The synthesis of new 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines 5a-e depicted on scheme 1 was carried out by the route described previously [19]. Compounds 5a-e are obtained by the sequence of reactions starting from available 2-aryl-4-dichloromethylene-1,3-oxazol- 5(4H)-ones 1a,b [18]. Treating of 1a,b with arylamidine hydrochlorides 2a,b in the presence of triethylamine followed by heating with pyridine afforded the cyclocondensation products [1,3]oxazolo[4,5-d]pyrimidines 3a-d. 1H-NMR of 3a-d showed the presense of NH at 12.82-13.06 ppm. The reaction of compounds 3a-d with trichlorophosphate in the presence N,N-dimethylaniline proceeded 2,5-diaryl-7-chloro[1,3]oxazolo[4,5- d]pyrimidines 4a-d. Compounds 4a-d were converted into the corresponding 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines 5a-e by reaction with piperazines. Structures of synthesized compounds were confirmed by the IR, 1H and 13C-NMR, and GC-MS spectra. IR spectra of compounds 5a-c showed the presence of NH absorption bands in the range 3412-2690 cm-1. 1H-NMR spectra for 5a-c revealed a singlet at 2.30- 2.43 ppm due to the NH-piperazine moiety.
Reagents and conditions: (i) TEA, THF, r.t., 72 h; Py, reflux, 10 h; (ii) POCl3, Me2NPh, reflux, 3 h; (iii) piperazine derivative, TEA, dioxane, reflux, 6 h.
Biology
The one dose assay
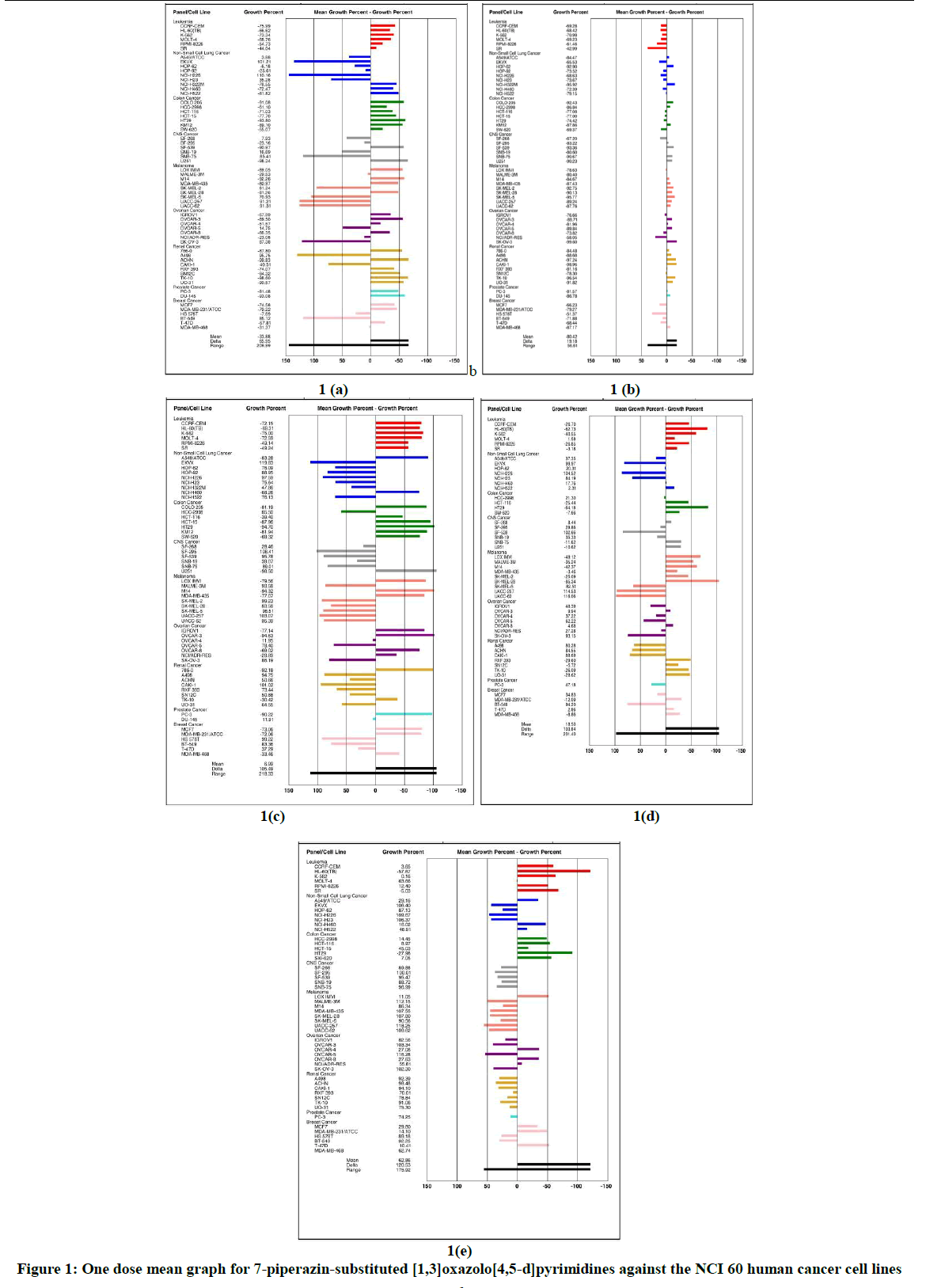
The tumor growth inhibition properties of the synthesized compounds were screened on human cancer cell lines at the NIH, Bethesda, Maryland, USA, under the drug discovery program of the NCI, for one dose anti-cancer assay. Results for each compound at a single dose concentration of 10 μM were reported as a mean graph of the percent growth of the treated cells when compared to the untreated control cells. The synthesized compounds showed a distinctive sensitivity against individual cell lines (Figure 1).
The compounds added at a concentration of 1·10-5 M and the culture incubated for 48 h. Results of each test agents are reported as percentage growth of the treated cells when compared with untreated control cells.
Compound 5a showed the growth percent ranging between -99.8 and 110.2%. The most sensitive cancer cell lines were ACHN, TK-10 and UO-31 (renal, lethality is 99.8, 98.6, and 90.6%, respectively), U-251 and SF-539 (CNS, 98.2 and 91.0%, respectively), HT-29, COLO-205 and KM-12 (colon, 93.8, 91.0 and 89.1%, respectively), DU-145 and PC-3 (prostate, 93.1% and 81.5%, respectively), M14, LOX IMVI, SK-MEL-28 and MDA-MB-435 (melanoma, 92.3, 81.3 and 81.0%, respectively), MDA-MB-231/ATCC and MCF-7 (breast, 79.2% and 74.6%, respectively), CCRF-CEM, K-562, MOLT-4 and HL-60(TB) (leukemia, 76.1, 73.3, 68.8% and 66.6%, respectively). This compound also exhibited the cell proliferation inhibition against Non-Small Cell Lung Cancer A549/ATCC (96.1%), CNS Cancer SF-268 and SNB-19 (92.1 and 83.3%, respectively), and Ovarian Cancer OVCAR-5 (85.2%) cell lines in one dose primary assay.
Compound 5b showed the growth percent ranges from -99.6 to -43.0% with a mean lethality of 80%, i.e. it displayed high cytotoxicity against all cancer lines tested. This compound revealed the highest activity (lethality ≥ 90%) against cancer cell lines of SK-OV-3 (ovarian), CACI-1, ACHN, NK-10 and UO-31 (renal), SK-MEL-5, SK-MEL-2 and SK-MEL-28 (melanoma), NCI-H322M and HOP-62 (lung), COLO-205 (colon), SNB-75 and U-251 (CNS).
Compound 5c showed the growth percent ranging from -98.5 to 119.8%, and displayed the best cytotoxicity against CNS Cancer U251 (-98.5%), Colon Cancer HT29 (-94.8%), Ovarian Cancer OVCAR-3 (-94.6%), Melanoma M14 (-94.3%), Renal cancer 786-0 (-92.2%), Prostate Cancer PC-3 (-90.2%), and Non-Small Cell Lung Cancer A549/ATCC (-83.3%) cell lines.
Compound 5d showed the growth percent ranging between -85.34 to 116.06. The most sensitive cancer cell lines were Leukemia (lethality for HL-60(TB), K-562, RPMI-8226, CCRF-CEM and SR cell lines is 62.7, 40.6, 26.9, 26.7 and 3.2%, respectively), Colon Cancer (HT-29-64.2, HCT-116-25.4 and SW-620-7.7%), CNS Cancer (SNB-75-11.6 and U251-10.6%), Melanoma (SK-MEL-28-85.3, LOX IMVI- 49.1, M14- 42.4, MALME-3M- 35.2, SK-MEL-2- 25.1 and MDA-MB-435-3.5%), Renal Cancer (RXF 393-29.6, UO-31-28.6, TK-10-26.1 and SN12C-5.7%), and Breast Cancer (MDA-MB-231/ATCC-12.1 and MDA-MB-468-8.9%). Compound 5d also exhibited the cell growth inhibition against Leukemia MOLT-4 (98.4%), Non-Small Cell Lung Cancer (NCI-H522-97.7%, NCI-H460-82.2, HOP-62-79.7 and A549/ATCC-62.6%), Colon Cancer HCC-2998 (78.7%), CNS Cancer (SF-268-91.6, SF-295-71.1 and SNB-19-64.7%), Ovarian Cancer (OVCAR-8-95.3, OVCAR-3-90.1, NCI/ADR-RES-72.7, OVCAR-4-62.8 and IGROV1-1.6%), Prostate Cancer PC-3 (52.8%), and Breast Cancer MCF-7 (65.2%) cell lines in one dose primary assay.
Compound 5e showed the growth percent ranging from -57.7 to 118.3%, and displayed the most cytotoxicity against HL-60(TB), SR (leukemia), and HT29 (colon) cell lines (-57.7, -5.0 and -27.98% respectively). This compound also showed the cell proliferation inhibition of following cancer cell lines: K-562 (99.8%), CCRF-CEM (96.3%) and RPMI-8226 (87.6%) (leukemia); NCI-H460 (84%), A549/ATCC (70.8%) and NCI-H522 (53.1%) (lung); SW-620 (92.9%), HCT-116 (91%), HCC-2998 (85.5%) and HCT-15 (55.0%) (colon); LOX IMVI (88.9%) (melanoma); OVCAR-4 (72.9%) and OVCAR-8 (72.4%) (ovarian); T-47D (89.6%) and MCF7 (70.2%) (breast). The cell lines of CNS, Renal and Prostate subpanels were practically insensitive to this drug.
The five-dose assay
All synthesized compounds satisfied the pre-determined threshold inhibition criteria of the NCI-60 One-Dose Screening were tested against the panels of 60 cancer cell lines of NCI. Figure 2 represents the results of the five-dose assay for anticancer activity of these compounds against each cancer cell line.
Note: The first column describes the subpanel and cell line involved. The next two columns list the mean optical densities (MOD) of cells at day 0 and the vehicle control, the next five columns list the MOD test for each of five different concentrations. Each concentration is expressed as the log10 (molar). The next five columns list the calculated PGs for each concentration. The response parameters GI50, TGI and LC50 were interpolated values representing the concentrations at which the PG is +50, 0 and -50 respectively. Sometimes these response parameters cannot be obtained by interpolation. If, for instance, all of the PGs in a given row exceed +50, then none of the three parameters can be obtained by interpolation. In such a case, the value given for each response parameter is the highest concentration tested and preceded by a “>” sign.
Compound 5a showed GI50 values ranging from 1.26 (Leukemia SR cell line) to 15.7 μm (Ovarian Cancer SK-OV-3 cell line), TGI – from 2.8 (Renal Cancer UO-31 cell line) to 31.6 μm (Breast Cancer BT-549 cell line), and LC50 – from 5.4 (Renal Cancer UO-31 cell line). LC50 of this compound for cancer cell lines of CCRF-CEM and RPMI-8226 (leukemia), EKVX and NCI-H23 (lung), HCC-2998 (colon), SF-268 (CNS), UACC-257 (melanoma), and HS 578T (breast) exceeded 100 μm. TGI for EKVX (lung), HCC-2998 (colon), and UACC-257 (melanoma) cancer cell lines was also more than 100 μm.
Compound 5b showed GI50 values ranging from 0.16 (Renal Cancer UO-31 cell line) to 2.0 μm (Breast Cancer HS 578T cell line), TGI – from 0.32 (Melanoma LOX IMVI and Renal Cancer UO-31 cell lines) to 4.2 μm (Breast Cancer HS 578T cell line), and LC50 – from 0.59 (Colon Cancer HCT-116 cell line) to 7.8 μm (Non-Small Cell Lung Cancer NCI-H226 cell line), with the exception of Leukemia CCRF-CEM cell line (>100 μm).
Compound 5c showed GI50 values ranging from 0.25 (Leukemia SR cell line) to 75.9 μm (Renal Cancer CAKI-1 cell line), with the exception of cancer lines with GI50 > 100 μm (Figure 2).
Level of TGI was changed from 0.74 (Leukemia SR cell line) to 38.2 μM (Renal Cancer TK-10 cell line) except cancer lines with GI50 > 100 μm. Value of LC50 was changed from 5.8 (Non-Small Cell Lung Cancer NCI-H322M and Renal Cancer UO-31 cell lines) to 83.6 (Breast Cancer BT-549 cell line) with the same exception.
Compound 5d showed GI50 values ranging from 0.47 (Renal Cancer A-498 cell line) to 9.35 μm (Ovarian Cancer OVCAR-5 cell line), TGI-from 2.5 (Renal Cancer A-498 cell line) to 11.1 μm (CNS Cancer SF-295 cell line), and LC50-from 6.25 (Colon Cancer COLO-205 cell line) to 61.1 μm (Ovarian Cancer SK-OV-3 cell line). It should be noted that LC50 for majority of cancer cell lines exceeded 100 μm, with the exception of above mentioned, and also, SF-539 and SNB-75 (CNS), LOX IMVI, M14, SK-MEL-28 and UACC-62 (melanoma), IRGOV-1 and OVCAR-3 (ovarian), 786-0 and RXF 393 (renal), BT-549 and MDA-MB-468 (breast).
Compound 5e showed GI50 values ranging from 1.77 (CNS Cancer SNB-75 cell line) to 30.0 μm (Melanoma UACC-257 cell line), TGI – from3.48 (Melanoma SK-MEL-28 and Ovarian Cancer OVCAR-3 cell lines) to 53.2 μm (Renal Cancer A-498 cell line), and LC50 – from 6.67 (Melanoma SK-MEL-5 cell line) to 8.82 μm (Leukemia HL-60(TB) cell line). LC50 of compound NSC-762197 for subpanels of Leukemia (with the exception of HL-60(TB) cell line), Non-Small Cell Lung Cancer, Colon Cancer (with the exception of COLO 205), CNS Cancer (with the exception of SF-539 and SNB-15 cell lines), Melanoma (with the exception of SK-MEL-5), Ovarian Cancer (with the exception of SK-)V-3), Renal Cancer (with the exception of CAKI-1), Prostate Cancer, Breast Cancer (with the exception of BT-549) exceeded 100 μm. Antitumor activity of compounds 5a-e against the particular cancer subpanels and selectivity index of anticancer activity of compounds show Table 2.
The order of decreasing antitumor activity of tested compounds (GI50, TGI and LC50) is: 5b > 5a > 5c > 5e ≈ 5d. It thus compound 5b exhibited the highest activity towards all tested cancer subpanels with the most selectivity to Leukemia and Colon Cancer subpanels (Table 2). The anticancer activity results showed that the presence of tolyl moiety, containing electron releasing methyl group (compound 5b), at 2 position of oxazolo[4,5-d]pyrimidine backbone instead of phenyl one (compound 5a) enhances its anticancer activity more than 5 times, while displacement of phenyl moiety at 5 position of one on tolyl group (compound 5c) reduces the activity an order of magnitude (Tables 1 and 2).
| Compound | Molecular weight | Chemical structure | Chemical name |
|---|---|---|---|
| 5a | 371.45 |  |
2-Phenyl-7-piperazin-1-yl-5-p-tolyl[1,3]oxazolo[4,5-d]pyrimidine |
| 5b | 371.45 |  |
5-Phenyl-7-piperazin-1-yl-2-p-tolyl[1,3]oxazolo[4,5-d]pyrimidine |
| 5c | 385.47 | 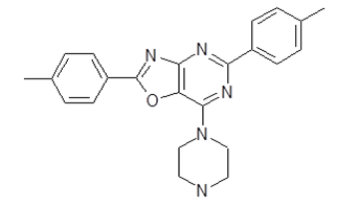 |
7-Piperazin-1-yl-2,5-di-p-tolyl[1,3]oxazolo[4,5-d]pyrimidine |
| 5d | 385.47 | 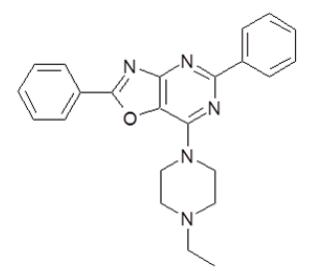 |
7-(4-Ethyl-piperazin-1-yl)-2,5-diphenyl[1,3]oxazolo[4,5-d]pyrimidine |
| 5e | 401.47 | 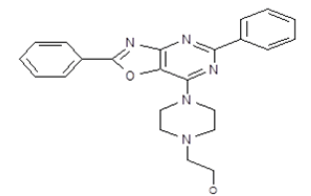 |
2-[4-(2,5-Diphenyl-[1,3]oxazolo[4,5-d]pyrimidin-7-yl)-piperazin-1-yl]-ethanol |
Table 1: Chemical structures of compounds 5a-e
| Indices | Leukemia | Non-Small Cell Lung Cancer | Colon Cancer | CNS Cancer | Mela-noma | Ovarian Cancer | Renal Cancer | Prostate Cancer | Breast Cancer | MG-MID |
|---|---|---|---|---|---|---|---|---|---|---|
| Compound 5a | ||||||||||
| GI50 | 1.7 | 2.04 | 3.24 | 1.78 | 3.16 | 2.57 | 2.19 | 1.7 | 2.63 | 2.34 |
| SIGI50 | 1.4 | 1.2 | 0.7 | 1.3 | 0.7 | 0.9 | 1.1 | 1.4 | 0.9 | |
| TGI | 3.8 | 6.17 | 5.62 | 3.47 | 5.5 | 5.13 | 4.07 | 3.16 | 5.89 | 4.9 |
| SITGI | 1.3 | 0.8 | 0.9 | 1.4 | 0.9 | 1 | 1.2 | 1.6 | 0.8 | |
| LC50 | 17.78 | 13.49 | 9.55 | 9.33 | 10.97 | 10.47 | 7.76 | 5.89 | 16.6 | 11.22 |
| SILC50 | 0.6 | 0.8 | 1.2 | 1.2 | 1 | 1.1 | 1.5 | 1.9 | 0.7 | |
| Compound 5b | ||||||||||
| GI50 | 0.22 | 0.83 | 0.3 | 0.55 | 1.32 | 0.45 | 0.5 | 0.5 | 0.55 | 0.53 |
| SIGI50 | 2.4 | 0.6 | 1.8 | 1 | 0.4 | 1.2 | 1.1 | 1.1 | 1 | |
| TGI | 0.51 | 2.09 | 0.66 | 1.26 | 2.51 | 1.12 | 1.15 | 1.66 | 1.7 | 1.29 |
| SITGI | 2.5 | 0.6 | 2 | 1 | 0.5 | 1.2 | 1.1 | 0.8 | 0.8 | |
| LC50 | 3.8 | 5.01 | 1.7 | 2.82 | 4.68 | 3.31 | 2.88 | 4.57 | 4.9 | 3.47 |
| SILC50 | 0.9 | 0.7 | 2 | 1.2 | 0.7 | 1.1 | 1.2 | 0.8 | 0.7 | |
| Compound 5c | ||||||||||
| GI50 | 0.51 | 10.96 | 1.78 | 8.71 | 9.78 | 190.55 | 6.02 | 1.7 | 10.72 | 5.01 |
| SIGI50 | 9.8 | 0.5 | 2.8 | 0.6 | 0.5 | 0.03 | 0.8 | 3 | 0.5 | |
| TGI | 1.66 | 22.39 | 5.13 | 24.55 | 17.38 | 10.72 | 12.02 | 3.31 | 27.54 | 11.75 |
| SITGI | 7.1 | 0.5 | 2.3 | 0.5 | 0.7 | 1.1 | 1 | 3.6 | 0.4 | |
| LC50 | 6.46 | 40.74 | 10.47 | 44.67 | 32.36 | 20.89 | 22.91 | 6.31 | 39.81 | 26.3 |
| SILC50 | 4.1 | 0.7 | 2.5 | 0.6 | 0.8 | 1.3 | 1.2 | 4.2 | 0.7 | |
| Compound 5d | ||||||||||
| GI50 | 2.13 | 5.13 | 3.02 | 2.46 | 4.37 | 3.24 | 2.09 | 4.68 | 2.4 | 3.09 |
| SIGI50 | 1.5 | 0.6 | 1 | 1.3 | 0.7 | 1 | 1.5 | 0.7 | 1.3 | |
| TGI | 21.38 | 30.2 | 24.55 | 17.38 | 20.42 | 18.2 | 14.45 | >100 | 8.91 | 19.5 |
| SITGI | 0.9 | 0.7 | 0.8 | 1.1 | 1 | 1.1 | 1.4 | £0.2 | 2.2 | |
| LC50 | >100 | >100 | 67.61 | 46.77 | 37.15 | 45.71 | 50.12 | >100 | 44.67 | 57.54 |
| SILC50 | £0.6 | £0.6 | 0.9 | 1.2 | 1.6 | 1.3 | 1.2 | £0.6 | 1.3 | |
| Compound 5e | ||||||||||
| GI50 | 3.02 | 2.69 | 2.63 | 2.29 | 5.62 | 2.29 | 5.01 | 3.31 | 2.09 | 3.16 |
| SIGI50 | 1.1 | 1.2 | 1.2 | 1.4 | 0.6 | 1.4 | 0.6 | 1 | 1.5 | |
| TGI | 14.45 | 25.7 | 33.11 | 14.45 | 56.23 | 20.89 | 24.55 | >100 | 4.47 | 22.39 |
| SITGI | 1.6 | 0.9 | 0.7 | 1.6 | 0.4 | 1.1 | 0.9 | £0.2 | 5 | |
| LC50 | 54.95 | >100 | 64.57 | 38.91 | 70.8 | 64.57 | 69.18 | >100 | 58.88 | 66.07 |
| SILC50 | 1.2 | £0.7 | 1 | 1.7 | 0.9 | 1 | 1 | £0.7 | 1.1 | |
Table 2: Antitumor activity of compounds 5a-e against the particular cancer subpanels: median growth inhibitory (GI50, μM), total growth inhibitory (TGI, μM), median lethal (LC50, μM), and selectivity index of anticancer activity of compounds
Conclusion
The novel series of 7-piperazin-substituted [1,3]oxazolo[4,5-d]pyrimidines have been synthesized in good yields and displayed high anticancer activity. Differently substituted oxazoles have different activity. Compound 5b demonstrated the anticancer activity against all cancer cell lines at submicromolar concentrations whereas compounds 5d and 5c had similar activity only against particular ones. The rests were active in micromolar and decimicromolar concentrations. Therefore, compounds 5b is the potent lead compounds for anticancer drug discovery and further research.
Acknowledgement
We would like to thank US Public Health Service and National Cancer Institute, Bethesda, MD, USA, for in vitro evaluation of anticancer activity (providing the NCI-60 cell testing) within the framework of Developmental Therapeutic Program (http://dtp.cancer.gov), and Enamine Ltd for the material and technical support.
Disclaimer
This material should not be interpreted as representing the viewpoint of the U.S. Department of Health and Human Services, the National Institutes of Health, or the National Cancer Institute.
References
- B. Cao, F. Bray, H. Beltrán-Sánchez, O. Ginsburg, S. Soneji, I. Soerjomataram, BMJ., 2017, 357, j2765.
- NCD mortality and morbidity, 2015.
- I. Ali, M.N. Lone, Z.A. Al-Othman, A. Al-Warthan, M.M. Sanagi, Curr. Drug Targets, 2015, 16(7), 711.
- M.M. Ghorab, F.A. Ragab, H.I. Heiba, A.M. Soliman, Eur. J. Med. Chem., 2016, 117, 8.
- P. Martins, J. Jesus, S. Santos, L.R. Raposo, C. Roma-Rodrigues, P.V. Baptista, A.R. Fernandes, Molecules., 2015, 20, 16852.
- M. Charehsaz, E.E. Gürdal, S. Helvacıoğlu, M. Yarım, Marmara Pharmaceutical Journal., 2017, 21/2, 243.
- A.A. Abdel-Aziz, A.S. El-Azab, A.M. Alanazi, Y.A. Asiri, I.A. Al-Suwaidan, A.R. Maarouf, R.R. Ayyad, T.Z. Shawer, J. Enzyme Inhib. Med. Chem., 2016, 31(5), 796.
- S. Sekar, N. Naik, A.M. Sridhara, S. Yogish, J. Pharm. Res., 2016, 10(6), 419.
- B. Chetan, M. Bunha, M. Jagrat, B.N. Sinha, P. Saiko, G. Graser, T. Szekeres, G. Raman, P. Rajendran, D. Moorthy, A. Basu, V. Jayaprakash, Bioorg. Med. Chem. Lett., 2010, 20(13), 3906.
- E.E. Gurdal, M. Yarim, I. Durmaz, R. Cetin-Atalay, Drug Res. (Stuttg), 2013, 63(3), 121.
- S.M. Ahmed, N.M. Awad, H.R. Haff, Pharmacophore., 2018, 9(1), 30.
- N.S.M. Ismail, E.M.H. Ali, D.A. Ibrahimc, R.A.T. Serya, D.A. Abou El Ella, Future Journal of Pharmaceutical Sciences (FJPS), 2016, 2(1), 20.
- X.H. Liu, H.F. Liu, C.X. Pan, J.X. Li, L.S. Bai, B.A. Song, X.F. Chu, Lett. Drug Des. Discov., 2010, 7, 238.
- R. Schobert, B. Biersack, A. Dietrich, K. Effenberger, S. Knauer, T. Mueller, J. Med. Chem., 2010, 53, 6595.
- A.S.A. El-All, S.A. Osman, H.M.F. Roaiah, M.M. Abdalla, A.A.A. El Aty, W.H. El-Hady, Med. Chem. Res., 2015, 24, 4093.
- H. Zhou, J.Q. Cheng, Z.S. Wang, F.H. Chen, X.H. Liu, Curr. Top. Med. Chem., 2016, 16, 3582.
- J.H. Suh, E.K. Yum, Y.S. Cho, Chem. Pharm. Bull. (Tokyo), 2015, 63, 573.
- B.S. Drach, G.N. Miskevich, Russ. J. Organ. Chem., 1974, 10, 2315. (Chem. Abstr., 1975, 82, 72843).
- V.M. Sviripa, A.A. Gakh, V S. Brovarets, A.V. Gutov, B.S. Drach, Synthesis., 2006, 20, 3462.