Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 3
The Relationship between Blood Counts and Serum Levels of Zinc and Copper in Children with β-thalassemia Living in Najaf Governorate, in Iraq
Rasha Shaker Nima1, Hannan Ali Ablaa1 and Hussain Jasem Mohammed2*
1Department of Laboratory Investigation, Faculty of Science, Kufa University, Iraq
2Department of Chemistry, Faculty of Science, Kufa University, Iraq
- *Corresponding Author:
- Hussain Jasem Mohammed
Department of Laboratory Investigation
Faculty of Science
Kufa University, Iraq
Abstract
Background: There are elaborate systems in the body that managing and regulating blood and stored levels of trace metals. The Supplement can prevent or decreases the severity of different diseases. While, several drugs altered zinc and copper levels, what can create different diseases.
Aim: To estimate the impact of serum micro-nutrients level; Zinc and copper on blood counts in β-thalassemia patients.
Methods: This analytical cross sectional study involved 108 diagnosed cases of β-thalassemia, (Age 5-15 year) live in the Najaf governorate in Iraq. Anthropometric data and the history therapies, as well as duration of blood transfusion were recorded. Blood samples collected for full blood count and hemoglobin electrophoresis evaluation. Zinc and copper in serum were measured by flame atomic absorption. Ferritin was analyzed by Enzyme-linked Immunosorbent Assay (ELISA) method.
Results: Serum zinc and copper levels reveled variable relations with blood counts and height for age Z-score. There were significant negative correlation between Zn values and height for age Z-score and ferritin levels. While serum copper levels were significantly associated with platelet count, Mean Corpuscular Volume (MCV), Mean Corpuscular Volume (MCH) and White Blood Cell (WBC) count.
Conclusion: The significant correlation of blood zinc level with high for age Z-score and ferritin levels may be pointed that zinc can reduce the complication of β-thalassemia. While an increase in the serum copper level can initiate the development of β-thalassemia syndrome.
Keywords
β-Thalassemia, Blood count, Serum copper level, Serum zinc level
Background
There are elaborate systems in the body that managing and regulating blood and stored levels of trace metals [1]. After iron, zinc is the second most abundant transition metal in organisms. It differs from all metal by appearing in all enzyme classes [2]. While, in every tissue of the body, copper is present. Primarily is stored in the liver. Also found, but in fewer amount of muscle, kidney, heart and brain [3]. Zinc participates in various phases of cellular metabolism. There are 200 enzymes needed zinc in their catalytic activity [4]. It plays an important role in wound healing, immune function, protein synthesis, in addition to their important structural role in zinc fingers. They are proteins that involved in RNA packaging, transcriptional activation, DNA recognition, protein folding and regulation of apoptosis [5]. In human metabolism pathway copper play an important role, because it permits many vital enzymes act properly. Copper is important in preserving the skin strength, blood vessels, epithelial and connective tissues all over the body [6]. Copper has a vital role in the synthesis of melanin, myelin, hemoglobin and also in the normal function of thyroid gland [7]. The balance between zinc and copper is more important than the concentration of them [1,8]. The Supplement can prevent or decreases the severity of different diseases. While, several drugs altered zinc and copper levels, what can create different diseases [9].
Hemoglobinopathies are the commonest inherited disorders worldwide. Unfortunately, the affected individual treatment manifests a sizable global disease burden [10]. Beta-thalassemia is characterized by a globin gene disorder by the reduced synthesis (β+) or absence (β0) of β-globin chains in hemoglobin a molecules, consequently, the excess of unbound α-globin chains in precursors of erythroid in the bone marrow and in mature erythrocytes, resulting in powerless erythropoiesis and peripheral hemolysis [11]. There are highly fluctuation in clinical features of β- thalassemia. They range from sever long lasting blood transfusion, to those with moderate and asymptomatic cases [12].
Subjects and Methods
This study was an analytical cross sectional study from 8st June 2016 to 20st May 2017. That was conducted among 108 diagnosed cases of β- thalassemia, on desfrioxamine for at least two years. They are regularly visited thalassemia center in Al-Zahraa hospital, in the Najaf governorate, in Iraq. None of the patients were chronic transfusion program at the point of recruitment. Blood samples were taken from the patients after written permission of the parent were performed, after oral simple explanation of the aim of the study. From each individual, seven milliliters of blood were drawn. From which 2.5 ml was transferred to the tube with Disodium Ethylenediaminetetraacetate Dihydrate (Na- EDTA), for full blood count determination (Hematology auto-analyzer) and hemoglobin electrophoresis via cellulose acetate hemoglobin electrophoresis. The resting 4.5 ml used for obtaining serum samples for ferritin (By Enzyme-linked Immunosorbent Assay (ELISA) method). Zinc and copper (By the atomic absorption method) estimations. It is very important to mention that the liver and kidney function tests were with the normal value.
Statistical analysis
Package for Social Science (SPSS) software version 20 was used for analyzing all data. Relationships between zinc and copper levels and blood counts were determined using the person's linear regression for bivalent correlation. National Center for Health Statistic (NCHS) growth diagram was used for determining height for age Z-score. The level of significance was set at P<0.05.
Results
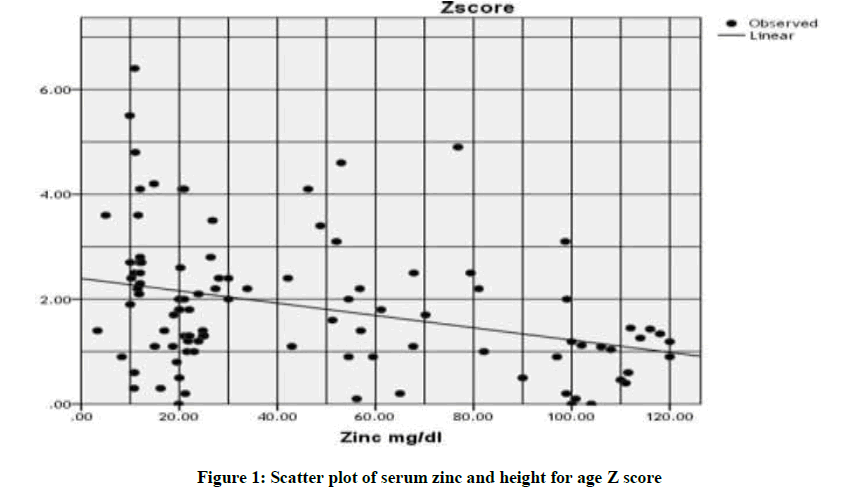
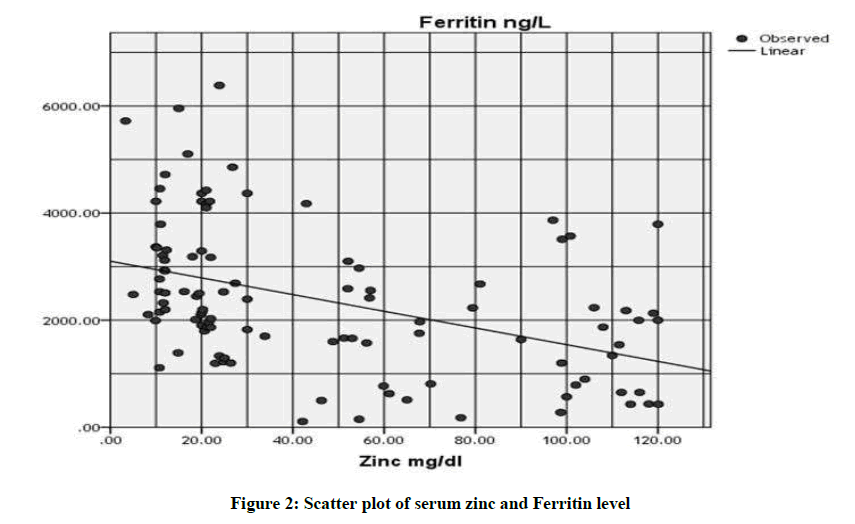
The study populations had a mean age 8.825 ± 2.664 years, involving 58 girls and 50 boys. The height for age Z-score and Body Mass Index (BMI) were (2.095 ± 1.623) and (16.076 ± 2.937) respectively. Correspondingly, Packed Cell Volume (PCV), the means of white Blood Cell Count (WBC), Red Blood Cell count (RBC), platelet count, Mean Corpuscular Volume (MCV), Mean Corpuscular Volume (MCH), Hemoglobin Concentration (Hb), Fetal hemoglobin percent and fetal hemoglobin concentration were; (24.231 ± 4.660%), (10.380 ± 57.340), (3.493 ± 1.178), (329.157 ± 142.243), (74.980 ± 7.818 FL), (24.507 ± 3.548 PG), (7.961 ± 1.431 g/dl), (62.965 ± 34.839%) and (4.972 ± 2.929 g/dl). The mean serum ferritin level was (2687.5 ± 10062 ng/dl). Mean serum zinc and copper levels were (34.510 ± 23.342 mg/dl), (97.417 ± 73.208 mg/dl) respectively (Table 1). It was found that Zinc level was significantly correlated with height for age Z-score (r2=0.112; P-value= 0.01) and with a ferritin level (r2=0.188; P-value=0.0001) (Table 2 and Figures 1, 2).
| Parameter | Patients (108) | |
|---|---|---|
| Mean ± SD | Range | |
| Age (year) | 8.825 ± 2.664 | 5-15 |
| Height for age Z-score | 2.095 ± 1.623 | 0.1 - 6.4 |
| BMI | 16.076 ± 2.937 | 8.3- 20.54 |
| PCV (%) | 24.231 ± 4.660 | 6 -31 |
| WBC | 10.380 ± 57.340 | 1.73 -37.2 |
| RBC | 3.493 ± 1.178 | 1.64 -10.45 |
| Platelets | 329.157 ± 142.243 | 25 - 627 |
| MCV (FL) | 74.980 ± 7.818 | 48.5 - 91.9 |
| MCH (PG) | 24.507 ± 3.548 | 13.3 - 29.7 |
| Hb (g/dl) | 7.961 ± 1.431 | 4.210 -11.340 |
| Ferritin (ng/l) | 2687.51 ± 1768.34 | 109 - 10062 |
| HbF (%) | 62.965 ± 34.839 | 0.4 - 98.1 |
| HbF (g/dl) | 4.972 ± 2.929 | 3.421 - 5.589 |
| Zinc (mg/dl) | 34.510 ± 23.342 | 10.75 - 111.52 |
| Copper (mg/dl) | 97.417 ± 73.208 | 19.23 - 189.3 |
| Duration of blood transfusion | 28.297 ± 14.6 | 10 - 60 |
Table 1: Demographic characteristics and full blood count parameters of study populations (Mean ± SD)
| Parameters | Correlation coefficients (r2) | P (value) |
|---|---|---|
| Zinc Vs Height for age Z-score | 0.112 | 0.01 |
| Zinc Vs PCV | 0.003 | 0.831 |
| Zinc Vs WBC | 0.037 | 0.203 |
| Zinc Vs RBC | 0.045 | 0.532 |
| Zinc Vs Platelets | 0.021 | 0.978 |
| Zinc Vs MCV | 0.058 | 0.679 |
| Zinc Vs MCH | 0.019 | 0.442 |
| Zinc Vs Hb | 0.041 | 0.342 |
| Zinc Vs Ferritin level | 0.188 | 0.0001 |
| Zinc Vs HbF (%) | 0.079 | 0.402 |
| Zinc Vs HbF (g/dl)) | 0.034 | 0.67 |
| Zinc Vs Duration of blood transfusion | 0.022 | 0.148 |
Table 2: Correlation between serum zinc and full blood count parameters
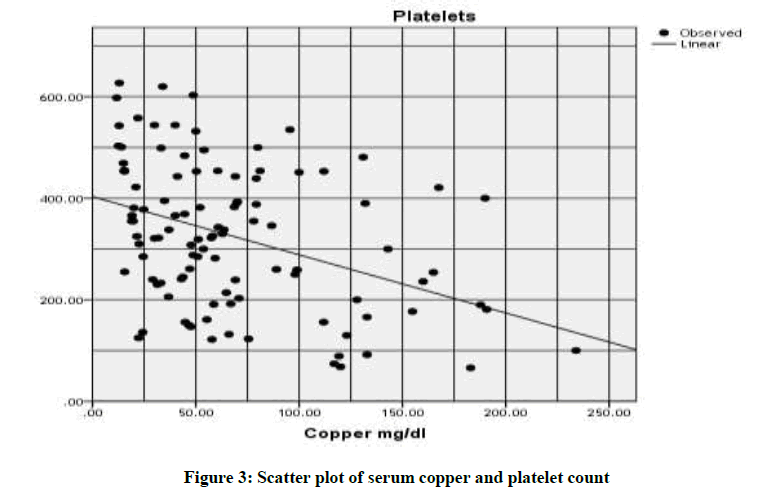
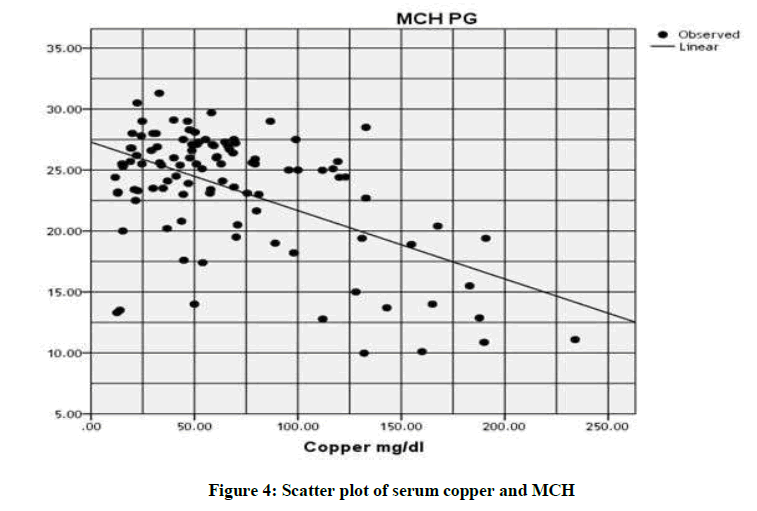
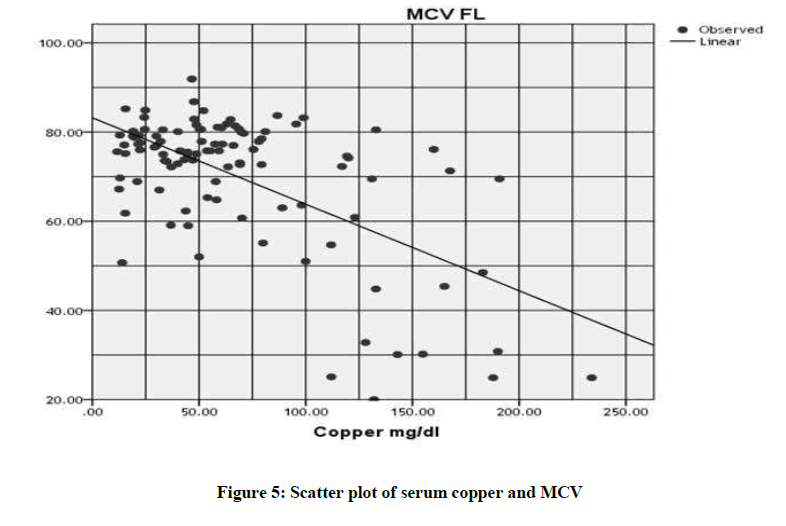
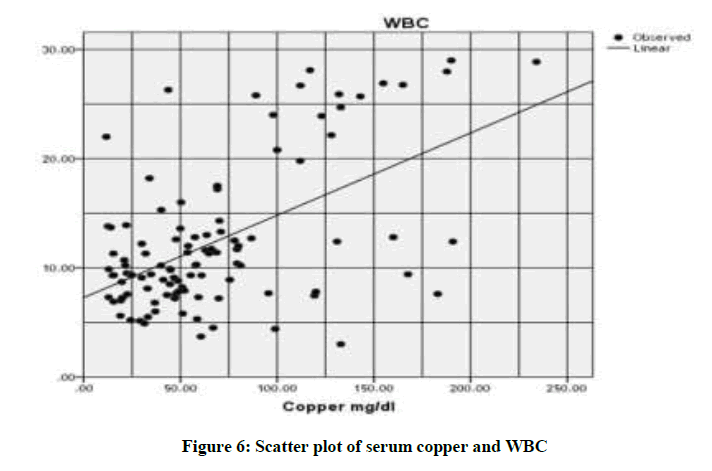
While, the correlation between copper level with platelet count (r2=0.154; P-value=0.0001), MCV (r2=0.370; P-value=0.0001) and MCH (r2=0.301; P-value=0.0001) (Table 3 and Figures 3-6).
| Parameters | Correlation coefficients (r2) | P (value) |
|---|---|---|
| Copper Vs Height for age Z-score | 0.013 | 0.328 |
| Copper Vs PCV | 0.201 | 0.744 |
| Copper Vs WBC | 0.299 | 0.000 |
| Copper Vs RBC | 0.234 | 0.653 |
| Copper Vs Platelets | 0.154 | 0.000 |
| Copper Vs MCV | 0.370 | 0.000 |
| Copper Vs MCH | 0.301 | 0.000 |
| Copper Vs Hb | 0.001 | 0.893 |
| Copper Vs Ferritin | 0.004 | 0.689 |
| Copper Vs HbF (%) | 0.017 | 0.264 |
| Zinc Vs HbF (g/dl) | 0.031 | 0.176 |
| Copper Vs Duration of blood transfusion | 0.002 | 0.689 |
Table 3: Correlation between serum copper and full blood count parameters
Discussion
In the current analytical cross-section study, serum zinc and copper levels reveled variable relations with blood counts and growth index (height for age Z-score). There was significant correlation between zinc levels and growth index and ferritin levels. Zinc is an essential microelement in the human body. It is mainly crucial for children during the growth duration. Many studies have shown that zinc plays a role in promoting physical growth and the development [13]. In β-thalassemia patients, defective synthesis of beta globin chains results in accumulation of unpaired alpha chain, that precipitates in erythroid precursors in the bone marrow and in the mature erythrocytes, leading to ineffective erythropoiesis and peripheral haemolysis [14,15]. Iron chelating treatment and regular blood transfusions are the management of β-thalassemia patients [16].
Iron chelating; desferrioxamine has a zinc binding affinity and amplify urinary zinc output, leading to gradual hypozincemia and slow growth rates [17]. Albumin and transferrin transported zinc in the blood. Therefore, accumulation iron can decrease the absorption of zinc [18]. Therefore, it is not surprising that serum zinc levels have been related with ferritin levels and height for age Z-score in our patients.
Hypercupremia is a principal complication of thalassemia [19]. Although, other studies pointed out no change in serum copper concentration [9]. Copper can act as both an antioxidant and a pro-oxidant. In the body, free radicals occur naturally. The free radicals can damage cell membrane combine with genetic material and promote of a number of health problems and diseases [20]. At the time, when copper acts as anti-oxidant, it nutralizes or scavenges the free radicals and eventually may prevent the damage they cause [21]. While, when copper acts as a pro-oxidant, it promotes free radical harmful effects [22]. In the present study, there was a significant correlation between serum copper levels and platelet counts. Although the deduction for this result was not fully clear, it is likely that copper improves destruction of platelets, by oxidant cellular damage, full filling for significant correlation resulted in this study. The important indices in the evaluation of patients with anemia are MCV and MCH. Previous studies had pointed that intracellular copper could prompt red blood cell hemolysis [23]. Several intravascular hemolysis, could create reticulocytosis; older red blood cells are known to have small size and hemoglobin content than younger red blood cells [24].
The previous information, may explain the results of a significant negative correlation between MCV and MCH with serum copper levels in the current study. The results of this study pointed out, there was a significant positive correlation between serum copper levels and white blood cells. Turnlund et al. determined the influence of copper supplementation on lymphocyte count [25]. This observation may be due to the copper's role in mediating lymphocyte maturation and controlling immune function. These results could interpret the relation between white blood cells and serum copper levels in our patients.
The serum concentration of copper in patients with thalassemia major depends on several factors including; the amount of copper intake in the daily diet, intestinal uptake of copper, iron accumulation, kidney function, copper to zinc ratio and administration of Desferal [26]. In the current study, there was a significant correlation between copper level and platelet count. Even though the reason for this observation was not entirely clear, it is possible that there could be a preferential destruction of platelets, induced by copper, via oxidant cellular damage, accounting for the significant negative correlation observed in this study.
The MCH represents the amount of hemoglobin per red blood cell and is an important index in the evaluation of patients with anemia [23]. Studies have shown that intracellular copper could trigger red cell hemolysis (Presumably through the generation of superoxide ions in the presence of sulfhydryl groups), as evident in Wilson’s disease [27]. The pre-existing chronic hemolytic anemia state of SCA could thus be potentially exacerbated by concurrent copper induced red cell lysis. Intravascular hemolysis, particularly when severe, could cause reticulocytosis; younger red blood cells are known to have higher hemoglobin content (Higher MCH) than older red cells [28]. The above explanation may thus explain the finding of a significant positive correlation between serum copper levels and MCH in this study. We did not observe any significant correlation between serum copper and mean cell volume (MCV) and neutrophil counts. This is in contrast to the report of Prasad et al. [29]. The reason for this discrepancy could not be explained from this study. There was a significant positive correlation between serum copper levels and blood lymphocyte count. Turnlund et al. reported a significant increase in circulating lymphocytes in individuals on copper supplementation [25]. This may be due to the established role of copper in mediating normal lymphocyte maturation and regulating immune function [30]. These observations could explain the association between serum copper and blood lymphocytes in this study.
Conclusion
The results of the current study highlighted the fact that zinc can reduce the complication of β-thalassemia. While an increase in the serum copper level can initiate the development of β-thalassemia syndrome.
References
- J. Osredak, N. Susta, J. Clinc. Toxicol., 2011, 3.
- H.G. Classen, U. Grober, D. Low, J. Schmidt, H. Stracke, Med. Monatasschr. Phirm., 2011, 34, 87-95.
- R.M. Lianos, J.F. Mercer, DNA cell Biol., 2002, 21, 259-270.
- N. Roohani, R. Hurrell, R. Kelishadi, R. Schulin, J. Res. Med. Sci., 2013, 18(2), 144-157.
- J. Jen, Y.C. Wang, J. Biomed. Sci., 2016, 23, 53.
- A. Morrell, S. Tallino, L. Yu, L. Jason, J.L. Burkhead, IUBMB Life., 2017, 69(4), 263-270.
- J.H. Laity, B.M. Lee, P.E. Wright, Curr. Opin. Struct. Biol., 2001, 11(1), 39-46.
- M. Malavolta, F. Piacenza, A. Basso, R. Giacconi, L. Costarelli, E. Mocchegiani, Mech. Ageing Dev., 2015, 151, 93-100.
- A.F. Mahyar, P. Ayazi, A. Asghar, H. Mojabi, M.R. Sehhat, A. Javadi, Iran J. Pediatric., 2010, 20(3), 297-302.
- P. Kountouris, C.W. Lederer, P. Fanis, X. Feleki, J. Old, M. Kleanthous, PloS One., 2014, 9(7), e103020.
- V.D. Sanctis, C. Kattams, D. Canatan, A.T. Soliman, H. Elsedfy, M. Karimi, S. Daar, Y. Wali, M. Yassin, N. Soliman, P. Sobti, S. Al-Jaouni, M. Kholy, B. Fiscina, M. Angastiniotis, Mediterr. J. Hematol. Infect. Dis., 2017, 9, e2017018.
- A. Cao, R. Galanello, Genetics in Medicine., 2010, 12(2), 61-76.
- R. Jha, S. Jha, J. Pathol. Nepal., 2014, 4, 663-671.
- H.B. Brom, J.M. Peerson, J. Rivera, L.H. Allen, Am. J. Clin. Nutr., 2002, 75, 1062-1071.
- R. Galanello, R. Origa, Orphanet J. Rare. Diseases., 2010, 5, 11.
- M. Petrou, Indian J. Hum. Genet., 2010, 16, 1-5.
- S. Sultan, S.M. Irfan, J. Kakar, R. Zeeshan, Malaysian J. Pathol., 2015, 37(1), 35-38.
- K. Orino, Biometals., 2017, 30(4), 615-621.
- N.S. Al-Allawi, K.M. Hassan, A.K. Sheikha, F.F. Nerweiy, R.S. Dawood, J. Jubrael, Mol. Biol. Int., 2010, 1-4.
- X.L. Zuo, J.M. Chen, X. Zjou, X.Z. Li, G.Y. Mei, Biotrace Elem. Res., 2006, 114(1-3), 41-53.
- A.C. Sande, E.M. Alvaez, R.S. Otero, J. Nerochem., 2017, 141(5), 738-749.
- M. Lisa, Toxicology., 2003, 189(1-2), 147-163.
- S. Tasneem, N. Sultana, A. Snoverm, K. Alan, Rawal Medical Journal., 2016, 41(4), 424-427.
- R. Malka, F.F. Delgado, S.R. Manalis, J.M. Higgins, PLoS Comput. Biol., 2014, 10(10), e1003839.
- J.R. Turnlund, R.A. Jacob, C.L. Keen, J.J. Strain, D.S. Kelley, J.M. Domek, W.R. Keyes, J.L. Ensunsa, J. Lykkesfeldt, J. Coulter, Am. J. Clin. Nutr., 2004, 79(6), 1037-1044.
- G. Rakhra, D. Masih, A. Vats, S.K. Verma, V.K. Singh, R.T. Rana, V. Kirar, S.N. Singh, Nutrition., 2017.
- E.A. Roberts, M.L. Schilsky, Hepatology., 2008, 47, 2089-2111.
- R.S. Franco, Am. J. Hematol.,2009, 84(2), 109-114.
- A.S. Prasad, J. Ortega, G.L. Brewer, D. Oberleas, E.B. Schoomaker, JAMA., 1976, 235(22), 2396-2398.
- S.L. Jarosz, A. Marek, M. Kwiecien, Z. Gradzki, B. Kaczmarek, J. Animal Physiol. Animal Nutr., 2017.