Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 4
Thermodynamic and Kinetic Studies for Adsorption of Methyl Violet Dye on to Creeson Seeds
Huda N Al-Ani*
Department of Chemistry, College of Science, University of Baghdad, Jadiriya, Baghdad, Iraq
- *Corresponding Author:
- Huda N Al-Ani
Department of Chemistry
College of Science
University of Baghdad
Jadiriya, Baghdad, Iraq
Abstract
The present investigation describes the adsorption of Methyl Violet (MV) dye onto Creeson (Lepidium sativum) (LS) seeds known as Garden cress from its aqueous solution, has been known centuries ago in eastern regions then spread worldwide. The using of creeson seeds as adsorbent for the removal MV dye was investigated in aqueous solution. Effect of experimental coefficient like connection time, primary concentration of adsorbate (MV) and amount of adsorbent seeds (LS) dose and temperature were evaluated to obtain the optimum condition for adsorption of MV onto Cresson seeds using batch adsorption. The adsorption information were mathematically analyzed using isotherms of adsorption like Langmuir, Freundlich and Dubinin-Radushkevich isotherms to study adsorption mechanism of MV dye onto creeson seed. The isotherms of adsorption were advanced and equilibrium data adjusted well to Freundlich isotherm model. The studies of kinetic indicated that the process of adsorption followed the model of second-order kinetic. The quantities (ΔG0, ΔH0 and ΔS0) were estimated and the obtained ΔG° negative values indicate a spontaneous adsorption process, ΔH° value obtained positive designate to endothermic properties of adsorption process and ΔS° value obtained positive during the adsorption indicate increased randomness.
Keywords
Creeson seed, Methyl violet dye, Adsorption kinetics
Introduction
Artificial dyes of great extent assortment and amount are used by various industries for coloring their eductors [1-3]. Plausible quantity of the dyes appended in the practicability to be left over semifinished and finally find their way to water collection. Presence of dyes in a water collection position earnest cause of danger to the environment like the change in color, nature of the water and makes it infelicitous for human exhaustion. The growing environmental problems caused by dye flowing out have driven plausible research exertions to decrease of dye wastewater. Methyl Violet (MV) is in particular important because of its extensive appliance in antibacterial [4], temper inks, textiles, bacteria categorization and in paints [5,6]. The intake of MV may cause excitation to the breathing apparatus, and swallowing exemplary causes distress to the digestive system [7]. Furthermore, MV dye is rebellious and hard to control to demean because of the existence of three aryl groups, which each one is bonded to N2 atom that interplay with one or two CH3 groups Figure 1 [8]. An extensive range of technologies had been developed for removal by artificial dyes with aqueous solutions to diminution their effect in the environment, inclusive membrane filtration processes [9], adsorption mechanics [10], clotting [11], developed processes of oxidation [12] and processes of ozonation [13]. Between the above-referred to techniques, sorption is higher quality to the different mechanisms in terms which have a low cost, elasticity, modesty of a plan, case of procedure and insensitivity to toxic contaminating [14]. The adsorption is one of the greatest qualified mechanisms can be valid for MV dye removal [15,16] and the mercantile adsorbents as plant seeds are costing much, to examine is perform by some researchers for recognizing low-priced adsorbents [17].
Agricultural by yields have been considerably studied for pollutant, these contain peat, wood, pine bark, peanut shells, plant seeds, wool, banana pith, compost and leaves [18]. Cresson seeds can employ like an origin of agriculturally-based. The seeds swell upon moistening and once swollen comprise of a rigid core with a cancellate swollen external layer. The gummous layer of blown seeds is a pectinous template, made up of plausible amounts of unesterified galacturonic acid with a large capaciousness of hydration [19,20]. The aim of this study is using Cresson seeds as an adsorbent for the polltant MV dye from waterish solution; study the effect of specific coefficients as, elementary concentration of MV, connection time, adsorbent dose and the temperature for maximum removal of MV dye in its aqueous solutions using UV-visible spectrophotometer; determined the kinetic equation best describe the data obtained; determined the adsorption equation which best describe the equilibrium uptake and finally to calculate the thermodynamic coefficients (ΔHo, ΔGo and ΔSo) for practical execution to Creeson seeds. Study the mechanisms of adsorption proses.
Experimental Section
Materials
Sorbent
Cresson seeds (Lepidium sativum seeds), brownish red in color, were purchased from the local market, prepared as whole seed, the plants producing these seeds grown extensively in Iraq Figure 2. The required quantity of seeds were washed with distilled water then allowed to swell for 30 min and directly used as adsorbent
Sorbate
The commercial Analytical MV dye appearance green to dark-green powder[with Molecular formula (C25H30ClN3) and Molecular weight- 407.986 g/mol; λmax was 590 nm, Mono isotopic mass 407.212830 Da, EC Number 208-953-6, Melting point: 205 to 215°C, Soluble in water and ethanol, CAS Registry Number: 548-62-9] was used for the preparation of stock solution of 40 mg/l, with dissolving 0.01 g from MV dye with 250 ml distilled water. A series of diluted solutions were prepared (30, 20, 10 mg/l) by fresh diluting of the stock solution, to compute the best λmax using UV-Visible spectrophotometer.
Methods
The experiments of adsorption were executed using batch equilibrium method [21]. Swollen seeds were taken in 250 ml round bottom flask containing 125 ml of MV dye solution. The flasks were placed on a hot plate with magnetic stirrer (Bibby Strlintd, UK) and shaken at 150 rpm at 25°C, Finally predetermined time intervals, adsorbent was dismissed by centrifugation with 150 rpm, the supernatant assayed using UV-visible spectrophotometer. Seeds uptake by the adsorbent at equilibrium (Qe) was computed by following equation:
Qe = (C0 – Ce) V/W (1)
Where, Qe=Amount sorbed in (mg/g) of MV dey, C0=Initial concentration in (mg/l) of MV dey, Ce=Equilibrium concentration in (mg/l) and V=Total volume of solution in (litre) and W=Adsorbent used mass of (Cresson seeds) (gram) [22,23]. Removal percentage or adsorption percentage (%R) were calculated using Equation 2:
%R = [(C0 – Ce)/C0] × 100 (2)
Results and Discussion
Effect of adsorption parameters
Adsorbent
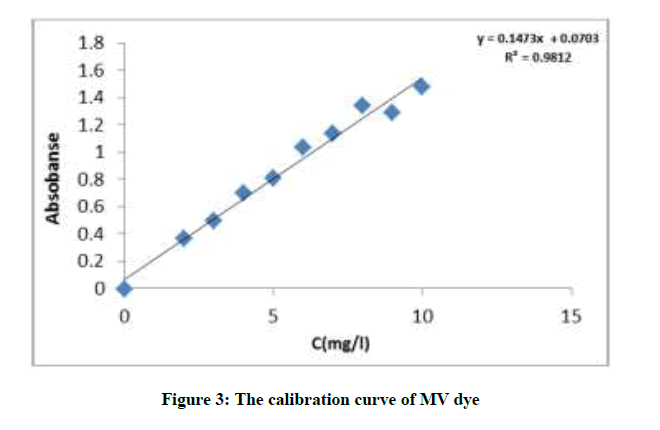
The chemical world phenomena and porous structure of adsorbent generally determines sorption activity. The results for the dye uptake using various amounts were carried out by prepared different concentration of MV dye (2, 3, 4, 5, 6, 7, 8, 9 and 10 mg/l) and calculated the absorbability using UV-Visible spectrophotometer then plot the calibration curve Figure 3.
Effect of connection time
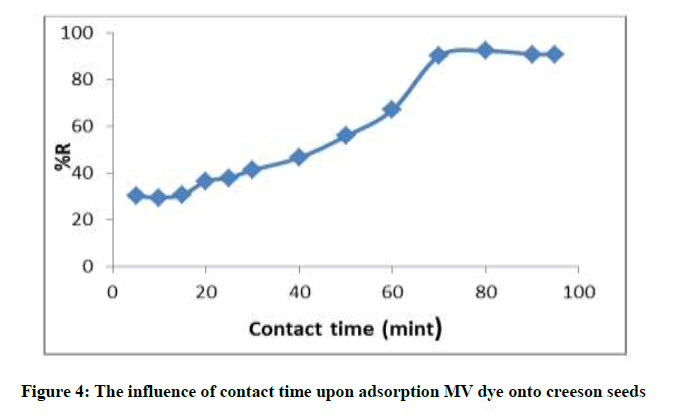
Cresson seeds mixture with (0.5 g) and (125 ml) volume solution with initial concentration (7 mg/l) of MV dye at 25°C for different time (5, 10, 20, 30, 40, 50, 60, 70 and 80 min) and centrifuged. MV concentration were determined as above the contact time effect on adsorption of MV are demonstrated in Figure 4, the equilibrium was attained after shaking for 80 min, there for 70 min was accepted as optimum time for adsorption of MV on Cresson seeds, additionally growth in contraction time did not exhibit any increase in adsorption due to a saturation in a surface sites [24].
Effect of adsorbent doses
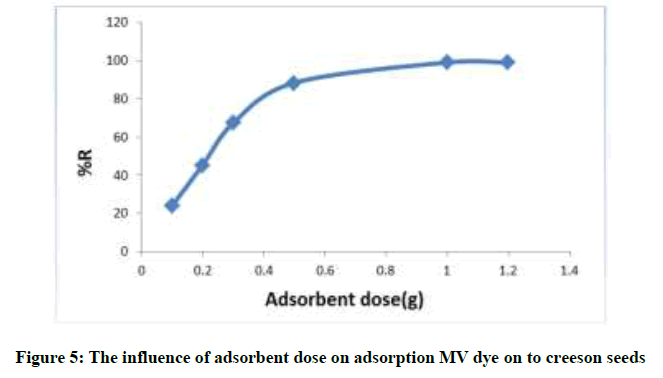
A primary concentration MV dye (40 mg/l) were used in conjunction with different amount of swollen creeson seeds of (0.1, 0.3, 0.5, 1 and 1.2 g), the other parameters were kept constant; contact time 70 min, agitation speed 150 rpm; temperature 40°C. MV uptake was found to increase with increase in LS dosage up to 1.2 g of seeds. Therefore optimum creeson (LS) dose was chosen as 1 g for the subsequent experiment as show in Figure 5.
Effect of primary concentrations
Experiential evaluates for adsorption by different concentration of MV (3, 4, 7, 8, 9 and 10) mg/l are shown in Figure 6. Such as the concentration for MV increase, more and more sites of surface are covered at higher concentration, the capacity of adsorbent obtain consumed caused by non-availability of surface location [21].
Effect of temperature
Experiments were executed at different temperature 25, 30, 35, 40 and 45°C in conjunction with the optimum other parameters, contact time 70 min, adsorbent dose 1 g, agitation speed 150 rpm Figure 7 shows that at 40°C there are the best removal R% and so this temperature was chosen for further experiments.
Adsorption isotherms
To determine the adsorption capacity and potential for selecting the adsorbent for elimination of MV dye, the topic of adsorption isotherm is essential in selecting the adsorbent. From the batch experiment executed, the most efficient state parameters chosen were; Cresson seeds dose 1 g, contact time 70 min and agitation speed 150 rpm. Adsorption isotherm study was carried out by five different temperatures which were 25, 30, 35, 40 and 45°C. The most common isotherm model was employed for describing the adsorption data, which was adsorption isotherm Figure 8.
The equilibrium values obtained are depicted in Table 1.
| At 25°C | At 30°C | At 35°C | At 40°C | At 45°C | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ce | Qe | Ce/Qe | Ce | Qe | Ce/Qe | Ce | Qe | Ce/Qe | Ce | Qe | Ce/Qe | Ce | Qe | Ce/Qe |
| 0. 033 | 0. 065 | 0. 505 | 0. 179 | 0. 064 | 0. 102 | 0. 027 | 0. 263 | 0. 102 | 0. 141 | 0. 052 | 2. 424 | 0. 130 | 0. 042 | 3. 076 |
| 0.062 | 0.075 | 0.744 | 0.224 | 0.062 | 1.468 | 0.170 | 0.116 | 1.468 | 0.131 | 0.047 | 2.787 | 0.1812 | 0.058 | 3.103 |
| 0.555 | 0.199 | 3.022 | 0.558 | 0.112 | 4.169 | 0.771 | 0.185 | 4.169 | 0.711 | 0.237 | 2.999 | 0.659 | 0.238 | 2.768 |
| 0.600 | 0.303 | 3.202 | 0.803 | 0.158 | 1.687 | 0.331 | 0.196 | 1.687 | 0.525 | 0.153 | 3.438 | 0.682 | 0.209 | 3.267 |
| 1.971 | 0.346 | 3.916 | 1.912 | 0.323 | 2.525 | 1.327 | 0.525 | 2.525 | 1.391 | 0.346 | 4.0179 | 1.399 | 0.379 | 3.684 |
| 2.110 | 0.454 | 4.867 | 1.925 | 0.291 | 4.494 | 1.591 | 0.429 | 4.494 | 1.951 | 0.383 | 5.094 | 1.632 | 0.485 | 3.367 |
| 1.810 | 0.304 | 5.950 | 2.109 | 0.272 | 2.266 | 1.156 | 0.510 | 2.265 | 1.150 | 0.316 | 3.636 | 1.626 | 0.458 | 3.654 |
| 2.170 | 0.276 | 7.876 | 3.129 | 0.315 | 9.331 | 3.000 | 0.369 | 9.331 | 2.320 | 0.514 | 4.518 | 0.950 | 0.300 | 3.166 |
| 1.590 | 0.199 | 7.969 | 1.701 | 0.185 | 2.176 | 1.129 | 0.519 | 2.176 | 2.388 | 0.543 | 4.402 | 2.871 | 0.662 | 4.340 |
| 3.626 | 0.276 | 14.038 | 2.131 | 0.251 | 3.006 | 0.714 | 0.237 | 3.006 | 0.741 | 0.182 | 3.071 | 1.434 | 0.407 | 3.526 |
| 3.990 | 0.269 | 14.805 | 4.330 | 0.363 | 7.458 | 2.506 | 0.352 | 7.458 | 1.911 | 0.471 | 4.124 | 2.591 | 0.572 | 4.532 |
Table 1: Equilibrium parameters on adsorption of MV dye on to cresson seeds
Langmuir adsorption isotherm
The Langmuir adsorption isotherm is legal for monolayer adsorption on to a level with a limited number of homogeneous situation [24]. It is founded on evaluation of adsorption identity, such as evenly plentiful adsorption locale, monolayer surface covering and no influence between adsorbed type [25,26]. In agreement with the Langmuir isotherm, the adsorption practicability be able to declared as:
Ce/Qe = Ce/Qm + KL/(Qm) (3)
Ce in (mg ⁄l) is equilibrium concentration in solution of MV dye, Qe in (mg/g)=Adsorbed amount at equilibrium per unit weight, Qm in (mg ⁄g)=The adsorption ability at the highest level denominated effective dissociation constant. The linear diagram of Ce⁄Qe VS. Ce propose the suitability of Langmuir isotherm (Figure 9). The assess of Qm and KL were computed by the slope and intercepts of the charts are listed in (Table 2). Langmuir constants relates to adsorption ability and rate of adsorption consecutively.
| T (°K) | Langmuir isotherm results | ||
|---|---|---|---|
| Qm | K L | R2 | |
| 298 | 0.275 | 9.676 | 0.963 |
| 303 | 0.478 | 0.630 | 0.878 |
| 308 | 0.479 | 2.762 | 0.831 |
| 313 | 1.103 | 0.352 | 0.840 |
| 318 | 2.109 | 0.167 | 0.795 |
Table 2: Langmuir parameters of adsorption isotherm with their correlation coefficient on adsorption of MV dye on cresson seeds
The linearity of the plots (0.795 → 0.963) at different temperature as show in Table 2 in Langmuir isotherm model.
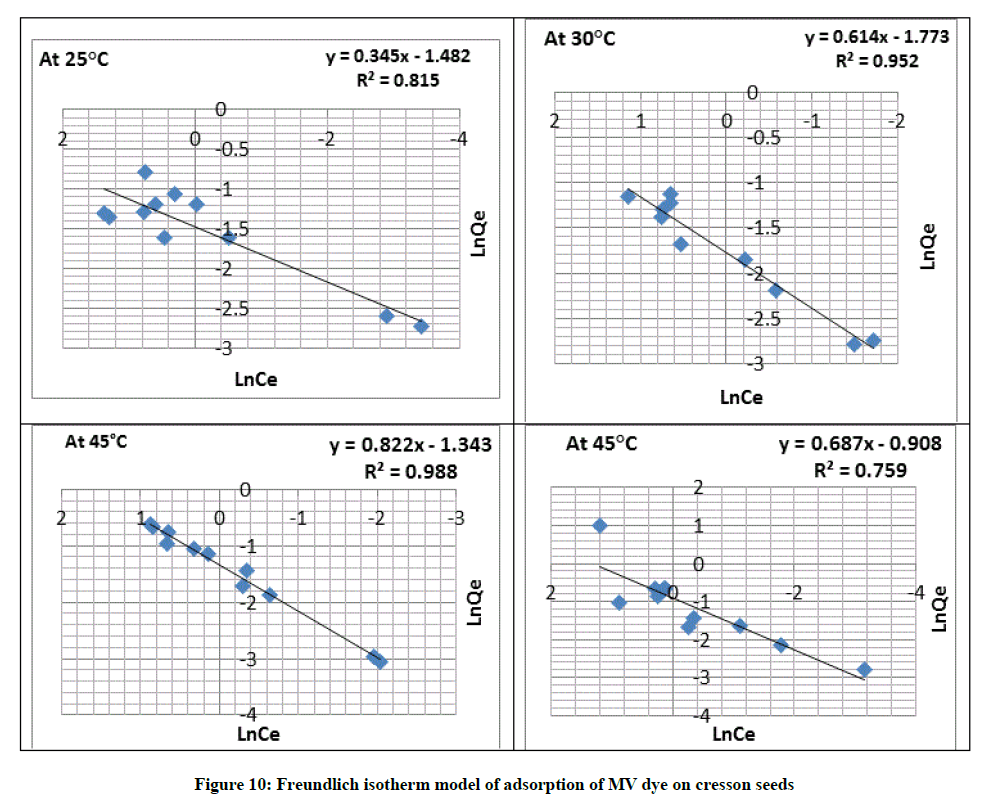
Freundlich adsorption isotherm
Batch isotherm data fitted to the linear format with the Freundlich adsorption isotherm (Table 3) which is commonly expressed by sequential Equation [27].
| Freundlich isotherm Results | |||
|---|---|---|---|
| T (°K) | Kf | n | R2 |
| 298 | 0.227 | 2.899 | 0.815 |
| 303 | 0.169 | 1.629 | 0.952 |
| 308 | 0.135 | 2.041 | 0.783 |
| 313 | 0.261 | 1.217 | 0.988 |
| 318 | 0.403 | 1.456 | 0.759 |
Table 3: Freundlich isotherm model parameters to the adsorption of MV dye on cresson seeds
LnQe=LnKf + (1/n) Ce (4)
Where, Kf and n are computed from Figure 10 and data are provided in Table 2. These are signal of the adsorption capacitance and adsorption strength consecutively. The linearity of the plots (0.759 → 0.988) at different temperature is evident and the result show that (n) is more than (1) that indicates that adsorption is favorable as show in Table 4, favorable sticking of adsorbate to adsorbent [28]. This assistance the suitability of Freundlich isotherm indicating that adsorption by swollen Creeson seeds may be governed by physisorption. From the values of the regression coefficient R2 listed in Table 4, Freundlich isotherm gave good and fitted better than Langmuir isotherm from the calculated results for experimental data R2.
| At (298)° K | At (303)° K | At (308) °K | At (313)°K | At (318) °K | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LnCe | LnQe | LnCe | LnQe | LnCe | LnQe | LnCe | LnQe | LnCe | LnQe | |
| -3.404 | -2.737 | -1.720 | -2.749 | -3.144 | -2.776 | -1.958 | -2.959 | -3.144 | -2.775 | |
| -2.891 | -2.597 | -1.496 | -2.785 | -1.772 | -2.157 | -2.032 | -3.0533 | -1.772 | -2.157 | |
| -0.511 | -1.162 | -0.584 | -2.187 | -0.261 | -1.688 | -0.340 | -1.439 | -0.260 | -1.688 | |
| -0.029 | -1.193 | -0.213 | -1.845 | -1.106 | -1.628 | -0.644 | -1.879 | -1.106 | -1.628 | |
| -0.302 | -1.063 | 0.648 | -1.130 | 0.283 | -0.643 | 0.330 | -1.061 | 0.283 | -0.643 | |
| 0.747 | -0.791 | 0.654 | -1.229 | 0.255 | -0.846 | 0.668 | -0.959 | 0.255 | -0.846 | |
| 0.593 | -1.190 | 0.746 | -1.302 | 0.145 | -0.673 | 0.139 | -1.152 | 0.145 | -0.673 | |
| 0.775 | -1.289 | 1.141 | -1.156 | 1.203 | -0.997 | 0.842 | -0.666 | 1.203 | 0.998 | |
| 0.464 | -1.612 | 0.531 | -1.686 | 0.121 | -0.656 | 0.870 | -0.616 | 0.121 | -0.656 | |
| 0.129 | -1.356 | 0.757 | -1.381 | -0.337 | -1.437 | -0.299 | -1.703 | -0.337 | -1.438 | |
| 1.384 | -1.311 | 1.312 | -1.998 | 0.893 | -1.045 | 0.648 | -0.752 | 0.892 | -1.044 | |
Table 4: Freundlich isotherm parameters of adsorption and their correlation coefficient on adsorption of MV dye on cresson seeds
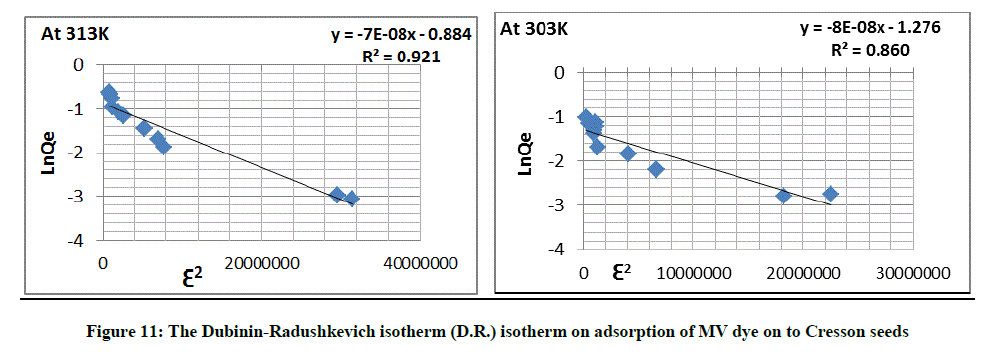
Dubinin-Radushkevich isotherm
This adsorption isotherm model was selected to calculate the quality porosity of biomass and the seeming energy of adsorption. The represented model is using sequential Equation 5:
Qe =Qs e-Kad Ԑ2 (5)
Ln Qe = Ln Qs - Kad Ԑ2 (6)
Where, Kad is the isotherm constant (mol2.J-2), Qs is the Dubinin-Radushkevich isotherm constant connected to level of sorbate sorption by sorbent level [29,30].
E (sorption energy) = 1/√2Kad (7)
Ԑ = R T Ln [1+1/Ce] (8)
A diagram between ln qe against Ԑ2 for sorbents, to submit straight lines to suggest an adjustment of isotherm to the experimental data. The obvious energy (E) of adsorption from Dubinin-Radushkevich isotherm model is able to calculate by relation given as Equation 7 [31]. In (Tables 5 and 6) show the height value of E=5000 J.mol-1, that indicates that the adsorption is physisorption show Figure 11.
| At (298) °K | At (303) °K | At (308) °K | At (313) °K | At (318) °K | |||||
|---|---|---|---|---|---|---|---|---|---|
| LnQe | Ɛ 2 | LnQe | Ɛ 2 | LnQe | Ɛ 2 | LnQe | Ɛ 2 | LnQe | Ɛ 2 |
| -2.737 | 73170151 | -2.7488 | 22549901 | -2.776 | 66577894 | -2.9599 | 29559650 | -2.775 | 32654557 |
| -2.5967 | 53252129 | -2.785 | 18302064 | -2.157 | 24398784 | -3.0533 | 31437740 | -2.157 | 24566648 |
| -1.1617 | 5902756 | -2.187 | 6693774 | -1.688 | 4538839 | -1.439 | 5215264 | -1.688 | 5952397 |
| --1.1931 | 3076678 | -1.845 | 4116652 | -1.628 | 12693844 | -1.879 | 7692924 | -1.628 | 5690521 |
| -1.063 | 1879657 | -1.130 | 1123131 | -0.643 | 2068555 | -1.061 | 1985137 | -0.643 | 2030991 |
| -0.791 | 923783 | -1.229 | 1110768 | -0.846 | 1559529 | -0.959 | 1158447 | -0.846 | 1595141 |
| -1.190 | 1187618 | -1.302 | 955793 | -0.673 | 2547418 | -1.152 | 2648691 | -0.673 | 1604506 |
| -1.289 | 881741 | -1.156 | 488060 | -0.997 | 452163 | -0.666 | 869008 | 0.998 | 3611289 |
| -1.612 | 1461358 | -1.686 | 1356912 | -0.6564 | 2638387 | -0.616 | 827732 | -0.656 | 62369 |
| -1.356 | 364144 | -1.381 | 939480 | -1.437 | 5028452 | -1.703 | 6936560 | -1.438 | 1954698 |
| -1.311 | 307022 | -1.998 | 273986 | -1.0435 | 773048 | -0.752 | 1198358 | -1.044 | 743912 |
Table 5: The experimental data of Dubinin-Radushkevich isotherm (D.R.) isotherm on adsorption of MV dye onto cresson seeds
| Temperature (°K) | Kad (mol2.J-2) | Qs (mg.g-1) | E (kJ/mol) | R2 |
|---|---|---|---|---|
| 298 | 2 × 10-8 | 0.2272 | 5000 | 0.859 |
| 303 | 8 × 10-8 | 0.2792 | 2500 | 0.860 |
| 308 | 3 × 10-8 | 0.3745 | 4100 | 0.742 |
| 313 | 7 × 10-8 | 0.4132 | 2700 | 0.921 |
| 318 | 6 × 10-8 | 0.5091 | 2900 | 0.491 |
Table 6: The calculated data of Dubinin-Radushkevich isotherm (D.R.) isotherm
Kinetics
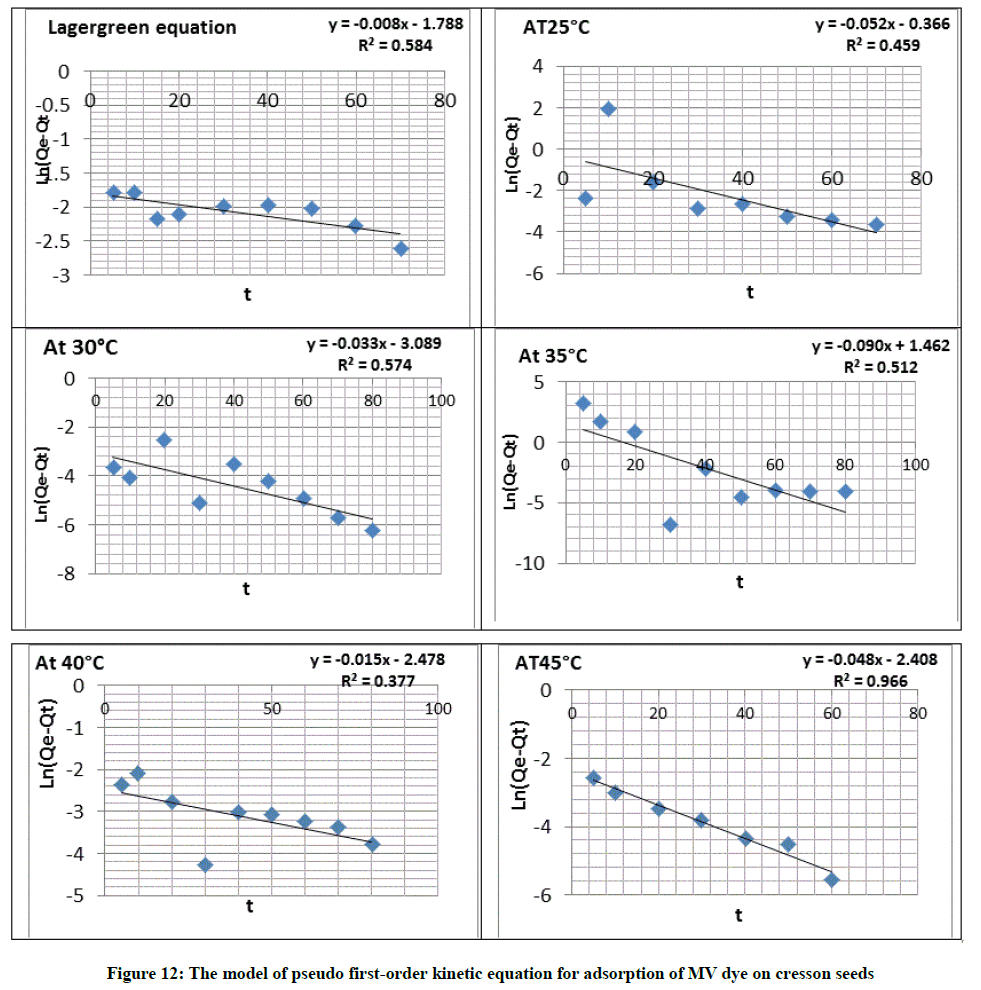
Lagergren kinetic rate equation
The equation of Lagergren kinetics has been mainly used for the adsorption of an adsorbate from an aqueous solution [32]. Lagergren authentic paper declared the pseudo-first order rate equation for liquid-solid adsorption array in 1898 and was summarize as follows Equation 9:
Ln (Qe-Qt) = LnQe-K1t (9)
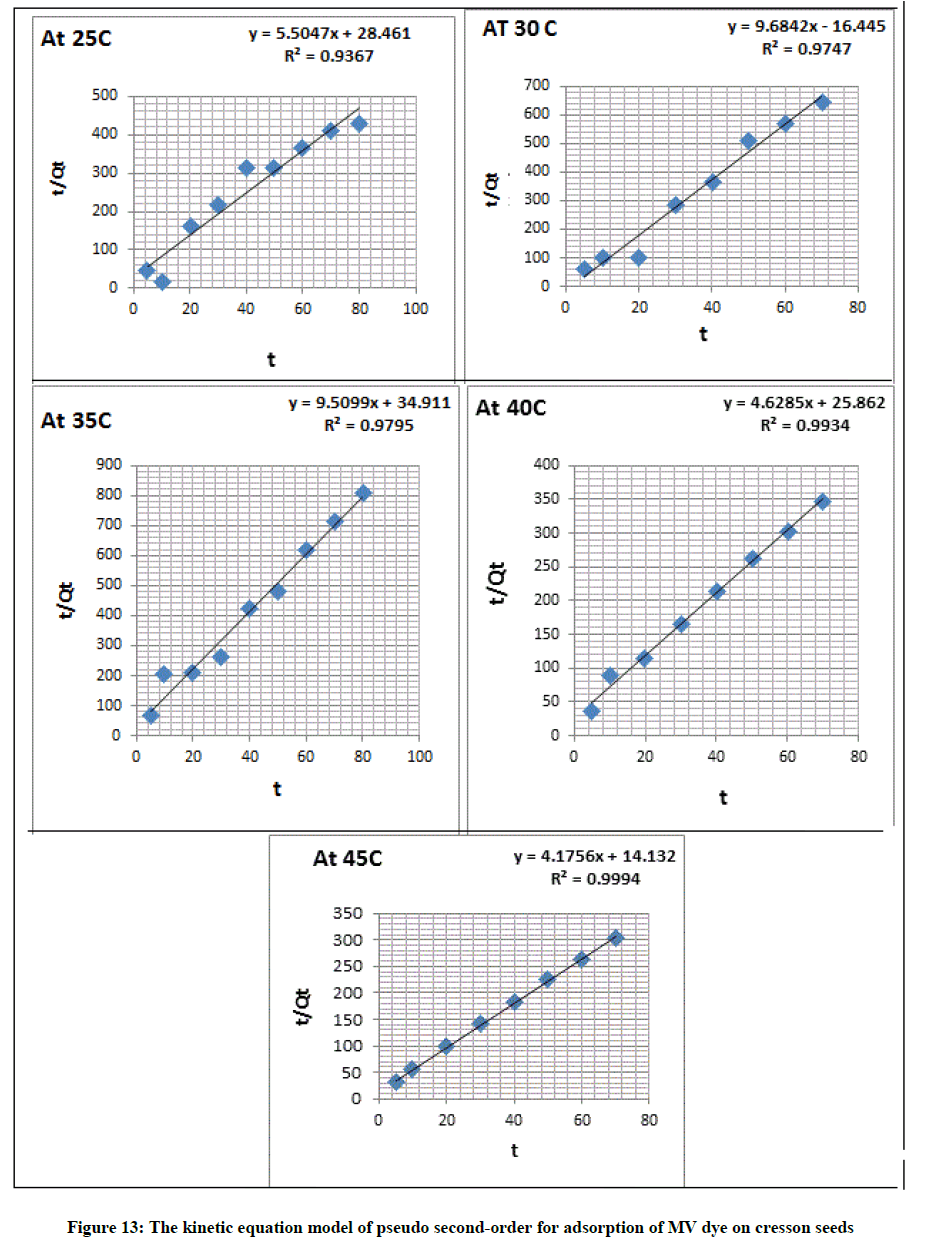
The pseudo second-order equation as equation (10), which applied to empirical data:
t/Qt = 1/K2 Qe2 + t/Qe (10)
Where, Qe and Qt respectively are the adsorption capacities at equilibrium and at (t) time in (mg g-1). K1 in (min-1)=Adsorption rate constant for pseudo-first order [33]. Where, K2=Constant of adsorption rate for pseudo second-order in (g/mg. min) and (t) is time in min show Figure 12. The parameters obtained by the application of the two kinetic models were reported in Table 7.
| At (298) °K | At (303) °K | At (308) °K | At (313) °K | At (318) °K | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time (min) |
Ln (Qe-Qt) |
t/Qt | Time (min) | Ln (Qe-Qt) |
t/Qt | Time (min) | Ln (Qe-Qt) |
t/Qt | Time (min) |
Ln (Qe-Qt) |
t/Qt | Time (min) |
Ln (Qe-Qt) |
t/Qt |
| 5 | -2.392 | 46.664 | 5 | -3.668 | 57.654 | 5 | 3.156 | 68.399 | 5 | -2.38 | 34.602 | 5 | -2.553 | 32.272 |
| 10 | 1.961 | 17.289 | 10 | -4.061 | 99.255 | 10 | 1.703 | 203.045 | 10 | -2.08 | 89.102 | 10 | -3.013 | 54.444 |
| 20 | -1.598 | 161.079 | 20 | -2.526 | 100.099 | 20 | 0.887 | 210.084 | 20 | -2.782 | 114.187 | 20 | -3.477 | 99.056 |
| 30 | -2.828 | 215.123 | 30 | -5.116 | 282.353 | 30 | -6.812 | 262.467 | 30 | -4.287 | 164.745 | 30 | -3.826 | 142.18 |
| 40 | -2.645 | 313.603 | 40 | -3.487 | 363.636 | 40 | -2.24 | 423.28 | 40 | -3.016 | 212.653 | 40 | -4.351 | 182.648 |
| 50 | -3.252 | 312.769 | 50 | -4.234 | 511.509 | 50 | -4.493 | 478.377 | 50 | -3.079 | 261.643 | 50 | -4.509 | 226.143 |
| 60 | -3.393 | 363.665 | 60 | -4.923 | 571.565 | 60 | -3.989 | 617.284 | 60 | -3.244 | 302.877 | 60 | -5.573 | 262.009 |
| 70 | -3.619 | 407.545 | 70 | -5.719 | 642.378 | 70 | -4.034 | 714.286 | 70 | -3.355 | 346.198 | 70 | -5.878 | 304.347 |
| 80 | -4.466 | 427.608 | 80 | -6.223 | 57.654 | 80 | -4.092 | 808.081 | 80 | -3.778 | 364.602 | 80 | -5.998 | 349.112 |
Table 7: The calculated data of Lagergren kinetic rate equation on adsorption of MV dye on to Cresson seeds
The value of the rate constant calculated from the equations of pseudo first-order and Pseudo second-order which found R2 value for equation of pseudo first-order is=0.578 and for the second order is 0.827 more than the value of pseudo first-order equation, that suggesting that the interaction between the seed and the MV dye follow the pseudo-second order mechanism as show in Figure 13 and Table 8.
| Pseudo first - order | Pseudo second - order | |||||
|---|---|---|---|---|---|---|
| T (°K) | K1 | Qe | R2 | K2 | Qe | R2 |
| 298 | 0.052 | 0.693 | 0.459 | 0.891 | 0.182 | 0.937 |
| 303 | 0.033 | 0.046 | 0.574 | -4.826 | 0.103 | 0.975 |
| 308 | 0906 | 4.317 | 0.512 | 2.139 | 0.105 | 0.979 |
| 313 | 0.016 | 0.084 | 0.377 | 0.688 | 0.216 | 0.993 |
| 318 | 0.049 | 0.089 | 0.966 | 1.306 | 0.239 | 0.999 |
Table 8: Parameters of kinetic models to adsorption with MV dye on cresson seeds
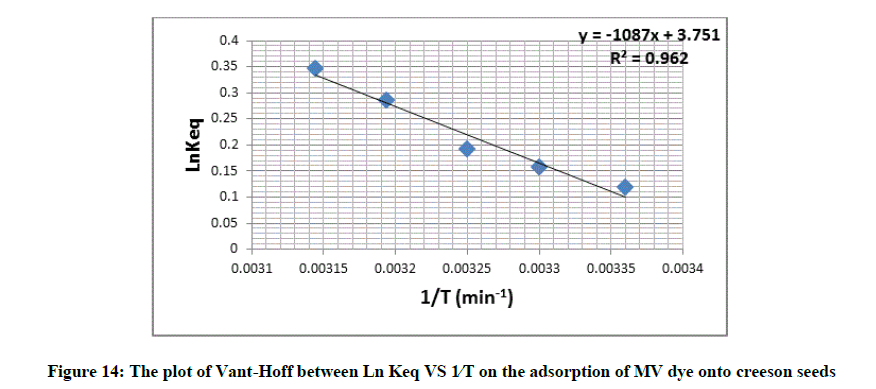
Where, Keq is the equilibrium constant, Qe is concentration at equilibrium of solid phase (mg⁄g), Ce is concentration at equilibrium for liquid phase (mg ⁄ l) and T=An absolute temperature and R=Gas constant [34]. The values of ΔH° and ΔS° computed from slope and intercept using Vant Hoff plots Figure 14 and are given in Table 9.
| T (°K) | 1/T | Keq | LnKeq | ∆H° (J.mol-1) |
∆G° (J.mol-1) |
∆S° (J.mol-1.K- 1) |
|---|---|---|---|---|---|---|
| 298 | 0.00336 | 1.1246 | 0.1174 | 9037.3180 | -290.377 | 31.3009 |
| 303 | 0.00330 | 1.1696 | 0.1566 | -393.959 | 31.1253 | |
| 308 | 0.00323 | 1.2112 | 0.1916 | -489.806 | 30.9322 | |
| 313 | 0.00319 | 1.3300 | 0.2852 | -740.921 | 31.2404 | |
| 318 | 0.00314 | 1.4130 | 0.3457 | -912.441 | 31.2885 |
Table 9: Thermodynamic parameters for adsorption of MV dye on creeson seeds
When the system temperature is increase, the degree of adsorption increase, this become clear from Keq values which increase with increase the temperature, which means endothermic process, this verified by ΔH° positive values. The positive value of ΔS° the change in entropy, propose by increased randomness during the process of adsorption. The ΔG° with negative value suggest that adsorption is spontaneous. The equilibrium values obtained are depicted in (Table 9).
Conclusions
This study confirmed that Creeson seeds can be adopted in reality for removal of MV dye in solution. The removal efficiency reaches 76% in some examples. The adsorption process based on solution with temperature effect. The adsorption process was best fitted with Freundlich adsorption model and kinetics fitted to pseudo-Second order model. According to experimental consequence, creeson seeds is recommended as an available and safe biosorbent to the removal. Parameters of thermodynamic as ΔH°, ΔS° and ΔG° were computed, which demonstrated that the adsorption was endothermic nature and spontaneous process, which was evident by increasing the randomness of the dye at the solid and liquid interface.
References
- Y. Bulut, H. Aydin, Desalination., 2006, 194(1-3), 259-267.
- R. Gong, Y. Jin, J. Chen, Y. Hu, J. Sun, Dyes Pigm., 2007, 73(3), 332-337.
- V.S. Mane, I.D. Mall, V.C. Srivastava, Dyes and Pigm., 2007, 73(3), 269-278.
- J. Lia, D.H.L. Ng, P. Song, C. Kong, Y. Song, Mater. Sci. Eng B., 2015, 194, 1-8.
- J. Pal, M. Deb, D. Deshmukh, B. Sen, Appl. Nanosci., 2014, 4(61-65).
- M. Saji, S. Taguchi, K. Uchiyama, E. Osono, N. Hayama, H. Ohkuni, J. Hosp. Infect., 1995, 31, 225-228.
- P. Li, Y.J. Su, Y. Wang, B. Liu, L.M. Sun, J. Hazard. Mater., 2010, 179, 43-48.
- L.R. Bonetto, F. Ferrarini, C. de Marco, J.S. Crespo, R. Guégan, M. Giovanela, J. Water Process. Eng., 2015, 6, 11-20.
- J. Wu, M.A. Eiteman, S.E. Law, J. Environ. Eng., 1998, 124, 272-277.
- V. Vimonses, S. Lei, B. Jin, C.W.K. Chow, C. Saint, Chem. Eng. J., 2009, 148, 354-364.
- S.S. Moghaddam, M.R.A. Moghaddam, M. Arami, J. Hazard. Mater., 2010, 175, 651-657.
- S. Hisaindee, M.A. Meetani, M.A. Rauf, TrAC Trends Anal. Chem., 2013, 49, 31-44.
- K. Turhan, I. Durukan, S.A. Ozturkcan, Z. Turgut, Dyes Pigm.,2012, 92, 897-901.
- L. Wang, J. Zhang, R. Zhao, C. Li, Y. Li, C. Zhang, Desalination., 2010, 254, 68-74.
- N. Yeddou, A. Bensmaili, Aicha Elsevier., 2005, 185, 1/3, 499-508.
- P. Nigam, I. M. Banat, D. Singh, R. Marchant, Proc. Biochem., 2007, 31(5), 435-442.
- T. Gedif, H.J. Hahn, J. Ethnopharmacol.,2003, 87, 155-161.
- T. Teklehaymanot, M. Giday, G. Medhin, Y. Mekonnen, J. Ethnopharmacol., 2007, 111, 271-283.
- M.M. Patel, G.M. Chauhan, L.D. Patel, Indian J. Hosp. Pharm., 1987, 2000202.
- S. Chandra, S. Babbar, Indian J. Chem. B., 1987, 26, 82-84.
- D.K. Mahmoud, M.A. Salleh, W.A. Wan, Abdul Karim, A. Idris, Z. Zainal Abidin, Chem. Eng. J., 2012, 181-182, 449-457.
- M. Ozacar, Adsorption., 2003, 9, 125-132.
- I.V. Szebeni, N. Kovács, Anal. Bioanal. Chem., 2005, 383(3), 476-482.
- A.C. John, Ibironke, O. Lara, Adedeji, Victor, O. Oladunni, American- Eurasian Journal of Scientific Research., 2011, 6(3), 123-130.
- I. Langmuir, J. Ame. Chem. Soc., 1918, 57, 1361.
- S. Gueu, Yao, B. Adouby, K. Ado, Int. J. Environmen. Sci. Technol., 2007, 4(1), 11-17.
- P.N. Dave, N. Subrahmanyam, S. Sharma, Indian J. Chem. Technol., 2009, 16(3), 234.
- B. Hameed, A. Rahman, J. Hazard. Mater., 2008, 160(2), 576-581.
- M.M. Dubinin, L.V. Radushkevich, Proc. Acad. Sci.: Phys. Chem. Sect., 1947, 55, 331-333.
- A.U. Itodo, F.W. Abdulrahman, L.G. Hassan, S.A. Maigandi, Int. J. Natural Appl. Sci.,2009a, 5(1), 92-97.
- B. Hameed, A. Ahmad, K. Latiff, Dyes Pigm., 2007, 75(1), 143-149.
- M. Kandah, Chem. Eng. J., 2001, 84, 543-549.
- S. Lagergren, Zur Theorieder Sogenannten, Adsorption Geloester Stoffe, Veternskapsakad, Handlingar, 1898, 24, 1-39.
- B. Mi Hwa, B.C.O. Ijagbemi, O.SeJin, D.S. Kim, J. Hazard. Mater., 2010, 176, 820-828.