Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 10
Wool and Silk Fabrics Dyeing by Mannitol-assisted Pigment Produced from Penicillium purpurogenum
Eman F Ahmed1*, Waill A. Elkhateeb1, Marwa. Abou Taleb2, Salwa I Mowafi2 and Ibrahim S Abdelsalam1
1Chemistry of Natural and Microbial Products Department, National Research Centre, 12622, Cairo, Egypt
2Proteinic and Man-made Fibers Department, Textile Industries Research Division, National Research Centre, 12622, Cairo, Egypt
- *Corresponding Author:
- Eman F Ahmed
Chemistry of Natural and Microbial Products Department
National Research Centre, 12622, Cairo, Egypt
Abstract
Penicillium purpurogenum, order Eurotiales was a superior producer to red, orange and yellow colorants. The fungus was grown in Dox media, it was found that Mannitol (30 g/l) and peptone (4 g/l) doubled the fungal productivity more than fourteen folds. The optimum conditions for pigment production was found to be at faint alkaline media (pH 8) at ambient temperature (30oC), at which a sharp increase of pigment yield was attained (263.63 g/l). During fermentation, the fungus was subjected to physical stress; Viz. high and low temperatures, magnetic field, and different wave lengths of light. The microorganism maintained its activity at the aforementioned physical effects. It also tolerated various amounts of potassium chloride from (1-9) g/l and produced high amounts of pigment (143.75-56.39 mg/ml). Among some available useless waste, the milled almond and the peels of sweet potato were the most effective in pigment production (74.32 and 120.79 mg/ml, respectively). The colorant was used in dyeing of wool and silk fabrics under various dyeing conditions; Viz. dye concentration, dyeing temperature, dyeing time, and pH. The dye ability of wool fabrics with the colorant was found to be higher than that of silk. The fastness properties of the dyed fabrics towards light and washing are satisfactory. The said colorant imparted ultraviolet protection to the dyed wool and silk fabrics.
Keywords
Penicillium purpurogenum, Red dyes, Physiological studies, Wool, Silk, fabrics, Dyeing.
Introduction
Recently, the way was opened to alternative pigment products from natural sources due to many reasons, one is the suffering of industry from the high cost feedstock of eco-friendly pigment. On the other hand, the disadvantages of synthetic food colorants which destroy the nutrients in food, cause many dangers to consumers that led to some health problems, most notably types of cancer, Sudden mood swings, hyperactivity in children, DNA damage or genotoxicity, attention-deficit disorder and detrimental effects on environment [1]. Some natural red colorants, for instance anthocyanins which was originated from plants can't tolerate high pH values. Other colorants from plants or animals origins are not available throughout the year, become faint and unstable against heat and light [2]. For these reasons, a strong need to microbial natural colorants was emerged.
A number of microorganisms have been reported by many authors as producer of large quantities of colorants including Paecilomyces, Monascus, Cordyceps, Serratia, Streptomyces, Penicillium atrovenetum and Penicillium herquei. [3-7].
Production of microbial colorant is advantageous due to the rapid growth of the microorganism that lead to high production rate of non-toxic ecofriendly compounds [8,9].
Pigments were considered as valuable compounds in food industry, they are used as antioxidants, additives and color intensifiers. Microorganisms produce wide varieties of water-soluble pigments. It was reported that the Genus Penicillium was a potent producer of polyketide pigments [10].
The textile industries are one of the most popular industrial sectors worldwide. In the meantime, this industry was classified as one of the most polluting industry that discharges effluents contaminated with many pollutants; among them colorants are most hazardous as they are not biodegradable [11].
Wool and silk are two major natural proteinic fibres used in textile industries and have considerable share in the global market. By virtue of their acidic and basic niches; they can be dyed with acid and reactive dyestuffs. Reactive dyes are usually used as colorant for both substrates due to its ability to form covalent bonds with wool and silk [12]. Synthetic colorants exist in the global market with a reasonable price and different hues. Nevertheless, their use in textile coloration has many disadvantages; e.g. skin allergy, and irritation, discharge of toxic chemicals during their synthesis, and carcinogenetic effects in minor cases. Accordingly, textile colorists were reinforced to find our alternatives to synthetic pigments, and the market demand and customer interest toward microbial colorants were mandatory [13]. Various microorganisms were used by many researchers to produce colorants; Viz marine bacteria [14], Monascus, Rhodotorula, Bacillus, Achromobacter, Yarrowia [15], bacteria from seafood [16].
Consequently, the recent study was carried out to optimize the concentration of the main and most significant nutrients in the medium for maximum production yield of pigment. Also to study the resistance of the fungus to physical stress and the effect of different cheap wastes and other chemical stress on the productivity of the fungus. A green technology was adopted to utilize Penicillium purpurogenum, together with mannitol to produce red dye. The produced colorant was used to dye wool and silk fabrics in an eco-friendly process.
Materials and Methods
Microorganism
The culture of Penicillium purpurogenum (2603) was purchased from Assiut University Mycology Centre (AUMC), the spore suspension; (2×107 spores/ml) was used to inoculate Czapeck Yeast Agar (CYA) growth media that contained the following ingredients (g/l): Glucose 30, yeast extract 2, peptone 10, NaNO3 3, KCl 0.5, MgSO4 0.5 and Agar 25(Pitt, 1985), incubated for 7 days at 28°C and stored at 4°C.
Fabrics
Light weight merino wool fabrics (140 g/m2) were provided by Misr Company for Spinning and Weaving, El-Mehalla El-Kobra, Egypt. Natural silk fabric (80 g/m2) was purchased from private sector in Akhmeem, Egypt. All other chemicals are of laboratory grades and used as provided without any further purification.
Culture agar medium and production medium
The fungus was grown in a rich Czapeck Yeast Agar medium, Dox production medium (50 ml) in 250 ml Erlenmeyer flasks was inoculated with the fungus cell suspension (2×107 spores/ml).The culture was shaked at 200 rpm and 28°C for 7 days. Dox production medium was prepared with the following nutrients (g/l distilled water): Sucrose (20), NaNO3 (3.0), KCl (0.5), MgSO4.7H2O (0.5), KH2PO4 (1.0), FeSO4.7H2O (0.1).
Pigments Extraction by different solvents
Cultures from Dox production media were filtered through filter paper (No. 1; Whatman), the biomass was dried at 50ºC for 48 h and weighed. Red pigment was extracted with Toluene, Methanol, n-hexane, Isopropanol and Ethyl acetate. The extract was concentrated till dryness and dissolved in 10 ml distilled water, absorbance was read at 500 nm by a UV-visible spectrophotometer.
Significance of Carbon nutrient on production yield
To choose the promising source of carbohydrates to optimize pigments concentrations; the carbon source in Dox broth medium was replaced by different saccharides (20 g/l); Mannitol, Dextrin, Lactose, glucose, sucrose and Fructose. Each nutrient was added as a sole carbon ingredient in the fermentation medium. All flasks were incubated at 28ºC and 200 rpm for 8 days.
Investigation of Pigment production at different levels of Mannitol
P. purpurogenum was grown in Dox medium, six concentrations of Mannitol (10-35) gl were used in the fermentation broth medium, the flasks were incubated at 28ºC and 200 rpm for 8 days. The absorbance of pigments was read at 500 nm and the concentration was calculated according to Beer's Law:
y = mx + b. whereas y is the absorbance of red pigments at 500 nm, m is the slope, x is the concentration in mg/ml and b is the value of the intercept.
Evaluation of pigments productivity by different nitrogen sources
Production medium was prepared with 30 g/l mannitol and the salts (KCl (1.0), K2HPO4 (0.5), MgSO4.7H2O (0.5) and FeSO4.7H2O (0.01) g/l). Different nitrogen sources (3 g) were added in the medium eg: NaNO3, yeast extract, peptone and ammonium sulphate.
Significance of peptone weight on Pigment yield
Different weights of peptone were added in the production medium of the fungus to cover the range from (1-6) g/l, the produced pigment was read at 500 nm by spectrometer and the production yield was calculated.
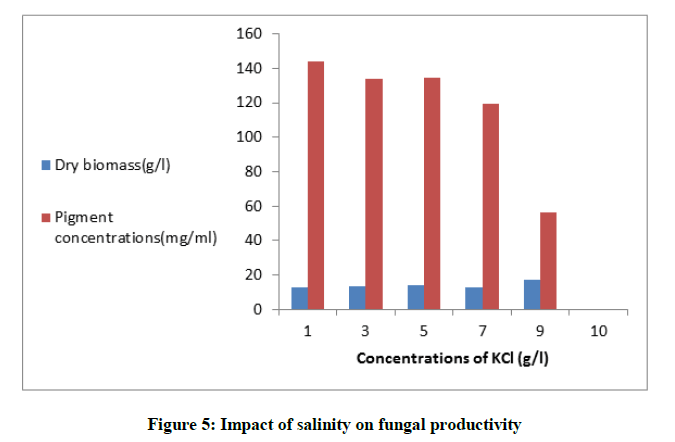
Impact of salinity on fungal productivity
Different concentrations of KCl (1-10) g/l were used in the fermentation medium, the cells were inoculated in 50 ml Dox medium for 8 days at 30ºC. The concentration of the pigment was evaluated.
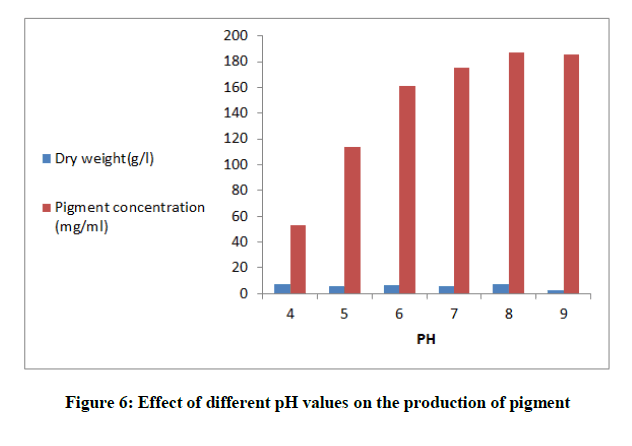
Effect of pH values on pigment production
Optimized broth media were adjusted at different pH values in the range (4.0 to 9.0) in shaking flasks at (200) rpm and incubated at 28ºC, inoculated with P. purpurogenum spore suspension and the absorbance was read at 500 nm to investigate the pigment yield.
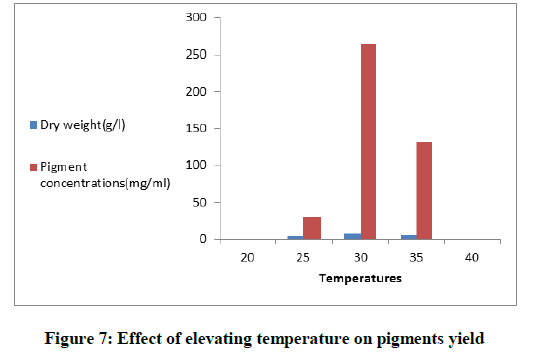
The relation between production and increasing temperatures
The medium was adjusted to pH 8 and the incubated fungus was exposed to different temperatures (20, 25, 30, 35 and 40)ºC for 8 days. The absorbance was read at 500 nm and the concentration of pigments was calculated.
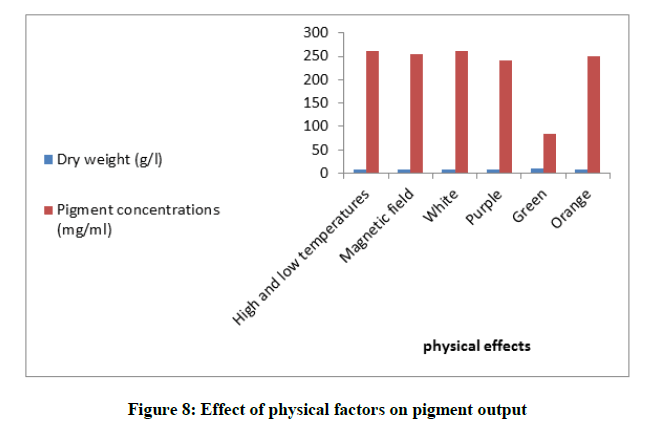
Investigation of production stability under physical stress
High and low temperatures
The fungus was used to inoculate 50 ml optimized fermentation medium in 250 ml flask. Medium was adjusted at optimum pH. The culture was incubated at different temperatures during the incubation period. At the first 24 h the culture was incubated at 20ºC, it was incubated at 30ºC at the second day, then at 40ºC at the third day. The temperature was changed after each 24 h of incubation. The flask was filtered and the pigment yield was evaluated.
Effect of the absorbed light and the magnetic field
The cultured media (50 ml) were incubated in 250 ml flasks, during the incubation period, the flasks were exposed to white light and covered with different transparent colored papers. The concentration of the produced pigment was evaluated.
The fungus was grown in the fermentation medium under a magnetic field for 7 days at 30ºC and 200 rpm before the yield of the pigment was assessed.
Effect of wastes on growth and pigment output
Different wastes (2 g/50 ml medium) were used as the main nutrient in the fermentation medium of the fungus in addition of traces of salts (K2HPO4 (0.5 g/l) and MgSO4.7H2O (0.5 g/l), peptone (0.5) g/l was also added. The flasks were incubated for 8 days at 30ºC and 200 rpm.
Dyeing
Wool and silk fabrics were dyed with the produced dye using various dye concentrations, dyeing temperatures, dyeing times, and pH. The dyeing process was applied to the fabric via exhaustion technique.
Effect of dye concentration
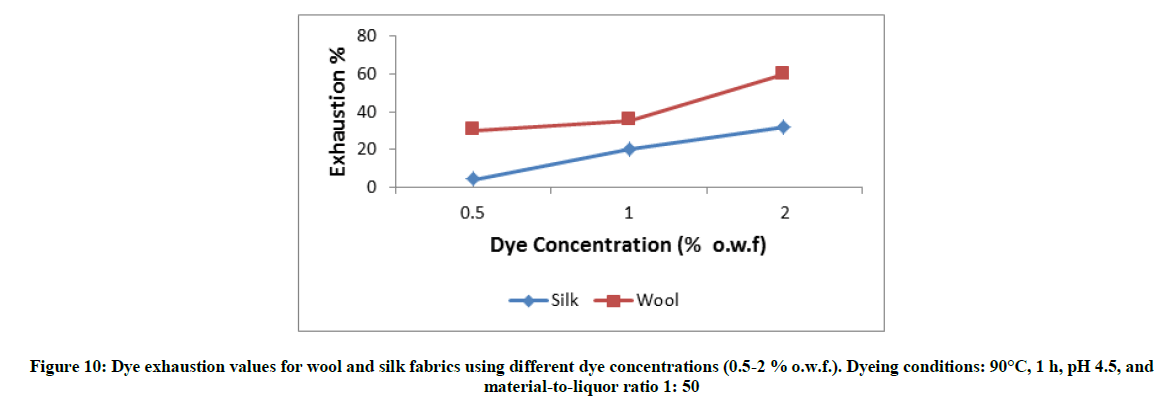
Wool or silk fabrics were immersed in the dye bath (0.5-2.0 % shade) at 90°C, pH 4.5 for 1 h; the material to liquor ratio (MLR) was 1: 50. After dyeing, the fabrics were removed, rinsed and dried at room temperature.
Effect of dyeing time
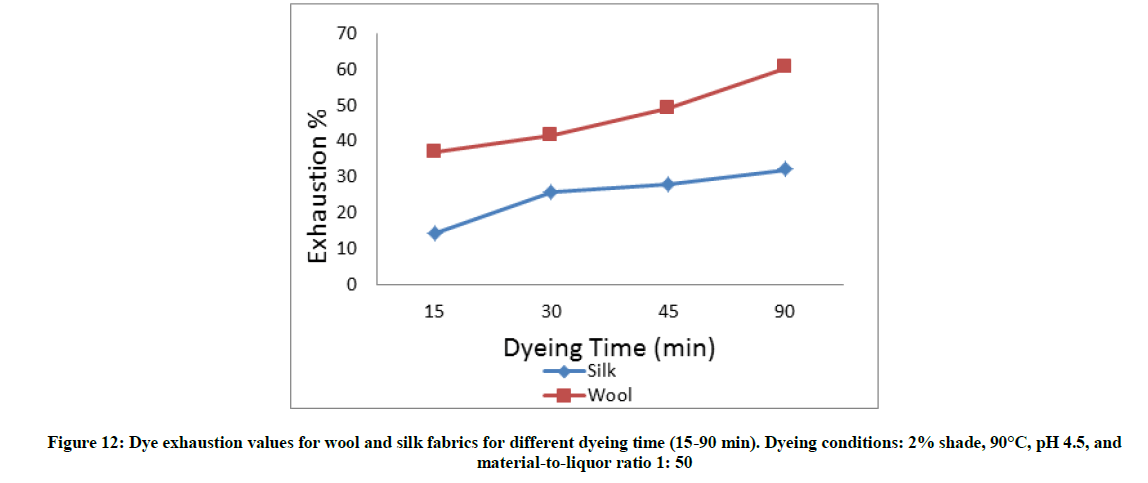
The fabric samples were dyed with 3% shade, pH 4.5, at the 90°C for (15-90 min), and the MLR is 1: 50. Once the dyeing time ended the fabric were removed, rinsed and dried at ambient temperature.
Effect of dyeing temperature
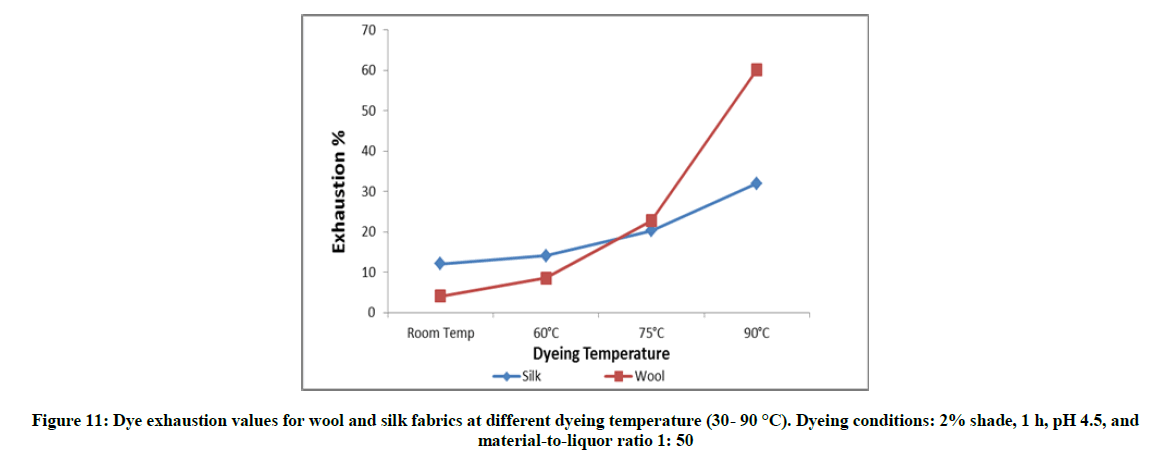
The samples were dyed with 3% shade, pH 4.5, for 30 min at different temperature (ca. 30-90°C); and the MLR is 1: 50. Once the dyeing time ended the fabric were removed, rinsed and dried at ambient temperature.
Effect of pH
Wool or silk fabrics were dyed using 3% shade at 90°C for 1 h at pH 2-9 (adjusted by acetic acid or sodium carbonate) and the MLR is 1: 50. Once the dyeing time ended the fabric were removed, rinsed and dried at ambient temperature.
Analysis
Infrared spectroscopy
Infrared spectroscopy was carried out for the dye on Nexus 670 Fourier transform infrared spectrometer (Nicolet, USA).
Dye exhaustion
The dye exhaustion was measured by measuring the absorbance of the dye bath at λmax 498 spectrophotometrically using T60 UV-VIS spectrophotometer, PG Instruments Ltd., Uk.
Washing fastness
The colour fastness to washing was determined in accordance to ISO standard methods. The color fastness to washing was determined according to the AATCC test method 36 (1972) using Lunder Ometer [17]. The washing fastness was assessed using the Grey Scale reference for color change.
Light fastness
Colorfastness to light was determined according to AATCC test method (16 A-1989). Evaluation was established using blue scale as reference of color change [18].
UV-protection
The transmission of ultraviolet (UV) radiation through dyed as well as un-dyed fabrics was evaluated with a Varian Cary 300 ultraviolet visible spectrophotometer (Mulgrave, Victoria 3170, Australia) at a wavelength range of 320-400 nm.
Results
Optimization of nutrients for maximum pigment yield
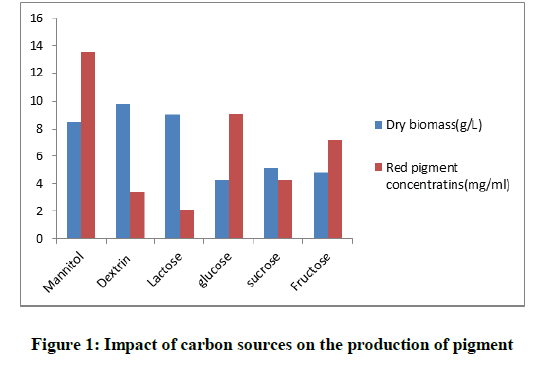
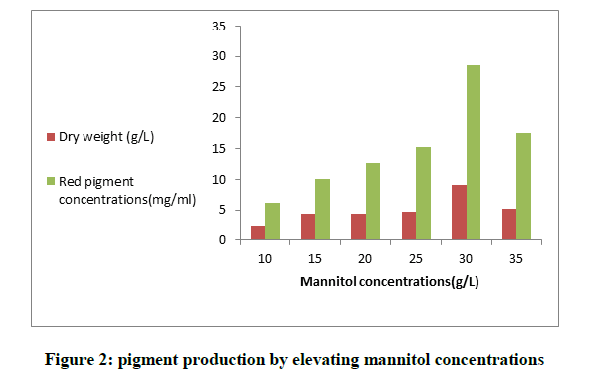
Different carbon and nitrogen sources were added in the fermentation medium and tested for maximum output of red pigment. The obtained results in Figure 1 showed that from all used saccharides; mannitol was the most significant for optimum production (13.5 mg/ml). From the presented data, it was proved that there wasn't any relation between the production of pigments and the growth of the fungus; although Dextrin medium didn't produce high concentration of pigment (3.4 mg/ml), it enhanced a high fungal biomass (9.8 g/l). On the contrary, the low biomass of the fungus (4.26 g/l) produced high amount of pigment (9.1 mg/ml) in glucose medium. To evaluate the optimum level of Mannitol for the highest yield of pigment; different concentrations were used, it was shown in Figure 2 that Mannitol (30 g/l) enhanced the production of maximum red pigment (28.63 mg/ml) and a decrease in pigmentation was noticed by decreasing the level of Mannitol. We concluded from this result the significance of Mannitol as a carbon source in the production medium of the fungus.
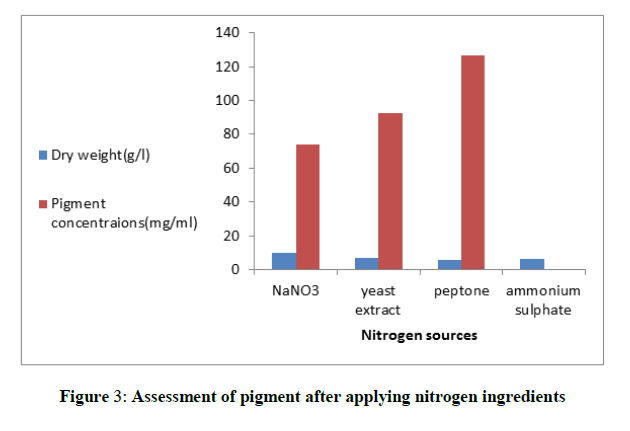
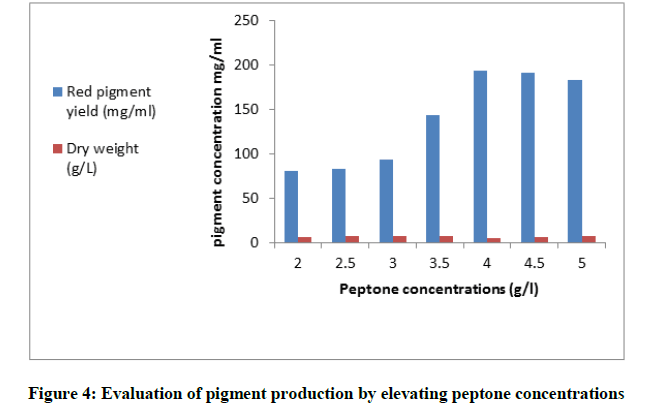
For the selection of the suitable source of nitrogen in the fermentation medium, NaNO3, yeast extract, peptone and ammonium sulphate (3 g/l) were chosen Figure 3. After the incubation time the pigment yield was assessed. Peptone was the most favorable for maximum pigment output (126.6 mg/ml). Interestingly, it was noticed that ammonium sulphate enhanced the production of yellow pigment in the medium and inhibited the red one. When peptone was used in the production medium in different levels from (2 to 5 g/l), maximum yield of pigment (193.62 mg/ml) was obtained in the basal medium with peptone concentration (4 g/l) Figure 4. Moreover, the tolerance of the fungus to salinity by increasing the levels of KCl from (1-10) g/l was studied. The fungus exhibited a resistance to the examined salt at most concentrations till (9 g/l) and produced the highest yield of pigment (143.75 mg/ml). Furthermore, the cells of the fungus not only tolerated the salinity in the medium but also a sharp increase in the fungus mass was obtained Figure 5.
The fermentation media were adjusted at different pH values to cover the range from (4-9) Figure 6. Maximum production of the pigment (187.39 mg/ml) was noticed at the alkaline medium at pH 8. The basal production medium contained (g/l); Mannitol (30), peptone (4), KCl (1), it was adjusted at pH 8. To study the effect of temperatures on pigment production, the medium was incubated for 8 days at different degrees of temperatures (20ºC-40ºC). Figure 7 showed that 30ºC was the optimum for both pigment yield and mycelial growth.
It was found that the pigment composition changed according to the absorbed light. In our results presented in Figure 8, it was found that the white, red, orange and purple lights influenced the production of red pigment (262.28, 260.57, 250.16 and 240.43) mg/ml respectively. Although the green spectrum reduced the yield of the pigment (83.19 mg/ml), it highly increased the mycelial mass of the fungus (9.12 g/l). Further stress was conducted on the mycelium of the fungus by the magnetic field effect Figure 8. A clear tolerance towards this effect was achieved. The production of red pigment was 254.20 mg/ml which was produced from (7.98 g/l) biomass weight.
Many reports searched on the use and recycle of the cheap by-products that cause detrimental problems in the environment [19]. A selection of some inexpensive substrates was used as a source of carbohydrates in the fermentation medium of the fungus (Table 1). Of all these wastes, only milled Almond enhanced pigment production (120.79 mg/ml) and the sweet potato (74.32 mg/ml).The data showed a reasonable amount of biomass at all waste media.
| Natural waste | Red pigment yield (mg/ml) |
|---|---|
| Control | 147.3 |
| Baggase | _ |
| Olive wastes | _ |
| Wheat bran | _ |
| Apricot seeds | _ |
| Rice bran | _ |
| Corn starch | _ |
| Cossi glance | _ |
| Sweet potato | 74.32 |
| Milled almond | 120.79 |
Table 1: Effect of different wastes on the production of pigment
Fourier transfer infrared (FTIR) spectroscopy
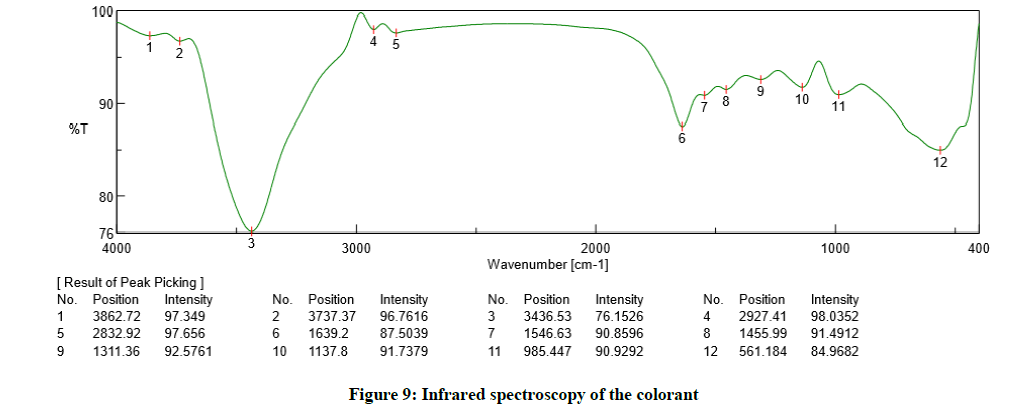
The FT-IR chart of the extracted dye, shown in Figure 9, involves the characteristic band of stretching vibration of free phenolic hydroxyl group at 3436 cm-1. Another distinct band appears at 1639 cm-1 which corresponds to the double bond in aromatic compounds. The broad medium intensity band at 561 cm-1 might be due to presence of C–X bond, where "X" is any halogen atom.
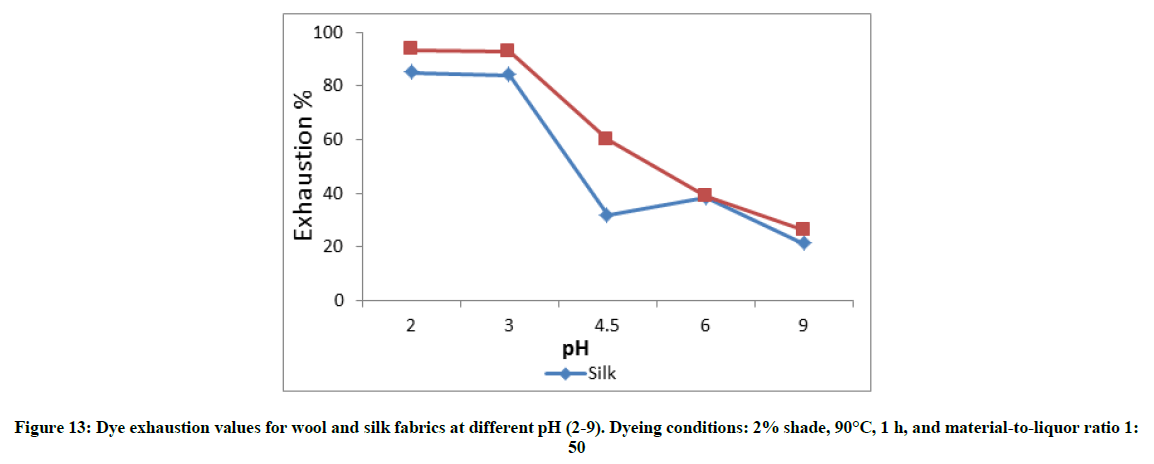
Effect of dyeing conditions on the dye ability of wool and silk fabrics
Figures 10-13 show the exhaustion percent of the said dye on wool and silk fabrics at different dye shade, dyeing temperature, dyeing time, and pH. Thorough inspection of these figures reveals that in most cases the dye ability of wool (α-keratin) fabrics towards the extracted dye is higher than that of silk (β-sheet) fabrics. This might be attributed to the higher crystallinity of the latter than the former, which leads to easier penetration of the dye molecule into wool fiber interior [20].
Moreover, the extent of dye exhaustion from the dyeing bath of wool and silk fabrics was found to be a function of the dye concentration, dyeing temperature, and dyeing time. The dyeing temperature seems to be the most effective parameter in enhancing the dye ability of wool and silk fabrics with the said dye. On the other hand, the dye ability of wool and silk fabrics show their maxima at pH 2-3. Further increase in the pH value within the acidic-slightly acidic range resulted in remarkable decrease in the exhaustion percent of dye. In alkaline medium (pH 9), the dye exhaustion was declined below to almost one fourth that in case of dyeing wool or silk fabrics at pH 2. This implies that the used dye is anionic in nature which is linked to the protonated amino groups-in acidic dyebath-along the silk and wool macromolecules.
Color fastness
To assign the durability of the dyed fabrics towards light and washing, the fastness properties of the dyed wool and silk fabrics to these factors was assessed. Results of this investigation, shown in Table 2, elucidate that the dyed silk and wool fabrics have moderate fastness to light, implying partial photocatalytic degradation of the dye. On the contrary, the dyed fabrics exhibited high fastness to washing which indicates that the bonding between the substrate and the dye molecules is strong enough to render the fabric adequate ability to withstand the alkaline conditions followed in the washing test.
| Fabric | Light fastness | Washing fastness | ||
|---|---|---|---|---|
| St* | St** | Alt. | ||
| Wool | 3-Feb | 5-Apr | 4 | 4 |
| Silk | 3 | 5-Apr | 4 | 4 |
St*: staining on cotton; St**: staining on wool; Alt.: Alteration
Table 2: Light and wash fastness of wool and silk fabrics dyed with red dye extracted from Penicillium purpurogenum (Dyeing conditions: 2% shade, 90°C, 1 h, pH 3, and material-to-liquor ratio 1: 50)
Ultraviolet Protection Factor (UPF)
The UPF of the dyed as well as undyed wool and silk fabrics was assessed to monitor any new function that can be imparted to the fabric from the extracted dye. Results of this investigation, abridged in Table 3, reveal that the used dye enhanced the UPF of wool and silk fabrics by about 225% and 60%, respectively.
| Sample | UPF |
|---|---|
| Undyed Silk | 7.6 |
| Dyed Silk | 12 |
| Undyed Wool | 55.1 |
| Dyed Wool | 179.3 |
Table 3: UPF of dyed as well as undyed wool and silk fabrics (Dyeing conditions: 2% shade, 90°C, 1 h, pH 3, and material-to-liquor ratio 1: 50)
Actually we tried to monitor any other functions that might be imparted to silk and fabrics by the dyeing with the extracted dye. Among others, these functions include microbial resistance of wool and silk fabrics, mothproofing of wool fabrics. However, the used dye has no sensible positive effect on the biological resistance of the said fabrics.
Discussion
It was reported that the use of synthetic dyes causes serious health and environment problems. Due to this fact, many published works searched on alternatives to synthetic pigments. It was discovered that natural colors that were extracted from plants and microorganisms are safe coloring agents. The advantages of pigment production from microorganisms include the easy and fast growth of the cells in cheap culture medium which is independent on weather conditions [21]. Culture conditions were known to influence the color of the pigment. Due to the fact that the red pigment was the most economically important, the nutrients of media were studied and optimized for maximum production of the red pigment. Glucose and Fructose were favorable carbon sources but Mannitol was the superior for maximum red pigment production. Other searches reported that high pigment production (670 mg l 1) was achieved in starch medium while high biomass (5.50 g) was grown in Fructose medium [2]. Peptone was the optimum nitrogen source for maximum yield of pigments. Our results were in accordance with, Cho et al. [22] and (Gunasekaran and Poorniammal [2] who reported the importance of peptone in supporting the production of pigments by different fungi. The examined fungus showed a high tolerance to different levels of KCl and high productivity at most concentrations of the salt. From this result, we can regard this behavior to the halophilic nature of this strain. The present study shed the light on the importance of temperature and pH on the pigments produced by the filamentous fungus. According to the concept that the pH of the culture medium had a big role in pigment formation. Different pH values were examined and the alkaline medium at pH 8 was the most appropriate to the production of the pigment. It was reported that another species of Penicillium produced the red pigment at PH 9 [2]. Another search, Cho et al. [22] reported the neutral conditions (PH 6) and others confirmed the acidic medium for pigments production. To evaluate the effect of temperature on the yield of the pigment, the fungus was incubated at different degrees of temperatures where 30ºC was the best for mycelium weight. The same result was reported by Patil et al. [21]. The previous temperature was also the most favorable for pigment production.
It was reported before that light regulates the growth, controls the reproduction and pigment formation [23]. The white, red, orange and purple influenced pigment productivity. We could explain this result to the presence of red spectrum included in the aforementioned lights which enhanced the production of red pigment. In contrast, the green spectrum decreased the production of pigment due to the absence of the red spectrum which could be considered as a vital factor for production process. Aiming to assign whether the colorant produced by P. Purpurogenum is dye or pigment, and to determine the class of this colorant; we carried out some investigations thereabout. The obtained colorant is water soluble material which emphasizes that this colorant is neither a pigment nor a disperse/vat dye. The dye couldn't be used for dyeing of cellulosic fabrics which implies that this dye is neither reactive nor direct dye. This colorant is not a basic (cationic dye) because it cannot dye acrylic fabrics. This colorant is capable of dyeing proteinic fabrics like wool and silk, which assures that this substance is an anionic in nature (acid dye).
In this study, the red pigment was produced in a high concentration under optimum amounts of nutrients and conditions of fermentation. The stability of the produced pigment was examined which showed a high tolerance against different stress and the red dye was found to be anionic in nature, it is an appropriate candidate for dyeing of wool and silk fabrics with acceptable light and wash fastness. The said colorant imparted ultraviolet protection to wool and silk fabrics.
Conclusion
The red pigment was produced in a high concentration under optimum amounts of nutrients and conditions of fermentation. The stability of the produced pigment was examined which showed a high tolerance against different stress. The red dye was found to be anionic in nature, and is an appropriate candidate for dyeing of wool and silk fabrics with acceptable light and wash fastness. The said colorant imparted ultraviolet protection to wool and silk fabrics.
Acknowledgement
The authors are indebted to Prof. Hosam El-Sayed, Head of the Proteinic and Man-made Fibres Department of the National Research Centre in Cairo, for his valuable guidance and discussions during preparation of dying application section in this work.
References
- M. Shahid, F. Moha mmad, J. Cleaner Produc., 2013, 53, 310-331.
- S. Gunasekaran, R. Poorniammal, Afr. J. Biotechnol., 2008, 7(12), 1894-1898.
- G.W. Engstrom, R.E. Stenkamp, D.J. McDorman, L.H. Jensen, J. Agric. Food Chem.,1982,30(2), 304-307.
- K.I. Suhr, I. Haasum, L.D. Streenstrup, T.O. Larsen, J Dairy Sci., 2002, 85(11), 2786-2794.
- L. Dufossé, Food Technol. Biotechnol., 2006, 44(3), 313-321.
- A. Méndez-Zavala, J. Contreras-Esquivel, F. Lara-Victoriano, R. Rodríguez-Herrera, C. Aguilar, Rev Mex Ing Quím., 2007, 6, 267-273.
- J. Hernández-Rivera, A. Méndez-Zavala, C. Pérez-Berúmen, J. Contreras-Esquivel, R. Rodríguez-Herrera, C. Aguilar, Mex. Asoc. Food Sci. Editions. Saltillo. Mexico, 2008, 108-112.
- C.H. Kim, S.W. Kim, S.I. Hong, Proc. Biochem., 1999, 35, 485- 490.
- Y. Jiang, H. Li, F. Chen, K. Hyde, J. Agric. Technol., 2005, 1(1), 113-126.
- P. Jûzlová, L. Martinková, V. Kren, J. Indus. Microbiol., 1996, 16, 163-170.
- T. Tuba, A. Pervin, J. Textile Engineering and Fashion Technol.,2017, 2(4), 429- 442.
- Md. K. Uddin, Int. J. Engg. Technol., 2010, 10(6).
- K. Abhishek, S. Hari, S. Jyoti, D. Shipra, K. Mahendra, Int. J. Pharmaceut. Chemi. Biol. Sci., 2015, 5(1), 203-212.
- K.P. Shiva, G. Krishna, R.S. Prakasham, M.A. Singara Charya, J. Marine Biosci., 2015, 1(1), 11-19.
- V. Joshi, D. Attri, A. Baja, S. Bhushan, Indian J. Biotechnol., 2003, 2(3), 362-369.
- T. Boontosaeng, S. Nimrat, V. Vuthiphandchai, J. Environ. Sci. Toxicol. Food Technol., 2016, 10(5), 30-34.
- AATCC Technical Manual, Method 36, 1972, 68(1993), 23.
- AATCC Technical Manual, Method 16, 1989, 68(1993), 30.
- J. Tinoi , N. Rakariyatham and R. Deming , Process Biochem., 2005,40, 2551-2557.
- N.H. Leon, J. Cosmetic Chemists of Great Britain., 1972, 23(7), 427-445.
- S. Patil, G. Sivanandhan, D. Thakare, Int. J. Curr. Microbiol. Appl. Sci, 2015, 4, 599-609.
- Y.J. Cho, J.P. Park, H.J. Hwang, S.W. Kim, J.W. Choi, J.W. Yun, Lett. Appl. Microbiol., 2002, 35(3), 195-202.
- S. Babitha, C. Julio, R. Carlos, P. Ashok, World J. Microbiol. Biotechnol., 2008, 24(11), 2671-2675.