Review Article - Der Pharma Chemica ( 2022) Volume 14, Issue 3
Wound Healing Efficacy of Some Plants: A Review
Pratibha S1*, Samriti F1, Praveen N2 and Sumitra N12Department Pharmaceutical Sciences, OM Sterling Global University, India
Pratibha S, Department of Pharmacy, Banasthali Vidyapith, Banasthali-304022, India, Email: faujdarsamriti@gmail.com
Received: 11-Feb-2022, Manuscript No. dpc-22-54190; Accepted Date: Feb 14, 2022 ; Editor assigned: 14-Feb-2022, Pre QC No. dpc-22-54190; Reviewed: 02-Mar-2022, QC No. dpc-22-54190; Revised: 08-Mar-2022, Manuscript No. dpc-22-54190; Published: 16-Mar-2022, DOI: 10.4172/0975-413X.14.3.28-33
Abstract
Plants are not only used for nutrient purpose but also for their therapeutic activity again various ailments since ancient times. Various researchers revealed the presence of various organic materials necessary to provide protection against microbial attack in wounds. This review article focused on the occurrence of wound and their treatment through various plants. Plants containing active constituents which are successfully researched for their wound healing activity were also discussed. Various herbal formulations are available in market for their wound healing activity. Herbal formulations are very effective than allopathic formulation because of cost effective and lack of resistance.
Keywords
Wound Injury; Inflammation; Polysaccharides; Extract
Introduction
Skin is the largest organ in the human body in respect of surface area. Internal tissues are protected by the skin from mechanical damage, microbial infection, UV light, and extreme temperatures. This renders it extremely vulnerable to harm with serious consequences for both individual patients and the healthcare system as a whole. Wounds remain a difficult clinical challenge in day to day pathology with early and late consequences causing a high rate of morbidity and mortality [1, 2]. Much has been deduced from the study of the physiology of healing and wound control, keeping control on innovative therapeutic approaches and the continuing development of various technologies which helps in acute and long-term wound protection trying to lessen the wound burden [3, 4].
Wound
Wound is a breakdown in the protective function of the skin or in the continuity of any body tissue. Wounds can form as a result of pathological processes that originate either outside or internally in the organ due to thermal, mechanical and physical damage. Acute wound is normal healthy individuals heal through an orderly sequence of physiological events that include hemostasis, inflammation, epithelialization, fibroplasia and maturation. When this process is altered, a chronic wound may develop and is more likely to occur in patients with may develop and is more likely to occur in patients with underlying disorders such as peripheral artery diseases, diabetes, venous insufficiency, nutritional deficiencies and other diabetes states. Wounding harms the tissue and disarray the local environment within it, without consideration of the cause and whatever the form. Symptoms of a physiological response to a noxious stimulus include bleeding, vascular constriction with coagulation, complement activation, and an inflammatory response [5, 6].
Classification of Wound
Wound are generally classified according to cause of development of wound
Acute Wounds
In acute wounds, there is tissue damage or injury that generally occurs through an orderly and time-reparative phase that results in the anatomical and functional integrity being restored sustainably. Acute wounds are typically caused by the cuts or surgical incisions [7].
Closed Wounds
The blood elude from the circulatory system in closed wounds but stays inside the body. It becomes evident in the form of bruises.
Open wounds
Blood leaks from the body through an open wound and bleeding is clearly noticeable. The open wound may be divided further into categories according to the source causing the wound.
Incised Wounds
This is a wound with no loss of tissue and minor damage to tissue. It is caused primarily by sharp objects like a scalpel or knife. This is the non-surgical injury in conjunction with other types of trauma which results in tissue loss and damage.
Puncture Wounds
These are caused by an object which, like a nail or a needle, which punctures the skin. Since dirt may penetrate deep into the wound, chances of infection are common in them. Abrasive or superficial wounds slip onto a rough surface induces abrasion. During this time, abrasion is scraped off the top layer of the skin, i.e., epidermis which exposes nerve endings resulting in a painful injury.
Penetration Wounds
Penetration wounds are chiefly caused by an object like a knife going in and out of the skin.
Gunshot Wounds
This type of wound is due to bullet or similar projectile which drives through or into the body.
Chronic Wounds
Chronic wounds are wounds that have not gone through the usual healing stages and hence reach a state of pathologic inflammation and they required expanded healing time [8].
Phases of Wound Healing
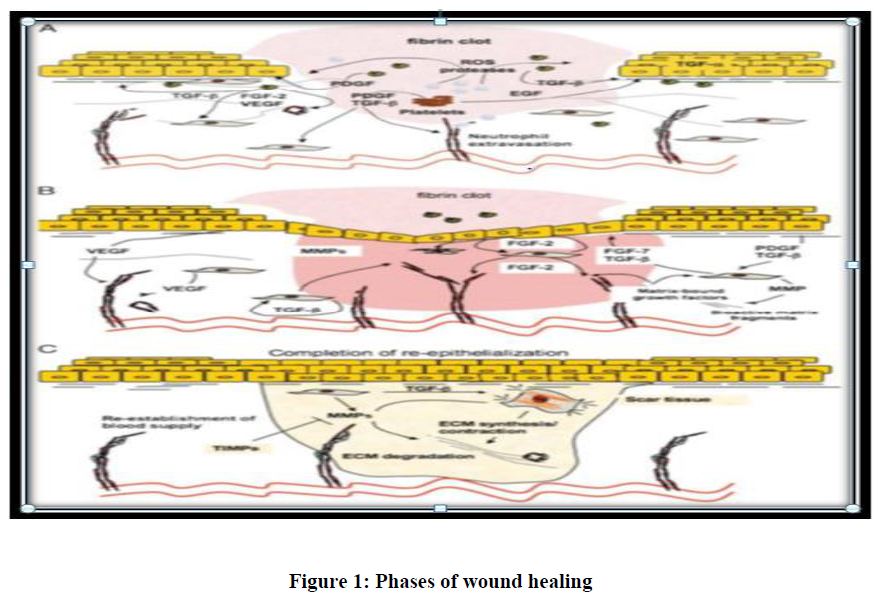
Wound healing occurs as a cellular response to tissue injury and involves activation of keratinocytes, fibroblasts, endothelial cells, macrophages and platelets the process involved organized cell migration and recruitment of endothelial cells for angiogenesis. Many growth factors and cytokines released by these cell types coordinate and maintain wound healing. Acute wounds transition through the stages of wound healing as a linear pathway; with clear start- and end points Chronic wounds are arrested in one of these stages, usually the inflammatory stages, and cannot progress further.
After inducing a superficial wound, the fundamental barrier function of the skin is gone and variety of mechanisms is provoked at the site in order to exterminate foreign material and finally restore the skin's normal structure. But this may only be successful to a limited extent; the wound will never achieve the utmost tensile strength of unwounded skin and to an upper limit only approximately 70 percent of the skin's fundamental functions may be restored to a wound most of the skin's essential functions will be restored to a wound [9]. However, this necessitates the transport of inflammatory cells, matrix molecules, cytokines, nutrients and chemokine’s at the site of wound as well as an augment in metabolic demand. These entire processes takes place simultaneously and also can be classified as proliferative, inflammatory and remodeling wound healing phases.
After hemostasis, the inflammatory phase of wound healing commences quickly and has the prime purpose of clearing germs and foreign material from the wound while also limiting damage to a small area. Vasodilation increases vascular permeability, allowing monocytes and neutrophils to migrate at the site of wound. The conversion of monocytes to macrophages which is typically considered to as the controller of this inflammatory phase of wound healing and this is aided by a complicated interplay of cytokines [10]. In addition to phagocytizing and digesting tissue debris and residual neutrophils, macrophages also produces growth factors and cytokines that enhance cell migration and tissue proliferation. After around three days from the very first wound, the proliferative phase focuses on fibroblasts as well as on collagen and ground substances formation which will serve as the foundation for the prior wound area's tissue scaffold. Meanwhile, within the granulation tissue, endothelial cells undergo a rapid development phase and angiogenesis develops that result in a dense vascular network that supplies this very active healing area. The wound enters a remodeling or maturation phase after about 2-3 weeks during which the collagen type is returned to normal [11] and wound tissue grows, ensuing in complete cross-linking and rebuilding of a quite similar ordinary structure, also the vascular network is swiftly regressing [12] Figure 1.
Plants Researched For Wound Healing
Azadirachta indica commonly called as Neem, belongs to Meliaceae family and it is a well-known versatile plant of India for more than 2000 years possess wide range of medicinal activities. It has Nimbidine, nimbin, sodium nimbidate and nimbolideas main active constituent extracted from oil of its seed kernels which showed antibacterial, anti-pyretic and anti-inflammatory properties [13]. Purohit et al. evaluated the wound healing activity of ethanolic extract of leaves of A. indica in male albino rats [14] Figure 2.
Centella asiatica is also known as indian pennywort which is used to facilitate healing of wounds. It is a member of Umbelliferae family. It is also known as Brahmi in Unani medicine and Mandookaparni in Ayurveda medicine system to facilitate the healing of the chronic ulcers in terms of their distance, depth, and scale, extracts from the Centella asiatica aerial sections are reported. Asiaticoside isolated from the Centella asiatica has been shown to promote epithelialization and collagen deposition in a punch type wound. The main therapeutic active constituents in centella plant are saponins containing triterpens namely, asiatic acid, madecassic acid, asiaticoside and madecassoside [15]. Centella asiatica isolated triterpenes improve collagen remodeling and the synthesis of glycosaminoglycans. In addition, it has been shown that oral administration of made cassoside from Centella asiatica promotes collagen synthesis and angiogenesis at the wound site [16, 7]. It has been reported that 1% C. asiatica extract cream improves chronic wound healing [17] Figure 3.
Curcuma longa is a well-known spice and it is a member of Zingiberaceae family. It is very useful herb of Ayurveda medicine. It has been used throughout India since thousands of years. It is also known as “Indian saffron” or turmeric 70. Major components of turmeric are named curcuminoids, which include mainly curcumin (diferuloyl methane), demethoxycurcumin, and bis demethoxycurcumin. Curcumin, a potent antioxidant is believed to be the most important bioactive component of the herb turmeric and has the properties like antioxidant, anti-inflammatory, anti-platelet, cholesterol lowering, antibacterial and anti-fungal effects [18, 12]. Purohit et al. investigated that ethanolic extract of C. longa rhizomes have better and faster wound healing effect than standard drug Povidone Iodine ointment on excision wound model [19] Figure 4.
Eucalyptus is also known as Dinkum oil and is belonging to family Myrtaceae. Eucalyptus contains some compounds such as cineole which is also known as eucalyptol. It not only contains cineole but contains other compounds such as pinene, camphene, and phellandrene, citronellal, geranyl acetate. It is traditionally used for skin care including burns, blisters, herpes, cuts, wounds, skin infections and insect bites [20] Figure 5.
Aloe Vera is a member of Asphodelaceae family and comprises of many natural bioactive compounds, including basic and complex such as glycosides, polysaccharides, saponins, pyrocatechol, anthraquinones, acemannan, phytol, oleic acid, and water-soluble polysaccharides. Acetone extracts from Aloe vera leaves show greater antimicrobial activity than that of alcohol and aqueous extracts. Aloe Vera tends to be more susceptible to gram-positive bacterial species than gram-negative species. Saponins, acemannan, and anthraquinone derivatives are compounds with a proven antimicrobial activity. Acemannan, a large Aloe Vera muco polysaccharide (mesoglycan), is an effective stimulator for the operation of macrophages and T cells and induces the transcription of proinflammatory mRNAs (including IL-1af, IL-1β, IL-6, TNF-af, PGE2, and nitrous oxide). Mesoglycan moieties bind and absorb endogenous mitogenic inhibitors and species of reactive oxygen, which promote phagocytosis. Coincidentally, glycan’s stabilize, prolonging their function, the secreted cytokines, growth factors, and other bio actives [21, 22]. Topically applied acemannan, acting through cyclin D1 and AKT/mTOR signaling pathways has been documented to significantly reduce the time for wound closure [23] Figure 6.
Calendula officinalis L. is so called pot marigold and is one of the medicinal plants in the Asteraceae family. Phytochemical evaluations of Calendula officinalis showed the presence of the flavonoids, flavonol glycosides, coumarins, saponins, triterpenes, alcohol triterpenes, fatty acid esters, carotenoids, essential oils, hydrocarbons, and fatty acids. Some studies have reported biological activities in Calendula officinalis including wound healing and anti-inflammatory effects [24, 25]. Calendula officinalis aerial part hydro alcoholic extracts, have ant nociceptive and anti-inflammatory activities in chemical pain and anti-inflammatory tests [26] Figure 7.
Dalbergia sissoo is a member of fabaceae family and is a wind-dispersed tropical tree [27, 28]. It is the third biggest angiosperm plant family, with roughly 730 genera and over 19400 species, including legumes. The plant is a home-grown of Afghanistan, Bangladesh, India, Bhutan, Malaysia, and Kenya etc [29]. It consists of different phytochemicals such as alkaloids, flavonoids, protease inhibitors, isoflavonoids, cyanogenic glycosides, and lectins. Non protein amino acids, amines, coumarins, phenylpropanoid, diterpenes, sesquiterpenes and triterpenes. Medicinal property of this plant contributes to important secondary metabolites present in the plant [30, 31]. Chemically, leaves include sissotrin as well as an isoflavon-O-glycoside [32]. Traditionally Dalbergia sissoo used to treat many diseases such as scabies, expectorant, antipyretic and disorders of leucoderma, blood and dysentery [28] D. sissoo leaves are used to treat animals suffering from non-specific diarrhea in India and Nepal and expectorant refrigerant and skin in ailments [28] Figure 8.
Henna is the popular name for Lawsonia inermis Linn, which is used in traditional medicine. It is a member of Lythraceae. Lawsonia inermis leaves are used to make an important cosmetic dye. Henna leaves has been utilize as a color for nails, hair, hands and textiles in the Far East, Middle East, and Northern Africa for ages. Henna was also used to cure skin issues, headaches, jaundice, amebiasis and spleen enlargement [33, 34].Flavonoids, coumarins, triterpenoids, steroids, and xanthones are among the phyto constituents found in it. It has been reported earlier that aqueous extract of plant consists of alkaloids, saponins, terpenoids, Quinone’s, xanthones, carbohydrates, coumarins, phenolic compounds, flavonoids, proteins, quinines, fat (6%), resin (2-3%) and tannins (7-8%) [35,36]. It is beneficial as coloring agent, bronchitis, boils, scabies, Spleen related disorders, dysuria, platelet function defect, skin diseases, diuretic and antibacterial. Leaves paste was used as antimicrobial, anti-inflammatory, trypsin inhibitory, wound healing, antioxidant, and anti-parasitic, analgesic, and antipyretic, tuber, anti-tumoral activity [37] Figure 9.
Conclusion
Wound healing remains a challenging clinical problem for long ago. Much research has been done on wound care, with emphasis on the advancement of acute and chronic wound treatment techniques in Ayurveda. Plants are the potent healers because they provoke their pair mechanism naturally. The aim of this review is to give data on medicinal plants which were already reported and to give information on the medicinal properties, ethno pharmacological uses of Indian medicinal plants.
REFERENCES
- Natarajan S, Williamson D, Stiltz AJ et al., Am J Clin Dermatol. 2000, 1(5): p.269-275.
- Alonso JE, Lee J, Burgess AR et al., Surg Clin North Am. 1996, 76(4): p.879-903.
- Lazarus GS, Cooper DM, Knighton DR et al., Wound Repair Regen. 1994, 2(3): p.165-170.
- Bischoff M, Kinzl L and Schmelz A. Der Unfallchirurg. 1999, 102(10): p.797-804.
- GS Schultz, NurMngmt. 1999, 2: 413-429.
- Badri PN and Renu S. Res J Med Plant. 2011, 5: 508-514.
- Sharma A, Khanna S, Kaur G et al., Future J Pharm Sci. 2021, 7(1): p.1-13.
- Robson MC. Surg Clin North Am. 1997, 77(3): p.637-650.
- Stadelmann WK, Digenis AG and Tobin GR. Am J Surg. 1998, 176(2): p.26S-38S.
- Wynn TA and Barron L. Semin Liver Dis. 2010, 30(3): p. 245-257).
- Gurtner GC, Werner S, Barrandon Y et al., Nature. 2008, 453(7193): p.314-321.
- Saini S, Dhiman A and Nanda S. Int J Pharm Sci Res. 2016, 7(5): p.1809.
- Biswas K, Chattopadhyay I, Banerjee RK et al., Curr Sci. 2002: p.1336-1345.
- Purohit SK, Solanki R, Soni R et al., AJPTech. 2013, 3(2): p.73-75.
- Hashim P, Sidek H, Helan MHM et al., mol. 2011, 16(2): p.1310-1322.
- Babu MK, Prasad OS and Murthy TE. J Chem Pharm Res. 2011, 3(3): p.353-62.
- Sharma A, Mittal A, Puri V et al., Biotech. 2020, 10(11): p.1-13.
- Naz S, Jabeen S, Ilyas S et al., Pak J Bot. 2010, 42(1): p.455-62.
- Purohit SK, Solanki R, Mathur V et al., Asian J. Pharm Res. 2013, 3(2): p.79-81.
- Hukkeri VI, Karadi RV, Akki KS et al., Indian drugs. 2002, 39(9): p.481-483.
- Galehdari H, Negahdari S, Kesmati M et al., BMC Complement Altern. 2016, 16(1): p.1-9.
- Choi S and Chung MH. In Seminars in integrative med. 2003, 1(1): p. 53-62.
- Oryan A, Naeini A, Nikahval B et al., Veterinarski arhiv. 2010, 80(4): p.509-522.
- Parente LML, Lino Júnior RDS, Tresvenzol LMF et al., Evid based Complement Altern Med. 2012, 2012.
- Preethi KC and Kuttan R. J Basic Clin Physiol Pha. 2009, 20(1): p.73-80.
- Farahpour MR. Inter J Pharm Tech Res. 2014, 1: p.974-4304.
- Wang BY, Shi L, Ruan ZY et al., Genet Mol Res. 2011, 10(1): p.114-120.
- Saini S and Sharma S. Int J Pharm Res. 2012, 3: 464-71.
- Lazarus GS, Cooper DM, Knighton DR et al., Wound Repair Regen. 1994, 2(3): p.165-170.
- Kapoor CS, Bamniya BR and Kapoor K. Environ Monit Assess. 2013, 185(9): p.7565-7580.
- Arabi Z and Sardari S. Avicenna J Med Biotechnol. 2010, 2(2): p.93.
- Rana V and Kumar V. Int J Chemtech Res. 2011, 3: p.483-487.
- Zumrutdaland E and Ozaslan M. Int J Pharmacol. 2012, 896: 483-9.
- Afzal M, Oriquat Al, Al-Hussan JM et al., Heterocycle. 1980, 14: 1973-6.
- Al-Snafi AE. Int J Curr Pharm Res. 2019, 11(6): p.1-10.
- Jain VC, Shah DP, Sonani NG et al., Romanian J Biol-Plant Biol. 2010, 55: p.127-133.
- Borade AS, Kale BN and Shete RV. Int J Pharm Life Sci. 2011, 2(1): p.536-541. Indexed at, Google Scholar
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref