Abstract
Decolouring of Synthetic Waste Water by Chemical Oxidation
Author(s): Rathinakumar Vedachalam* and Sugumaran KaruppiahDischarge of waste water from the textile industry into the natural water bodies causes serious problem to the aquatic environment. This waste water contains dyes which are mainly made of chemicals in which majority of these chemicals categorized as inorganic. General methods used to treat this waste water can remove organic chemicals, also these methods consumes more time. Hence treating this waste water has become a great challenge to the chemists and environmental engineers. As the dyes which are used in the textile industry are inorganic in nature, the common practices used to treat this is waste water are no longer useful. Hence new methods are required to treat these inorganic chemicals and these methods must be must faster than the common practices. Hence chemical oxidation methods came into existence. The paper deals with chemical oxidation process for the color removal of waste water by using Fenton’s reagent. In this process Hydrogen Peroxide is used as oxidant. Fenton’s reagent uses ferrous ions to make OH- radicals. These OH-oxidizes the chemicals present in waste water and decolorizes it. So, in this paper focuses on preparation of Fenton reagent and optimum time requirement that can remove methylene blue (MB) color.
Select your language of interest to view the total content in your interested language
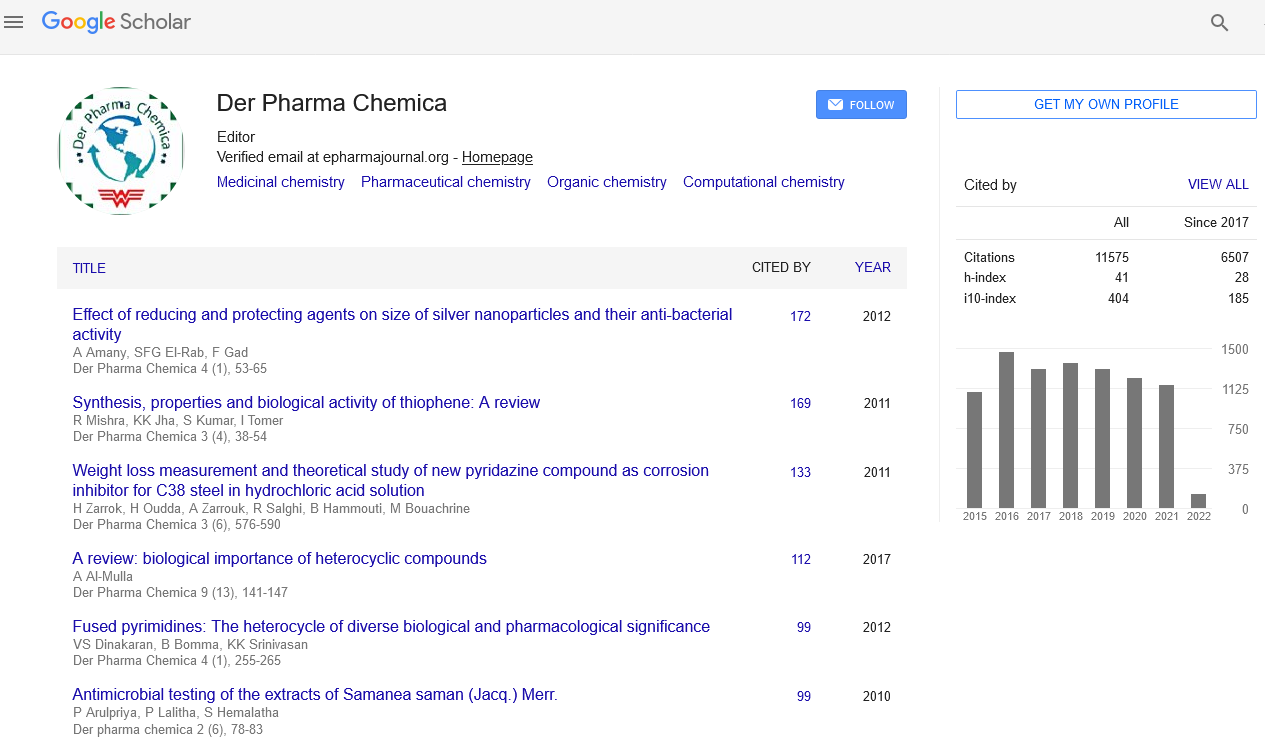
Google Scholar citation report
Citations : 25868
Der Pharma Chemica received 25868 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons
DOWNLOADS