Abstract
Evaluation of Antioxidant Property and Binding Constant with DNA using Quinones by Electrochemical Technique
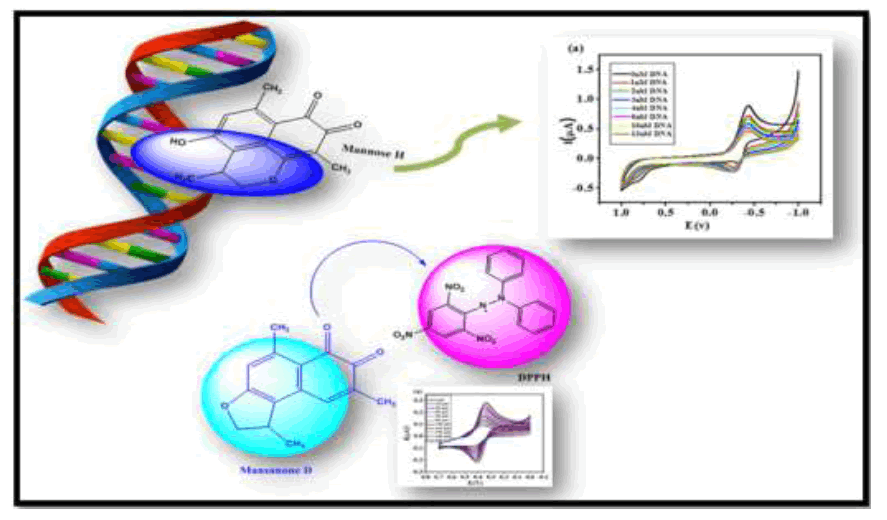
Author(s): Ramaraj R Deepaa, Abraham A Daniel Arulrajb, Abdul Kather A. Sheik Mideenc, Rathinasamy R Gandhidasanb , Vairathevar Sivasamy VS VasanthaAntioxidant property and interaction with Deoxyribonucleic Acid (DNA) of the following four quinones: Mansanone-D, Mansanone-H, Sissoidenone and 4-methoxy dalbergione, were investigated at pH=7.4 using cyclic voltammetry technique. The parameters like binding constant, binding site size and binding free energy were also determined from voltammetric data. On comparison of binding constant and binding site size among all quinones, Mansanone-H reveals strong binding capacity (1.04 x 109) with DNA than others. The negative sign of binding constants (ΔG) indicate the spontaneity of binding of quinones with DNA. The electrochemical behavior of DPPH radical was investigated in Dimethyl Sulfoxide (DMSO) and used this radical to determine antioxidant activity of quinones by cyclic voltammetry method. The scavenging effect of the radical was monitored by the decrease in peak currents with increase in the concentration of quinones. The binding constant and Gibbs free energy values were calculated from voltammetric data.
Graphical Abstract
Select your language of interest to view the total content in your interested language
Google Scholar citation report
Citations : 15261
Der Pharma Chemica received 15261 citations as per Google Scholar report
Der Pharma Chemica peer review process verified at publons



