Research Article - Der Pharma Chemica ( 2022) Volume 14, Issue 3
Ethanolic Leaf Extract of Gongronema Latifolium Prevents Depletion of Nitric Oxide and Anti-Oxidants Levels in Streptozotocin Induced Diabetic Rats.
Gabriel O Ujong1*, Justin A Beshel2, Idara Asuquo Okon3 and Daniel D Owu22Department of Human Physiology, University of Calabar, Nigeria
3Department of Physiology, PAMO University of Medical Sciences Port Harcourt, Rivers State, Nigeria
Gabriel O Ujong, Department of Human Physiology, Cross River University of Technology, Okuku Campus, Nigeria, Email: ujong4gab44@yahoo.com
Received: 06-Feb-2022, Manuscript No. dpc-22-53635; Accepted Date: Feb 08, 2022 ; Editor assigned: 08-Feb-2022, Pre QC No. dpc-22-53635; Reviewed: 24-Feb-2022, QC No. dpc-22-53635; Revised: 02-Mar-2022, Manuscript No. dpc-22-53635; Published: 10-Mar-2022, DOI: 10.4172/0975-413X.14.3.22-27
Abstract
Lipid peroxidation is a major disorder that disrupts cellular integrity, resulting in many pathological conditions. This work was designed to evaluate the effect of ethanolic leaf extract of Gongronema latifolium (GL) on nitric oxide, anti-oxidants (SOD, total anti-oxidant capacity) and lipid peroxidation product (MDA) in Streptozotocin-induced diabetic rats. 40 Wistar rats of both sexes (150g-200g) were divided into 5 groups (n=8). Group 1 (control) received 1mL normal saline orally. Group 2 (GL) received 200 mg/kg b.wt. Of GL orally. Group 3 (DM) received 65mg/kg b.wt. of streptozotocin (STZ) i.p. for two days with an interval of one day in between. Group 4 (DM+GL) received 65mg/kg b.wt. Of STZ + 200 mg/kg GL o.p. Group 5 (DM + Insulin) received 65mg/kg bw of STZ insulin (s.c.). Animals with blood glucose above 200mg/dL were declared diabetic. The regiment lasted for 28days. The result of the study shows a significant (P<0.05) increase in the glucose level of the diabetic group compared to the control. Intervention with GL and insulin significantly reduced the glucose level towards normal. TAC, SOD and nitric oxide levels were significantly (p<0.01) decreased with a corresponding increase of MDA in the diabetic group. Treatment with GL and insulin significantly (p<0.01) reversed these changes by increasing the levels of TAC, SOD, NO and decreases MDA levels towards normal. In conclusion, Gongronema Latifolium as well as insulin possess anti-diabetic, anti-oxidant properties and protect the cell membrane against lipid peroxidation in diabetic model.
Keywords
Gongronema Latifolium; Diabetes Mellitus; SOD; MDA; TAC; NO; insulin
Introduction
Diabetes mellitus (DM) is a pathological and metabolic condition characterized by impaired glucose metabolism caused by inadequate insulin action or insulin resistance [1, 2]. Clinically, it is defined as a fasting plasma glucose level >7.8 mmol/l (140mg/dl) or a 2hour post-prandial plasma glucose >11 mmol/l (200 mg/dl) [3]. In DM, blood glucose level is persistently raised above normal range (80-100mg/dl). Diabetes mellitus is generally classified into two Type 1 and Type 2 diabetes with Type 2 being 10 times more common than Type 1 [4]. It is a complicated and chronic disease with complex etiologies [5] which can lead to reduced glucose tolerance, nerve damage, kidney failure, atherosclerosis, stroke, blindness and heart disease.
There is increased prevalence of DM due to population growth, aging, urbanization and lifestyle. Although lifestyle modification plays a greater role in the prevention of diabetes, effective clinical management of diabetes relies on adequate control of blood glucose, which must take into consideration the need to maintain adequate energy in the face of intermittent food intake along with variable exercise and thus variable demand [6]. It is a chronic lifelong condition that affects the body's ability to use the energy found in food. The total number of people with diabetes is projected to rise from 171 million in 2000 to 366 million in 2030 [7].
Gongronema latifolium (Asclepiadaceous) is a herbaceous climber with yellow flowers and stem that yields characteristic milky exudates. It is widespread in Tropical Africa and can be found from Senegal, Chad and Democratic Republic of Congo. It occurs in rainforest, deciduous, and secondary forest, and also in mangrove and disturbed roadside forests, from sea level up to 900m altitude [8]. The leafy vegetable can be propagated by seed. Its common name is ‘amaranth globe’. In Nigeria, G. latifolium is known by different local names. In Nigeria, it has different names such as ‘utasi’ by the Efiks/Ibibios, ‘utazi’ by the Igbos and arokeke by the Yorubas [9]. G. latifolium crude leaf extract is used in the treatment of malaria, diabetes, hypertension, and as a laxative [10].
It possesses antioxidant activity by increasing superoxide dismutase and glutathione peroxidase activities [11] and also reduces renal and hepatic oxidative stress, lipid peroxidation, and increases the glutathione/glutathione disulphide (GSH/GSSG) ratio [12, 13].
Hyperglycemia could suppress or reduce local NO production or act by one of several hypothetical mechanisms. Several studies suggest that microvascular deregulations and impaired endothelial dependent aortic relaxation in DM may primarily involve the endothelial depended nitric oxide-mediated component [14]. Nitric oxide with its increasingly appreciated functional diversity in cell metabolism, microvascular regulation, neuro modulation and diabetes, provides a strong theoretical bridge between Aldose Reductase and Na+/K+ ATPase and the vascular components of early experimental diabetic neuropathy. Nitric oxide deficiency might perturb nerve conduction velocity (NCV) and nerve Na+/K+ ATPase through a non-ischemic mechanism, perhaps interacting with other more well established AR2-related defects in signal transduction [15], involved in modulation of (Na+,K+)-ATPase activity [15].
It is well documented that the diabetic state induces increased cellular oxygen consumption [16]. NO regulates the delivery of oxygen to tissue by setting the level of vascular tone and blood pressure and by inhibiting cellular oxygen consumption. Thus, alterations in NO activity might contribute to the development of diabetes-induced renal hypoxia [17].
In pancreatic β-cells, NO exerts positive and negative regulation of insulin secretion and anti- and pro-apoptotic activity at low and high concentrations, respectively. The deregulation of NO production or metabolism in β-cells participates in the pathogenesis and natural course of Type-2 DM. Inflammatory cytokines produced during the process of Type-1 DM induce the expression of induced NOS. Micromolar levels of NO produced by induced NOS lead to impaired insulin secretion and β-cell apoptosis [18]. In a study using human umbilical vein endothelial cells (HUVEC), showed that high glucose exposure enhanced NO production at early time point but reduced subsequently after exposure for a longer duration. They observed increased nitric oxide levels in diabetic patients and diabetic patients with coronary artery diseases, and no significant change in nitric oxide levels in diabetic patients with hypertension [19].
Preparation of Gongronema latifolium Extract
Gongronema latifolium was harvested in a local farm in Ugep, yakurr Local Government, Cross River State. It was identified and authenticated in the Department of Botany and Ecological Studies, University of Calabar, Calabar. The leaves were washed and dried under shade, for seven days then blended into fine powder and stored in a cool dry place away from light until required for use. The powdered leaves (400g) was dissolved in 1250ml of ethanol (BDH Ltd Poole, England) in the evening, and allowed to stay overnight. The mixture was then centrifuged in the morning of the next day and the supernatant collected. The supernatant was suction filtered, first, using Whatmann no. 1 filter paper, and then a second time using cellulose filter paper. The filtrate was evaporated to dryness at 30°C using a vacuum rotatory evaporator (Caframo, VV2000, and Ohio) and water bath (Caframo, WB2000). This extraction gave a percentage yield of about 4.3% using a digital sensitive weighing balance. The extract was stored at 4°C till further use [12].
Experiment Animal
Forty adult Wistar rats of both sexes equal number weighing 150-200 g were used for the study. The animals were purchased from the animal house of the Department of Human Physiology, Cross River University of Technology, Okuku Campus, Nigeria. The animals were kept under experimentally controlled conditions (27±2°C, with 12hours light-dark cycle). All the animals were acclimatized for one week before the commencement of the experiment. They were kept in plastic cages and fed with normal rat chow and tap water ad libitum. Ethical approval was obtained from the Faculty of Basic Medical Sciences University of Calabar Animal Research and Ethical Committee with ethical number No: 019PY20317).
Experimental Design
The Wistar rats were divided into five groups equal no of both sexes of eight rats each and were placed in different rat cages for proper identification. The rats were acclimatized for one week and grouping is shown below.
Group 1: Control
Group 2: STZ Diabetic (DM)
Group 3: Gongronema latifolium (GL)
Group 4: DM+GL
Group 5: DM + Insulin
Administration of Gongronema Latifolium Extract and Insulin
The plant extracts reconstituted in distilled water (vehicle) were administered via oral gastric intubation at a dose of 200 mg/kg body weight daily to groups 3 and 4 animals. Insulin (10 IU/kg body weight) was administered subcutaneously once daily to group 5, [11]. Treatment lasted for 28 days.
Induction of Diabetes Mellitus
A mixture of 7-acetoxy-4-methyl coumarin (0.01 mol) and anhydrous AlCl3 (0.03 mol) was heated under anhydrous conditions in an oil bath at 125oC and the temperature was raised during 2.5 hr period to 170ºC. To the reaction mixture, crushed ice and dilute HCl were added with stirring and the mixture was left for 2-3 hr in order to decompose the complex. The separated product was filtered, washed with water and recrystallized from ethanol. A single spot in TLC indicated the purity of the compound. m.p. 159-160ºC, yield 68%. Diabetes was induced in overnight fasted rats by a single intra peritoneal injection of a freshly prepared solution of streptozotocin (STZ) 65 mg/kg b.wt. i.p. USA in citrate buffer (0.1 M, pH 4.5). STZ was obtained from Sigma Aldrich Chemicals Company; St. Louis, MO. two days after the induction of diabetes mellitus, a confirmatory test was done on fasting blood obtained via tail puncture using a portable glucometer and strips (Accu-Chek, Roche, Germany). Animals with fasting glucose level above 200 mg/dl were considered diabetic.
Collection of Blood Samples
After 28 days of treatment, the animals were fasted for 12hours overnight and fasting blood glucose level determined using Accu-chek Glucometer. The animals were anaesthetized using 5% chloroform soaked in cotton wool and blood samples collected via cardiac puncture using sterile needles into plane and EDTA sample bottles. The blood samples in plane tubes were then centrifuged at 1000rpm for 10 minutes for serum collection and stored for subsequent biochemical analysis.
Determination of Nitric Oxide
An alcoholic solution (30ml) of Schiff bases (I-IV) 1 mmol was refluxed with 1 mmol of CoCl2.6H2O/NiCl2.6H2O/CuCl2.2H2O in 30ml ethanol solution on steam bath for 1h. Then, to the reaction mixture 2 mmol of sodium acetate was added and refluxtion was continued for 3h. The separated complex was filtered, washed thoroughly with water, Ethanol, Ether and finally dried in vacuum over fused CaCl2. The Griess assay was employed to assess total levels of the metabolites NO2- and NO3 in AD and control plasma samples. Plasma sample were deproteinized by the addition of zinc sulphate and centrifugation at 10,000g for 10 minutes, and the supernatant removed for analysis any NO3 present in the supernatant was reduced to NO2 by the addition of vanadium (III) chloride. Griess reagent was subsequently added and a colour change which is directly proportional to the level of NO2 was observed. This colour change was measured by a spectrophotometer at 540nm and the amount of nitrate in the samples can be determined by comparison to a standard curve of known nitrite concentrations. Total nitrite and nitrite levels were represented as total nitrite oxide metabolites (NOx) and measurement of NOx was considered a direct marker of in vivo NO production [20, 21].
Determination of Malondialdehyde (MDA) Level
MDA was determined following TBARS kit manufacturer’s procedure (Calbiochem Chemicals, USA). 100μl of sample or standard and 100μl of sodium dodecyl sulphate solution were added to 5ml vial and vortex to mix. The reagent (4ml) was added down the side of each vial. The vials were placed in vigorously boiling water for 1 hour, and later removed and placed in ice bath for 10 minutes. The 96-well plate was loaded with 150μl of sample or standard, and the absorbance read at 540nm using the microplate reader within 30 minutes [22].
MDA (μM) = [(corrected absorbance) – (y-intercept)/slope]
Determination of Total Antioxidant Capacity
The cell Biolabs’ OxiSelectTM total Antioxidant Capacity assay kit manufacturer’s procedure was used to estimate total antioxidant capacity. 20 μl of the diluted uric acid and sample were added to the 96-well microtiter. The reaction buffer (180 μl) was lso added to each well using a multichannel pipette and shake vigorously. The initial absorbance wa obtained by reading the plate over the spectrophotometer at 490nm. To initiate the reaction, 50 μl of copper ion reagent was added to each well and incubated for 5 minutes. 50 μl of the buffer reagent was again added to terminate the reaction, and the plate read off at 490nm [23].
Total antioxidant capacity = [(corrected absorbance) – (y-intercept)/slope].
Data Analysis
Results are expressed as mean ± SEM. Data was analyzed using the Graph Pad Prism software (version 6.0). Analysis of variance (ANOVA) followed by Turkey post hoc test was used for comparison among and between the different groups. Probability level of p<0.05 was accepted as significant.
Result
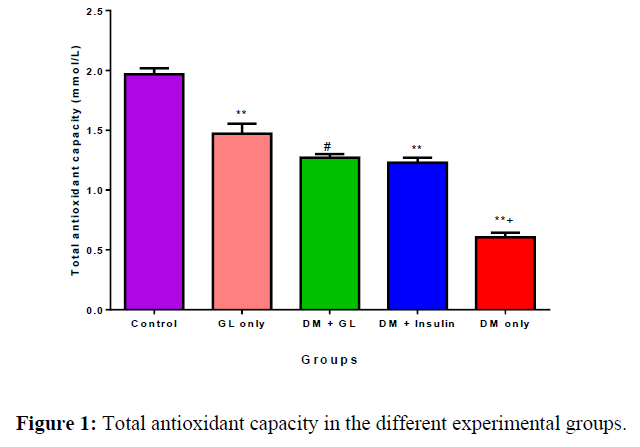
Effect of GL and Insulin on Total Antioxidant Capacity (TAC) in Diabetic Rats
The mean serum TAC concentration (mmol/L) in the control, GL only, DM+GL, DM + Insulin, and DM only groups were 1.97 ± 0.05, 147 ± 0.08, 1.27 ± 0.03, 1.23 ± 0.04, and 0.60 ± 0.04 respectively. TAC was significantly (p<0.05) reduced in the DM group compared with the control. Treatment with GL and insulin increased TAC towards normal (Figure 1).
Values are expressed as mean ± SEM, n = 8.
* = p<0.05 compared with control;
** = p<0.01 compared with control;
a = p<0.05 compared with DM+ insulin.
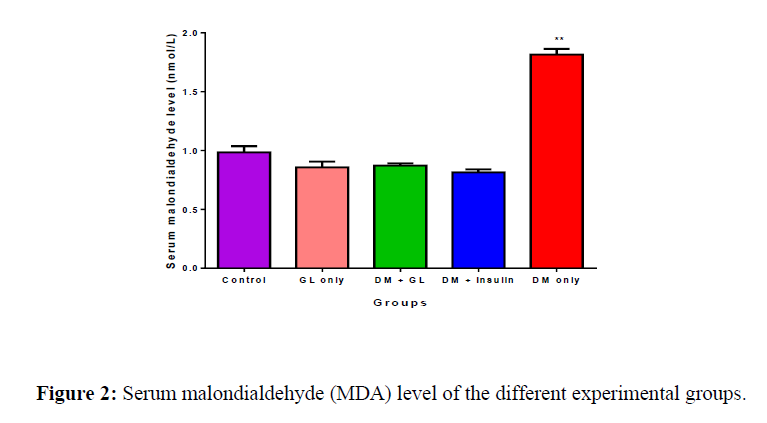
Effect of GL and Insulin on Malondialdehyde (MDA) Level in Diabetic Rats
The mean serum MDA in the control, GL only, DM+GL, DM+Insulin, and DM only groups was 0.986 ± 0.0508nmol/L, 0.857 ± 0.0481nmol/L, 0.871 ± 0.0184nmol/L, 0.814 ± 0.0261nmol/L and 1.87 ± 0.0477nmol/L respectively. MDA was significantly (p<0.05) increased in the DM group compared with the control. Treatment with GL reduced MDA level towards normal (Figure 2).
Values are expressed as mean ± SEM, n = 8.
**=p<0.01 compared to all other groups
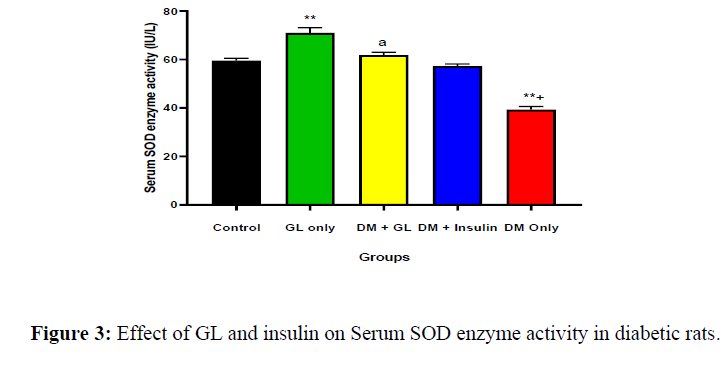
Effect of GL and Insulin on Superoxide Dismutase in Diabetic Rats
The mean serum levels in the control, GL only, DM+GL, DM+Insulin, and DM only was 60 ± 1.0 IU/L, 71 ± 2.1 IU/L, 62 ± 1.1 IU/L, 57 ± 0.87 IU/L, and 39 ± 1.3 IU/L, respectively. The result showed a significant (p<0.01) decrease in the DM only group compared with the control. But treatment with GL increased the SOD level towards normal. This is presented in (Figure 3).
Values are expressed as mean ± SEM, n = 8.
* = p<0.05 compared with control;
** = p<0.01 compared with control;
a = p<0.05 compared with DM+ insulin
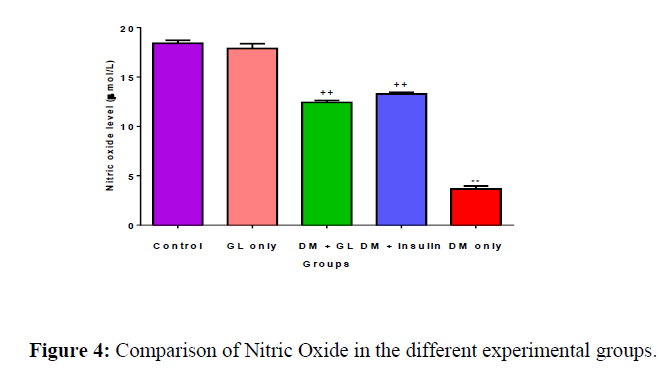
Effect of GL and Insulin on Nitric Oxide Levels in Diabetic Rats
The mean serum levels in control, GL only, DM+GL, DM+Insulin, and DM only was 18.4 ± 0.303 μmol/L, 15.9 ± 0.317 μmol/L, 12.4 ± 0.202 μmol/L, 13.3 ± 0.184 μmol/L, and 2.83 ± 0.0989 μmol/L respectively. The result shows a significant (p<0.05) decrease in the DM group compared with the control. Treatment with GL significantly increased it towards normal. This is presented in (Figure 4).
Values are expressed as mean ± SEM, n = 8.
** = p<0.01 compared with other groups;
++ = p<0.01 compared with control
Discussion
This study was aimed at investigating the role of ethanolic leaf extract of Gongronema latifolium on anti-oxidant and anti-per oxidative effect in streptozotocin-induced diabetic rats. And the effect of the extract on blood glucose levels of rats treated with G. latifolium and insulin were also determined.
The decrease in blood glucose levels seen in animals treated with insulin and GL is in agreement with previous studies that reported hypoglycaemic effect of GL in diabetic condition [24, 25]. Gongronema latifolium contain phytochemicals with a combine potential for reducing blood glucose level. Phytochemicals present in Gongronema Latifolium include flavonoid, polyphenols, coumarins, saponins, tanins and alkaloids. Studies have shown that phytochemicals like flavonoid and glycosides have hypoglycaemic effect by inhibiting alpha glucosidase enzyme or by regeneration of the damaged pancreatic beta cells [26]. G. latifolium can ameliorate this damage due to its antioxidant properties.
Oxidative stress due to increase production of free radicals in diabetic condition can also increase levels of inflammatory markers or cytokines [27]. Nitric oxide is a vasodilator produced by an enzyme, endothelium nitric oxide synthetase (eNOS) in the endothelium and in the brain by inducible nitric oxide synthatase (iNOS) [28]. Reduction in NO synthesis is one of the risk factors for cardiovascular diseases including hypertension [29]. An increase in the production of asymmetric dimethyl arginine (ADMA) an inhibitor to nitric oxide synthase has been reported during diabetes, thus resulting in a decreased availability of NO in diabetic conditions [30]. Increased reactive oxygen species (ROS) production reduce the bioavailability of nitric oxide (NO) to form peroxynitrite which uncoupled the endothelial nitric oxide (NO) synthase to form a harmful superoxide generating enzymes which in turn contributes to vascular damage. Measurement of NO concentration is a useful adjunct in the laboratory diagnosis of hypertension and in the complication of DM [31].
The results from this work showed a significant decrease in NO concentration in the diabetic condition that was increased with Gongronema latifolium (DM+GL) treatment. The group treated with insulin, standard ant diabetic drugs also shows a significant increase in NO concentration when compared to oxidative stress induced diabetic group without treatment. The decrease in serum NO level in DM group is in agreement with a previous study that reported a decrease in level of serum NO in this disease condition [30]. This increase in NO concentration in Gongronema latifolium group is probably due to the action of alkaloid and tannins which are present in the plant extract. A study has shown that tannins and related compounds induce nitric oxide synthase in infection [32].
Superoxide dismutase (SODs) constitutes the first line of defence mechanism against ROS. It is produced at any location where an electron transport chain is present in the cell such as mitochondria, chloroplasts, microsomes, glyoxysomes, peroxisomes and the cytosol. The results of our present study showed a significant increase in SOD activity in the two treated groups DM+GL group and DM+ insulin group when compared to the oxidative stress induced group (DM Only). The SOD activity also increased significantly in Gongronema latifolium only group when compared to the normal control group. The results are in agreement with previous study [9]. The significant decrease in SOD activity in DM group is probably due to increase production of free radicals causing its inhibition. The increase in SOD activity is therefore probably due to antioxidant property of G. latifolium which can also minimize the extent of damage caused by free radicals [24].
Malondialdehyde (MDA) is found in biological tissues and fluids and is used as a biomarker of lipid peroxidation [33]. It is one of the end products of oxidative reactions [34]. The results for MDA level show a significant increase in diabetic induced oxidative stress group (DM only). The increase MDA level in animals induced with DM suggests that there was increase lipid peroxidation. The result in the GL only group when compared to the control group (NC) shows the ability of G. latifolium to combat lipid peroxidation. This decrease in lipid peroxidation may be due to the present of active anti-oxidant phytochemicals such as coumarins and polyphenols in G. latifolium leaf extract [35].
REFERENCES
- American Diabetes Association. Diabetes care. 2013, 36(Suppl 1): p.S67.
- Kumar S, Behl T, Sachdeva M et al., Life Sci. 2021, 264: p.118661.
- Maher A, Jermeen EY, Manar SM et al., Med J Cairo Univ. 2019, 87(3): p.909-917.
- Barron E, Bakhai C, Kar P et al., Lancet Diabetes Endocrinol. 2020, 8(10): p.813-822.
- Norris CM, Yip CY, Nerenberg KA et al., J Am Heart Assoc. 2020, 9(4): p.e015634.
- Green ME, Shah BR, Slater M et al., CMAJ. 2020, 192(33): p.E937-E945.
- Wild S, Roglic G, Green A et al., Diabetes care. 2004, 27(5): p.1047-1053.
- Ujong GO, Beshel JA, Nkanu E et al., J drug deliv ther. 2022, 12(1): p.45-50.
- Ugochukwu NH and Babady NE. Fitoterapia. 2002, 73(7-8): p.612-618.
- Morebise O. European J Med Plants. 2015, 10(1): p.1-9.
- Ugochukwu NH, Babady NE, Cobourne M et al., J Biosci. 2003, 28(1): p.1-5.
- Ugochukwu NH and Cobourne MK. Clin Chim Acta. 2003, 336(1-2): p.73-81.
- Okon IA, Ufot UF, Onoyeraye UG et al., J pharm biomed res. 2019.
- Ross M, Lithgow H, Hayes L et al., Biochem. Cell Biol of Ageing: Part II Clin Sci. 2019, p.311-338.
- Tsou KANG, Snyder GL. Proc Natl Acad Sci. 1993, 90(8): p.3462-3465.
- Buerk DG, Lamkin-Kennard K and Jaron D. Free Radic Biol Med. 2003, 34(11): p.1488-1503.
- Palm F, Buerk DG, Carlsson PO et al., Diabetes. 2005, 54(11): p.3282-3287.
- Kaneko YK and Ishikawa T. J Pharmacol Sci. 2013, 123(4): p.295-300.
- Adela R, Nethi SK, Bagul PK et al., PloS one. 2015, 10(4): p.e0125270.
- Moshage H, Kok B, Huizenga JR et al., Clin Chem. 1995, 41(6): p.892-896.
- Miranda KM, Espey MG and Wink DA. Nitric oxide. 2001, 5(1): p.62-71.
- Ohkawa H, Ohishi N and Yagi K. Anal Biochem. 1979, 95(2): p.351-358.
- Frei B, Stocker R and Ames BN. Proc Natl Acad Sci. 1988, 85(24): p.9748-9752.
- Owu DU, Nwokocha CR, Obembe AO et al., West Indian Med J. 2012, 61(9).
- Adeeyo AO, Odelade KA, Msagati TA et al., J King Saud Univ Sci. 2020, 32(4): p.2349-2357.
- Ojo OA, Osukoya OA, Ekakitie LI et al., J Diabetes Metab Disord. 2020, 19(1): p.469-481.
- Anvarifard P, Anbari M, Ostadrahimi A et al., Nutr Metab. 2022, 19(1): p.1-23.
- Alp NJ and Channon KM. Arterioscler Thromb Vasc Biol. 2004, 24(3): p.413-420.
- Liu X, Xu X, Shang R et al., Nitric oxide. 2018, 78: p.113-120.
- Sacks DB. Diabetes care. 2011, 34(2): p.518-523.
- Kolodziej H, Burmeister A, Trun W et al., Bioorg Med.Chem. 2005, 13(23): p.6470-6476.
- Tsikas D. Anal Biochem. 2017, 524: p.13-30.
- Huszar G and Vigue L. J Androl. 1994, 15(1): p.71-77.
- Todorova I, Simeonova G, Kyuchukova D et al., J Comp Pathol. 2005, 13(4): p.190-194.
- Beshel JA, Palacios J, Beshel FN et al., J Basic Clin Physiol Pha. 2020, 31(1).
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar , Crossref
Indexed at , Google Scholar, Crossref
Indexed at , Google Scholar, Crossref